User login
MSBase study validates therapy for relapse in secondary progressive MS
An observational cohort study of a prospective international database of patients with multiple sclerosis has reported that medical therapy can reduce disability progression in patients with active secondary progressive MS (SPMS) who are prone to inflammatory relapses.
The international researchers, led by Nathanial Lizak, MMBS, of the University of Melbourne, conducted the cohort study of 1,621 patients with SPMS from the MSBase international registry, which prospectively collected the information from 1995 to 2018. Their findings were published in JAMA Neurology.
“ wrote Dr. Lizak and colleagues of the MSBase Study Group.
Therapy’s impact on disease progression
To ensure that they had timely data on the early disease course of all study patients, they researchers only included patients whose diagnosis and first documented Expanded Disability Status Scale (EDSS) score were no more than 24 months apart. At SPMS conversion, 1,494 patients had an EDSS score of less than 7 (on a scale of 0-10); during the follow-up period, 267 of them (17.9%) crossed over the threshold of 7.
Dr. Lizak and colleagues noted that early treatment during relapsing remitting MS didn’t impact outcomes after SPMS conversion.
For evaluating the MS Severity Score (MSSS), the study split the cohort into three groups depending on the efficacy of therapy: low-efficacy treatments, mostly consisting of interferon-beta and glatiramer acetate; medium-efficacy treatments, mostly fingolimod and dimethyl fumarate; and high-efficacy treatments, predominately natalizumab and mitoxantrone.
The MSSS progression slope in the cohort had an average reduction of 0.02 points per year. “For patients who experienced superimposed relapses during SPMS, a reduced MSSS progression slope during SPMS was observed among those who received disease-modifying therapies for a greater proportion of time during SPMS,” Dr. Lizak and colleagues wrote.
MSSS progression slope reduction was more pronounced in the medium- and high-efficacy groups, with a reduction of beta 0.22 (P = .06) and 0.034 (P = .002), respectively.
“Based on our models, the level of disability in patients with active SPMS who are continuously treated with high-efficacy immunotherapies would progress more slowly in comparison with the general population with MS by a mean (standard deviation) of 1.56 (4.60) deciles over 10 years,” Dr. Lizak and colleagues wrote.
While the researchers cited a number of studies that didn’t support immunotherapy for SPMS, they also did cite the EXPAND trial to support treatment with siponimod in SPMS patients (Lancet. 2018;391:1263-73). The ASCEND trial of natalizumab enrolled a largely relapse-free cohort and found no link between treatment and disability progression in SPMS (Lancet Neurol. 2018;17:405-15), and a previous report by the MSBase Study Group found no benefit of therapy when adjusted for SPMS relapse rates (Neurology. 2017;89:1050-9).
“Together with the present study, the existing data converge on the suggestion that relapses during SPMS provide a therapeutic target and a marker of future response to immunotherapy during SPMS,” Dr. Lizak and colleagues wrote.
Challenging dogma
Commenting on the research, Mark Freedman, HBSc, MSc, MD, said that the MSBase study makes an important contribution to the literature on management of SPMS. “Up until this point we’ve been basing our assumptions on secondary progressive MS on natural history studies, which are actually quite old, dating back 20-30 years.” Dr. Freedman is senior scientist in the Neuroscience Program at the Ottawa Hospital Research Institute and professor of medicine in neurology at the University of Ottawa.
He said “the most damaging” of those studies was by the late Christian Confavreaux, MD, and colleagues in Lyon, France (N Engl J Med. 2000;343:1430-8), that reported relapses didn’t alter the progression of disability. “In other words, once you’re in EDSS of 4, it’s a runaway train; it doesn’t matter what you do,” Dr. Freedman said.
“That was kind of dogma for years,” he said. “The reason this publication is important is because it’s suggesting that’s not the case.” In other words, the MSBase cohort study is validating what neurologists have been doing in the real world for years: treating patients with SPMS who have relapses.
Dr Lizak reported receiving travel compensation from Merck outside the scope of the study. His coauthors reported numerous financial relationships.
SOURCE: Lizak N et al. JAMA Neurol. 2020 Jul 27. doi: 10.1001/jamaneurol.2020.2453
An observational cohort study of a prospective international database of patients with multiple sclerosis has reported that medical therapy can reduce disability progression in patients with active secondary progressive MS (SPMS) who are prone to inflammatory relapses.
The international researchers, led by Nathanial Lizak, MMBS, of the University of Melbourne, conducted the cohort study of 1,621 patients with SPMS from the MSBase international registry, which prospectively collected the information from 1995 to 2018. Their findings were published in JAMA Neurology.
“ wrote Dr. Lizak and colleagues of the MSBase Study Group.
Therapy’s impact on disease progression
To ensure that they had timely data on the early disease course of all study patients, they researchers only included patients whose diagnosis and first documented Expanded Disability Status Scale (EDSS) score were no more than 24 months apart. At SPMS conversion, 1,494 patients had an EDSS score of less than 7 (on a scale of 0-10); during the follow-up period, 267 of them (17.9%) crossed over the threshold of 7.
Dr. Lizak and colleagues noted that early treatment during relapsing remitting MS didn’t impact outcomes after SPMS conversion.
For evaluating the MS Severity Score (MSSS), the study split the cohort into three groups depending on the efficacy of therapy: low-efficacy treatments, mostly consisting of interferon-beta and glatiramer acetate; medium-efficacy treatments, mostly fingolimod and dimethyl fumarate; and high-efficacy treatments, predominately natalizumab and mitoxantrone.
The MSSS progression slope in the cohort had an average reduction of 0.02 points per year. “For patients who experienced superimposed relapses during SPMS, a reduced MSSS progression slope during SPMS was observed among those who received disease-modifying therapies for a greater proportion of time during SPMS,” Dr. Lizak and colleagues wrote.
MSSS progression slope reduction was more pronounced in the medium- and high-efficacy groups, with a reduction of beta 0.22 (P = .06) and 0.034 (P = .002), respectively.
“Based on our models, the level of disability in patients with active SPMS who are continuously treated with high-efficacy immunotherapies would progress more slowly in comparison with the general population with MS by a mean (standard deviation) of 1.56 (4.60) deciles over 10 years,” Dr. Lizak and colleagues wrote.
While the researchers cited a number of studies that didn’t support immunotherapy for SPMS, they also did cite the EXPAND trial to support treatment with siponimod in SPMS patients (Lancet. 2018;391:1263-73). The ASCEND trial of natalizumab enrolled a largely relapse-free cohort and found no link between treatment and disability progression in SPMS (Lancet Neurol. 2018;17:405-15), and a previous report by the MSBase Study Group found no benefit of therapy when adjusted for SPMS relapse rates (Neurology. 2017;89:1050-9).
“Together with the present study, the existing data converge on the suggestion that relapses during SPMS provide a therapeutic target and a marker of future response to immunotherapy during SPMS,” Dr. Lizak and colleagues wrote.
Challenging dogma
Commenting on the research, Mark Freedman, HBSc, MSc, MD, said that the MSBase study makes an important contribution to the literature on management of SPMS. “Up until this point we’ve been basing our assumptions on secondary progressive MS on natural history studies, which are actually quite old, dating back 20-30 years.” Dr. Freedman is senior scientist in the Neuroscience Program at the Ottawa Hospital Research Institute and professor of medicine in neurology at the University of Ottawa.
He said “the most damaging” of those studies was by the late Christian Confavreaux, MD, and colleagues in Lyon, France (N Engl J Med. 2000;343:1430-8), that reported relapses didn’t alter the progression of disability. “In other words, once you’re in EDSS of 4, it’s a runaway train; it doesn’t matter what you do,” Dr. Freedman said.
“That was kind of dogma for years,” he said. “The reason this publication is important is because it’s suggesting that’s not the case.” In other words, the MSBase cohort study is validating what neurologists have been doing in the real world for years: treating patients with SPMS who have relapses.
Dr Lizak reported receiving travel compensation from Merck outside the scope of the study. His coauthors reported numerous financial relationships.
SOURCE: Lizak N et al. JAMA Neurol. 2020 Jul 27. doi: 10.1001/jamaneurol.2020.2453
An observational cohort study of a prospective international database of patients with multiple sclerosis has reported that medical therapy can reduce disability progression in patients with active secondary progressive MS (SPMS) who are prone to inflammatory relapses.
The international researchers, led by Nathanial Lizak, MMBS, of the University of Melbourne, conducted the cohort study of 1,621 patients with SPMS from the MSBase international registry, which prospectively collected the information from 1995 to 2018. Their findings were published in JAMA Neurology.
“ wrote Dr. Lizak and colleagues of the MSBase Study Group.
Therapy’s impact on disease progression
To ensure that they had timely data on the early disease course of all study patients, they researchers only included patients whose diagnosis and first documented Expanded Disability Status Scale (EDSS) score were no more than 24 months apart. At SPMS conversion, 1,494 patients had an EDSS score of less than 7 (on a scale of 0-10); during the follow-up period, 267 of them (17.9%) crossed over the threshold of 7.
Dr. Lizak and colleagues noted that early treatment during relapsing remitting MS didn’t impact outcomes after SPMS conversion.
For evaluating the MS Severity Score (MSSS), the study split the cohort into three groups depending on the efficacy of therapy: low-efficacy treatments, mostly consisting of interferon-beta and glatiramer acetate; medium-efficacy treatments, mostly fingolimod and dimethyl fumarate; and high-efficacy treatments, predominately natalizumab and mitoxantrone.
The MSSS progression slope in the cohort had an average reduction of 0.02 points per year. “For patients who experienced superimposed relapses during SPMS, a reduced MSSS progression slope during SPMS was observed among those who received disease-modifying therapies for a greater proportion of time during SPMS,” Dr. Lizak and colleagues wrote.
MSSS progression slope reduction was more pronounced in the medium- and high-efficacy groups, with a reduction of beta 0.22 (P = .06) and 0.034 (P = .002), respectively.
“Based on our models, the level of disability in patients with active SPMS who are continuously treated with high-efficacy immunotherapies would progress more slowly in comparison with the general population with MS by a mean (standard deviation) of 1.56 (4.60) deciles over 10 years,” Dr. Lizak and colleagues wrote.
While the researchers cited a number of studies that didn’t support immunotherapy for SPMS, they also did cite the EXPAND trial to support treatment with siponimod in SPMS patients (Lancet. 2018;391:1263-73). The ASCEND trial of natalizumab enrolled a largely relapse-free cohort and found no link between treatment and disability progression in SPMS (Lancet Neurol. 2018;17:405-15), and a previous report by the MSBase Study Group found no benefit of therapy when adjusted for SPMS relapse rates (Neurology. 2017;89:1050-9).
“Together with the present study, the existing data converge on the suggestion that relapses during SPMS provide a therapeutic target and a marker of future response to immunotherapy during SPMS,” Dr. Lizak and colleagues wrote.
Challenging dogma
Commenting on the research, Mark Freedman, HBSc, MSc, MD, said that the MSBase study makes an important contribution to the literature on management of SPMS. “Up until this point we’ve been basing our assumptions on secondary progressive MS on natural history studies, which are actually quite old, dating back 20-30 years.” Dr. Freedman is senior scientist in the Neuroscience Program at the Ottawa Hospital Research Institute and professor of medicine in neurology at the University of Ottawa.
He said “the most damaging” of those studies was by the late Christian Confavreaux, MD, and colleagues in Lyon, France (N Engl J Med. 2000;343:1430-8), that reported relapses didn’t alter the progression of disability. “In other words, once you’re in EDSS of 4, it’s a runaway train; it doesn’t matter what you do,” Dr. Freedman said.
“That was kind of dogma for years,” he said. “The reason this publication is important is because it’s suggesting that’s not the case.” In other words, the MSBase cohort study is validating what neurologists have been doing in the real world for years: treating patients with SPMS who have relapses.
Dr Lizak reported receiving travel compensation from Merck outside the scope of the study. His coauthors reported numerous financial relationships.
SOURCE: Lizak N et al. JAMA Neurol. 2020 Jul 27. doi: 10.1001/jamaneurol.2020.2453
FROM JAMA NEUROLOGY
All NSAIDs raise post-MI risk but some are safer than others: Next chapter
Patients on antithrombotics after an acute MI will face a greater risk for bleeding and secondary cardiovascular (CV) events if they start taking any nonaspirin NSAID, confirms a large observational study.
Like other research before it, the new study suggests those risks will be much lower for some nonaspirin NSAIDs than others. But it may also challenge at least some conventional thinking about the safety of these drugs, and is based solely on a large cohort in South Korea, a group for which such NSAID data has been in short supply.
“It was intriguing that our study presented better safety profiles with celecoxib and meloxicam versus other subtypes of NSAIDs,” noted the report, published online July 27 in the Journal of the American College of Cardiology.
Most of the NSAIDs included in the analysis, “including naproxen, conferred a significantly higher risk for cardiovascular and bleeding events, compared with celecoxib and meloxicam,” wrote the authors, led by Dong Oh Kang, MD, Korea University Guro Hospital, Seoul, South Korea.
A main contribution of the study “is the thorough and comprehensive evaluation of the Korean population by use of the nationwide prescription claims database that reflects real-world clinical practice,” senior author Cheol Ung Choi, MD, PhD, of the same institution, said in an interview.
“Because we included the largest number of patients of any comparable clinical studies on NSAID treatment after MI thus far, our study may allow the generalizability of the adverse events of NSAIDs to all patients by constituting global evidence encompassing different population groups,” Dr. Choi said.
The analysis has limitations along with its strengths, the authors acknowledged, including its observational design and potential for confounding not addressed in statistical adjustments.
Observers of the study concurred, but some cited evidence pointing to such confounding that is serious enough to question the entire study’s validity.
Among the cohort of more than 100,000 patients followed for an average of about 2.3 years after their MI, the adjusted risk of thromboembolic CV events went up almost 7 times for those who took any NSAID for at least 4 consecutive weeks, compared with those who didn’t take NSAIDs, based on prescription records.
Their adjusted risk of bleeding events – which included gastrointestinal, intracranial, respiratory, or urinary tract bleeding or posthemorrhagic anemia, the group writes – was increased 300%.
There was wide variance in the adjusted hazard ratios for outcomes by type of NSAID. The risk of CV events climbed from a low of about 3 with meloxicam and almost 5 for celecoxib to more than 10 and 12 for naproxen and dexibuprofen, respectively.
The hazard ratios for bleeding ranged from about 3 for both meloxicam and celecoxib to more than 6 for naproxen.
Of note, celecoxib and meloxicam both preferentially target the cyclooxygenase type 2 (COX-2) pathway, and naproxen among NSAIDs once had a reputation for relative cardiac safety, although subsequent studies have challenged that notion.
“On the basis of the contemporary guidelines, NSAID treatment should be limited as much as possible after MI; however, our data suggest that celecoxib and meloxicam could be considered possible alternative choices in patients with MI when NSAID prescription is unavoidable,” the group wrote.
They acknowledged some limitations of the analysis, including an observational design and the possibility of unidentified confounders; that mortality outcomes were not available from the National Health Insurance Service database used in the study; and that the 2009-2013 span for the data didn’t allow consideration of more contemporary antiplatelet agents and direct oral anticoagulants.
Also, NSAID use was based on prescriptions without regard to over-the-counter usage. Although use of over-the-counter NSAIDs is common in Korea, “most MI patients in Korea are prescribed most medications, including NSAIDs, in the hospital. So I think that usage of over-the-counter NSAIDs did not change the results,” Dr. Choi said.
“This study breaks new ground by demonstrating cardiovascular safety of meloxicam (and not only of celecoxib), probably because of its higher COX-2 selectivity,” wrote the authors of an accompanying editorial, Juan J. Badimon, PhD, and Carlos G. Santos-Gallego, MD, both of the Icahn School of Medicine at Mount Sinai, New York.
Notably, “this paper rejects the cardiovascular safety of naproxen, which had been suggested classically and in the previous Danish data, but that was not evident in this study.” The finding is consistent with the PRECISION trial, in which both bleeding and CV risk were increased with naproxen versus other NSAIDs, observed Dr. Badimon and Dr. Santos-Gallego.
They agreed with the authors in recommending that, “although NSAID treatment should be avoided in patients with MI, if the use of NSAIDs is inevitable due to comorbidities, the prescription of celecoxib and meloxicam could be considered as alternative options.”
But, “as no study is perfect, this article also presents some limitations,” the editorial agreed, citing some of the same issues noted by Dr. Kang and associates, along with potential confounding by indication and the lack of “clinical information to adjust (e.g., angiographic features, left ventricular function).”
“There’s undoubtedly residual confounding,” James M. Brophy, MD, PhD, a pharmacoepidemiologist at McGill University, Montreal, said in an interview.
The 400%-900% relative risks for CV events “are just too far in left field, compared to everything else we know,” he said. “There has never been a class of drugs that have shown this sort of magnitude of effect for adverse events.”
Even in PRECISION with its more than 24,000 high-coronary-risk patients randomized and followed for 5 years, Dr. Brophy observed, relative risks for the different NSAIDs varied by an order of magnitude of only 1-2.
“You should be interpreting things in the context of what is already known,” Dr. Brophy said. “The only conclusion I would draw is the paper is fatally flawed.”
The registry included 108,232 primarily male patients followed from their first diagnosed MI for CV and bleeding events. About 1.9% were prescribed at least one NSAID for 4 or more consecutive weeks during the follow-up period averaging 2.3 years, the group reported.
The most frequently prescribed NSAID was diclofenac, at about 72% of prescribed NSAIDs in the analysis for CV events and about 69% in the bleeding-event analysis.
Adding any NSAID to post-MI antithrombotic therapy led to an adjusted HR of 6.96 (P < .001) for CV events and 4.08 (P < .001) for bleeding events, compared with no NSAID treatment.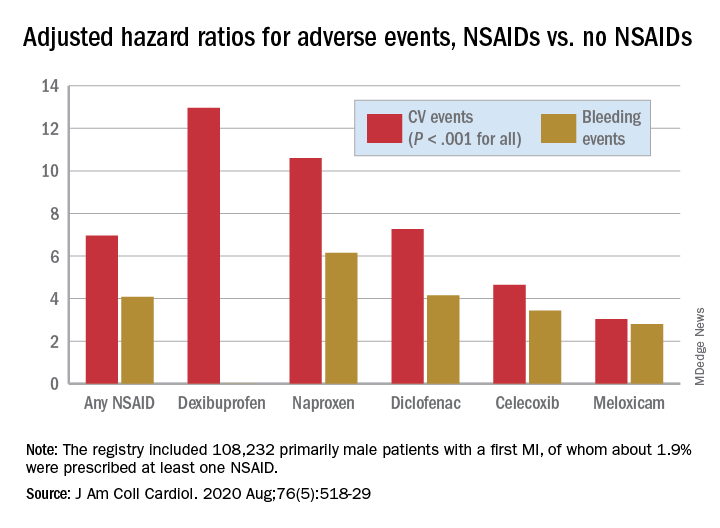
The 88% of the cohort who were on dual-antiplatelet therapy with aspirin and clopidogrel showed very nearly the same risk increases for both endpoints.
Further studies are needed to confirm the results “and ensure their generalizability to other populations,” Dr. Choi said. They should be validated especially using the claims data bases of countries near Korea, “such as Japan and Taiwan, to examine the reproducibility of the results in similar ethnic populations.”
That the study focused on a cohort in Korea is a strength, contended the authors as well as Dr. Badimon and Dr. Santos-Gallego, given “that most data about NSAIDs were extracted from Western populations, but the risk of thrombosis/bleeding post-MI varies according to ethnicity,” according to the editorial
Dr. Brophy agreed, but doubted that ethnic differences are responsible for variation in relative risks between the current results and other studies. “There are pharmacogenomic differences between different ethnicities as to how they activate these drugs. But I suspect that sort of difference is really minor. Maybe it leads to a 2% or a 5% difference in risks.”
Dr. Kang and associates, Dr. Badimon, Dr. Santos-Gallego, and Dr. Brophy disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients on antithrombotics after an acute MI will face a greater risk for bleeding and secondary cardiovascular (CV) events if they start taking any nonaspirin NSAID, confirms a large observational study.
Like other research before it, the new study suggests those risks will be much lower for some nonaspirin NSAIDs than others. But it may also challenge at least some conventional thinking about the safety of these drugs, and is based solely on a large cohort in South Korea, a group for which such NSAID data has been in short supply.
“It was intriguing that our study presented better safety profiles with celecoxib and meloxicam versus other subtypes of NSAIDs,” noted the report, published online July 27 in the Journal of the American College of Cardiology.
Most of the NSAIDs included in the analysis, “including naproxen, conferred a significantly higher risk for cardiovascular and bleeding events, compared with celecoxib and meloxicam,” wrote the authors, led by Dong Oh Kang, MD, Korea University Guro Hospital, Seoul, South Korea.
A main contribution of the study “is the thorough and comprehensive evaluation of the Korean population by use of the nationwide prescription claims database that reflects real-world clinical practice,” senior author Cheol Ung Choi, MD, PhD, of the same institution, said in an interview.
“Because we included the largest number of patients of any comparable clinical studies on NSAID treatment after MI thus far, our study may allow the generalizability of the adverse events of NSAIDs to all patients by constituting global evidence encompassing different population groups,” Dr. Choi said.
The analysis has limitations along with its strengths, the authors acknowledged, including its observational design and potential for confounding not addressed in statistical adjustments.
Observers of the study concurred, but some cited evidence pointing to such confounding that is serious enough to question the entire study’s validity.
Among the cohort of more than 100,000 patients followed for an average of about 2.3 years after their MI, the adjusted risk of thromboembolic CV events went up almost 7 times for those who took any NSAID for at least 4 consecutive weeks, compared with those who didn’t take NSAIDs, based on prescription records.
Their adjusted risk of bleeding events – which included gastrointestinal, intracranial, respiratory, or urinary tract bleeding or posthemorrhagic anemia, the group writes – was increased 300%.
There was wide variance in the adjusted hazard ratios for outcomes by type of NSAID. The risk of CV events climbed from a low of about 3 with meloxicam and almost 5 for celecoxib to more than 10 and 12 for naproxen and dexibuprofen, respectively.
The hazard ratios for bleeding ranged from about 3 for both meloxicam and celecoxib to more than 6 for naproxen.
Of note, celecoxib and meloxicam both preferentially target the cyclooxygenase type 2 (COX-2) pathway, and naproxen among NSAIDs once had a reputation for relative cardiac safety, although subsequent studies have challenged that notion.
“On the basis of the contemporary guidelines, NSAID treatment should be limited as much as possible after MI; however, our data suggest that celecoxib and meloxicam could be considered possible alternative choices in patients with MI when NSAID prescription is unavoidable,” the group wrote.
They acknowledged some limitations of the analysis, including an observational design and the possibility of unidentified confounders; that mortality outcomes were not available from the National Health Insurance Service database used in the study; and that the 2009-2013 span for the data didn’t allow consideration of more contemporary antiplatelet agents and direct oral anticoagulants.
Also, NSAID use was based on prescriptions without regard to over-the-counter usage. Although use of over-the-counter NSAIDs is common in Korea, “most MI patients in Korea are prescribed most medications, including NSAIDs, in the hospital. So I think that usage of over-the-counter NSAIDs did not change the results,” Dr. Choi said.
“This study breaks new ground by demonstrating cardiovascular safety of meloxicam (and not only of celecoxib), probably because of its higher COX-2 selectivity,” wrote the authors of an accompanying editorial, Juan J. Badimon, PhD, and Carlos G. Santos-Gallego, MD, both of the Icahn School of Medicine at Mount Sinai, New York.
Notably, “this paper rejects the cardiovascular safety of naproxen, which had been suggested classically and in the previous Danish data, but that was not evident in this study.” The finding is consistent with the PRECISION trial, in which both bleeding and CV risk were increased with naproxen versus other NSAIDs, observed Dr. Badimon and Dr. Santos-Gallego.
They agreed with the authors in recommending that, “although NSAID treatment should be avoided in patients with MI, if the use of NSAIDs is inevitable due to comorbidities, the prescription of celecoxib and meloxicam could be considered as alternative options.”
But, “as no study is perfect, this article also presents some limitations,” the editorial agreed, citing some of the same issues noted by Dr. Kang and associates, along with potential confounding by indication and the lack of “clinical information to adjust (e.g., angiographic features, left ventricular function).”
“There’s undoubtedly residual confounding,” James M. Brophy, MD, PhD, a pharmacoepidemiologist at McGill University, Montreal, said in an interview.
The 400%-900% relative risks for CV events “are just too far in left field, compared to everything else we know,” he said. “There has never been a class of drugs that have shown this sort of magnitude of effect for adverse events.”
Even in PRECISION with its more than 24,000 high-coronary-risk patients randomized and followed for 5 years, Dr. Brophy observed, relative risks for the different NSAIDs varied by an order of magnitude of only 1-2.
“You should be interpreting things in the context of what is already known,” Dr. Brophy said. “The only conclusion I would draw is the paper is fatally flawed.”
The registry included 108,232 primarily male patients followed from their first diagnosed MI for CV and bleeding events. About 1.9% were prescribed at least one NSAID for 4 or more consecutive weeks during the follow-up period averaging 2.3 years, the group reported.
The most frequently prescribed NSAID was diclofenac, at about 72% of prescribed NSAIDs in the analysis for CV events and about 69% in the bleeding-event analysis.
Adding any NSAID to post-MI antithrombotic therapy led to an adjusted HR of 6.96 (P < .001) for CV events and 4.08 (P < .001) for bleeding events, compared with no NSAID treatment.
The 88% of the cohort who were on dual-antiplatelet therapy with aspirin and clopidogrel showed very nearly the same risk increases for both endpoints.
Further studies are needed to confirm the results “and ensure their generalizability to other populations,” Dr. Choi said. They should be validated especially using the claims data bases of countries near Korea, “such as Japan and Taiwan, to examine the reproducibility of the results in similar ethnic populations.”
That the study focused on a cohort in Korea is a strength, contended the authors as well as Dr. Badimon and Dr. Santos-Gallego, given “that most data about NSAIDs were extracted from Western populations, but the risk of thrombosis/bleeding post-MI varies according to ethnicity,” according to the editorial
Dr. Brophy agreed, but doubted that ethnic differences are responsible for variation in relative risks between the current results and other studies. “There are pharmacogenomic differences between different ethnicities as to how they activate these drugs. But I suspect that sort of difference is really minor. Maybe it leads to a 2% or a 5% difference in risks.”
Dr. Kang and associates, Dr. Badimon, Dr. Santos-Gallego, and Dr. Brophy disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients on antithrombotics after an acute MI will face a greater risk for bleeding and secondary cardiovascular (CV) events if they start taking any nonaspirin NSAID, confirms a large observational study.
Like other research before it, the new study suggests those risks will be much lower for some nonaspirin NSAIDs than others. But it may also challenge at least some conventional thinking about the safety of these drugs, and is based solely on a large cohort in South Korea, a group for which such NSAID data has been in short supply.
“It was intriguing that our study presented better safety profiles with celecoxib and meloxicam versus other subtypes of NSAIDs,” noted the report, published online July 27 in the Journal of the American College of Cardiology.
Most of the NSAIDs included in the analysis, “including naproxen, conferred a significantly higher risk for cardiovascular and bleeding events, compared with celecoxib and meloxicam,” wrote the authors, led by Dong Oh Kang, MD, Korea University Guro Hospital, Seoul, South Korea.
A main contribution of the study “is the thorough and comprehensive evaluation of the Korean population by use of the nationwide prescription claims database that reflects real-world clinical practice,” senior author Cheol Ung Choi, MD, PhD, of the same institution, said in an interview.
“Because we included the largest number of patients of any comparable clinical studies on NSAID treatment after MI thus far, our study may allow the generalizability of the adverse events of NSAIDs to all patients by constituting global evidence encompassing different population groups,” Dr. Choi said.
The analysis has limitations along with its strengths, the authors acknowledged, including its observational design and potential for confounding not addressed in statistical adjustments.
Observers of the study concurred, but some cited evidence pointing to such confounding that is serious enough to question the entire study’s validity.
Among the cohort of more than 100,000 patients followed for an average of about 2.3 years after their MI, the adjusted risk of thromboembolic CV events went up almost 7 times for those who took any NSAID for at least 4 consecutive weeks, compared with those who didn’t take NSAIDs, based on prescription records.
Their adjusted risk of bleeding events – which included gastrointestinal, intracranial, respiratory, or urinary tract bleeding or posthemorrhagic anemia, the group writes – was increased 300%.
There was wide variance in the adjusted hazard ratios for outcomes by type of NSAID. The risk of CV events climbed from a low of about 3 with meloxicam and almost 5 for celecoxib to more than 10 and 12 for naproxen and dexibuprofen, respectively.
The hazard ratios for bleeding ranged from about 3 for both meloxicam and celecoxib to more than 6 for naproxen.
Of note, celecoxib and meloxicam both preferentially target the cyclooxygenase type 2 (COX-2) pathway, and naproxen among NSAIDs once had a reputation for relative cardiac safety, although subsequent studies have challenged that notion.
“On the basis of the contemporary guidelines, NSAID treatment should be limited as much as possible after MI; however, our data suggest that celecoxib and meloxicam could be considered possible alternative choices in patients with MI when NSAID prescription is unavoidable,” the group wrote.
They acknowledged some limitations of the analysis, including an observational design and the possibility of unidentified confounders; that mortality outcomes were not available from the National Health Insurance Service database used in the study; and that the 2009-2013 span for the data didn’t allow consideration of more contemporary antiplatelet agents and direct oral anticoagulants.
Also, NSAID use was based on prescriptions without regard to over-the-counter usage. Although use of over-the-counter NSAIDs is common in Korea, “most MI patients in Korea are prescribed most medications, including NSAIDs, in the hospital. So I think that usage of over-the-counter NSAIDs did not change the results,” Dr. Choi said.
“This study breaks new ground by demonstrating cardiovascular safety of meloxicam (and not only of celecoxib), probably because of its higher COX-2 selectivity,” wrote the authors of an accompanying editorial, Juan J. Badimon, PhD, and Carlos G. Santos-Gallego, MD, both of the Icahn School of Medicine at Mount Sinai, New York.
Notably, “this paper rejects the cardiovascular safety of naproxen, which had been suggested classically and in the previous Danish data, but that was not evident in this study.” The finding is consistent with the PRECISION trial, in which both bleeding and CV risk were increased with naproxen versus other NSAIDs, observed Dr. Badimon and Dr. Santos-Gallego.
They agreed with the authors in recommending that, “although NSAID treatment should be avoided in patients with MI, if the use of NSAIDs is inevitable due to comorbidities, the prescription of celecoxib and meloxicam could be considered as alternative options.”
But, “as no study is perfect, this article also presents some limitations,” the editorial agreed, citing some of the same issues noted by Dr. Kang and associates, along with potential confounding by indication and the lack of “clinical information to adjust (e.g., angiographic features, left ventricular function).”
“There’s undoubtedly residual confounding,” James M. Brophy, MD, PhD, a pharmacoepidemiologist at McGill University, Montreal, said in an interview.
The 400%-900% relative risks for CV events “are just too far in left field, compared to everything else we know,” he said. “There has never been a class of drugs that have shown this sort of magnitude of effect for adverse events.”
Even in PRECISION with its more than 24,000 high-coronary-risk patients randomized and followed for 5 years, Dr. Brophy observed, relative risks for the different NSAIDs varied by an order of magnitude of only 1-2.
“You should be interpreting things in the context of what is already known,” Dr. Brophy said. “The only conclusion I would draw is the paper is fatally flawed.”
The registry included 108,232 primarily male patients followed from their first diagnosed MI for CV and bleeding events. About 1.9% were prescribed at least one NSAID for 4 or more consecutive weeks during the follow-up period averaging 2.3 years, the group reported.
The most frequently prescribed NSAID was diclofenac, at about 72% of prescribed NSAIDs in the analysis for CV events and about 69% in the bleeding-event analysis.
Adding any NSAID to post-MI antithrombotic therapy led to an adjusted HR of 6.96 (P < .001) for CV events and 4.08 (P < .001) for bleeding events, compared with no NSAID treatment.
The 88% of the cohort who were on dual-antiplatelet therapy with aspirin and clopidogrel showed very nearly the same risk increases for both endpoints.
Further studies are needed to confirm the results “and ensure their generalizability to other populations,” Dr. Choi said. They should be validated especially using the claims data bases of countries near Korea, “such as Japan and Taiwan, to examine the reproducibility of the results in similar ethnic populations.”
That the study focused on a cohort in Korea is a strength, contended the authors as well as Dr. Badimon and Dr. Santos-Gallego, given “that most data about NSAIDs were extracted from Western populations, but the risk of thrombosis/bleeding post-MI varies according to ethnicity,” according to the editorial
Dr. Brophy agreed, but doubted that ethnic differences are responsible for variation in relative risks between the current results and other studies. “There are pharmacogenomic differences between different ethnicities as to how they activate these drugs. But I suspect that sort of difference is really minor. Maybe it leads to a 2% or a 5% difference in risks.”
Dr. Kang and associates, Dr. Badimon, Dr. Santos-Gallego, and Dr. Brophy disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Study finds no link between platelet count, surgery bleed risk in cirrhosis
The findings raise questions about current recommendations that call for transfusing platelet concentrates to reduce bleeding risk during surgery in cirrhosis patients with extremely low platelet counts, Gian Marco Podda, MD, PhD, said at the International Society on Thrombosis and Haemostasis virtual congress.
The overall rate of perioperative bleeding was 8.9% in 996 patients who underwent excision of hepatocellular carcinoma by resection (42%) or radiofrequency ablation (58%) without platelet transfusion between 1998 and 2018. The rates were slightly higher among 65 patients with platelet count of fewer than 50 × 109/L indicating severe thrombocytopenia, and in 292 patients with counts of 50-100 × 109/L, indicating moderate thrombocytopenia (10.8% and 10.2%, respectively), compared with those with a platelet count of higher than 100 × 109/L (8.1%), but the differences were not statistically significant, said Dr. Podda of the University of Milan (Italy).
The corresponding rates among those who underwent radiofrequency ablation were 8.6%, 5.9%, and 5%, and among those who underwent resection, they were 18.8%, 17.7%, and 15.9%.
On multivariate analysis, factors associated with an increased incidence of major bleeding were low hemoglobin level (odds ratio, 0.57), age over 65 years (OR, 1.19), aspartate aminotransferase level greater than twice the upper limit of normal (OR, 2.12), hepatitis B or C cirrhosis versus cryptogenic cirrhosis (OR, 0.08), and resection versus radiofrequency ablation (OR, 3.74), he noted. Logistic regression analysis showed no significant association between platelet count and major bleeding events.
Mortality, a secondary outcome measure, was significantly higher among those with moderate or severe thrombocytopenia (rate of 5.5% for each), compared with those with mild or no thrombocytopenia (2.4%), Dr. Podda said.
Factors associated with mortality on multivariate analysis were severe liver dysfunction as demonstrated by Model for End-Stage Liver Disease score of 10 or greater versus less than 10 (OR, 3.13) and Child-Pugh B and C score versus Child-Pugh A score (OR, 16.72), advanced tumor status as measured by Barcelona-Clínic Liver Cancer staging greater than A4 versus A1 (OR, 5.78), major bleeding (OR, 4.59), and resection versus radiofrequency ablation (OR, 3.31).
“Low platelet count was associated with an increased risk of mortality at 3 months. However, this association disappeared at the multivariate analysis, which took into account markers of severity of liver cirrhosis,” he said.
Dr. Podda and his colleagues conducted the study in light of a recommendation from a consensus conference of the Italian Association for the Study of Liver Disease and the Italian Society of Internal Medicine that called for increasing platelet count by platelet transfusions in patients with cirrhosis who undergo an invasive procedure and who have a platelet count lower than 50 × 109/L.
“This recommendation mostly stemmed from consideration of biological plausibility prospects rather than being based on hard experimental evidence,” he explained, noting that such severe thrombocytopenia affects about 10% of patients with liver cirrhosis.
Based on the findings of this study, the practice is not supported, he concluded.
Dr. Podda reported honoraria from Sanofi, Boehringer Ingelheim.
SOURCE: Ronca V et al. ISTH 2020, Abstract OC 13.4.
The findings raise questions about current recommendations that call for transfusing platelet concentrates to reduce bleeding risk during surgery in cirrhosis patients with extremely low platelet counts, Gian Marco Podda, MD, PhD, said at the International Society on Thrombosis and Haemostasis virtual congress.
The overall rate of perioperative bleeding was 8.9% in 996 patients who underwent excision of hepatocellular carcinoma by resection (42%) or radiofrequency ablation (58%) without platelet transfusion between 1998 and 2018. The rates were slightly higher among 65 patients with platelet count of fewer than 50 × 109/L indicating severe thrombocytopenia, and in 292 patients with counts of 50-100 × 109/L, indicating moderate thrombocytopenia (10.8% and 10.2%, respectively), compared with those with a platelet count of higher than 100 × 109/L (8.1%), but the differences were not statistically significant, said Dr. Podda of the University of Milan (Italy).
The corresponding rates among those who underwent radiofrequency ablation were 8.6%, 5.9%, and 5%, and among those who underwent resection, they were 18.8%, 17.7%, and 15.9%.
On multivariate analysis, factors associated with an increased incidence of major bleeding were low hemoglobin level (odds ratio, 0.57), age over 65 years (OR, 1.19), aspartate aminotransferase level greater than twice the upper limit of normal (OR, 2.12), hepatitis B or C cirrhosis versus cryptogenic cirrhosis (OR, 0.08), and resection versus radiofrequency ablation (OR, 3.74), he noted. Logistic regression analysis showed no significant association between platelet count and major bleeding events.
Mortality, a secondary outcome measure, was significantly higher among those with moderate or severe thrombocytopenia (rate of 5.5% for each), compared with those with mild or no thrombocytopenia (2.4%), Dr. Podda said.
Factors associated with mortality on multivariate analysis were severe liver dysfunction as demonstrated by Model for End-Stage Liver Disease score of 10 or greater versus less than 10 (OR, 3.13) and Child-Pugh B and C score versus Child-Pugh A score (OR, 16.72), advanced tumor status as measured by Barcelona-Clínic Liver Cancer staging greater than A4 versus A1 (OR, 5.78), major bleeding (OR, 4.59), and resection versus radiofrequency ablation (OR, 3.31).
“Low platelet count was associated with an increased risk of mortality at 3 months. However, this association disappeared at the multivariate analysis, which took into account markers of severity of liver cirrhosis,” he said.
Dr. Podda and his colleagues conducted the study in light of a recommendation from a consensus conference of the Italian Association for the Study of Liver Disease and the Italian Society of Internal Medicine that called for increasing platelet count by platelet transfusions in patients with cirrhosis who undergo an invasive procedure and who have a platelet count lower than 50 × 109/L.
“This recommendation mostly stemmed from consideration of biological plausibility prospects rather than being based on hard experimental evidence,” he explained, noting that such severe thrombocytopenia affects about 10% of patients with liver cirrhosis.
Based on the findings of this study, the practice is not supported, he concluded.
Dr. Podda reported honoraria from Sanofi, Boehringer Ingelheim.
SOURCE: Ronca V et al. ISTH 2020, Abstract OC 13.4.
The findings raise questions about current recommendations that call for transfusing platelet concentrates to reduce bleeding risk during surgery in cirrhosis patients with extremely low platelet counts, Gian Marco Podda, MD, PhD, said at the International Society on Thrombosis and Haemostasis virtual congress.
The overall rate of perioperative bleeding was 8.9% in 996 patients who underwent excision of hepatocellular carcinoma by resection (42%) or radiofrequency ablation (58%) without platelet transfusion between 1998 and 2018. The rates were slightly higher among 65 patients with platelet count of fewer than 50 × 109/L indicating severe thrombocytopenia, and in 292 patients with counts of 50-100 × 109/L, indicating moderate thrombocytopenia (10.8% and 10.2%, respectively), compared with those with a platelet count of higher than 100 × 109/L (8.1%), but the differences were not statistically significant, said Dr. Podda of the University of Milan (Italy).
The corresponding rates among those who underwent radiofrequency ablation were 8.6%, 5.9%, and 5%, and among those who underwent resection, they were 18.8%, 17.7%, and 15.9%.
On multivariate analysis, factors associated with an increased incidence of major bleeding were low hemoglobin level (odds ratio, 0.57), age over 65 years (OR, 1.19), aspartate aminotransferase level greater than twice the upper limit of normal (OR, 2.12), hepatitis B or C cirrhosis versus cryptogenic cirrhosis (OR, 0.08), and resection versus radiofrequency ablation (OR, 3.74), he noted. Logistic regression analysis showed no significant association between platelet count and major bleeding events.
Mortality, a secondary outcome measure, was significantly higher among those with moderate or severe thrombocytopenia (rate of 5.5% for each), compared with those with mild or no thrombocytopenia (2.4%), Dr. Podda said.
Factors associated with mortality on multivariate analysis were severe liver dysfunction as demonstrated by Model for End-Stage Liver Disease score of 10 or greater versus less than 10 (OR, 3.13) and Child-Pugh B and C score versus Child-Pugh A score (OR, 16.72), advanced tumor status as measured by Barcelona-Clínic Liver Cancer staging greater than A4 versus A1 (OR, 5.78), major bleeding (OR, 4.59), and resection versus radiofrequency ablation (OR, 3.31).
“Low platelet count was associated with an increased risk of mortality at 3 months. However, this association disappeared at the multivariate analysis, which took into account markers of severity of liver cirrhosis,” he said.
Dr. Podda and his colleagues conducted the study in light of a recommendation from a consensus conference of the Italian Association for the Study of Liver Disease and the Italian Society of Internal Medicine that called for increasing platelet count by platelet transfusions in patients with cirrhosis who undergo an invasive procedure and who have a platelet count lower than 50 × 109/L.
“This recommendation mostly stemmed from consideration of biological plausibility prospects rather than being based on hard experimental evidence,” he explained, noting that such severe thrombocytopenia affects about 10% of patients with liver cirrhosis.
Based on the findings of this study, the practice is not supported, he concluded.
Dr. Podda reported honoraria from Sanofi, Boehringer Ingelheim.
SOURCE: Ronca V et al. ISTH 2020, Abstract OC 13.4.
REPORTING FROM THE 2020 ISTH CONGRESS
Anti-CD8a, anti-IL-17A antibodies improved immune disruption in mice with history of NASH
Changes in a variety of T cells in the liver and visceral adipose tissue play a key role in the pathogenesis of nonalcoholic steatohepatitis, according to the results of a murine study.
Mikhaïl A. Van Herck, of the University of Antwerp (Belgium), and associates fed 8-week old mice a high-fat, high-fructose diet for 20 weeks, and then switched the mice to standard mouse chow for 12 weeks. The high-fat, high-fructose diet induced the metabolic syndrome and nonalcoholic steatohepatitis (NASH), accompanied by shifts in T cells. Interleukin-17–producing (Th17 cells increased in the liver, visceral adipose tissue, and blood, while regulatory T cells decreased in visceral adipose tissue, and cytotoxic T (Tc) cells rose in visceral adipose tissue while dropping in the blood and spleen.
These are “important immune disruptions,” the researchers wrote in Cellular and Molecular Gastroenterology and Hepatology. “In particular, visceral adipose tissue Tc cells are critically involved in NASH pathogenesis, linking adipose tissue inflammation to liver disease.”
After the mice were switched from the high-fat, high-fructose diet to standard mouse chow, their body weight, body fat, and plasma cholesterol significantly decreased and their glucose tolerance and insulin sensitivity improved to resemble the metrics of mice fed standard mouse chow throughout the study. Mice who underwent diet reversal also had significantly decreased liver weight and levels of plasma ALT, compared with mice that remained on the high-fat, high-fructose diet. Diet reversal also improved liver histology (nonalcoholic fatty liver disease activity scores), compared with the high-fat, high-fructose diet, the researchers wrote. “Importantly, the NASH was not significantly different between diet-reversal mice and mice fed the control diet for 32 weeks.”
Genetic tests supported these findings. On multiplex RNA analysis, hepatic expression of Acta2, Col1a1, and Col1a3 reverted to normal with diet reversal, indicating a normalization of hepatic collagen. Hepatic expression of the metabolic genes Ppara, Pparg, and Fgf21 also returned to normal, while visceral adipose tissue showed a decrease in Lep and Fgf21 expression and resolution of adipocyte hypertrophy.
However, diet reversal did not reverse inflammatory changes in T-cell subsets. Administering anti-CD8a antibodies after diet reversal decreased Tc cells in all tissue types that were tested, signifying “a biochemical and histologic attenuation of the high-fat, high-fructose diet-induced NASH,” the investigators wrote. Treating the mice with antibodies targeting IL-17A did not attenuate NASH but did reduce hepatic inflammation.
The fact that “the most pronounced effect” on NASH resulted from correcting immune disruption in visceral adipose tissue underscored “the immense importance of adipose tissue inflammation in [NASH] pathogenesis,” the researchers wrote. The finding that diet reversal alone did not reverse inflammation in hepatic or visceral adipose tissue “challeng[es] our current understanding of the reversibility of NASH and other obesity-related conditions.” They called for studies of underlying mechanisms as part of “the search for a medical treatment for NASH.”
Funders included the University Research Fund, University of Antwerp, and Research Foundation Flanders. The researchers reported having no conflicts of interest except that one coinvestigator is the chief science officer at Biocellvia, which performed some histologic analyses.
SOURCE: Van Herck MA et al. Cell Molec Gastroenterol Hepatol. 2020 Apr 20. doi: 10.1016/j.jcmgh.2020.04.010.
The trajectory of nonalcoholic steatohepatitis (NASH) is a public health watershed moment in gastroenterology and hepatology causing unparalleled morbidity, mortality, and societal costs. This study by Van Herck et al. advances our understanding of just how important a two-pronged environmental and biologic approach is to turn the NASH tide. The authors demonstrate that both dietary environmental exposure and biologic tissue-specific T-cell responses are involved in NASH pathogenesis, and that targeting one part of the equation is insufficient to fully mitigate disease. They observed that mice with more severe diet-induced NASH had more Th17 cells in the liver and visceral adipose tissue and more cytotoxic T cells in VAT. Conversely, there were fewer VAT T-regulatory cells in mice with more liver inflammation. The major novelty of this study is that simply changing the diet to a metabolically healthier and weight-reducing diet failed to correct T-cell dysregulation. Only T cell–directed therapies improved this abnormality.
Rotonya M. Carr, MD, is an assistant professor of medicine in the division of gastroenterology at the University of Pennsylvania, Philadelphia. She is a hepatologist, director of the liver metabolism and fatty liver program, and codirector of the human metabolic tissue resource. Dr. Carr receives research and salary support from Intercept Pharmaceuticals.
The trajectory of nonalcoholic steatohepatitis (NASH) is a public health watershed moment in gastroenterology and hepatology causing unparalleled morbidity, mortality, and societal costs. This study by Van Herck et al. advances our understanding of just how important a two-pronged environmental and biologic approach is to turn the NASH tide. The authors demonstrate that both dietary environmental exposure and biologic tissue-specific T-cell responses are involved in NASH pathogenesis, and that targeting one part of the equation is insufficient to fully mitigate disease. They observed that mice with more severe diet-induced NASH had more Th17 cells in the liver and visceral adipose tissue and more cytotoxic T cells in VAT. Conversely, there were fewer VAT T-regulatory cells in mice with more liver inflammation. The major novelty of this study is that simply changing the diet to a metabolically healthier and weight-reducing diet failed to correct T-cell dysregulation. Only T cell–directed therapies improved this abnormality.
Rotonya M. Carr, MD, is an assistant professor of medicine in the division of gastroenterology at the University of Pennsylvania, Philadelphia. She is a hepatologist, director of the liver metabolism and fatty liver program, and codirector of the human metabolic tissue resource. Dr. Carr receives research and salary support from Intercept Pharmaceuticals.
The trajectory of nonalcoholic steatohepatitis (NASH) is a public health watershed moment in gastroenterology and hepatology causing unparalleled morbidity, mortality, and societal costs. This study by Van Herck et al. advances our understanding of just how important a two-pronged environmental and biologic approach is to turn the NASH tide. The authors demonstrate that both dietary environmental exposure and biologic tissue-specific T-cell responses are involved in NASH pathogenesis, and that targeting one part of the equation is insufficient to fully mitigate disease. They observed that mice with more severe diet-induced NASH had more Th17 cells in the liver and visceral adipose tissue and more cytotoxic T cells in VAT. Conversely, there were fewer VAT T-regulatory cells in mice with more liver inflammation. The major novelty of this study is that simply changing the diet to a metabolically healthier and weight-reducing diet failed to correct T-cell dysregulation. Only T cell–directed therapies improved this abnormality.
Rotonya M. Carr, MD, is an assistant professor of medicine in the division of gastroenterology at the University of Pennsylvania, Philadelphia. She is a hepatologist, director of the liver metabolism and fatty liver program, and codirector of the human metabolic tissue resource. Dr. Carr receives research and salary support from Intercept Pharmaceuticals.
Changes in a variety of T cells in the liver and visceral adipose tissue play a key role in the pathogenesis of nonalcoholic steatohepatitis, according to the results of a murine study.
Mikhaïl A. Van Herck, of the University of Antwerp (Belgium), and associates fed 8-week old mice a high-fat, high-fructose diet for 20 weeks, and then switched the mice to standard mouse chow for 12 weeks. The high-fat, high-fructose diet induced the metabolic syndrome and nonalcoholic steatohepatitis (NASH), accompanied by shifts in T cells. Interleukin-17–producing (Th17 cells increased in the liver, visceral adipose tissue, and blood, while regulatory T cells decreased in visceral adipose tissue, and cytotoxic T (Tc) cells rose in visceral adipose tissue while dropping in the blood and spleen.
These are “important immune disruptions,” the researchers wrote in Cellular and Molecular Gastroenterology and Hepatology. “In particular, visceral adipose tissue Tc cells are critically involved in NASH pathogenesis, linking adipose tissue inflammation to liver disease.”
After the mice were switched from the high-fat, high-fructose diet to standard mouse chow, their body weight, body fat, and plasma cholesterol significantly decreased and their glucose tolerance and insulin sensitivity improved to resemble the metrics of mice fed standard mouse chow throughout the study. Mice who underwent diet reversal also had significantly decreased liver weight and levels of plasma ALT, compared with mice that remained on the high-fat, high-fructose diet. Diet reversal also improved liver histology (nonalcoholic fatty liver disease activity scores), compared with the high-fat, high-fructose diet, the researchers wrote. “Importantly, the NASH was not significantly different between diet-reversal mice and mice fed the control diet for 32 weeks.”
Genetic tests supported these findings. On multiplex RNA analysis, hepatic expression of Acta2, Col1a1, and Col1a3 reverted to normal with diet reversal, indicating a normalization of hepatic collagen. Hepatic expression of the metabolic genes Ppara, Pparg, and Fgf21 also returned to normal, while visceral adipose tissue showed a decrease in Lep and Fgf21 expression and resolution of adipocyte hypertrophy.
However, diet reversal did not reverse inflammatory changes in T-cell subsets. Administering anti-CD8a antibodies after diet reversal decreased Tc cells in all tissue types that were tested, signifying “a biochemical and histologic attenuation of the high-fat, high-fructose diet-induced NASH,” the investigators wrote. Treating the mice with antibodies targeting IL-17A did not attenuate NASH but did reduce hepatic inflammation.
The fact that “the most pronounced effect” on NASH resulted from correcting immune disruption in visceral adipose tissue underscored “the immense importance of adipose tissue inflammation in [NASH] pathogenesis,” the researchers wrote. The finding that diet reversal alone did not reverse inflammation in hepatic or visceral adipose tissue “challeng[es] our current understanding of the reversibility of NASH and other obesity-related conditions.” They called for studies of underlying mechanisms as part of “the search for a medical treatment for NASH.”
Funders included the University Research Fund, University of Antwerp, and Research Foundation Flanders. The researchers reported having no conflicts of interest except that one coinvestigator is the chief science officer at Biocellvia, which performed some histologic analyses.
SOURCE: Van Herck MA et al. Cell Molec Gastroenterol Hepatol. 2020 Apr 20. doi: 10.1016/j.jcmgh.2020.04.010.
Changes in a variety of T cells in the liver and visceral adipose tissue play a key role in the pathogenesis of nonalcoholic steatohepatitis, according to the results of a murine study.
Mikhaïl A. Van Herck, of the University of Antwerp (Belgium), and associates fed 8-week old mice a high-fat, high-fructose diet for 20 weeks, and then switched the mice to standard mouse chow for 12 weeks. The high-fat, high-fructose diet induced the metabolic syndrome and nonalcoholic steatohepatitis (NASH), accompanied by shifts in T cells. Interleukin-17–producing (Th17 cells increased in the liver, visceral adipose tissue, and blood, while regulatory T cells decreased in visceral adipose tissue, and cytotoxic T (Tc) cells rose in visceral adipose tissue while dropping in the blood and spleen.
These are “important immune disruptions,” the researchers wrote in Cellular and Molecular Gastroenterology and Hepatology. “In particular, visceral adipose tissue Tc cells are critically involved in NASH pathogenesis, linking adipose tissue inflammation to liver disease.”
After the mice were switched from the high-fat, high-fructose diet to standard mouse chow, their body weight, body fat, and plasma cholesterol significantly decreased and their glucose tolerance and insulin sensitivity improved to resemble the metrics of mice fed standard mouse chow throughout the study. Mice who underwent diet reversal also had significantly decreased liver weight and levels of plasma ALT, compared with mice that remained on the high-fat, high-fructose diet. Diet reversal also improved liver histology (nonalcoholic fatty liver disease activity scores), compared with the high-fat, high-fructose diet, the researchers wrote. “Importantly, the NASH was not significantly different between diet-reversal mice and mice fed the control diet for 32 weeks.”
Genetic tests supported these findings. On multiplex RNA analysis, hepatic expression of Acta2, Col1a1, and Col1a3 reverted to normal with diet reversal, indicating a normalization of hepatic collagen. Hepatic expression of the metabolic genes Ppara, Pparg, and Fgf21 also returned to normal, while visceral adipose tissue showed a decrease in Lep and Fgf21 expression and resolution of adipocyte hypertrophy.
However, diet reversal did not reverse inflammatory changes in T-cell subsets. Administering anti-CD8a antibodies after diet reversal decreased Tc cells in all tissue types that were tested, signifying “a biochemical and histologic attenuation of the high-fat, high-fructose diet-induced NASH,” the investigators wrote. Treating the mice with antibodies targeting IL-17A did not attenuate NASH but did reduce hepatic inflammation.
The fact that “the most pronounced effect” on NASH resulted from correcting immune disruption in visceral adipose tissue underscored “the immense importance of adipose tissue inflammation in [NASH] pathogenesis,” the researchers wrote. The finding that diet reversal alone did not reverse inflammation in hepatic or visceral adipose tissue “challeng[es] our current understanding of the reversibility of NASH and other obesity-related conditions.” They called for studies of underlying mechanisms as part of “the search for a medical treatment for NASH.”
Funders included the University Research Fund, University of Antwerp, and Research Foundation Flanders. The researchers reported having no conflicts of interest except that one coinvestigator is the chief science officer at Biocellvia, which performed some histologic analyses.
SOURCE: Van Herck MA et al. Cell Molec Gastroenterol Hepatol. 2020 Apr 20. doi: 10.1016/j.jcmgh.2020.04.010.
FROM CELLULAR AND MOLECULAR GASTROENTEROLOGY AND HEPATOLOGY
Exploring cannabis use by older adults
Older Americans – people aged 65 or older – make up 15% of the U.S. population, according to the Census Bureau. By the end of this decade, or the year 2030, this proportion will increase to 21% – and all “baby boomers,” those born between 1946 and 1964, will be older than 65.1 Those demographic developments are occurring alongside a change in societal, legal, and public attitudes on cannabis.
Liberalization of cannabis laws across the United States allows for ever easier access to medicinal and recreational cannabis. Traditionally, cannabis use, its effects, and related considerations in the adolescent and young adult populations have commanded significant research attention. Cannabis use in older adults, however, is not as well studied.2 An exploration of trends in cannabis use by older adults and potential impact in terms of health is timely and important.
According to data from the National Survey on Drug Use and Health, cannabis use in adults aged 65 years and older appears to have been increasing steadily over the past 2 decades. Use among this group rose from 0.4% in 2006 and 2007, to 2.9% in 2015 and 2016.2 And, most recently, use climbed from 3.7% in 2017 to 4.2% in 2018.2
Cannabis use also has risen among other adults. For those aged 50-64, cannabis use increased from 2.8% in 2006-2007 to 4.8% in 2012-2013.2,3 Meanwhile, from 2015 to 2016, that number increased to 9.0%.3,4
Past-year cannabis use in the groups of those aged 50-64 and those aged 65 and older appears to be higher in individuals with mental health problems, alcohol use disorder, and nicotine dependence.5,6 Being male and being unmarried appear to be correlated with past-year cannabis use. Multimorbidity does not appear to be associated with past-year cannabis use. Those using cannabis tend to be long-term users and have first use at a much younger age, typically before age 21.
Older adults use cannabis for both recreational and perceived medical benefits. Arthritis, chronic back pain, anxiety, depression, relaxation, stress reduction, and enhancement in terms of creativity are all purported reasons for use. However, there is limited to no evidence for the efficacy of cannabis in helping with those conditions and purposes. Clinical trials have shown that cannabis can be beneficial in managing pain and nausea, but those trials have not been conducted in older adults.7,8
There is a real risk of cannabis use having a negative impact on the health of older adults. To begin with, the cannabis consumed today is significantly higher in potency than the cannabis that baby boomers were introduced to in their youth. The higher potency, combined with an age-related decline in function experienced by some older adults, makes them vulnerable to its known side effects, such as anxiety, dry mouth, tachycardia, high blood pressure, palpitations, wheezing, confusion, and dizziness.
Cannabis use is reported to bring a fourfold increase in cardiac events within the first hour of ingestion.9 Cognitive decline and memory impairment are well known adverse effects of cannabis use. Research has shown significant self-reported cognitive decline in older adults in relation to cannabis use.Cannabis metabolites are known to have an effect on cytochrome P450 enzymes, affecting the metabolism of medication, and increasing the susceptibility of older adults who use cannabis to adverse effects of polypharmacy. Finally, as research on emergency department visits by older adults shows, cannabis use can increase the risk of injury among this cohort.
As in the United States, cannabis use among older adults in Canada has increased significantly. The percentage of older adults who use cannabis in the Canadian province of Ontario, for example, reportedly doubled from 2005 to 2015. In response to this increase, and in anticipation of a rise in problematic use of cannabis and cannabis use disorder in older adults, the Canadian Coalition for Seniors’ Mental Health (through financial support from Substance Use and Addictions Program of Health Canada) has created guidelines on the prevention, assessment, and management of cannabis use disorder in older adults.
In the absence of a set of guidelines specific to the United States, the recommendations made by the coalition should be helpful in the care of older Americans. Among other recommendations, the guidelines highlight the needs for primary care physicians to build a better knowledge base around the use of cannabis in older adults, to screen older adults for cannabis use, and to educate older adults and their families about the risk of cannabis use.9
Cannabis use is increasingly popular among older adults10 for both medicinal and recreational purposes. Research and data supporting its medical benefits are limited, and the potential of harm from its use among older adults is present and significant. Importantly, many older adults who use marijuana have co-occurring mental health issues and substance use disorder(s).
Often, our older patients learn about benefits and harms of cannabis from friends and the Internet rather than from physicians and other clinicians.9 We must do our part to make sure that older patients understand the potential negative health impact that cannabis can have on their health. Physicians should screen older adults for marijuana use. Building a better knowledge base around changing trends and views in/on the use and accessibility of cannabis will help physicians better address cannabis use in older adults.
Mr. Kaleka is a medical student in the class of 2021 at Central Michigan University College of Medicine, Mount Pleasant. He has no disclosures. Mr. Kaleka would like to thank his mentor, Furhut Janssen, DO, for her continued guidance and support in research on mental health in vulnerable populations.
References
1. Vespa J et al. Demographic turning points for the United States: Population projections for 2020 to 2060. Current Population Reports. Washington: U.S. Census Bureau. 2020 Feb.
2. Han BH et al. Addiction. 2016 Oct 21. doi: 10.1111/add.13670.
3. Han BH and Palamar JJ. Drug Alcohol Depend. 2018 Oct;191:374-81.
4. Han BH and Palamar JJ. JAMA Intern Med. 2020 Feb 4;180(4):609-11.
5. Choi NG et al. Drug Alcohol Abuse. 2018;44(2):215-23.
6. Reynolds IR et al. J Am Griatr Soc. 2018 Nov;66(11):2167-71.
7. Ahmed AIA et al. J Am Geriatr Soc. 2014 Feb;62(2):410-1.
8. Lum HD et al. Gerontol Geriatr Med. 2019 Jan-Dec;5:2333721419843707.
9. Bertram JR et al. Can Geriatr J. 2020 Mar;23(1):135-42.
10. Baumbusch J and Yip IS. Clin Gerontol. 2020 Mar 29;1-7.
Older Americans – people aged 65 or older – make up 15% of the U.S. population, according to the Census Bureau. By the end of this decade, or the year 2030, this proportion will increase to 21% – and all “baby boomers,” those born between 1946 and 1964, will be older than 65.1 Those demographic developments are occurring alongside a change in societal, legal, and public attitudes on cannabis.
Liberalization of cannabis laws across the United States allows for ever easier access to medicinal and recreational cannabis. Traditionally, cannabis use, its effects, and related considerations in the adolescent and young adult populations have commanded significant research attention. Cannabis use in older adults, however, is not as well studied.2 An exploration of trends in cannabis use by older adults and potential impact in terms of health is timely and important.
According to data from the National Survey on Drug Use and Health, cannabis use in adults aged 65 years and older appears to have been increasing steadily over the past 2 decades. Use among this group rose from 0.4% in 2006 and 2007, to 2.9% in 2015 and 2016.2 And, most recently, use climbed from 3.7% in 2017 to 4.2% in 2018.2
Cannabis use also has risen among other adults. For those aged 50-64, cannabis use increased from 2.8% in 2006-2007 to 4.8% in 2012-2013.2,3 Meanwhile, from 2015 to 2016, that number increased to 9.0%.3,4
Past-year cannabis use in the groups of those aged 50-64 and those aged 65 and older appears to be higher in individuals with mental health problems, alcohol use disorder, and nicotine dependence.5,6 Being male and being unmarried appear to be correlated with past-year cannabis use. Multimorbidity does not appear to be associated with past-year cannabis use. Those using cannabis tend to be long-term users and have first use at a much younger age, typically before age 21.
Older adults use cannabis for both recreational and perceived medical benefits. Arthritis, chronic back pain, anxiety, depression, relaxation, stress reduction, and enhancement in terms of creativity are all purported reasons for use. However, there is limited to no evidence for the efficacy of cannabis in helping with those conditions and purposes. Clinical trials have shown that cannabis can be beneficial in managing pain and nausea, but those trials have not been conducted in older adults.7,8
There is a real risk of cannabis use having a negative impact on the health of older adults. To begin with, the cannabis consumed today is significantly higher in potency than the cannabis that baby boomers were introduced to in their youth. The higher potency, combined with an age-related decline in function experienced by some older adults, makes them vulnerable to its known side effects, such as anxiety, dry mouth, tachycardia, high blood pressure, palpitations, wheezing, confusion, and dizziness.
Cannabis use is reported to bring a fourfold increase in cardiac events within the first hour of ingestion.9 Cognitive decline and memory impairment are well known adverse effects of cannabis use. Research has shown significant self-reported cognitive decline in older adults in relation to cannabis use.Cannabis metabolites are known to have an effect on cytochrome P450 enzymes, affecting the metabolism of medication, and increasing the susceptibility of older adults who use cannabis to adverse effects of polypharmacy. Finally, as research on emergency department visits by older adults shows, cannabis use can increase the risk of injury among this cohort.
As in the United States, cannabis use among older adults in Canada has increased significantly. The percentage of older adults who use cannabis in the Canadian province of Ontario, for example, reportedly doubled from 2005 to 2015. In response to this increase, and in anticipation of a rise in problematic use of cannabis and cannabis use disorder in older adults, the Canadian Coalition for Seniors’ Mental Health (through financial support from Substance Use and Addictions Program of Health Canada) has created guidelines on the prevention, assessment, and management of cannabis use disorder in older adults.
In the absence of a set of guidelines specific to the United States, the recommendations made by the coalition should be helpful in the care of older Americans. Among other recommendations, the guidelines highlight the needs for primary care physicians to build a better knowledge base around the use of cannabis in older adults, to screen older adults for cannabis use, and to educate older adults and their families about the risk of cannabis use.9
Cannabis use is increasingly popular among older adults10 for both medicinal and recreational purposes. Research and data supporting its medical benefits are limited, and the potential of harm from its use among older adults is present and significant. Importantly, many older adults who use marijuana have co-occurring mental health issues and substance use disorder(s).
Often, our older patients learn about benefits and harms of cannabis from friends and the Internet rather than from physicians and other clinicians.9 We must do our part to make sure that older patients understand the potential negative health impact that cannabis can have on their health. Physicians should screen older adults for marijuana use. Building a better knowledge base around changing trends and views in/on the use and accessibility of cannabis will help physicians better address cannabis use in older adults.
Mr. Kaleka is a medical student in the class of 2021 at Central Michigan University College of Medicine, Mount Pleasant. He has no disclosures. Mr. Kaleka would like to thank his mentor, Furhut Janssen, DO, for her continued guidance and support in research on mental health in vulnerable populations.
References
1. Vespa J et al. Demographic turning points for the United States: Population projections for 2020 to 2060. Current Population Reports. Washington: U.S. Census Bureau. 2020 Feb.
2. Han BH et al. Addiction. 2016 Oct 21. doi: 10.1111/add.13670.
3. Han BH and Palamar JJ. Drug Alcohol Depend. 2018 Oct;191:374-81.
4. Han BH and Palamar JJ. JAMA Intern Med. 2020 Feb 4;180(4):609-11.
5. Choi NG et al. Drug Alcohol Abuse. 2018;44(2):215-23.
6. Reynolds IR et al. J Am Griatr Soc. 2018 Nov;66(11):2167-71.
7. Ahmed AIA et al. J Am Geriatr Soc. 2014 Feb;62(2):410-1.
8. Lum HD et al. Gerontol Geriatr Med. 2019 Jan-Dec;5:2333721419843707.
9. Bertram JR et al. Can Geriatr J. 2020 Mar;23(1):135-42.
10. Baumbusch J and Yip IS. Clin Gerontol. 2020 Mar 29;1-7.
Older Americans – people aged 65 or older – make up 15% of the U.S. population, according to the Census Bureau. By the end of this decade, or the year 2030, this proportion will increase to 21% – and all “baby boomers,” those born between 1946 and 1964, will be older than 65.1 Those demographic developments are occurring alongside a change in societal, legal, and public attitudes on cannabis.
Liberalization of cannabis laws across the United States allows for ever easier access to medicinal and recreational cannabis. Traditionally, cannabis use, its effects, and related considerations in the adolescent and young adult populations have commanded significant research attention. Cannabis use in older adults, however, is not as well studied.2 An exploration of trends in cannabis use by older adults and potential impact in terms of health is timely and important.
According to data from the National Survey on Drug Use and Health, cannabis use in adults aged 65 years and older appears to have been increasing steadily over the past 2 decades. Use among this group rose from 0.4% in 2006 and 2007, to 2.9% in 2015 and 2016.2 And, most recently, use climbed from 3.7% in 2017 to 4.2% in 2018.2
Cannabis use also has risen among other adults. For those aged 50-64, cannabis use increased from 2.8% in 2006-2007 to 4.8% in 2012-2013.2,3 Meanwhile, from 2015 to 2016, that number increased to 9.0%.3,4
Past-year cannabis use in the groups of those aged 50-64 and those aged 65 and older appears to be higher in individuals with mental health problems, alcohol use disorder, and nicotine dependence.5,6 Being male and being unmarried appear to be correlated with past-year cannabis use. Multimorbidity does not appear to be associated with past-year cannabis use. Those using cannabis tend to be long-term users and have first use at a much younger age, typically before age 21.
Older adults use cannabis for both recreational and perceived medical benefits. Arthritis, chronic back pain, anxiety, depression, relaxation, stress reduction, and enhancement in terms of creativity are all purported reasons for use. However, there is limited to no evidence for the efficacy of cannabis in helping with those conditions and purposes. Clinical trials have shown that cannabis can be beneficial in managing pain and nausea, but those trials have not been conducted in older adults.7,8
There is a real risk of cannabis use having a negative impact on the health of older adults. To begin with, the cannabis consumed today is significantly higher in potency than the cannabis that baby boomers were introduced to in their youth. The higher potency, combined with an age-related decline in function experienced by some older adults, makes them vulnerable to its known side effects, such as anxiety, dry mouth, tachycardia, high blood pressure, palpitations, wheezing, confusion, and dizziness.
Cannabis use is reported to bring a fourfold increase in cardiac events within the first hour of ingestion.9 Cognitive decline and memory impairment are well known adverse effects of cannabis use. Research has shown significant self-reported cognitive decline in older adults in relation to cannabis use.Cannabis metabolites are known to have an effect on cytochrome P450 enzymes, affecting the metabolism of medication, and increasing the susceptibility of older adults who use cannabis to adverse effects of polypharmacy. Finally, as research on emergency department visits by older adults shows, cannabis use can increase the risk of injury among this cohort.
As in the United States, cannabis use among older adults in Canada has increased significantly. The percentage of older adults who use cannabis in the Canadian province of Ontario, for example, reportedly doubled from 2005 to 2015. In response to this increase, and in anticipation of a rise in problematic use of cannabis and cannabis use disorder in older adults, the Canadian Coalition for Seniors’ Mental Health (through financial support from Substance Use and Addictions Program of Health Canada) has created guidelines on the prevention, assessment, and management of cannabis use disorder in older adults.
In the absence of a set of guidelines specific to the United States, the recommendations made by the coalition should be helpful in the care of older Americans. Among other recommendations, the guidelines highlight the needs for primary care physicians to build a better knowledge base around the use of cannabis in older adults, to screen older adults for cannabis use, and to educate older adults and their families about the risk of cannabis use.9
Cannabis use is increasingly popular among older adults10 for both medicinal and recreational purposes. Research and data supporting its medical benefits are limited, and the potential of harm from its use among older adults is present and significant. Importantly, many older adults who use marijuana have co-occurring mental health issues and substance use disorder(s).
Often, our older patients learn about benefits and harms of cannabis from friends and the Internet rather than from physicians and other clinicians.9 We must do our part to make sure that older patients understand the potential negative health impact that cannabis can have on their health. Physicians should screen older adults for marijuana use. Building a better knowledge base around changing trends and views in/on the use and accessibility of cannabis will help physicians better address cannabis use in older adults.
Mr. Kaleka is a medical student in the class of 2021 at Central Michigan University College of Medicine, Mount Pleasant. He has no disclosures. Mr. Kaleka would like to thank his mentor, Furhut Janssen, DO, for her continued guidance and support in research on mental health in vulnerable populations.
References
1. Vespa J et al. Demographic turning points for the United States: Population projections for 2020 to 2060. Current Population Reports. Washington: U.S. Census Bureau. 2020 Feb.
2. Han BH et al. Addiction. 2016 Oct 21. doi: 10.1111/add.13670.
3. Han BH and Palamar JJ. Drug Alcohol Depend. 2018 Oct;191:374-81.
4. Han BH and Palamar JJ. JAMA Intern Med. 2020 Feb 4;180(4):609-11.
5. Choi NG et al. Drug Alcohol Abuse. 2018;44(2):215-23.
6. Reynolds IR et al. J Am Griatr Soc. 2018 Nov;66(11):2167-71.
7. Ahmed AIA et al. J Am Geriatr Soc. 2014 Feb;62(2):410-1.
8. Lum HD et al. Gerontol Geriatr Med. 2019 Jan-Dec;5:2333721419843707.
9. Bertram JR et al. Can Geriatr J. 2020 Mar;23(1):135-42.
10. Baumbusch J and Yip IS. Clin Gerontol. 2020 Mar 29;1-7.
IBD: Anti-TNF doses often increased without evidence of inflammation
Clinicians frequently increase doses of anti–tumor necrosis factor (anti-TNF) agents for patients with inflammatory bowel disease (IBD) without objective evidence of active inflammation, based on a retrospective study.
This strategy may be ineffective and expensive, according to lead author Evan D. Elias, MD, of the University of Manitoba, Winnipeg, Canada, and colleagues, who noted that worsening symptoms may have noninflammatory causes, such as coexisting functional disorders.
“Objective evidence of active inflammation is a key part of [the dose escalation] process,” the investigators wrote in the Journal of Clinical Gastroenterology, “however, there are no data assessing whether an adequate evaluation of the cause of persistent gastrointestinal symptoms is routinely undertaken in clinical practice.”
To address this knowledge gap, Dr. Elias and colleagues conducted a retrospective study involving 23 physicians in Manitoba who initiated anti-TNF therapy in 838 patients with IBD during 2005-2016. The primary outcome was rate of dose escalation within 1 year. Secondarily, the investigators audited 11 out of 23 prescribers to determine how frequently patients receiving increased doses underwent objective assessment of inflammation, both in general and in the 90 days preceding dose escalation.
Of note, therapeutic drug monitoring, fecal calprotectin testing, and transabdominal ultrasound were not commonly available to prescribers during the study period.
Within 1 year, almost one-third of patients (32.9%) received increased doses of either adalimumab or infliximab, with no significant difference in escalation practices between the two agents. After approximately 2 years, increased doses were prescribed for almost half of the patients (49%), with a median time to dose increase of 17.4 months.
Patients with ulcerative colitis were 83% more likely to receive increased doses than were those with Crohn’s disease (P = .0001), and doses were typically escalated at an earlier date (7.1 vs. 11.1 months). In contrast, dose escalation was not associated with a variety of other patient factors, such as sex, age, or baseline laboratory values.
Comprehensive data from 11 prescribers showed that 70.8% of patients who received increased doses underwent some form of testing to assess inflammation, but only 24.7% of them had objective evidence of inflammatory activity that supported the dose increase.
Raymond K. Cross Jr., MD, MS, AGAF, FACG, director of the IBD program at the University of Maryland, Baltimore County, said that the findings were “not surprising.”
Dr. Cross said that patients with IBD should ideally undergo testing for drug levels and inflammatory activity prior to dose escalation, but this one-size-fits-all approach doesn’t always translate to real-world practice.
“There’s the science, and there’s the practical,” he said, noting that dose escalation may precede testing “in an effort to be efficient and decrease suffering.” This includes cases in which patients receive an earlier or increased dose to bridge the gap between worsening of symptoms and completion of diagnostics.
Dr. Cross also pointed out that patients with ulcerative colitis typically have a strong correlation between symptoms and level of inflammation.
“If [a patient with ulcerative colitis] has bloody diarrhea, it’s 90% or so that they’re going to have active inflammation in their colon, so I feel less obligated to do those checks in an ulcerative colitis patient who has classic symptoms,” he said.
Byron Vaughn, MD, codirector of the IBD program at the University of Minnesota, Minneapolis, suggested that the study by Elias and colleagues “serves as an important reminder to follow the tenets of a treat-to-target strategy, which include regular assessment of objective markers of inflammation specific to IBD before and after a dose adjustment.”
Still, like Dr. Cross, Dr. Vaughn noted that dose escalation without additional testing “may be reasonable” in certain scenarios, such as when patients with ulcerative colitis have bloody diarrhea and tenesmus or for patients with perianal Crohn’s disease.
“A key limitation of this study is the lack of drug concentration monitoring,” Dr. Vaughn said. “Due to the immunogenicity of anti-TNF medications there is a strong rationale for proactive dose adjustments to avoid low or undetectable concentrations and subsequent antibody formation. However, as the authors point out, this is unlikely to be a major factor in this particular study as drug concentration measurement was not routinely available over the study period.”
The investigators disclosed relationships with AbbVie, Ferring, Janssen, and others. Dr. Vaughn disclosed relationships with AbbVie, Celgene, Genentech, Takeda, and others. Dr. Cross disclosed relationships with AbbVie, Janssen, and Samsung Bioepis.
SOURCE: Elias ED et al. J Clin Gastroenterol. 2020 Jul 17. doi: 10.1097/MCG.0000000000001391.
Clinicians frequently increase doses of anti–tumor necrosis factor (anti-TNF) agents for patients with inflammatory bowel disease (IBD) without objective evidence of active inflammation, based on a retrospective study.
This strategy may be ineffective and expensive, according to lead author Evan D. Elias, MD, of the University of Manitoba, Winnipeg, Canada, and colleagues, who noted that worsening symptoms may have noninflammatory causes, such as coexisting functional disorders.
“Objective evidence of active inflammation is a key part of [the dose escalation] process,” the investigators wrote in the Journal of Clinical Gastroenterology, “however, there are no data assessing whether an adequate evaluation of the cause of persistent gastrointestinal symptoms is routinely undertaken in clinical practice.”
To address this knowledge gap, Dr. Elias and colleagues conducted a retrospective study involving 23 physicians in Manitoba who initiated anti-TNF therapy in 838 patients with IBD during 2005-2016. The primary outcome was rate of dose escalation within 1 year. Secondarily, the investigators audited 11 out of 23 prescribers to determine how frequently patients receiving increased doses underwent objective assessment of inflammation, both in general and in the 90 days preceding dose escalation.
Of note, therapeutic drug monitoring, fecal calprotectin testing, and transabdominal ultrasound were not commonly available to prescribers during the study period.
Within 1 year, almost one-third of patients (32.9%) received increased doses of either adalimumab or infliximab, with no significant difference in escalation practices between the two agents. After approximately 2 years, increased doses were prescribed for almost half of the patients (49%), with a median time to dose increase of 17.4 months.
Patients with ulcerative colitis were 83% more likely to receive increased doses than were those with Crohn’s disease (P = .0001), and doses were typically escalated at an earlier date (7.1 vs. 11.1 months). In contrast, dose escalation was not associated with a variety of other patient factors, such as sex, age, or baseline laboratory values.
Comprehensive data from 11 prescribers showed that 70.8% of patients who received increased doses underwent some form of testing to assess inflammation, but only 24.7% of them had objective evidence of inflammatory activity that supported the dose increase.
Raymond K. Cross Jr., MD, MS, AGAF, FACG, director of the IBD program at the University of Maryland, Baltimore County, said that the findings were “not surprising.”
Dr. Cross said that patients with IBD should ideally undergo testing for drug levels and inflammatory activity prior to dose escalation, but this one-size-fits-all approach doesn’t always translate to real-world practice.
“There’s the science, and there’s the practical,” he said, noting that dose escalation may precede testing “in an effort to be efficient and decrease suffering.” This includes cases in which patients receive an earlier or increased dose to bridge the gap between worsening of symptoms and completion of diagnostics.
Dr. Cross also pointed out that patients with ulcerative colitis typically have a strong correlation between symptoms and level of inflammation.
“If [a patient with ulcerative colitis] has bloody diarrhea, it’s 90% or so that they’re going to have active inflammation in their colon, so I feel less obligated to do those checks in an ulcerative colitis patient who has classic symptoms,” he said.
Byron Vaughn, MD, codirector of the IBD program at the University of Minnesota, Minneapolis, suggested that the study by Elias and colleagues “serves as an important reminder to follow the tenets of a treat-to-target strategy, which include regular assessment of objective markers of inflammation specific to IBD before and after a dose adjustment.”
Still, like Dr. Cross, Dr. Vaughn noted that dose escalation without additional testing “may be reasonable” in certain scenarios, such as when patients with ulcerative colitis have bloody diarrhea and tenesmus or for patients with perianal Crohn’s disease.
“A key limitation of this study is the lack of drug concentration monitoring,” Dr. Vaughn said. “Due to the immunogenicity of anti-TNF medications there is a strong rationale for proactive dose adjustments to avoid low or undetectable concentrations and subsequent antibody formation. However, as the authors point out, this is unlikely to be a major factor in this particular study as drug concentration measurement was not routinely available over the study period.”
The investigators disclosed relationships with AbbVie, Ferring, Janssen, and others. Dr. Vaughn disclosed relationships with AbbVie, Celgene, Genentech, Takeda, and others. Dr. Cross disclosed relationships with AbbVie, Janssen, and Samsung Bioepis.
SOURCE: Elias ED et al. J Clin Gastroenterol. 2020 Jul 17. doi: 10.1097/MCG.0000000000001391.
Clinicians frequently increase doses of anti–tumor necrosis factor (anti-TNF) agents for patients with inflammatory bowel disease (IBD) without objective evidence of active inflammation, based on a retrospective study.
This strategy may be ineffective and expensive, according to lead author Evan D. Elias, MD, of the University of Manitoba, Winnipeg, Canada, and colleagues, who noted that worsening symptoms may have noninflammatory causes, such as coexisting functional disorders.
“Objective evidence of active inflammation is a key part of [the dose escalation] process,” the investigators wrote in the Journal of Clinical Gastroenterology, “however, there are no data assessing whether an adequate evaluation of the cause of persistent gastrointestinal symptoms is routinely undertaken in clinical practice.”
To address this knowledge gap, Dr. Elias and colleagues conducted a retrospective study involving 23 physicians in Manitoba who initiated anti-TNF therapy in 838 patients with IBD during 2005-2016. The primary outcome was rate of dose escalation within 1 year. Secondarily, the investigators audited 11 out of 23 prescribers to determine how frequently patients receiving increased doses underwent objective assessment of inflammation, both in general and in the 90 days preceding dose escalation.
Of note, therapeutic drug monitoring, fecal calprotectin testing, and transabdominal ultrasound were not commonly available to prescribers during the study period.
Within 1 year, almost one-third of patients (32.9%) received increased doses of either adalimumab or infliximab, with no significant difference in escalation practices between the two agents. After approximately 2 years, increased doses were prescribed for almost half of the patients (49%), with a median time to dose increase of 17.4 months.
Patients with ulcerative colitis were 83% more likely to receive increased doses than were those with Crohn’s disease (P = .0001), and doses were typically escalated at an earlier date (7.1 vs. 11.1 months). In contrast, dose escalation was not associated with a variety of other patient factors, such as sex, age, or baseline laboratory values.
Comprehensive data from 11 prescribers showed that 70.8% of patients who received increased doses underwent some form of testing to assess inflammation, but only 24.7% of them had objective evidence of inflammatory activity that supported the dose increase.
Raymond K. Cross Jr., MD, MS, AGAF, FACG, director of the IBD program at the University of Maryland, Baltimore County, said that the findings were “not surprising.”
Dr. Cross said that patients with IBD should ideally undergo testing for drug levels and inflammatory activity prior to dose escalation, but this one-size-fits-all approach doesn’t always translate to real-world practice.
“There’s the science, and there’s the practical,” he said, noting that dose escalation may precede testing “in an effort to be efficient and decrease suffering.” This includes cases in which patients receive an earlier or increased dose to bridge the gap between worsening of symptoms and completion of diagnostics.
Dr. Cross also pointed out that patients with ulcerative colitis typically have a strong correlation between symptoms and level of inflammation.
“If [a patient with ulcerative colitis] has bloody diarrhea, it’s 90% or so that they’re going to have active inflammation in their colon, so I feel less obligated to do those checks in an ulcerative colitis patient who has classic symptoms,” he said.
Byron Vaughn, MD, codirector of the IBD program at the University of Minnesota, Minneapolis, suggested that the study by Elias and colleagues “serves as an important reminder to follow the tenets of a treat-to-target strategy, which include regular assessment of objective markers of inflammation specific to IBD before and after a dose adjustment.”
Still, like Dr. Cross, Dr. Vaughn noted that dose escalation without additional testing “may be reasonable” in certain scenarios, such as when patients with ulcerative colitis have bloody diarrhea and tenesmus or for patients with perianal Crohn’s disease.
“A key limitation of this study is the lack of drug concentration monitoring,” Dr. Vaughn said. “Due to the immunogenicity of anti-TNF medications there is a strong rationale for proactive dose adjustments to avoid low or undetectable concentrations and subsequent antibody formation. However, as the authors point out, this is unlikely to be a major factor in this particular study as drug concentration measurement was not routinely available over the study period.”
The investigators disclosed relationships with AbbVie, Ferring, Janssen, and others. Dr. Vaughn disclosed relationships with AbbVie, Celgene, Genentech, Takeda, and others. Dr. Cross disclosed relationships with AbbVie, Janssen, and Samsung Bioepis.
SOURCE: Elias ED et al. J Clin Gastroenterol. 2020 Jul 17. doi: 10.1097/MCG.0000000000001391.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Complete endoscopic healing tied to better Crohn’s disease outcomes
Patients with Crohn’s disease who experienced complete endoscopic healing with biologic therapy had significantly lower subsequent rates of treatment failure, intestinal resection, and hospitalization, compared with patients who experienced only partial mucosal healing, according to the findings of a two-center retrospective study.
Over a median of 4.8 years of follow-up (interquartile range, 2.1-7.2 years) rates of treatment failure were 25% in patients with complete mucosal healing at baseline (that is, a Crohn’s Disease Endoscopic Index of Severity [CDEIS] score of 0) and 48% in patients with partial healing (CDEIS score greater than 0 but less than 4). The difference was statistically significant (P = .045). No patient with a baseline CDEIS score of 0 required intestinal resection, compared with 11% of patients with scores greater than 0 but less than 4 (P = .031). Rates of hospitalization because of Crohn’s disease were 3.5% versus 18.5%, respectively (P = .013). Clara Yzet, MD, of Amiens (France) University Hospital, Université de Picardie Jules Verne, reported the findings together with her associates in Clinical Gastroenterology and Hepatology.
Mucosal healing is a key therapeutic target in Crohn’s disease that has been linked to desirable outcomes, such as steroid-free remission and a lower rate of intestinal resection. However, prior observational studies have inconsistently defined mucosal healing, and clinical trials of biologics have used any of at least seven different definitions, the researchers wrote. Recently, in patients with ulcerative colitis, a Scandinavian Journal of Gastroenterology and another in the Journal of Crohn’s and Colitis linked a stricter definition of mucosal healing (an endoscopic Mayo score of 0, or histologic healing) with superior long-term clinical outcomes. In patients with Crohn’s disease, however, there has been no established threshold for mucosal healing based on either the CDEIS or the Simplified Endoscopic Score for Crohn’s disease (SES-CD).
Therefore, Dr. Yzet and her associates identified and reviewed the medical records of 84 consecutive adults with clinically remitted Crohn’s disease who received anti–tumor necrosis factor therapies (infliximab and adalimumab) or vedolizumab at two university hospitals in France between 2008 and 2015. All patients received baseline and follow-up colonoscopies, with results scored on the CDEIS. In all cases, the second CDEIS score was less than 4 (the CDEIS ranges from 0 to 44). The primary outcome was treatment failure, defined as the need for biologic optimization (increasing the dose or shortening the dosing interval of the biologic), corticosteroids, or immunosuppressants; a Harvey-Bradshaw score greater than 4 associated with a change in treatment; or the need for intestinal resection or hospitalization because of Crohn’s disease.
At baseline, 57 patients had CDEIS scores of 0 (complete mucosal healing) and 27 patients had scores greater than 0 but less than 4 (partial mucosal healing). The two groups were otherwise similar except that patients with complete mucosal healing had a shorter median duration of Crohn’s disease (10.3 vs. 15.1 years in the partial healing group; P = .029) and a lower prevalence of Crohn’s disease phenotype B2 (stricturing) according to the Vienna classification (1.8% vs. 14.8%; P = .035). In the multivariate analysis, CDEIS score was the only factor associated with treatment failure (hazard ratio, 2.61; 95% confidence interval, 1.16-5.88; P = .02).
“Our findings suggest that we should be more ambitious in clinical practice to change patients’ life and disease course by achieving complete endoscopic healing. However, this strategy could be limited by the ability of current drugs to achieve complete mucosal healing,” the researchers wrote. “Obtaining a complete mucosal healing would require today a significant need for optimization or change of biologics.”
No external funding sources were reported. Two coinvestigators disclosed ties to AbbVie, Amgen, Biogaran, Biogen, Ferring, Janssen, MSD, Pfizer, Takeda, and several other pharmaceutical companies. The remaining investigators reported having no relevant conflicts of interest.
SOURCE: Yzet C et al. Clin Gastroenterol Hepatol. 2019 Nov 16. doi: 10.1016/j.cgh.2019.11.025.
Patients with Crohn’s disease who experienced complete endoscopic healing with biologic therapy had significantly lower subsequent rates of treatment failure, intestinal resection, and hospitalization, compared with patients who experienced only partial mucosal healing, according to the findings of a two-center retrospective study.
Over a median of 4.8 years of follow-up (interquartile range, 2.1-7.2 years) rates of treatment failure were 25% in patients with complete mucosal healing at baseline (that is, a Crohn’s Disease Endoscopic Index of Severity [CDEIS] score of 0) and 48% in patients with partial healing (CDEIS score greater than 0 but less than 4). The difference was statistically significant (P = .045). No patient with a baseline CDEIS score of 0 required intestinal resection, compared with 11% of patients with scores greater than 0 but less than 4 (P = .031). Rates of hospitalization because of Crohn’s disease were 3.5% versus 18.5%, respectively (P = .013). Clara Yzet, MD, of Amiens (France) University Hospital, Université de Picardie Jules Verne, reported the findings together with her associates in Clinical Gastroenterology and Hepatology.
Mucosal healing is a key therapeutic target in Crohn’s disease that has been linked to desirable outcomes, such as steroid-free remission and a lower rate of intestinal resection. However, prior observational studies have inconsistently defined mucosal healing, and clinical trials of biologics have used any of at least seven different definitions, the researchers wrote. Recently, in patients with ulcerative colitis, a Scandinavian Journal of Gastroenterology and another in the Journal of Crohn’s and Colitis linked a stricter definition of mucosal healing (an endoscopic Mayo score of 0, or histologic healing) with superior long-term clinical outcomes. In patients with Crohn’s disease, however, there has been no established threshold for mucosal healing based on either the CDEIS or the Simplified Endoscopic Score for Crohn’s disease (SES-CD).
Therefore, Dr. Yzet and her associates identified and reviewed the medical records of 84 consecutive adults with clinically remitted Crohn’s disease who received anti–tumor necrosis factor therapies (infliximab and adalimumab) or vedolizumab at two university hospitals in France between 2008 and 2015. All patients received baseline and follow-up colonoscopies, with results scored on the CDEIS. In all cases, the second CDEIS score was less than 4 (the CDEIS ranges from 0 to 44). The primary outcome was treatment failure, defined as the need for biologic optimization (increasing the dose or shortening the dosing interval of the biologic), corticosteroids, or immunosuppressants; a Harvey-Bradshaw score greater than 4 associated with a change in treatment; or the need for intestinal resection or hospitalization because of Crohn’s disease.
At baseline, 57 patients had CDEIS scores of 0 (complete mucosal healing) and 27 patients had scores greater than 0 but less than 4 (partial mucosal healing). The two groups were otherwise similar except that patients with complete mucosal healing had a shorter median duration of Crohn’s disease (10.3 vs. 15.1 years in the partial healing group; P = .029) and a lower prevalence of Crohn’s disease phenotype B2 (stricturing) according to the Vienna classification (1.8% vs. 14.8%; P = .035). In the multivariate analysis, CDEIS score was the only factor associated with treatment failure (hazard ratio, 2.61; 95% confidence interval, 1.16-5.88; P = .02).
“Our findings suggest that we should be more ambitious in clinical practice to change patients’ life and disease course by achieving complete endoscopic healing. However, this strategy could be limited by the ability of current drugs to achieve complete mucosal healing,” the researchers wrote. “Obtaining a complete mucosal healing would require today a significant need for optimization or change of biologics.”
No external funding sources were reported. Two coinvestigators disclosed ties to AbbVie, Amgen, Biogaran, Biogen, Ferring, Janssen, MSD, Pfizer, Takeda, and several other pharmaceutical companies. The remaining investigators reported having no relevant conflicts of interest.
SOURCE: Yzet C et al. Clin Gastroenterol Hepatol. 2019 Nov 16. doi: 10.1016/j.cgh.2019.11.025.
Patients with Crohn’s disease who experienced complete endoscopic healing with biologic therapy had significantly lower subsequent rates of treatment failure, intestinal resection, and hospitalization, compared with patients who experienced only partial mucosal healing, according to the findings of a two-center retrospective study.
Over a median of 4.8 years of follow-up (interquartile range, 2.1-7.2 years) rates of treatment failure were 25% in patients with complete mucosal healing at baseline (that is, a Crohn’s Disease Endoscopic Index of Severity [CDEIS] score of 0) and 48% in patients with partial healing (CDEIS score greater than 0 but less than 4). The difference was statistically significant (P = .045). No patient with a baseline CDEIS score of 0 required intestinal resection, compared with 11% of patients with scores greater than 0 but less than 4 (P = .031). Rates of hospitalization because of Crohn’s disease were 3.5% versus 18.5%, respectively (P = .013). Clara Yzet, MD, of Amiens (France) University Hospital, Université de Picardie Jules Verne, reported the findings together with her associates in Clinical Gastroenterology and Hepatology.
Mucosal healing is a key therapeutic target in Crohn’s disease that has been linked to desirable outcomes, such as steroid-free remission and a lower rate of intestinal resection. However, prior observational studies have inconsistently defined mucosal healing, and clinical trials of biologics have used any of at least seven different definitions, the researchers wrote. Recently, in patients with ulcerative colitis, a Scandinavian Journal of Gastroenterology and another in the Journal of Crohn’s and Colitis linked a stricter definition of mucosal healing (an endoscopic Mayo score of 0, or histologic healing) with superior long-term clinical outcomes. In patients with Crohn’s disease, however, there has been no established threshold for mucosal healing based on either the CDEIS or the Simplified Endoscopic Score for Crohn’s disease (SES-CD).
Therefore, Dr. Yzet and her associates identified and reviewed the medical records of 84 consecutive adults with clinically remitted Crohn’s disease who received anti–tumor necrosis factor therapies (infliximab and adalimumab) or vedolizumab at two university hospitals in France between 2008 and 2015. All patients received baseline and follow-up colonoscopies, with results scored on the CDEIS. In all cases, the second CDEIS score was less than 4 (the CDEIS ranges from 0 to 44). The primary outcome was treatment failure, defined as the need for biologic optimization (increasing the dose or shortening the dosing interval of the biologic), corticosteroids, or immunosuppressants; a Harvey-Bradshaw score greater than 4 associated with a change in treatment; or the need for intestinal resection or hospitalization because of Crohn’s disease.
At baseline, 57 patients had CDEIS scores of 0 (complete mucosal healing) and 27 patients had scores greater than 0 but less than 4 (partial mucosal healing). The two groups were otherwise similar except that patients with complete mucosal healing had a shorter median duration of Crohn’s disease (10.3 vs. 15.1 years in the partial healing group; P = .029) and a lower prevalence of Crohn’s disease phenotype B2 (stricturing) according to the Vienna classification (1.8% vs. 14.8%; P = .035). In the multivariate analysis, CDEIS score was the only factor associated with treatment failure (hazard ratio, 2.61; 95% confidence interval, 1.16-5.88; P = .02).
“Our findings suggest that we should be more ambitious in clinical practice to change patients’ life and disease course by achieving complete endoscopic healing. However, this strategy could be limited by the ability of current drugs to achieve complete mucosal healing,” the researchers wrote. “Obtaining a complete mucosal healing would require today a significant need for optimization or change of biologics.”
No external funding sources were reported. Two coinvestigators disclosed ties to AbbVie, Amgen, Biogaran, Biogen, Ferring, Janssen, MSD, Pfizer, Takeda, and several other pharmaceutical companies. The remaining investigators reported having no relevant conflicts of interest.
SOURCE: Yzet C et al. Clin Gastroenterol Hepatol. 2019 Nov 16. doi: 10.1016/j.cgh.2019.11.025.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Oculostenotic reflex still holds sway, survey shows
Most interventional cardiologists still rely solely upon angiography in making revascularization decisions about intermediate stenoses in the setting of stable coronary artery disease – and in doing so they end up making the wrong call nearly 40% of the time, according to the results of an international survey presented at the virtual annual meeting of the European Association of Percutaneous Cardiovascular Interventions.
“We saw a strong tendency to visually overestimate the percent diameter stenosis,” reported Gabor G. Toth, MD, an interventional cardiologist at the Medical University of Graz (Austria).
The same tendency has been highlighted in numerous randomized trials and observational studies. That’s why both European and U.S. guidelines now strongly recommend invasive functional assessment, such as fractional-flow reserve (FFR) testing, in evaluating the significance of intermediate stenoses in the absence of noninvasive evidence of ischemia. The new survey findings point to an important disconnect between these guideline recommendations and current clinical practice, he noted.
Dr. Toth presented the results of the second web-based, international survey on interventional decision-making strategy sponsored by the European Association of Percutaneous Cardiovascular Interventions. He contrasted the findings with those of the previously reported first international online survey, conducted 6 years earlier, for which he was first author (Circ Cardiovasc Interv. 2014 Dec;7[6]:751-9).
The two surveys were identically designed. In both, participants answered questions that enabled investigators to place them into one of four categories based upon the extent of their experience in interventional cardiology. The participants were also presented with 5 angiograms of focal intermediate stenoses and asked to determine the stenosis significance of each lesion. No information on the functional significance of the stenoses was included; however, the respondents could request additional diagnostic information by “ordering” adjunctive invasive functional assessment tests, including FFR, quantitative coronary angiography, intravascular ultrasound, or optical coherence tomography. Importantly, participating cardiologists were asked to make their decisions based upon best possible clinical practice in a hypothetical scenario where financial constraints had no role.
The second international survey was conducted during the latter half of 2019. The 334 interventional cardiologists who responded performed a total of 978 case evaluations including 2,054 coronary lesion assessments.
About 59% of all decisions were made solely on the basis of angiographic appearance without any information as to the functional significance of a given stenosis: Indeed, 13% of all stenoses were thereby declared to be “certainly” nonsignificant, and 46% were deemed “certainly” significant. In total, that figure was down significantly from the 71% rate in the first survey. In the first survey, 47% of decisions based upon angiographic appearance alone were discordant with FFR results known to the investigators, compared with a 39% discordance rate in the second survey.
Of the physician decisions made in the second survey, 10% involved a request for intravascular imaging, essentially unchanged from the 9% rate in the first survey. However, there was a significant increase over time in requests for invasive functional assessment tests: 25% in the first survey, rising to 31% in the second. This increase was entirely driven by additional requests for data on nonhyperemic pressure ratios; there was no difference in requests for FFR testing between the 2013 and 2019 surveys.
Clinician experience played an interesting role in decision-making: “Experience does not have an impact on the accuracy of angiographically based decisions, but experience does have an impact on understanding the need for adjunctive functional diagnostic testing,” Dr. Toth explained.
Indeed, 21% of decisions made by the least-experienced interventional cardiologists involved a request for adjunctive invasive functional assessment, compared with 24% of decisions by physicians in the third quartile of experience, 32% in the second, and 37% of decisions made by the most experienced clinicians.
Discussant Michael Haude, MD, PhD, said that “these results clearly show that eyeball angioguidance is still the dominant tool used in decision-making, and that this eyeball angioguidance continuously overestimates the stenosis when you compare the results to quantitative coronary angiography.
“These results, surprisingly for me, show a quite low uptake of the invasive functional assessments despite overwhelming scientific data leading to clear guideline-based recommendations. Why is this the case, even after financial constraints are ruled out? Probably because FFR is still a complex invasive procedure. Maybe, in the future, quantitative flow-ratio angiography [which requires no pressure wire] or CT-based FFR will be more popular,” said Dr. Haude, an interventional cardiologist at the Rheinland Clinic in Neuss, Germany.
He reported receiving research grants from Biotronik and serving as a paid consultant to that company as well as Cardiac Dimensions, Orbus Neich, and Philips. Dr. Toth reported having no financial conflicts regarding the international survey.
Most interventional cardiologists still rely solely upon angiography in making revascularization decisions about intermediate stenoses in the setting of stable coronary artery disease – and in doing so they end up making the wrong call nearly 40% of the time, according to the results of an international survey presented at the virtual annual meeting of the European Association of Percutaneous Cardiovascular Interventions.
“We saw a strong tendency to visually overestimate the percent diameter stenosis,” reported Gabor G. Toth, MD, an interventional cardiologist at the Medical University of Graz (Austria).
The same tendency has been highlighted in numerous randomized trials and observational studies. That’s why both European and U.S. guidelines now strongly recommend invasive functional assessment, such as fractional-flow reserve (FFR) testing, in evaluating the significance of intermediate stenoses in the absence of noninvasive evidence of ischemia. The new survey findings point to an important disconnect between these guideline recommendations and current clinical practice, he noted.
Dr. Toth presented the results of the second web-based, international survey on interventional decision-making strategy sponsored by the European Association of Percutaneous Cardiovascular Interventions. He contrasted the findings with those of the previously reported first international online survey, conducted 6 years earlier, for which he was first author (Circ Cardiovasc Interv. 2014 Dec;7[6]:751-9).
The two surveys were identically designed. In both, participants answered questions that enabled investigators to place them into one of four categories based upon the extent of their experience in interventional cardiology. The participants were also presented with 5 angiograms of focal intermediate stenoses and asked to determine the stenosis significance of each lesion. No information on the functional significance of the stenoses was included; however, the respondents could request additional diagnostic information by “ordering” adjunctive invasive functional assessment tests, including FFR, quantitative coronary angiography, intravascular ultrasound, or optical coherence tomography. Importantly, participating cardiologists were asked to make their decisions based upon best possible clinical practice in a hypothetical scenario where financial constraints had no role.
The second international survey was conducted during the latter half of 2019. The 334 interventional cardiologists who responded performed a total of 978 case evaluations including 2,054 coronary lesion assessments.
About 59% of all decisions were made solely on the basis of angiographic appearance without any information as to the functional significance of a given stenosis: Indeed, 13% of all stenoses were thereby declared to be “certainly” nonsignificant, and 46% were deemed “certainly” significant. In total, that figure was down significantly from the 71% rate in the first survey. In the first survey, 47% of decisions based upon angiographic appearance alone were discordant with FFR results known to the investigators, compared with a 39% discordance rate in the second survey.
Of the physician decisions made in the second survey, 10% involved a request for intravascular imaging, essentially unchanged from the 9% rate in the first survey. However, there was a significant increase over time in requests for invasive functional assessment tests: 25% in the first survey, rising to 31% in the second. This increase was entirely driven by additional requests for data on nonhyperemic pressure ratios; there was no difference in requests for FFR testing between the 2013 and 2019 surveys.
Clinician experience played an interesting role in decision-making: “Experience does not have an impact on the accuracy of angiographically based decisions, but experience does have an impact on understanding the need for adjunctive functional diagnostic testing,” Dr. Toth explained.
Indeed, 21% of decisions made by the least-experienced interventional cardiologists involved a request for adjunctive invasive functional assessment, compared with 24% of decisions by physicians in the third quartile of experience, 32% in the second, and 37% of decisions made by the most experienced clinicians.
Discussant Michael Haude, MD, PhD, said that “these results clearly show that eyeball angioguidance is still the dominant tool used in decision-making, and that this eyeball angioguidance continuously overestimates the stenosis when you compare the results to quantitative coronary angiography.
“These results, surprisingly for me, show a quite low uptake of the invasive functional assessments despite overwhelming scientific data leading to clear guideline-based recommendations. Why is this the case, even after financial constraints are ruled out? Probably because FFR is still a complex invasive procedure. Maybe, in the future, quantitative flow-ratio angiography [which requires no pressure wire] or CT-based FFR will be more popular,” said Dr. Haude, an interventional cardiologist at the Rheinland Clinic in Neuss, Germany.
He reported receiving research grants from Biotronik and serving as a paid consultant to that company as well as Cardiac Dimensions, Orbus Neich, and Philips. Dr. Toth reported having no financial conflicts regarding the international survey.
Most interventional cardiologists still rely solely upon angiography in making revascularization decisions about intermediate stenoses in the setting of stable coronary artery disease – and in doing so they end up making the wrong call nearly 40% of the time, according to the results of an international survey presented at the virtual annual meeting of the European Association of Percutaneous Cardiovascular Interventions.
“We saw a strong tendency to visually overestimate the percent diameter stenosis,” reported Gabor G. Toth, MD, an interventional cardiologist at the Medical University of Graz (Austria).
The same tendency has been highlighted in numerous randomized trials and observational studies. That’s why both European and U.S. guidelines now strongly recommend invasive functional assessment, such as fractional-flow reserve (FFR) testing, in evaluating the significance of intermediate stenoses in the absence of noninvasive evidence of ischemia. The new survey findings point to an important disconnect between these guideline recommendations and current clinical practice, he noted.
Dr. Toth presented the results of the second web-based, international survey on interventional decision-making strategy sponsored by the European Association of Percutaneous Cardiovascular Interventions. He contrasted the findings with those of the previously reported first international online survey, conducted 6 years earlier, for which he was first author (Circ Cardiovasc Interv. 2014 Dec;7[6]:751-9).
The two surveys were identically designed. In both, participants answered questions that enabled investigators to place them into one of four categories based upon the extent of their experience in interventional cardiology. The participants were also presented with 5 angiograms of focal intermediate stenoses and asked to determine the stenosis significance of each lesion. No information on the functional significance of the stenoses was included; however, the respondents could request additional diagnostic information by “ordering” adjunctive invasive functional assessment tests, including FFR, quantitative coronary angiography, intravascular ultrasound, or optical coherence tomography. Importantly, participating cardiologists were asked to make their decisions based upon best possible clinical practice in a hypothetical scenario where financial constraints had no role.
The second international survey was conducted during the latter half of 2019. The 334 interventional cardiologists who responded performed a total of 978 case evaluations including 2,054 coronary lesion assessments.
About 59% of all decisions were made solely on the basis of angiographic appearance without any information as to the functional significance of a given stenosis: Indeed, 13% of all stenoses were thereby declared to be “certainly” nonsignificant, and 46% were deemed “certainly” significant. In total, that figure was down significantly from the 71% rate in the first survey. In the first survey, 47% of decisions based upon angiographic appearance alone were discordant with FFR results known to the investigators, compared with a 39% discordance rate in the second survey.
Of the physician decisions made in the second survey, 10% involved a request for intravascular imaging, essentially unchanged from the 9% rate in the first survey. However, there was a significant increase over time in requests for invasive functional assessment tests: 25% in the first survey, rising to 31% in the second. This increase was entirely driven by additional requests for data on nonhyperemic pressure ratios; there was no difference in requests for FFR testing between the 2013 and 2019 surveys.
Clinician experience played an interesting role in decision-making: “Experience does not have an impact on the accuracy of angiographically based decisions, but experience does have an impact on understanding the need for adjunctive functional diagnostic testing,” Dr. Toth explained.
Indeed, 21% of decisions made by the least-experienced interventional cardiologists involved a request for adjunctive invasive functional assessment, compared with 24% of decisions by physicians in the third quartile of experience, 32% in the second, and 37% of decisions made by the most experienced clinicians.
Discussant Michael Haude, MD, PhD, said that “these results clearly show that eyeball angioguidance is still the dominant tool used in decision-making, and that this eyeball angioguidance continuously overestimates the stenosis when you compare the results to quantitative coronary angiography.
“These results, surprisingly for me, show a quite low uptake of the invasive functional assessments despite overwhelming scientific data leading to clear guideline-based recommendations. Why is this the case, even after financial constraints are ruled out? Probably because FFR is still a complex invasive procedure. Maybe, in the future, quantitative flow-ratio angiography [which requires no pressure wire] or CT-based FFR will be more popular,” said Dr. Haude, an interventional cardiologist at the Rheinland Clinic in Neuss, Germany.
He reported receiving research grants from Biotronik and serving as a paid consultant to that company as well as Cardiac Dimensions, Orbus Neich, and Philips. Dr. Toth reported having no financial conflicts regarding the international survey.
REPORTING FROM EUROPCR 2020
Value of palliative care shines clearly in a crisis
Hospitalists have played a key role
For some palliative care professionals, the COVID-19 pandemic, particularly in viral hot spots like New York City, represents a “moment” that could lead to greater awareness of what this service offers to seriously ill patients in a crisis.
They say it has provided an opportunity to show what palliative care teams can contribute to the difficult circumstances of patients with severe symptoms, isolated and alone in quarantined hospitals, with poor survival rates, perhaps sedated for extended stays on scarce ventilators – and for their family members, who are able to visit them only virtually via telephone or tablet.
But it has also highlighted gaps – including insufficient staffing for some palliative care teams. Hospitalists and other clinicians in the hospital need to learn the basics of primary palliative care, such as how to communicate bad news, initiate goals of care conversations, and address common symptoms of serious illness, such as pain. That way, they could shoulder more of the demand for this kind of care when palliative care specialists are in short supply.
Hospitalists, some of whom also have pursued a specialization in palliative care, have played key roles in clarifying and redefining the new role for palliative care, whom it is meant for, and who should provide it. Central to this new role is the greater use of telemedicine – for talking to hospitalized patients without increasing viral exposure, for linking up with family members who can’t visit their loved ones in the hospital, and for helping frontline hospital staff who need a palliative care consultation – or just a chance to debrief on what they are seeing.
A pandemic wake-up call
Elizabeth Gundersen, MD, FHM, FAAHPM, director of the hospice and palliative medicine fellowship program at the Charles E. Schmidt College of Medicine at Florida Atlantic University (FAU) in Boca Raton, practiced hospital medicine for 10 years before pursuing a fellowship in hospice and palliative medicine and working as an academic palliative medicine physician. She calls the pandemic a wake-up call for gaps in care and all the things that weren’t working well in the health care system.
“Now we are seeing more clearly what’s lacking – or broken – and what we will carry forward from this experience into the post-COVID world,” she said. Some hospitalists do palliative care very well, and others don’t feel as comfortable in having these difficult conversations with patients. But in the uncertain course of the virus they get thrust into it.
Although FAU’s associated hospitals were not as inundated with COVID-19 patients in the early weeks of the pandemic as were other regions, the volume of other patients plummeted, Dr. Gundersen said, adding that “there’s still been incredible intensity and worry about the virus. For me, the basic role of palliative care hasn’t changed, and the phrase I have always used when introducing myself – ‘we’re an extra layer of support for the patient and family’ – still holds true,” she said.
“I try to make it clear to people that palliative care is not synonymous with end-of-life care. We don’t want people to think that a palliative care referral implies imminent death. The goal is not to get more people to have a do not attempt resuscitation (DNAR) order, but to determine the patient and family’s treatment goals and whether a DNAR order fits those goals.”
The tough conversations
Dr. Gundersen is cochair of SHM’s Palliative Care Special Interest Group, along with Rab Razzak, MD, clinical director of palliative medicine at University Hospitals Cleveland Medical Center, one of the hospitals affiliated with Case Western University in Cleveland. (Connect with them on Twitter: @Top_Gundersen and @rabrazzak.)
Dr. Razzak also transitioned from hospital medicine to palliative medicine 10 years ago. “As a hospitalist, I enjoyed the tough conversations and bringing the human element into my health care interactions,” he explained. “To me, palliative care is a philosophy of care that puts the person we call the patient at the center of the interaction, while we try to figure out how to best care for them as a person.”
When the pandemic hit, University Hospitals made 20 ICU beds available for COVID-19 patients, Dr. Razzak said. This unit has since been full but not overflowing, while overall hospital census went down. The palliative care team at the hospital includes four inpatient doctors, nurse practitioners, and a chaplain, as well as an outpatient team primarily focused on oncology.
“In some settings, palliative care has been at the forefront of difficult conversations, when things aren’t going well for the patient and there’s much uncertainty,” Dr. Razzak said. The interface between hospital medicine and palliative care can be complementary, he added. “We talk about primary palliative care, which we want every discipline to be able to do – lead meaningful conversations, help manage symptoms.”
The take-home message for hospitalists, he said, is to get training in how to have these discussions, using such resources as VitalTalk (https://www.vitaltalk.org/), a nonprofit organization that disseminates education in communication skills for difficult conversations, and the Center to Advance Palliative Care (www.capc.org) at Icahn School of Medicine at Mount Sinai in New York City. “Once you’ve mastered the conversation, it will get easier. But ask for help when you need it, and learn how to know when you need it.”
Dr. Gundersen added that hospital medicine groups and palliative care teams could reach out to each other and talk about what they did in the crisis and how they can work together in the future. She recommends frequent ongoing support and collaboration that could range from formal conferences or training sessions to informal team interactions, perhaps with sandwiches in the doctor’s lounge – provided that there’s room for social distancing. She has recently started giving talks in the community and grand rounds presentations in hospitals about palliative care.
Other approaches and applications
In New York City, the initial epicenter for the pandemic in the United States, the adult palliative care service of Columbia University Medical Center (CUMC) experienced a sevenfold increase in consultation requests at the apex of the crisis, said its director, Craig Blinderman, MD. That demand was impossible to meet with existing staff. So Dr. Blinderman and colleagues established a virtual consultation model, recruiting and deploying volunteer out-of-state palliative care specialists to staff it.
An eight-bed palliative care unit was opened at CUMC for COVID-19 patients whose surrogates had opted not to initiate or continue intubation or life-sustaining treatments. This helped to relieve some of the pressures on the ICUs while making it possible for in-person visits to the hospice unit by families – in full PPE. Palliative care staff were embedded in various units in the hospital.
A palliative care response team composed of a hospice and palliative medicine fellow and four psychiatry residents or fellows, based in the emergency department and with supervision from the palliative care team, provided time-critical goals of care conversations with families using telemedicine – and a forum for listening to their suffering. Dr. Blinderman and colleagues also have found time to write up their experience for medical journals.1,2
There’s no reason to think that hospitalists, with a little basic training, couldn’t be having these same goals of care conversations, Dr. Blinderman said. “But the fact that hospitalists, at the pandemic’s peak, along with ICU doctors, were seeing an unprecedented magnitude of dying on a daily basis generated a lot of moral distress for them.”
Palliative care professionals, because they engage with these issues in a different way, may be somewhat better equipped to deal with the sheer emotional demands when so many are dying, as at the peak of the surge in New York. “We don’t see dying as a failure on our part but an opportunity to relieve suffering,” Dr. Blinderman said. And the palliative care field also emphasizes the importance of self-care for its practitioners.
“How do we meet the incredible palliative care needs in the epicenter of a pandemic? That question also applies to other kinds of crises we could imagine, for example, climate-related disasters,” Dr. Blinderman said. “What lessons have we learned about the value of palliative care and how to start incorporating it more integrally into the delivery of hospital care? Here we showed that we could work collaboratively with our colleagues at other major medical centers, bringing together their expertise to help us when we didn’t have the bandwidth to meet the demand,” he said.
Scripts can help
“Also, it won’t make sense to just go back to normal (after the crisis fades),” Dr. Blinderman said. “We need to take a close look at how our society is functioning in the wake of the pandemic and the ways the health care system has failed us. We have learned that we’re all interconnected and we need to work together to serve our communities – locally and nationally – applying basic distributive justice.”
Could there be, for example, a national infrastructure for mobilizing and deploying palliative care resources to areas of greatest need, similar to what was done in New York?
At Northwestern Medicine in Chicago, a number of palliative care clinicians at the system’s hospitals worked together to develop scripts designed to help other clinicians start goals of care conversations with patients and families, for use in the hospital as well as in outpatient primary care and other settings, with results integrated into the system’s electronic health record.
Front-line clinicians may not have the time to ask for formal consults from palliative care because of high volume and rapidly changing patient status, explained Eytan Szmuilowicz, MD, director of the section of palliative medicine at Northwestern Memorial Hospital. Or they may not have access to specialty-level palliative care in their settings.
The scripts are aimed at primary care, emergency physicians, and hospitalists needing to consider critical care placement or attempted resuscitation and to ICU clinicians helping families make decisions about life-sustaining treatments. They also can help facilitate advance care planning discussions. An example is “CALMER,” a six-step mnemonic guide to promote goals of care discussions with hospitalized patients. For more information on these scripts, contact Dr. Szmuilowicz: Eytan.Szmuilowicz@nm.org.
Eerily quiet
The COVID-19 crisis has been quite a whirlwind for hospital medicine, said Jeanie Youngwerth, MD, a hospitalist and program director of the palliative care service at the University of Colorado in Denver, which was a significant viral hotspot early on.
“When it first started, things seemed to change almost overnight – starting on Friday, March 13. People had to take action right away to develop work flows and the technology to allow us to see as many patients as possible,” she said. By the time Monday came, it was a whole new ballgame.
Dr. Youngwerth and two colleagues worked quickly to develop inpatient telemedicine capacity where none existed. “We knew we would not be going into patients’ rooms, but most of our team showed up in the hospital to work with the primary care teams. Our job was to see what we could do that actually made a difference,” she said.
“The hospital became a very strange place. You’d walk down the hallway and it was eerily quiet. Everybody you came across was being so nice to each other.” Televisits became a powerful way to bring the human connection back to medical care.
“What we learned from families was that they were thirsting to have some kind of connection with their loved one, and to be able to talk about their loved one and who they were as a person,” she said. “We’d contact the family through video visits and then, when the family meeting ended, the nurse would bring an iPad into the patient’s room so the family could see their loved one on a ventilator. They would immediately start communicating with their loved one, praying aloud, singing, playing music. It would make a huge difference for the family – and for the staff.”
References
1. Nakagawa S et al. Pandemic palliative care consultations spanning state and institutional borders. J Am Geriatr Soc. 2020 May 22. doi: 10.1111/jgs.16643.
2. Lee J Abrukin L, Flores S. Early intervention of palliative care in the emergency department during the COVID-19 pandemic. JAMA Intern Med. 2020 Jun 5. doi: 10.1001/jamainternmed.2020.2713.
Hospitalists have played a key role
Hospitalists have played a key role
For some palliative care professionals, the COVID-19 pandemic, particularly in viral hot spots like New York City, represents a “moment” that could lead to greater awareness of what this service offers to seriously ill patients in a crisis.
They say it has provided an opportunity to show what palliative care teams can contribute to the difficult circumstances of patients with severe symptoms, isolated and alone in quarantined hospitals, with poor survival rates, perhaps sedated for extended stays on scarce ventilators – and for their family members, who are able to visit them only virtually via telephone or tablet.
But it has also highlighted gaps – including insufficient staffing for some palliative care teams. Hospitalists and other clinicians in the hospital need to learn the basics of primary palliative care, such as how to communicate bad news, initiate goals of care conversations, and address common symptoms of serious illness, such as pain. That way, they could shoulder more of the demand for this kind of care when palliative care specialists are in short supply.
Hospitalists, some of whom also have pursued a specialization in palliative care, have played key roles in clarifying and redefining the new role for palliative care, whom it is meant for, and who should provide it. Central to this new role is the greater use of telemedicine – for talking to hospitalized patients without increasing viral exposure, for linking up with family members who can’t visit their loved ones in the hospital, and for helping frontline hospital staff who need a palliative care consultation – or just a chance to debrief on what they are seeing.
A pandemic wake-up call
Elizabeth Gundersen, MD, FHM, FAAHPM, director of the hospice and palliative medicine fellowship program at the Charles E. Schmidt College of Medicine at Florida Atlantic University (FAU) in Boca Raton, practiced hospital medicine for 10 years before pursuing a fellowship in hospice and palliative medicine and working as an academic palliative medicine physician. She calls the pandemic a wake-up call for gaps in care and all the things that weren’t working well in the health care system.
“Now we are seeing more clearly what’s lacking – or broken – and what we will carry forward from this experience into the post-COVID world,” she said. Some hospitalists do palliative care very well, and others don’t feel as comfortable in having these difficult conversations with patients. But in the uncertain course of the virus they get thrust into it.
Although FAU’s associated hospitals were not as inundated with COVID-19 patients in the early weeks of the pandemic as were other regions, the volume of other patients plummeted, Dr. Gundersen said, adding that “there’s still been incredible intensity and worry about the virus. For me, the basic role of palliative care hasn’t changed, and the phrase I have always used when introducing myself – ‘we’re an extra layer of support for the patient and family’ – still holds true,” she said.
“I try to make it clear to people that palliative care is not synonymous with end-of-life care. We don’t want people to think that a palliative care referral implies imminent death. The goal is not to get more people to have a do not attempt resuscitation (DNAR) order, but to determine the patient and family’s treatment goals and whether a DNAR order fits those goals.”
The tough conversations
Dr. Gundersen is cochair of SHM’s Palliative Care Special Interest Group, along with Rab Razzak, MD, clinical director of palliative medicine at University Hospitals Cleveland Medical Center, one of the hospitals affiliated with Case Western University in Cleveland. (Connect with them on Twitter: @Top_Gundersen and @rabrazzak.)
Dr. Razzak also transitioned from hospital medicine to palliative medicine 10 years ago. “As a hospitalist, I enjoyed the tough conversations and bringing the human element into my health care interactions,” he explained. “To me, palliative care is a philosophy of care that puts the person we call the patient at the center of the interaction, while we try to figure out how to best care for them as a person.”
When the pandemic hit, University Hospitals made 20 ICU beds available for COVID-19 patients, Dr. Razzak said. This unit has since been full but not overflowing, while overall hospital census went down. The palliative care team at the hospital includes four inpatient doctors, nurse practitioners, and a chaplain, as well as an outpatient team primarily focused on oncology.
“In some settings, palliative care has been at the forefront of difficult conversations, when things aren’t going well for the patient and there’s much uncertainty,” Dr. Razzak said. The interface between hospital medicine and palliative care can be complementary, he added. “We talk about primary palliative care, which we want every discipline to be able to do – lead meaningful conversations, help manage symptoms.”
The take-home message for hospitalists, he said, is to get training in how to have these discussions, using such resources as VitalTalk (https://www.vitaltalk.org/), a nonprofit organization that disseminates education in communication skills for difficult conversations, and the Center to Advance Palliative Care (www.capc.org) at Icahn School of Medicine at Mount Sinai in New York City. “Once you’ve mastered the conversation, it will get easier. But ask for help when you need it, and learn how to know when you need it.”
Dr. Gundersen added that hospital medicine groups and palliative care teams could reach out to each other and talk about what they did in the crisis and how they can work together in the future. She recommends frequent ongoing support and collaboration that could range from formal conferences or training sessions to informal team interactions, perhaps with sandwiches in the doctor’s lounge – provided that there’s room for social distancing. She has recently started giving talks in the community and grand rounds presentations in hospitals about palliative care.
Other approaches and applications
In New York City, the initial epicenter for the pandemic in the United States, the adult palliative care service of Columbia University Medical Center (CUMC) experienced a sevenfold increase in consultation requests at the apex of the crisis, said its director, Craig Blinderman, MD. That demand was impossible to meet with existing staff. So Dr. Blinderman and colleagues established a virtual consultation model, recruiting and deploying volunteer out-of-state palliative care specialists to staff it.
An eight-bed palliative care unit was opened at CUMC for COVID-19 patients whose surrogates had opted not to initiate or continue intubation or life-sustaining treatments. This helped to relieve some of the pressures on the ICUs while making it possible for in-person visits to the hospice unit by families – in full PPE. Palliative care staff were embedded in various units in the hospital.
A palliative care response team composed of a hospice and palliative medicine fellow and four psychiatry residents or fellows, based in the emergency department and with supervision from the palliative care team, provided time-critical goals of care conversations with families using telemedicine – and a forum for listening to their suffering. Dr. Blinderman and colleagues also have found time to write up their experience for medical journals.1,2
There’s no reason to think that hospitalists, with a little basic training, couldn’t be having these same goals of care conversations, Dr. Blinderman said. “But the fact that hospitalists, at the pandemic’s peak, along with ICU doctors, were seeing an unprecedented magnitude of dying on a daily basis generated a lot of moral distress for them.”
Palliative care professionals, because they engage with these issues in a different way, may be somewhat better equipped to deal with the sheer emotional demands when so many are dying, as at the peak of the surge in New York. “We don’t see dying as a failure on our part but an opportunity to relieve suffering,” Dr. Blinderman said. And the palliative care field also emphasizes the importance of self-care for its practitioners.
“How do we meet the incredible palliative care needs in the epicenter of a pandemic? That question also applies to other kinds of crises we could imagine, for example, climate-related disasters,” Dr. Blinderman said. “What lessons have we learned about the value of palliative care and how to start incorporating it more integrally into the delivery of hospital care? Here we showed that we could work collaboratively with our colleagues at other major medical centers, bringing together their expertise to help us when we didn’t have the bandwidth to meet the demand,” he said.
Scripts can help
“Also, it won’t make sense to just go back to normal (after the crisis fades),” Dr. Blinderman said. “We need to take a close look at how our society is functioning in the wake of the pandemic and the ways the health care system has failed us. We have learned that we’re all interconnected and we need to work together to serve our communities – locally and nationally – applying basic distributive justice.”
Could there be, for example, a national infrastructure for mobilizing and deploying palliative care resources to areas of greatest need, similar to what was done in New York?
At Northwestern Medicine in Chicago, a number of palliative care clinicians at the system’s hospitals worked together to develop scripts designed to help other clinicians start goals of care conversations with patients and families, for use in the hospital as well as in outpatient primary care and other settings, with results integrated into the system’s electronic health record.
Front-line clinicians may not have the time to ask for formal consults from palliative care because of high volume and rapidly changing patient status, explained Eytan Szmuilowicz, MD, director of the section of palliative medicine at Northwestern Memorial Hospital. Or they may not have access to specialty-level palliative care in their settings.
The scripts are aimed at primary care, emergency physicians, and hospitalists needing to consider critical care placement or attempted resuscitation and to ICU clinicians helping families make decisions about life-sustaining treatments. They also can help facilitate advance care planning discussions. An example is “CALMER,” a six-step mnemonic guide to promote goals of care discussions with hospitalized patients. For more information on these scripts, contact Dr. Szmuilowicz: Eytan.Szmuilowicz@nm.org.
Eerily quiet
The COVID-19 crisis has been quite a whirlwind for hospital medicine, said Jeanie Youngwerth, MD, a hospitalist and program director of the palliative care service at the University of Colorado in Denver, which was a significant viral hotspot early on.
“When it first started, things seemed to change almost overnight – starting on Friday, March 13. People had to take action right away to develop work flows and the technology to allow us to see as many patients as possible,” she said. By the time Monday came, it was a whole new ballgame.
Dr. Youngwerth and two colleagues worked quickly to develop inpatient telemedicine capacity where none existed. “We knew we would not be going into patients’ rooms, but most of our team showed up in the hospital to work with the primary care teams. Our job was to see what we could do that actually made a difference,” she said.
“The hospital became a very strange place. You’d walk down the hallway and it was eerily quiet. Everybody you came across was being so nice to each other.” Televisits became a powerful way to bring the human connection back to medical care.
“What we learned from families was that they were thirsting to have some kind of connection with their loved one, and to be able to talk about their loved one and who they were as a person,” she said. “We’d contact the family through video visits and then, when the family meeting ended, the nurse would bring an iPad into the patient’s room so the family could see their loved one on a ventilator. They would immediately start communicating with their loved one, praying aloud, singing, playing music. It would make a huge difference for the family – and for the staff.”
References
1. Nakagawa S et al. Pandemic palliative care consultations spanning state and institutional borders. J Am Geriatr Soc. 2020 May 22. doi: 10.1111/jgs.16643.
2. Lee J Abrukin L, Flores S. Early intervention of palliative care in the emergency department during the COVID-19 pandemic. JAMA Intern Med. 2020 Jun 5. doi: 10.1001/jamainternmed.2020.2713.
For some palliative care professionals, the COVID-19 pandemic, particularly in viral hot spots like New York City, represents a “moment” that could lead to greater awareness of what this service offers to seriously ill patients in a crisis.
They say it has provided an opportunity to show what palliative care teams can contribute to the difficult circumstances of patients with severe symptoms, isolated and alone in quarantined hospitals, with poor survival rates, perhaps sedated for extended stays on scarce ventilators – and for their family members, who are able to visit them only virtually via telephone or tablet.
But it has also highlighted gaps – including insufficient staffing for some palliative care teams. Hospitalists and other clinicians in the hospital need to learn the basics of primary palliative care, such as how to communicate bad news, initiate goals of care conversations, and address common symptoms of serious illness, such as pain. That way, they could shoulder more of the demand for this kind of care when palliative care specialists are in short supply.
Hospitalists, some of whom also have pursued a specialization in palliative care, have played key roles in clarifying and redefining the new role for palliative care, whom it is meant for, and who should provide it. Central to this new role is the greater use of telemedicine – for talking to hospitalized patients without increasing viral exposure, for linking up with family members who can’t visit their loved ones in the hospital, and for helping frontline hospital staff who need a palliative care consultation – or just a chance to debrief on what they are seeing.
A pandemic wake-up call
Elizabeth Gundersen, MD, FHM, FAAHPM, director of the hospice and palliative medicine fellowship program at the Charles E. Schmidt College of Medicine at Florida Atlantic University (FAU) in Boca Raton, practiced hospital medicine for 10 years before pursuing a fellowship in hospice and palliative medicine and working as an academic palliative medicine physician. She calls the pandemic a wake-up call for gaps in care and all the things that weren’t working well in the health care system.
“Now we are seeing more clearly what’s lacking – or broken – and what we will carry forward from this experience into the post-COVID world,” she said. Some hospitalists do palliative care very well, and others don’t feel as comfortable in having these difficult conversations with patients. But in the uncertain course of the virus they get thrust into it.
Although FAU’s associated hospitals were not as inundated with COVID-19 patients in the early weeks of the pandemic as were other regions, the volume of other patients plummeted, Dr. Gundersen said, adding that “there’s still been incredible intensity and worry about the virus. For me, the basic role of palliative care hasn’t changed, and the phrase I have always used when introducing myself – ‘we’re an extra layer of support for the patient and family’ – still holds true,” she said.
“I try to make it clear to people that palliative care is not synonymous with end-of-life care. We don’t want people to think that a palliative care referral implies imminent death. The goal is not to get more people to have a do not attempt resuscitation (DNAR) order, but to determine the patient and family’s treatment goals and whether a DNAR order fits those goals.”
The tough conversations
Dr. Gundersen is cochair of SHM’s Palliative Care Special Interest Group, along with Rab Razzak, MD, clinical director of palliative medicine at University Hospitals Cleveland Medical Center, one of the hospitals affiliated with Case Western University in Cleveland. (Connect with them on Twitter: @Top_Gundersen and @rabrazzak.)
Dr. Razzak also transitioned from hospital medicine to palliative medicine 10 years ago. “As a hospitalist, I enjoyed the tough conversations and bringing the human element into my health care interactions,” he explained. “To me, palliative care is a philosophy of care that puts the person we call the patient at the center of the interaction, while we try to figure out how to best care for them as a person.”
When the pandemic hit, University Hospitals made 20 ICU beds available for COVID-19 patients, Dr. Razzak said. This unit has since been full but not overflowing, while overall hospital census went down. The palliative care team at the hospital includes four inpatient doctors, nurse practitioners, and a chaplain, as well as an outpatient team primarily focused on oncology.
“In some settings, palliative care has been at the forefront of difficult conversations, when things aren’t going well for the patient and there’s much uncertainty,” Dr. Razzak said. The interface between hospital medicine and palliative care can be complementary, he added. “We talk about primary palliative care, which we want every discipline to be able to do – lead meaningful conversations, help manage symptoms.”
The take-home message for hospitalists, he said, is to get training in how to have these discussions, using such resources as VitalTalk (https://www.vitaltalk.org/), a nonprofit organization that disseminates education in communication skills for difficult conversations, and the Center to Advance Palliative Care (www.capc.org) at Icahn School of Medicine at Mount Sinai in New York City. “Once you’ve mastered the conversation, it will get easier. But ask for help when you need it, and learn how to know when you need it.”
Dr. Gundersen added that hospital medicine groups and palliative care teams could reach out to each other and talk about what they did in the crisis and how they can work together in the future. She recommends frequent ongoing support and collaboration that could range from formal conferences or training sessions to informal team interactions, perhaps with sandwiches in the doctor’s lounge – provided that there’s room for social distancing. She has recently started giving talks in the community and grand rounds presentations in hospitals about palliative care.
Other approaches and applications
In New York City, the initial epicenter for the pandemic in the United States, the adult palliative care service of Columbia University Medical Center (CUMC) experienced a sevenfold increase in consultation requests at the apex of the crisis, said its director, Craig Blinderman, MD. That demand was impossible to meet with existing staff. So Dr. Blinderman and colleagues established a virtual consultation model, recruiting and deploying volunteer out-of-state palliative care specialists to staff it.
An eight-bed palliative care unit was opened at CUMC for COVID-19 patients whose surrogates had opted not to initiate or continue intubation or life-sustaining treatments. This helped to relieve some of the pressures on the ICUs while making it possible for in-person visits to the hospice unit by families – in full PPE. Palliative care staff were embedded in various units in the hospital.
A palliative care response team composed of a hospice and palliative medicine fellow and four psychiatry residents or fellows, based in the emergency department and with supervision from the palliative care team, provided time-critical goals of care conversations with families using telemedicine – and a forum for listening to their suffering. Dr. Blinderman and colleagues also have found time to write up their experience for medical journals.1,2
There’s no reason to think that hospitalists, with a little basic training, couldn’t be having these same goals of care conversations, Dr. Blinderman said. “But the fact that hospitalists, at the pandemic’s peak, along with ICU doctors, were seeing an unprecedented magnitude of dying on a daily basis generated a lot of moral distress for them.”
Palliative care professionals, because they engage with these issues in a different way, may be somewhat better equipped to deal with the sheer emotional demands when so many are dying, as at the peak of the surge in New York. “We don’t see dying as a failure on our part but an opportunity to relieve suffering,” Dr. Blinderman said. And the palliative care field also emphasizes the importance of self-care for its practitioners.
“How do we meet the incredible palliative care needs in the epicenter of a pandemic? That question also applies to other kinds of crises we could imagine, for example, climate-related disasters,” Dr. Blinderman said. “What lessons have we learned about the value of palliative care and how to start incorporating it more integrally into the delivery of hospital care? Here we showed that we could work collaboratively with our colleagues at other major medical centers, bringing together their expertise to help us when we didn’t have the bandwidth to meet the demand,” he said.
Scripts can help
“Also, it won’t make sense to just go back to normal (after the crisis fades),” Dr. Blinderman said. “We need to take a close look at how our society is functioning in the wake of the pandemic and the ways the health care system has failed us. We have learned that we’re all interconnected and we need to work together to serve our communities – locally and nationally – applying basic distributive justice.”
Could there be, for example, a national infrastructure for mobilizing and deploying palliative care resources to areas of greatest need, similar to what was done in New York?
At Northwestern Medicine in Chicago, a number of palliative care clinicians at the system’s hospitals worked together to develop scripts designed to help other clinicians start goals of care conversations with patients and families, for use in the hospital as well as in outpatient primary care and other settings, with results integrated into the system’s electronic health record.
Front-line clinicians may not have the time to ask for formal consults from palliative care because of high volume and rapidly changing patient status, explained Eytan Szmuilowicz, MD, director of the section of palliative medicine at Northwestern Memorial Hospital. Or they may not have access to specialty-level palliative care in their settings.
The scripts are aimed at primary care, emergency physicians, and hospitalists needing to consider critical care placement or attempted resuscitation and to ICU clinicians helping families make decisions about life-sustaining treatments. They also can help facilitate advance care planning discussions. An example is “CALMER,” a six-step mnemonic guide to promote goals of care discussions with hospitalized patients. For more information on these scripts, contact Dr. Szmuilowicz: Eytan.Szmuilowicz@nm.org.
Eerily quiet
The COVID-19 crisis has been quite a whirlwind for hospital medicine, said Jeanie Youngwerth, MD, a hospitalist and program director of the palliative care service at the University of Colorado in Denver, which was a significant viral hotspot early on.
“When it first started, things seemed to change almost overnight – starting on Friday, March 13. People had to take action right away to develop work flows and the technology to allow us to see as many patients as possible,” she said. By the time Monday came, it was a whole new ballgame.
Dr. Youngwerth and two colleagues worked quickly to develop inpatient telemedicine capacity where none existed. “We knew we would not be going into patients’ rooms, but most of our team showed up in the hospital to work with the primary care teams. Our job was to see what we could do that actually made a difference,” she said.
“The hospital became a very strange place. You’d walk down the hallway and it was eerily quiet. Everybody you came across was being so nice to each other.” Televisits became a powerful way to bring the human connection back to medical care.
“What we learned from families was that they were thirsting to have some kind of connection with their loved one, and to be able to talk about their loved one and who they were as a person,” she said. “We’d contact the family through video visits and then, when the family meeting ended, the nurse would bring an iPad into the patient’s room so the family could see their loved one on a ventilator. They would immediately start communicating with their loved one, praying aloud, singing, playing music. It would make a huge difference for the family – and for the staff.”
References
1. Nakagawa S et al. Pandemic palliative care consultations spanning state and institutional borders. J Am Geriatr Soc. 2020 May 22. doi: 10.1111/jgs.16643.
2. Lee J Abrukin L, Flores S. Early intervention of palliative care in the emergency department during the COVID-19 pandemic. JAMA Intern Med. 2020 Jun 5. doi: 10.1001/jamainternmed.2020.2713.
ACC panel defines, advises on heart failure with ‘recovered’ EF
Because heart failure patients with recovered ejection fraction are a complex and diverse group, little consensus has emerged on how to define, diagnose, and manage this growing population.
To provide some clarity for identifying and treating these patients, a Journal of the American College of Cardiology scientific expert panel has issued a consensus document. Published Aug. 3 in the Journal of the American College of Cardiology, it provides a working definition of heart failure with recovered ejection fraction (HFrecEF) and recommends approaches for treatment and follow-up.
Defining a new class of HF
“Part of the impetus of this was to bring attention to what we think is a new class of heart failure, and it requires different treatment modalities and different ways of thinking about it,” expert panel member Douglas L. Mann, MD, cardiologist-in-chief at Barnes Jewish Hospital in St. Louis, said in an interview. “It’s a newly discovered HF biology about which we know very little and it’s very confusing to just go on the ejection fraction alone.”
The panel, led by Jane E. Wilcox, MD, of Northwestern University, Chicago, recommends three components for a working definition of HFrecEF: 1) documentation of a decreased left ventricle ejection fraction (LVEF) of less than 40% at baseline; 2) a 10% or greater absolute improvement in LVEF; and 3) a second measurement of LVEF >40%.
“We try to give it a nomenclature that clearly indicates what it is,” Dr. Mann said. “There has been a lot of confusing terminology.” Among the terms the panel calls out in the lexicon of modestly recovered EF, in addition to HFrecEF, are HF improved EF, HF with preserved EF (HFpEF), borderline HFpEF and HF with mid-range EF (HFmrEF).
The panel also recommends that guideline-based medical and device therapies for HFrecEF should continue indefinitely until there’s a better understanding of the biology and clinical epidemiology of HFrecEF, and that these patients should have close clinical follow-up because of the high risk of HF relapse.
Determining EF’s ‘trajectory’
The findings presented in the statement should help cardiologists distinguish HFrecEF from HFpEF, Dr. Mann noted. “Because EF is moving, we also have to emphasize the importance of following the trajectory of EF,” he said. “It’s not enough to know where the EF is; you have to know where it came from – if it had been from a higher number or a lower number – because that will help inform you about the patient’s disease.”
In that regard, the panel states that the level of change in LVEF – the “trajectory” – will provide clues to the nature and extent of myocardial injury, degree and duration of LV remodeling, and the type of therapy that’s indicated. Clinicians should consider HFmrEF, a description the European Society of Cardiology has endorsed, as an entity different from HFrecEF without data on LVEF trajectory.
The statement delves into the biology of HFrecEF, defining reverse LV remodeling as the restoration of some normalization to cardiac myocyte size and LV chamber geometry that results in a leftward shift toward normalization of end-diastolic pressure volume. The panel also noted that cardiac remodeling in reverse LV remodeling and recovery of LV function is bidirectional and involves multiple molecular and cellular changes that contribute to changes in the heart’s size, shape and function, and explains the role gene expression has in HF-related LV changes.
The statement explores the recovery of LV function, noting that spontaneous recovery often occurs when the cause of the myocardial dysfunction resolves. Common causes are chronic tachycardia and thyroid disease.
That panel noted that “super responders” to cardiac resynchronization therapy can provide insight into HFrecEF. Favorable responders include women, patients with nonischemic HF, very wide ECG ventricular depolarization wavelength with left-bundle branch block morphology, and dyssynchrony on ECG.
The panel states that, “regardless of the definition of HFrecEF,” the evidence suggests that younger patients, women, and those with nonischemic disease, shorter disease duration, and relatively few comorbidities are more likely to recover LVEF – and their outcomes are typically better than those of patients with HF reduced EF (HFrEF) and HFpEF.
Clinicians should bear in mind, however, that patients on guideline-directed medical therapy (GDMT) who achieve complete normalization of LV structure and function are prone to recurrent LV dysfunction and HF. The panel explored the role of potential treatment for three different etiologies of HF. Little is known about Takotsubo cardiomyopathy, considered a transient form of LV dysfunction, in terms of how many of these patients will develop HFrEF or if they’ll benefit from GDMT. Alcohol-induced cardiomyopathy patients should continue on medical therapy even if they have HFrecEF, as should patients with fulminant and nonfulminant myocarditis.
Managing HFrecEF
Management should include assessment of jugular vein distention and signs of volume overload – “particularly concerning in HFrecEF” – the panel noted. ECG is cost effective, and signs of left-bundle branch block are predictors of low success with GDMT alone. The panel also recommended a family history going back three generations and consideration of genetic testing to determine the risk for sudden cardiac death. Two-dimensional ECG can help predict GDMT response and cardiovascular magnetic resonance imaging can provide information about myocardial substrate at the time of diagnosis of HFrEF.
The panel suggested four areas for future research: 1) improved phenotyping of HFrEF; 2) use of inception cohorts to better understand the natural history of HFrecEF; 3) clinical trials to better define those clinical care components most effective at maintaining remission; and 4) basic studies to better define the biology of HFrecEF. “The goal,” wrote Dr. Wilcox and colleagues, “is to develop new therapeutic targets that will enable patients with HFrecEF to experience a durable remission from HF.”
Dr. Wilcox reported receiving funding from the National Institutes of Health and the American Heart Association, and financial relationships with Abbott, Medtronic, and Cytokinetics. Dr. Mann has received funding from NIH and reports financial relationships with MyoKardia and Novartis. Coauthors reported funding from NIH and AHA and financial relationships with Novartis, Amgen, AstraZeneca, Thoratec Corporation (Abbott), Sanofi, Pfizer, MyoKardia and American Regent.
SOURCE: Wilcox JE et al. J Am Coll Cardiol. 2020;76:719-34.
Because heart failure patients with recovered ejection fraction are a complex and diverse group, little consensus has emerged on how to define, diagnose, and manage this growing population.
To provide some clarity for identifying and treating these patients, a Journal of the American College of Cardiology scientific expert panel has issued a consensus document. Published Aug. 3 in the Journal of the American College of Cardiology, it provides a working definition of heart failure with recovered ejection fraction (HFrecEF) and recommends approaches for treatment and follow-up.
Defining a new class of HF
“Part of the impetus of this was to bring attention to what we think is a new class of heart failure, and it requires different treatment modalities and different ways of thinking about it,” expert panel member Douglas L. Mann, MD, cardiologist-in-chief at Barnes Jewish Hospital in St. Louis, said in an interview. “It’s a newly discovered HF biology about which we know very little and it’s very confusing to just go on the ejection fraction alone.”
The panel, led by Jane E. Wilcox, MD, of Northwestern University, Chicago, recommends three components for a working definition of HFrecEF: 1) documentation of a decreased left ventricle ejection fraction (LVEF) of less than 40% at baseline; 2) a 10% or greater absolute improvement in LVEF; and 3) a second measurement of LVEF >40%.
“We try to give it a nomenclature that clearly indicates what it is,” Dr. Mann said. “There has been a lot of confusing terminology.” Among the terms the panel calls out in the lexicon of modestly recovered EF, in addition to HFrecEF, are HF improved EF, HF with preserved EF (HFpEF), borderline HFpEF and HF with mid-range EF (HFmrEF).
The panel also recommends that guideline-based medical and device therapies for HFrecEF should continue indefinitely until there’s a better understanding of the biology and clinical epidemiology of HFrecEF, and that these patients should have close clinical follow-up because of the high risk of HF relapse.
Determining EF’s ‘trajectory’
The findings presented in the statement should help cardiologists distinguish HFrecEF from HFpEF, Dr. Mann noted. “Because EF is moving, we also have to emphasize the importance of following the trajectory of EF,” he said. “It’s not enough to know where the EF is; you have to know where it came from – if it had been from a higher number or a lower number – because that will help inform you about the patient’s disease.”
In that regard, the panel states that the level of change in LVEF – the “trajectory” – will provide clues to the nature and extent of myocardial injury, degree and duration of LV remodeling, and the type of therapy that’s indicated. Clinicians should consider HFmrEF, a description the European Society of Cardiology has endorsed, as an entity different from HFrecEF without data on LVEF trajectory.
The statement delves into the biology of HFrecEF, defining reverse LV remodeling as the restoration of some normalization to cardiac myocyte size and LV chamber geometry that results in a leftward shift toward normalization of end-diastolic pressure volume. The panel also noted that cardiac remodeling in reverse LV remodeling and recovery of LV function is bidirectional and involves multiple molecular and cellular changes that contribute to changes in the heart’s size, shape and function, and explains the role gene expression has in HF-related LV changes.
The statement explores the recovery of LV function, noting that spontaneous recovery often occurs when the cause of the myocardial dysfunction resolves. Common causes are chronic tachycardia and thyroid disease.
That panel noted that “super responders” to cardiac resynchronization therapy can provide insight into HFrecEF. Favorable responders include women, patients with nonischemic HF, very wide ECG ventricular depolarization wavelength with left-bundle branch block morphology, and dyssynchrony on ECG.
The panel states that, “regardless of the definition of HFrecEF,” the evidence suggests that younger patients, women, and those with nonischemic disease, shorter disease duration, and relatively few comorbidities are more likely to recover LVEF – and their outcomes are typically better than those of patients with HF reduced EF (HFrEF) and HFpEF.
Clinicians should bear in mind, however, that patients on guideline-directed medical therapy (GDMT) who achieve complete normalization of LV structure and function are prone to recurrent LV dysfunction and HF. The panel explored the role of potential treatment for three different etiologies of HF. Little is known about Takotsubo cardiomyopathy, considered a transient form of LV dysfunction, in terms of how many of these patients will develop HFrEF or if they’ll benefit from GDMT. Alcohol-induced cardiomyopathy patients should continue on medical therapy even if they have HFrecEF, as should patients with fulminant and nonfulminant myocarditis.
Managing HFrecEF
Management should include assessment of jugular vein distention and signs of volume overload – “particularly concerning in HFrecEF” – the panel noted. ECG is cost effective, and signs of left-bundle branch block are predictors of low success with GDMT alone. The panel also recommended a family history going back three generations and consideration of genetic testing to determine the risk for sudden cardiac death. Two-dimensional ECG can help predict GDMT response and cardiovascular magnetic resonance imaging can provide information about myocardial substrate at the time of diagnosis of HFrEF.
The panel suggested four areas for future research: 1) improved phenotyping of HFrEF; 2) use of inception cohorts to better understand the natural history of HFrecEF; 3) clinical trials to better define those clinical care components most effective at maintaining remission; and 4) basic studies to better define the biology of HFrecEF. “The goal,” wrote Dr. Wilcox and colleagues, “is to develop new therapeutic targets that will enable patients with HFrecEF to experience a durable remission from HF.”
Dr. Wilcox reported receiving funding from the National Institutes of Health and the American Heart Association, and financial relationships with Abbott, Medtronic, and Cytokinetics. Dr. Mann has received funding from NIH and reports financial relationships with MyoKardia and Novartis. Coauthors reported funding from NIH and AHA and financial relationships with Novartis, Amgen, AstraZeneca, Thoratec Corporation (Abbott), Sanofi, Pfizer, MyoKardia and American Regent.
SOURCE: Wilcox JE et al. J Am Coll Cardiol. 2020;76:719-34.
Because heart failure patients with recovered ejection fraction are a complex and diverse group, little consensus has emerged on how to define, diagnose, and manage this growing population.
To provide some clarity for identifying and treating these patients, a Journal of the American College of Cardiology scientific expert panel has issued a consensus document. Published Aug. 3 in the Journal of the American College of Cardiology, it provides a working definition of heart failure with recovered ejection fraction (HFrecEF) and recommends approaches for treatment and follow-up.
Defining a new class of HF
“Part of the impetus of this was to bring attention to what we think is a new class of heart failure, and it requires different treatment modalities and different ways of thinking about it,” expert panel member Douglas L. Mann, MD, cardiologist-in-chief at Barnes Jewish Hospital in St. Louis, said in an interview. “It’s a newly discovered HF biology about which we know very little and it’s very confusing to just go on the ejection fraction alone.”
The panel, led by Jane E. Wilcox, MD, of Northwestern University, Chicago, recommends three components for a working definition of HFrecEF: 1) documentation of a decreased left ventricle ejection fraction (LVEF) of less than 40% at baseline; 2) a 10% or greater absolute improvement in LVEF; and 3) a second measurement of LVEF >40%.
“We try to give it a nomenclature that clearly indicates what it is,” Dr. Mann said. “There has been a lot of confusing terminology.” Among the terms the panel calls out in the lexicon of modestly recovered EF, in addition to HFrecEF, are HF improved EF, HF with preserved EF (HFpEF), borderline HFpEF and HF with mid-range EF (HFmrEF).
The panel also recommends that guideline-based medical and device therapies for HFrecEF should continue indefinitely until there’s a better understanding of the biology and clinical epidemiology of HFrecEF, and that these patients should have close clinical follow-up because of the high risk of HF relapse.
Determining EF’s ‘trajectory’
The findings presented in the statement should help cardiologists distinguish HFrecEF from HFpEF, Dr. Mann noted. “Because EF is moving, we also have to emphasize the importance of following the trajectory of EF,” he said. “It’s not enough to know where the EF is; you have to know where it came from – if it had been from a higher number or a lower number – because that will help inform you about the patient’s disease.”
In that regard, the panel states that the level of change in LVEF – the “trajectory” – will provide clues to the nature and extent of myocardial injury, degree and duration of LV remodeling, and the type of therapy that’s indicated. Clinicians should consider HFmrEF, a description the European Society of Cardiology has endorsed, as an entity different from HFrecEF without data on LVEF trajectory.
The statement delves into the biology of HFrecEF, defining reverse LV remodeling as the restoration of some normalization to cardiac myocyte size and LV chamber geometry that results in a leftward shift toward normalization of end-diastolic pressure volume. The panel also noted that cardiac remodeling in reverse LV remodeling and recovery of LV function is bidirectional and involves multiple molecular and cellular changes that contribute to changes in the heart’s size, shape and function, and explains the role gene expression has in HF-related LV changes.
The statement explores the recovery of LV function, noting that spontaneous recovery often occurs when the cause of the myocardial dysfunction resolves. Common causes are chronic tachycardia and thyroid disease.
That panel noted that “super responders” to cardiac resynchronization therapy can provide insight into HFrecEF. Favorable responders include women, patients with nonischemic HF, very wide ECG ventricular depolarization wavelength with left-bundle branch block morphology, and dyssynchrony on ECG.
The panel states that, “regardless of the definition of HFrecEF,” the evidence suggests that younger patients, women, and those with nonischemic disease, shorter disease duration, and relatively few comorbidities are more likely to recover LVEF – and their outcomes are typically better than those of patients with HF reduced EF (HFrEF) and HFpEF.
Clinicians should bear in mind, however, that patients on guideline-directed medical therapy (GDMT) who achieve complete normalization of LV structure and function are prone to recurrent LV dysfunction and HF. The panel explored the role of potential treatment for three different etiologies of HF. Little is known about Takotsubo cardiomyopathy, considered a transient form of LV dysfunction, in terms of how many of these patients will develop HFrEF or if they’ll benefit from GDMT. Alcohol-induced cardiomyopathy patients should continue on medical therapy even if they have HFrecEF, as should patients with fulminant and nonfulminant myocarditis.
Managing HFrecEF
Management should include assessment of jugular vein distention and signs of volume overload – “particularly concerning in HFrecEF” – the panel noted. ECG is cost effective, and signs of left-bundle branch block are predictors of low success with GDMT alone. The panel also recommended a family history going back three generations and consideration of genetic testing to determine the risk for sudden cardiac death. Two-dimensional ECG can help predict GDMT response and cardiovascular magnetic resonance imaging can provide information about myocardial substrate at the time of diagnosis of HFrEF.
The panel suggested four areas for future research: 1) improved phenotyping of HFrEF; 2) use of inception cohorts to better understand the natural history of HFrecEF; 3) clinical trials to better define those clinical care components most effective at maintaining remission; and 4) basic studies to better define the biology of HFrecEF. “The goal,” wrote Dr. Wilcox and colleagues, “is to develop new therapeutic targets that will enable patients with HFrecEF to experience a durable remission from HF.”
Dr. Wilcox reported receiving funding from the National Institutes of Health and the American Heart Association, and financial relationships with Abbott, Medtronic, and Cytokinetics. Dr. Mann has received funding from NIH and reports financial relationships with MyoKardia and Novartis. Coauthors reported funding from NIH and AHA and financial relationships with Novartis, Amgen, AstraZeneca, Thoratec Corporation (Abbott), Sanofi, Pfizer, MyoKardia and American Regent.
SOURCE: Wilcox JE et al. J Am Coll Cardiol. 2020;76:719-34.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY

















