User login
Caring for patients on probation or parole
Mr. A, age 35, presents to your outpatient community mental health practice. He has a history of psychosis that began in his late teens. Since then, his symptoms have included derogatory auditory hallucinations, a recurrent persecutory delusion that governmental agencies are tracking his movements, and intermittent disorganized speech. At age 30, Mr. A assaulted a stranger out of fear that the individual was a government agent. He was arrested and experienced a severe psychotic decompensation while awaiting trial. He was found incompetent to stand trial and sent to a state hospital for restoration.
After 6 months of treatment and observation, Mr. A was deemed competent to proceed and returned to jail. He was subsequently convicted of assault and sentenced to 7 years in prison. While in prison, he received regular mental health care with infrequent recurrence of minor psychotic symptoms. He was released on parole due to his good behavior, but as part of his conditions of parole, he was mandated to follow up with an outpatient mental health clinician.
After telling you the story of how he ended up in your office, Mr. A says he needs you to speak regularly with his parole officer to verify his attendance at appointments and to discuss any mental health concerns you may have. Since you have not worked with a patient on parole before, your mind is full of questions: What are the expectations regarding your communication with his parole officer? Could Mr. A return to prison if you express concerns about his mental health? What can you do to improve his chances of success in the community?
Given the high rates of mental illness among individuals incarcerated in the United States, it shouldn’t be surprising that there are similarly high rates of mental illness among those on supervised release from jails and prisons. Clinicians who work with patients on community release need to understand basic concepts related to probation and parole, and how to promote patients’ stability in the community to reduce recidivism and re-incarceration. The court may require individuals on probation or parole to adhere to certain conditions of release, which could include seeing a psychiatrist or psychotherapist, participating in substance abuse treatment, and/or taking psychotropic medication. The court usually closely monitors the probationer or parolee’s adherence, and noncompliance can be grounds for probation or parole violation and revocation.
This article reviews the concepts of probation and parole (Box1,2), describes the prevalence of mental illness among probationers and parolees, and discusses the unique challenges and opportunities psychiatrists and other mental health professionals face when working with individuals on community supervision.
Box
The US Bureau of Justice Statistics (BJS) defines probation as a “court-ordered period of correctional supervision in the community, generally as an alternative to incarceration.” Probation allows individuals to be released from jail to community supervision, with the potential for dismissal or lowering of charges if they adhere to the conditions of probation. Conditions of probation may include participating in substance abuse or mental health treatment programs, abstaining from drugs and alcohol, and avoiding contact with known felons. Failure to comply with conditions of probation can lead to re-incarceration and probation revocation.1 If probation is revoked, a probationer may be sentenced, potentially to prison, depending on the severity of the original offense.2
The BJS defines parole as “a period of conditional supervised release in the community following a term in state or federal prison.”2 Parole allows for the community supervision of individuals who have already been convicted of and sentenced to prison for a crime. Individuals may be released on parole if they demonstrate good behavior while incarcerated. Similar to probationers, parolees must adhere to the conditions of parole, and violation of these may lead to re-incarceration.1
As of December 31, 2016, there were more than 4.5 million adults on community supervision in the United States, representing 1 out of every 55 adults in the US population. Individuals on probation accounted for 81% of adults on community supervision. The number of people on community supervision has dropped continuously over the last decade, a trend driven by 2% annual decreases in the probation population. In contrast, the parolee population has continued to grow over time and was approximately 900,000 individuals at the end of 2016.2
Mental illness among probationers and parolees
Research on mental illness in people involved in the criminal justice system has largely focused on those who are incarcerated. Studies have documented high rates of severe mental illness (SMI), such as schizophrenia and bipolar disorder, among those who are incarcerated; some estimate the rates to be 3 times as high as those of community samples.3,4 In addition to SMI, substance use disorders and personality disorders (in particular, antisocial personality disorder) are common among people who are incarcerated.5,6
Comparatively little is known about mental illness among probationers and parolees, although presumably there would be a similarly high prevalence of SMI, substance use disorders, and other psychiatric disorders among this population. A 1997 Bureau of Justice Statistics (BJS) survey of approximately 3.4 million probationers found that 13.8% self-reported a mental or emotional condition and 8.2% self-reported a history of an “overnight stay in a mental hospital.”7 The BJS estimated that there were approximately 550,000 probationers with mental illness in the United States. The study’s author noted that probationers with mental illness were more likely to have a history of prior offenses and more likely to be violent recidivists. In terms of substance use, compared with other probationers, those with mental illness were more likely to report using drugs in the month before their most recent offense and at the time of the offense.7
Continue to: More recent research...
More recent research, although limited, has shed some light on the role of mental health services for individuals on probation and parole. In 2009, Crilly et al8 reported that 23% of probationers reported accessing mental health services within the past year. Other studies have found that probationer and parolee engagement in mental health care reduces the risk of recidivism.9,10 A 2011 study evaluated 100 individuals on probation and parole in 2 counties in a southeastern state. The authors found that 75% of participants reported that they needed counseling for a mental health concern in the past year, but that only approximately 30% of them actually sought help. Individuals reporting higher levels of posttraumatic stress disorder symptomatology or greater drug use before being on probation or parole were more likely to seek counseling in the past year.11
An alternative: Problem-solving courts
Problem-solving courts (PSCs) offer an alternative to standard probation and/or sentencing. Problem-solving courts are founded on the concept of therapeutic jurisprudence, which seeks to change “the behavior of litigants and [ensure] the future well-being of communities.”12 Types of PSCs include drug court (the most common type in the United States), domestic violence court, veterans court, and mental health court (MHC), among others.
An individual may choose a PSC over standard probation because participants usually receive more assistance in obtaining treatment and closer supervision with an emphasis on rehabilitation rather than incapacitation or retribution. The success of PSCs relies heavily on the judge, as he/she plays a pivotal role in developing relationships with the participants, considering therapeutic alternatives to “bad” behaviors, determining sanctions, and relying on community mental health partners to assist participants in complying with conditions of the court.13-15
Psychiatrists and other mental health clinicians should be aware of MHCs, which are a type of PSC that provides for the community supervision of individuals with mental illness. Mental health courts vary in terms of eligibility criteria. Some accept individuals who merely report a history of mental illness, whereas others have specific diagnostic requirements.16 Some accept individuals accused of minor violations such as ordinance violations or misdemeanor offenses, while others accept individuals accused of felonies. Like other PSCs, participation in an MHC is voluntary, and most require a participant to enter a guilty plea upon entry.17 Participants may choose to enter an MHC to avoid prison time or to reduce or expunge charges after completing the program. Many MHCs also assign a probation officer to follow the participant in the community, similar to a standard probation model. Participants are usually expected to engage in psychiatric treatment, including psychotherapy, substance abuse counseling, medication management, and other services. If they do not comply with these conditions, they face sanctions that could include jail “shock” time, enhanced supervision, or an increase in psychiatric services.
Outpatient mental health professionals play an integral role in MHCs. Depending on the model, he/she may be asked to communicate treatment recommendations, attend weekly meetings at the court, and provide suggestions for interventions when the participant relapses, recidivates, and/or decompensates psychiatrically. This collaborative model can work well and allow the clinician unique opportunities to educate the court and advocate for his/her patient. However, clinicians who participate in an MHC need to remain aware of the potential to become a de facto probation officer, and need to maintain appropriate boundaries and roles. They should ensure that the patient provides initial and ongoing consent for them to communicate with the court, and share their programmatic recommendations with the patient to preserve the therapeutic alliance.
Continue to: Challenges upon re-entering the community
Challenges upon re-entering the community
Individuals recently released from jail or prison face unique challenges when re-entering the community. An individual who has been incarcerated, particularly for months to years, has likely lost his/her job, housing, health insurance, and access to primary supports. People with mental illness with a history of incarceration have higher rates of homelessness, substance use disorders, and unemployment than those with no history of incarceration.7,18 For individuals with mental illness, these additional stressors lead to further psychiatric decompensation, recidivism, and overutilization of emergency and crisis services upon release from prison or jail. The loss of health insurance presents great challenges: when someone is incarcerated, his/her Medicaid is suspended or terminated.19 This can happen at any point during incarceration. In states that terminate rather than suspend Medicaid, former prisoners face even longer waits to re-establish access to needed health care.
The period immediately after release is a critical time for individuals to be linked with substance and mental health treatment. Binswanger et al20 found former prisoners were at highest risk of mortality in the 2 weeks following release from prison; the highest rates of death were from drug overdose, cardiovascular disease, homicide, and suicide. A subsequent study found that women were at increased risk of drug overdose and opioid-related deaths.21 One explanation for the increase in drug-related deaths is the loss of physiologic tolerance while incarcerated; however, a lack of treatment while incarcerated, high levels of stress upon re-entry, and poor linkage to aftercare also may be contributing factors. Among prisoners recently released from New York City jails, Lim et al22 found that those with a history of homelessness and previous incarceration had the highest rates of drug-related deaths and homicides in the first 2 weeks after release. Non-Hispanic white men had the highest risk of drug-related deaths and suicides. While the risk of death is greatest immediately after release, former prisoners face increased mortality from multiple causes for multiple years after release.20-22
Clinicians who work with recently released prisoners should be aware of these individuals’ risks and actively work with them and other members of the mental health team to ensure these patients have access to social services, employment training, housing, and substance use resources, including medication-assisted treatment. Patients with SMI should be considered for more intensive services, such as assertive community treatment (ACT) or even forensic ACT (FACT) services, given that FACTs have a modest impact in reducing recidivism.23
Knowing whether the patient is on probation or parole and the terms of his/her supervision can also be useful in creating and executing a collaborative treatment plan. The clinician can assist the patient in meeting conditions of probation/parole such as:
- creating a stable home plan with a permanent address
- planning routine check-ins with probation/parole officers, and
- keeping documentation of ongoing mental health and substance use treatment.
Being aware of other terms of supervision, such as abstaining from alcohol and drugs, or remaining in one’s jurisdiction, also can help the patient avoid technical violations and a return to jail or prison.
Continue to: How to best help patients on community supervision
How to best help patients on community supervision
There are some clinical recommendations when working with patients on community supervision. First, do not assume that someone who has been incarcerated has antisocial personality disorder. Behaviors primarily related to seeking or using drugs or survival-type crimes should not be considered “antisocial” without additional evidence of pervasive and persistent conduct demonstrating impulsivity, lack of empathy, dishonesty, or repeated disregard for social norms and others’ rights. To meet criteria for antisocial personality disorder, these behaviors must have begun during childhood or adolescence.
If a patient does meet criteria for antisocial personality disorder, remember that he/she may also have a psychotic, mood, substance use, or other disorder that could lead to a greater likelihood of violence, recidivism, or other poor outcomes if left untreated. Treating any co-occurring disorders could enhance the patient’s engagement with treatment. There is some evidence that certain psychotropic medications, su
In addition to promoting patients’ mental health, such efforts can prevent re-arrest and re-incarceration and make a lasting positive impact on patients’ lives.
CASE CONTINUED
Mr. A signs a release-of-information form and you call his parole officer. His parole officer states that he would like to speak with you every few months to check on Mr. A’s treatment adherence. Within a few months, you transition Mr. A from an oral antipsychotic medication to a long-acting injectable antipsychotic medication to manage his psychotic disorder. He presents on time each month to your clinic to receive the injection.
Five months later, Mr. A receives 2 weeks of “shock time” at the local county jail for “dropping a dirty urine” that was positive for cannabinoids at a meeting with his parole officer. During his time in jail, he receives no treatment and he misses his monthly long-acting injectable dose.
Continue to: Upon release...
Upon release, he demonstrates the recurrence of some mild persecutory fears and hallucinations, but you resume him on his prior treatment regimen, and he recovers.
You encourage the parole officer to notify you if Mr. A violates parole and is incarcerated so that you can speak with clinicians in the jail to ensure that Mr. A remains adequately treated while incarcerated.
In the coming years, you continue to work with Mr. A and his parole officer to manage his mental health condition and to navigate his parole requirements in order to reduce his risk of relapse and recidivism. After Mr. A completes his time on parole, you continue to see him for outpatient follow-up.
Bottom Line
Clinicians may provide psychiatric care to probationers and parolees in traditional outpatient settings or in collaboration with a mental health court (MHC) or forensic assertive community treatment team. It is crucial to be aware of the legal expectations of individuals on community supervision, as well as the unique mental health risks and challenges they face. You can help reduce probationers’ and parolees’ risk of relapse and recidivism and support their recovery in the community by engaging in collaborative treatment planning involving the patient, the court, and/or MHCs.
Related Resources
- Lamb HR. Weinberger LE. Understanding and treating offenders with serious mental illness in public sector mental health. Behav Sci Law. 2017;35(4):303-318.
- Worcester S. Mental illness and the criminal justice system: Reducing the risks. Clinical Psychiatry News. https://www.mdedge.com/psychiatry/article/173208/schizophrenia-other-psychotic-disorders/mental-illness-and-criminal. Published August 22, 2018.
1. Bureau of Justice Statistics. FAQ detail: What is the difference between probation and parole? U.S. Department of Justice. https://www.bjs.gov/index.cfm?ty=qa&iid=324. Accessed November 17, 2018.
2. Kaeble D. Probation and parole in the United States, 2016. U.S. Department of Justice. https://www.bjs.gov/content/pub/pdf/ppus16.pdf. Published April 2018. Accessed April 23, 2019.
3. Kessler RC, Chiu WT, Demler O, et al. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):617-627.
4. Diamond, P.M., et al., The prevalence of mental illness in prison. Adm Policy Ment Health. 2001;29(1):21-40.
5. MacDonald R, Kaba F, Rosner Z, et al. The Rikers Island hot spotters: defining the needs of the most frequently incarcerated. Am J Public Health. 2015;105(11):2262-2268.
6. Trestman RL, Ford J, Zhang W, et al. Current and lifetime psychiatric illness among inmates not identified as acutely mentally ill at intake in Connecticut’s jails. J Am Acad Psychiatry Law. 2007;35(4):490-500.
7. Ditton PM. Bureau of Justice Statistics special report: mental health and treatment of inmates and probationers. U.S. Department of Justice. https://www.bjs.gov/content/pub/pdf/mhtip.pdf. Published July 1999. Accessed April 24, 2019.
8. Crilly JF, Caine ED, Lamberti JS, et al. Mental health services use and symptom prevalence in a cohort of adults on probation. Psychiatr Serv. 2009;60(4):542-544.
9. Herinckx HA, Swart SC, Ama SM, et al. Rearrest and linkage to mental health services among clients of the Clark County mental health court program. Psychiatr Serv. 2005;56(7):853-857.
10. Solomon P, Draine J, Marcus SC. Predicting incarceration of clients of a psychiatric probation and parole service. Psychiatr Serv. 2002;53(1):50-56.
11. Owens GP, Rogers SM, Whitesell AA. Use of mental health services and barriers to care for individuals on probation or parole. J Offender Rehabil. 2011;50(1):35-47.
12. Berman G, Feinblatt J. Problem‐solving courts: a brief primer. Law and Policy. 2001;23(2):126.
13. The Council of State Governments Justice Center. Mental health courts: a guide to research-informed policy and practice. U.S. Department of Justice. https://www.bja.gov/Publications/CSG_MHC_Research.pdf. Published 2009. Accessed November 22, 2018.
14. Landess J, Holoyda B. Mental health courts and forensic assertive community treatment teams as correctional diversion programs. Behav Sci Law. 2017;35(5-6):501-511.
15. Sammon KC. Therapeutic jurisprudence: an examination of problem‐solving justice in New York. Journal of Civil Rights and Economic Development. 2008;23:923.
16. Sarteschi CM, Vaughn MG, Kim, K. Assessing the effectiveness of mental health courts: a quantitative review. Journal of Criminal Justice. 2011;39(1):12-20.
17. Strong SM, Rantala RR. Census of problem-solving courts, 2012. U.S. Department of Justice, Bureau of Justice Assistance. http://www.bjs.gov/content/pub/pdf/cpsc12.pdf. Revised October 12, 2016. Accessed April 24, 2019.
18. McGuire JF, Rosenheck RA. Criminal history as a prognostic indicator in the treatment of homeless people with severe mental illness. Psychiatr Serv. 2004;55(1):42-48.
19. Families USA. Medicaid suspension policies for incarcerated people: a 50-state map. Families USA. https://familiesusa.org/product/medicaid-suspension-policies-incarcerated-people-50-state-map. Published July 2016. Accessed December 7, 2018.
20. Binswanger IA, Stern MF, Deyo RA, et al. Release from prison—a high risk of death for former inmates. N Engl J Med. 2007;356(2):157-165.
21. Binswanger IA, Blatchford PJ, Mueller SR, et al. Mortality after prison release: opioid overdose and other causes of death, risk factors, and time trends from 1999 to 2009. Ann Intern Med. 2013;159(9):592-600.
22. Lim S, Seligson AL, Parvez FM, et al. Risks of drug-related death, suicide, and homicide during the immediate post-release period among people released from New York City Jails, 2001-2005. Am J Epidemiol. 2012;175(6):519-526.
23. Cusack KJ, Morrissey JP, Cuddeback GS, et al. Criminal justice involvement, behavioral health service use, and costs of forensic assertive community treatment: a randomized trial. Community Ment Health J. 2010;46(4):356-363.
24. Felthous AR, Stanford MS. A proposed algorithm for the pharmacotherapy of impulsive aggression. J Am Acad Psychiatry Law. 2015:43(4);456-467.
Mr. A, age 35, presents to your outpatient community mental health practice. He has a history of psychosis that began in his late teens. Since then, his symptoms have included derogatory auditory hallucinations, a recurrent persecutory delusion that governmental agencies are tracking his movements, and intermittent disorganized speech. At age 30, Mr. A assaulted a stranger out of fear that the individual was a government agent. He was arrested and experienced a severe psychotic decompensation while awaiting trial. He was found incompetent to stand trial and sent to a state hospital for restoration.
After 6 months of treatment and observation, Mr. A was deemed competent to proceed and returned to jail. He was subsequently convicted of assault and sentenced to 7 years in prison. While in prison, he received regular mental health care with infrequent recurrence of minor psychotic symptoms. He was released on parole due to his good behavior, but as part of his conditions of parole, he was mandated to follow up with an outpatient mental health clinician.
After telling you the story of how he ended up in your office, Mr. A says he needs you to speak regularly with his parole officer to verify his attendance at appointments and to discuss any mental health concerns you may have. Since you have not worked with a patient on parole before, your mind is full of questions: What are the expectations regarding your communication with his parole officer? Could Mr. A return to prison if you express concerns about his mental health? What can you do to improve his chances of success in the community?
Given the high rates of mental illness among individuals incarcerated in the United States, it shouldn’t be surprising that there are similarly high rates of mental illness among those on supervised release from jails and prisons. Clinicians who work with patients on community release need to understand basic concepts related to probation and parole, and how to promote patients’ stability in the community to reduce recidivism and re-incarceration. The court may require individuals on probation or parole to adhere to certain conditions of release, which could include seeing a psychiatrist or psychotherapist, participating in substance abuse treatment, and/or taking psychotropic medication. The court usually closely monitors the probationer or parolee’s adherence, and noncompliance can be grounds for probation or parole violation and revocation.
This article reviews the concepts of probation and parole (Box1,2), describes the prevalence of mental illness among probationers and parolees, and discusses the unique challenges and opportunities psychiatrists and other mental health professionals face when working with individuals on community supervision.
Box
The US Bureau of Justice Statistics (BJS) defines probation as a “court-ordered period of correctional supervision in the community, generally as an alternative to incarceration.” Probation allows individuals to be released from jail to community supervision, with the potential for dismissal or lowering of charges if they adhere to the conditions of probation. Conditions of probation may include participating in substance abuse or mental health treatment programs, abstaining from drugs and alcohol, and avoiding contact with known felons. Failure to comply with conditions of probation can lead to re-incarceration and probation revocation.1 If probation is revoked, a probationer may be sentenced, potentially to prison, depending on the severity of the original offense.2
The BJS defines parole as “a period of conditional supervised release in the community following a term in state or federal prison.”2 Parole allows for the community supervision of individuals who have already been convicted of and sentenced to prison for a crime. Individuals may be released on parole if they demonstrate good behavior while incarcerated. Similar to probationers, parolees must adhere to the conditions of parole, and violation of these may lead to re-incarceration.1
As of December 31, 2016, there were more than 4.5 million adults on community supervision in the United States, representing 1 out of every 55 adults in the US population. Individuals on probation accounted for 81% of adults on community supervision. The number of people on community supervision has dropped continuously over the last decade, a trend driven by 2% annual decreases in the probation population. In contrast, the parolee population has continued to grow over time and was approximately 900,000 individuals at the end of 2016.2
Mental illness among probationers and parolees
Research on mental illness in people involved in the criminal justice system has largely focused on those who are incarcerated. Studies have documented high rates of severe mental illness (SMI), such as schizophrenia and bipolar disorder, among those who are incarcerated; some estimate the rates to be 3 times as high as those of community samples.3,4 In addition to SMI, substance use disorders and personality disorders (in particular, antisocial personality disorder) are common among people who are incarcerated.5,6
Comparatively little is known about mental illness among probationers and parolees, although presumably there would be a similarly high prevalence of SMI, substance use disorders, and other psychiatric disorders among this population. A 1997 Bureau of Justice Statistics (BJS) survey of approximately 3.4 million probationers found that 13.8% self-reported a mental or emotional condition and 8.2% self-reported a history of an “overnight stay in a mental hospital.”7 The BJS estimated that there were approximately 550,000 probationers with mental illness in the United States. The study’s author noted that probationers with mental illness were more likely to have a history of prior offenses and more likely to be violent recidivists. In terms of substance use, compared with other probationers, those with mental illness were more likely to report using drugs in the month before their most recent offense and at the time of the offense.7
Continue to: More recent research...
More recent research, although limited, has shed some light on the role of mental health services for individuals on probation and parole. In 2009, Crilly et al8 reported that 23% of probationers reported accessing mental health services within the past year. Other studies have found that probationer and parolee engagement in mental health care reduces the risk of recidivism.9,10 A 2011 study evaluated 100 individuals on probation and parole in 2 counties in a southeastern state. The authors found that 75% of participants reported that they needed counseling for a mental health concern in the past year, but that only approximately 30% of them actually sought help. Individuals reporting higher levels of posttraumatic stress disorder symptomatology or greater drug use before being on probation or parole were more likely to seek counseling in the past year.11
An alternative: Problem-solving courts
Problem-solving courts (PSCs) offer an alternative to standard probation and/or sentencing. Problem-solving courts are founded on the concept of therapeutic jurisprudence, which seeks to change “the behavior of litigants and [ensure] the future well-being of communities.”12 Types of PSCs include drug court (the most common type in the United States), domestic violence court, veterans court, and mental health court (MHC), among others.
An individual may choose a PSC over standard probation because participants usually receive more assistance in obtaining treatment and closer supervision with an emphasis on rehabilitation rather than incapacitation or retribution. The success of PSCs relies heavily on the judge, as he/she plays a pivotal role in developing relationships with the participants, considering therapeutic alternatives to “bad” behaviors, determining sanctions, and relying on community mental health partners to assist participants in complying with conditions of the court.13-15
Psychiatrists and other mental health clinicians should be aware of MHCs, which are a type of PSC that provides for the community supervision of individuals with mental illness. Mental health courts vary in terms of eligibility criteria. Some accept individuals who merely report a history of mental illness, whereas others have specific diagnostic requirements.16 Some accept individuals accused of minor violations such as ordinance violations or misdemeanor offenses, while others accept individuals accused of felonies. Like other PSCs, participation in an MHC is voluntary, and most require a participant to enter a guilty plea upon entry.17 Participants may choose to enter an MHC to avoid prison time or to reduce or expunge charges after completing the program. Many MHCs also assign a probation officer to follow the participant in the community, similar to a standard probation model. Participants are usually expected to engage in psychiatric treatment, including psychotherapy, substance abuse counseling, medication management, and other services. If they do not comply with these conditions, they face sanctions that could include jail “shock” time, enhanced supervision, or an increase in psychiatric services.
Outpatient mental health professionals play an integral role in MHCs. Depending on the model, he/she may be asked to communicate treatment recommendations, attend weekly meetings at the court, and provide suggestions for interventions when the participant relapses, recidivates, and/or decompensates psychiatrically. This collaborative model can work well and allow the clinician unique opportunities to educate the court and advocate for his/her patient. However, clinicians who participate in an MHC need to remain aware of the potential to become a de facto probation officer, and need to maintain appropriate boundaries and roles. They should ensure that the patient provides initial and ongoing consent for them to communicate with the court, and share their programmatic recommendations with the patient to preserve the therapeutic alliance.
Continue to: Challenges upon re-entering the community
Challenges upon re-entering the community
Individuals recently released from jail or prison face unique challenges when re-entering the community. An individual who has been incarcerated, particularly for months to years, has likely lost his/her job, housing, health insurance, and access to primary supports. People with mental illness with a history of incarceration have higher rates of homelessness, substance use disorders, and unemployment than those with no history of incarceration.7,18 For individuals with mental illness, these additional stressors lead to further psychiatric decompensation, recidivism, and overutilization of emergency and crisis services upon release from prison or jail. The loss of health insurance presents great challenges: when someone is incarcerated, his/her Medicaid is suspended or terminated.19 This can happen at any point during incarceration. In states that terminate rather than suspend Medicaid, former prisoners face even longer waits to re-establish access to needed health care.
The period immediately after release is a critical time for individuals to be linked with substance and mental health treatment. Binswanger et al20 found former prisoners were at highest risk of mortality in the 2 weeks following release from prison; the highest rates of death were from drug overdose, cardiovascular disease, homicide, and suicide. A subsequent study found that women were at increased risk of drug overdose and opioid-related deaths.21 One explanation for the increase in drug-related deaths is the loss of physiologic tolerance while incarcerated; however, a lack of treatment while incarcerated, high levels of stress upon re-entry, and poor linkage to aftercare also may be contributing factors. Among prisoners recently released from New York City jails, Lim et al22 found that those with a history of homelessness and previous incarceration had the highest rates of drug-related deaths and homicides in the first 2 weeks after release. Non-Hispanic white men had the highest risk of drug-related deaths and suicides. While the risk of death is greatest immediately after release, former prisoners face increased mortality from multiple causes for multiple years after release.20-22
Clinicians who work with recently released prisoners should be aware of these individuals’ risks and actively work with them and other members of the mental health team to ensure these patients have access to social services, employment training, housing, and substance use resources, including medication-assisted treatment. Patients with SMI should be considered for more intensive services, such as assertive community treatment (ACT) or even forensic ACT (FACT) services, given that FACTs have a modest impact in reducing recidivism.23
Knowing whether the patient is on probation or parole and the terms of his/her supervision can also be useful in creating and executing a collaborative treatment plan. The clinician can assist the patient in meeting conditions of probation/parole such as:
- creating a stable home plan with a permanent address
- planning routine check-ins with probation/parole officers, and
- keeping documentation of ongoing mental health and substance use treatment.
Being aware of other terms of supervision, such as abstaining from alcohol and drugs, or remaining in one’s jurisdiction, also can help the patient avoid technical violations and a return to jail or prison.
Continue to: How to best help patients on community supervision
How to best help patients on community supervision
There are some clinical recommendations when working with patients on community supervision. First, do not assume that someone who has been incarcerated has antisocial personality disorder. Behaviors primarily related to seeking or using drugs or survival-type crimes should not be considered “antisocial” without additional evidence of pervasive and persistent conduct demonstrating impulsivity, lack of empathy, dishonesty, or repeated disregard for social norms and others’ rights. To meet criteria for antisocial personality disorder, these behaviors must have begun during childhood or adolescence.
If a patient does meet criteria for antisocial personality disorder, remember that he/she may also have a psychotic, mood, substance use, or other disorder that could lead to a greater likelihood of violence, recidivism, or other poor outcomes if left untreated. Treating any co-occurring disorders could enhance the patient’s engagement with treatment. There is some evidence that certain psychotropic medications, su
In addition to promoting patients’ mental health, such efforts can prevent re-arrest and re-incarceration and make a lasting positive impact on patients’ lives.
CASE CONTINUED
Mr. A signs a release-of-information form and you call his parole officer. His parole officer states that he would like to speak with you every few months to check on Mr. A’s treatment adherence. Within a few months, you transition Mr. A from an oral antipsychotic medication to a long-acting injectable antipsychotic medication to manage his psychotic disorder. He presents on time each month to your clinic to receive the injection.
Five months later, Mr. A receives 2 weeks of “shock time” at the local county jail for “dropping a dirty urine” that was positive for cannabinoids at a meeting with his parole officer. During his time in jail, he receives no treatment and he misses his monthly long-acting injectable dose.
Continue to: Upon release...
Upon release, he demonstrates the recurrence of some mild persecutory fears and hallucinations, but you resume him on his prior treatment regimen, and he recovers.
You encourage the parole officer to notify you if Mr. A violates parole and is incarcerated so that you can speak with clinicians in the jail to ensure that Mr. A remains adequately treated while incarcerated.
In the coming years, you continue to work with Mr. A and his parole officer to manage his mental health condition and to navigate his parole requirements in order to reduce his risk of relapse and recidivism. After Mr. A completes his time on parole, you continue to see him for outpatient follow-up.
Bottom Line
Clinicians may provide psychiatric care to probationers and parolees in traditional outpatient settings or in collaboration with a mental health court (MHC) or forensic assertive community treatment team. It is crucial to be aware of the legal expectations of individuals on community supervision, as well as the unique mental health risks and challenges they face. You can help reduce probationers’ and parolees’ risk of relapse and recidivism and support their recovery in the community by engaging in collaborative treatment planning involving the patient, the court, and/or MHCs.
Related Resources
- Lamb HR. Weinberger LE. Understanding and treating offenders with serious mental illness in public sector mental health. Behav Sci Law. 2017;35(4):303-318.
- Worcester S. Mental illness and the criminal justice system: Reducing the risks. Clinical Psychiatry News. https://www.mdedge.com/psychiatry/article/173208/schizophrenia-other-psychotic-disorders/mental-illness-and-criminal. Published August 22, 2018.
Mr. A, age 35, presents to your outpatient community mental health practice. He has a history of psychosis that began in his late teens. Since then, his symptoms have included derogatory auditory hallucinations, a recurrent persecutory delusion that governmental agencies are tracking his movements, and intermittent disorganized speech. At age 30, Mr. A assaulted a stranger out of fear that the individual was a government agent. He was arrested and experienced a severe psychotic decompensation while awaiting trial. He was found incompetent to stand trial and sent to a state hospital for restoration.
After 6 months of treatment and observation, Mr. A was deemed competent to proceed and returned to jail. He was subsequently convicted of assault and sentenced to 7 years in prison. While in prison, he received regular mental health care with infrequent recurrence of minor psychotic symptoms. He was released on parole due to his good behavior, but as part of his conditions of parole, he was mandated to follow up with an outpatient mental health clinician.
After telling you the story of how he ended up in your office, Mr. A says he needs you to speak regularly with his parole officer to verify his attendance at appointments and to discuss any mental health concerns you may have. Since you have not worked with a patient on parole before, your mind is full of questions: What are the expectations regarding your communication with his parole officer? Could Mr. A return to prison if you express concerns about his mental health? What can you do to improve his chances of success in the community?
Given the high rates of mental illness among individuals incarcerated in the United States, it shouldn’t be surprising that there are similarly high rates of mental illness among those on supervised release from jails and prisons. Clinicians who work with patients on community release need to understand basic concepts related to probation and parole, and how to promote patients’ stability in the community to reduce recidivism and re-incarceration. The court may require individuals on probation or parole to adhere to certain conditions of release, which could include seeing a psychiatrist or psychotherapist, participating in substance abuse treatment, and/or taking psychotropic medication. The court usually closely monitors the probationer or parolee’s adherence, and noncompliance can be grounds for probation or parole violation and revocation.
This article reviews the concepts of probation and parole (Box1,2), describes the prevalence of mental illness among probationers and parolees, and discusses the unique challenges and opportunities psychiatrists and other mental health professionals face when working with individuals on community supervision.
Box
The US Bureau of Justice Statistics (BJS) defines probation as a “court-ordered period of correctional supervision in the community, generally as an alternative to incarceration.” Probation allows individuals to be released from jail to community supervision, with the potential for dismissal or lowering of charges if they adhere to the conditions of probation. Conditions of probation may include participating in substance abuse or mental health treatment programs, abstaining from drugs and alcohol, and avoiding contact with known felons. Failure to comply with conditions of probation can lead to re-incarceration and probation revocation.1 If probation is revoked, a probationer may be sentenced, potentially to prison, depending on the severity of the original offense.2
The BJS defines parole as “a period of conditional supervised release in the community following a term in state or federal prison.”2 Parole allows for the community supervision of individuals who have already been convicted of and sentenced to prison for a crime. Individuals may be released on parole if they demonstrate good behavior while incarcerated. Similar to probationers, parolees must adhere to the conditions of parole, and violation of these may lead to re-incarceration.1
As of December 31, 2016, there were more than 4.5 million adults on community supervision in the United States, representing 1 out of every 55 adults in the US population. Individuals on probation accounted for 81% of adults on community supervision. The number of people on community supervision has dropped continuously over the last decade, a trend driven by 2% annual decreases in the probation population. In contrast, the parolee population has continued to grow over time and was approximately 900,000 individuals at the end of 2016.2
Mental illness among probationers and parolees
Research on mental illness in people involved in the criminal justice system has largely focused on those who are incarcerated. Studies have documented high rates of severe mental illness (SMI), such as schizophrenia and bipolar disorder, among those who are incarcerated; some estimate the rates to be 3 times as high as those of community samples.3,4 In addition to SMI, substance use disorders and personality disorders (in particular, antisocial personality disorder) are common among people who are incarcerated.5,6
Comparatively little is known about mental illness among probationers and parolees, although presumably there would be a similarly high prevalence of SMI, substance use disorders, and other psychiatric disorders among this population. A 1997 Bureau of Justice Statistics (BJS) survey of approximately 3.4 million probationers found that 13.8% self-reported a mental or emotional condition and 8.2% self-reported a history of an “overnight stay in a mental hospital.”7 The BJS estimated that there were approximately 550,000 probationers with mental illness in the United States. The study’s author noted that probationers with mental illness were more likely to have a history of prior offenses and more likely to be violent recidivists. In terms of substance use, compared with other probationers, those with mental illness were more likely to report using drugs in the month before their most recent offense and at the time of the offense.7
Continue to: More recent research...
More recent research, although limited, has shed some light on the role of mental health services for individuals on probation and parole. In 2009, Crilly et al8 reported that 23% of probationers reported accessing mental health services within the past year. Other studies have found that probationer and parolee engagement in mental health care reduces the risk of recidivism.9,10 A 2011 study evaluated 100 individuals on probation and parole in 2 counties in a southeastern state. The authors found that 75% of participants reported that they needed counseling for a mental health concern in the past year, but that only approximately 30% of them actually sought help. Individuals reporting higher levels of posttraumatic stress disorder symptomatology or greater drug use before being on probation or parole were more likely to seek counseling in the past year.11
An alternative: Problem-solving courts
Problem-solving courts (PSCs) offer an alternative to standard probation and/or sentencing. Problem-solving courts are founded on the concept of therapeutic jurisprudence, which seeks to change “the behavior of litigants and [ensure] the future well-being of communities.”12 Types of PSCs include drug court (the most common type in the United States), domestic violence court, veterans court, and mental health court (MHC), among others.
An individual may choose a PSC over standard probation because participants usually receive more assistance in obtaining treatment and closer supervision with an emphasis on rehabilitation rather than incapacitation or retribution. The success of PSCs relies heavily on the judge, as he/she plays a pivotal role in developing relationships with the participants, considering therapeutic alternatives to “bad” behaviors, determining sanctions, and relying on community mental health partners to assist participants in complying with conditions of the court.13-15
Psychiatrists and other mental health clinicians should be aware of MHCs, which are a type of PSC that provides for the community supervision of individuals with mental illness. Mental health courts vary in terms of eligibility criteria. Some accept individuals who merely report a history of mental illness, whereas others have specific diagnostic requirements.16 Some accept individuals accused of minor violations such as ordinance violations or misdemeanor offenses, while others accept individuals accused of felonies. Like other PSCs, participation in an MHC is voluntary, and most require a participant to enter a guilty plea upon entry.17 Participants may choose to enter an MHC to avoid prison time or to reduce or expunge charges after completing the program. Many MHCs also assign a probation officer to follow the participant in the community, similar to a standard probation model. Participants are usually expected to engage in psychiatric treatment, including psychotherapy, substance abuse counseling, medication management, and other services. If they do not comply with these conditions, they face sanctions that could include jail “shock” time, enhanced supervision, or an increase in psychiatric services.
Outpatient mental health professionals play an integral role in MHCs. Depending on the model, he/she may be asked to communicate treatment recommendations, attend weekly meetings at the court, and provide suggestions for interventions when the participant relapses, recidivates, and/or decompensates psychiatrically. This collaborative model can work well and allow the clinician unique opportunities to educate the court and advocate for his/her patient. However, clinicians who participate in an MHC need to remain aware of the potential to become a de facto probation officer, and need to maintain appropriate boundaries and roles. They should ensure that the patient provides initial and ongoing consent for them to communicate with the court, and share their programmatic recommendations with the patient to preserve the therapeutic alliance.
Continue to: Challenges upon re-entering the community
Challenges upon re-entering the community
Individuals recently released from jail or prison face unique challenges when re-entering the community. An individual who has been incarcerated, particularly for months to years, has likely lost his/her job, housing, health insurance, and access to primary supports. People with mental illness with a history of incarceration have higher rates of homelessness, substance use disorders, and unemployment than those with no history of incarceration.7,18 For individuals with mental illness, these additional stressors lead to further psychiatric decompensation, recidivism, and overutilization of emergency and crisis services upon release from prison or jail. The loss of health insurance presents great challenges: when someone is incarcerated, his/her Medicaid is suspended or terminated.19 This can happen at any point during incarceration. In states that terminate rather than suspend Medicaid, former prisoners face even longer waits to re-establish access to needed health care.
The period immediately after release is a critical time for individuals to be linked with substance and mental health treatment. Binswanger et al20 found former prisoners were at highest risk of mortality in the 2 weeks following release from prison; the highest rates of death were from drug overdose, cardiovascular disease, homicide, and suicide. A subsequent study found that women were at increased risk of drug overdose and opioid-related deaths.21 One explanation for the increase in drug-related deaths is the loss of physiologic tolerance while incarcerated; however, a lack of treatment while incarcerated, high levels of stress upon re-entry, and poor linkage to aftercare also may be contributing factors. Among prisoners recently released from New York City jails, Lim et al22 found that those with a history of homelessness and previous incarceration had the highest rates of drug-related deaths and homicides in the first 2 weeks after release. Non-Hispanic white men had the highest risk of drug-related deaths and suicides. While the risk of death is greatest immediately after release, former prisoners face increased mortality from multiple causes for multiple years after release.20-22
Clinicians who work with recently released prisoners should be aware of these individuals’ risks and actively work with them and other members of the mental health team to ensure these patients have access to social services, employment training, housing, and substance use resources, including medication-assisted treatment. Patients with SMI should be considered for more intensive services, such as assertive community treatment (ACT) or even forensic ACT (FACT) services, given that FACTs have a modest impact in reducing recidivism.23
Knowing whether the patient is on probation or parole and the terms of his/her supervision can also be useful in creating and executing a collaborative treatment plan. The clinician can assist the patient in meeting conditions of probation/parole such as:
- creating a stable home plan with a permanent address
- planning routine check-ins with probation/parole officers, and
- keeping documentation of ongoing mental health and substance use treatment.
Being aware of other terms of supervision, such as abstaining from alcohol and drugs, or remaining in one’s jurisdiction, also can help the patient avoid technical violations and a return to jail or prison.
Continue to: How to best help patients on community supervision
How to best help patients on community supervision
There are some clinical recommendations when working with patients on community supervision. First, do not assume that someone who has been incarcerated has antisocial personality disorder. Behaviors primarily related to seeking or using drugs or survival-type crimes should not be considered “antisocial” without additional evidence of pervasive and persistent conduct demonstrating impulsivity, lack of empathy, dishonesty, or repeated disregard for social norms and others’ rights. To meet criteria for antisocial personality disorder, these behaviors must have begun during childhood or adolescence.
If a patient does meet criteria for antisocial personality disorder, remember that he/she may also have a psychotic, mood, substance use, or other disorder that could lead to a greater likelihood of violence, recidivism, or other poor outcomes if left untreated. Treating any co-occurring disorders could enhance the patient’s engagement with treatment. There is some evidence that certain psychotropic medications, su
In addition to promoting patients’ mental health, such efforts can prevent re-arrest and re-incarceration and make a lasting positive impact on patients’ lives.
CASE CONTINUED
Mr. A signs a release-of-information form and you call his parole officer. His parole officer states that he would like to speak with you every few months to check on Mr. A’s treatment adherence. Within a few months, you transition Mr. A from an oral antipsychotic medication to a long-acting injectable antipsychotic medication to manage his psychotic disorder. He presents on time each month to your clinic to receive the injection.
Five months later, Mr. A receives 2 weeks of “shock time” at the local county jail for “dropping a dirty urine” that was positive for cannabinoids at a meeting with his parole officer. During his time in jail, he receives no treatment and he misses his monthly long-acting injectable dose.
Continue to: Upon release...
Upon release, he demonstrates the recurrence of some mild persecutory fears and hallucinations, but you resume him on his prior treatment regimen, and he recovers.
You encourage the parole officer to notify you if Mr. A violates parole and is incarcerated so that you can speak with clinicians in the jail to ensure that Mr. A remains adequately treated while incarcerated.
In the coming years, you continue to work with Mr. A and his parole officer to manage his mental health condition and to navigate his parole requirements in order to reduce his risk of relapse and recidivism. After Mr. A completes his time on parole, you continue to see him for outpatient follow-up.
Bottom Line
Clinicians may provide psychiatric care to probationers and parolees in traditional outpatient settings or in collaboration with a mental health court (MHC) or forensic assertive community treatment team. It is crucial to be aware of the legal expectations of individuals on community supervision, as well as the unique mental health risks and challenges they face. You can help reduce probationers’ and parolees’ risk of relapse and recidivism and support their recovery in the community by engaging in collaborative treatment planning involving the patient, the court, and/or MHCs.
Related Resources
- Lamb HR. Weinberger LE. Understanding and treating offenders with serious mental illness in public sector mental health. Behav Sci Law. 2017;35(4):303-318.
- Worcester S. Mental illness and the criminal justice system: Reducing the risks. Clinical Psychiatry News. https://www.mdedge.com/psychiatry/article/173208/schizophrenia-other-psychotic-disorders/mental-illness-and-criminal. Published August 22, 2018.
1. Bureau of Justice Statistics. FAQ detail: What is the difference between probation and parole? U.S. Department of Justice. https://www.bjs.gov/index.cfm?ty=qa&iid=324. Accessed November 17, 2018.
2. Kaeble D. Probation and parole in the United States, 2016. U.S. Department of Justice. https://www.bjs.gov/content/pub/pdf/ppus16.pdf. Published April 2018. Accessed April 23, 2019.
3. Kessler RC, Chiu WT, Demler O, et al. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):617-627.
4. Diamond, P.M., et al., The prevalence of mental illness in prison. Adm Policy Ment Health. 2001;29(1):21-40.
5. MacDonald R, Kaba F, Rosner Z, et al. The Rikers Island hot spotters: defining the needs of the most frequently incarcerated. Am J Public Health. 2015;105(11):2262-2268.
6. Trestman RL, Ford J, Zhang W, et al. Current and lifetime psychiatric illness among inmates not identified as acutely mentally ill at intake in Connecticut’s jails. J Am Acad Psychiatry Law. 2007;35(4):490-500.
7. Ditton PM. Bureau of Justice Statistics special report: mental health and treatment of inmates and probationers. U.S. Department of Justice. https://www.bjs.gov/content/pub/pdf/mhtip.pdf. Published July 1999. Accessed April 24, 2019.
8. Crilly JF, Caine ED, Lamberti JS, et al. Mental health services use and symptom prevalence in a cohort of adults on probation. Psychiatr Serv. 2009;60(4):542-544.
9. Herinckx HA, Swart SC, Ama SM, et al. Rearrest and linkage to mental health services among clients of the Clark County mental health court program. Psychiatr Serv. 2005;56(7):853-857.
10. Solomon P, Draine J, Marcus SC. Predicting incarceration of clients of a psychiatric probation and parole service. Psychiatr Serv. 2002;53(1):50-56.
11. Owens GP, Rogers SM, Whitesell AA. Use of mental health services and barriers to care for individuals on probation or parole. J Offender Rehabil. 2011;50(1):35-47.
12. Berman G, Feinblatt J. Problem‐solving courts: a brief primer. Law and Policy. 2001;23(2):126.
13. The Council of State Governments Justice Center. Mental health courts: a guide to research-informed policy and practice. U.S. Department of Justice. https://www.bja.gov/Publications/CSG_MHC_Research.pdf. Published 2009. Accessed November 22, 2018.
14. Landess J, Holoyda B. Mental health courts and forensic assertive community treatment teams as correctional diversion programs. Behav Sci Law. 2017;35(5-6):501-511.
15. Sammon KC. Therapeutic jurisprudence: an examination of problem‐solving justice in New York. Journal of Civil Rights and Economic Development. 2008;23:923.
16. Sarteschi CM, Vaughn MG, Kim, K. Assessing the effectiveness of mental health courts: a quantitative review. Journal of Criminal Justice. 2011;39(1):12-20.
17. Strong SM, Rantala RR. Census of problem-solving courts, 2012. U.S. Department of Justice, Bureau of Justice Assistance. http://www.bjs.gov/content/pub/pdf/cpsc12.pdf. Revised October 12, 2016. Accessed April 24, 2019.
18. McGuire JF, Rosenheck RA. Criminal history as a prognostic indicator in the treatment of homeless people with severe mental illness. Psychiatr Serv. 2004;55(1):42-48.
19. Families USA. Medicaid suspension policies for incarcerated people: a 50-state map. Families USA. https://familiesusa.org/product/medicaid-suspension-policies-incarcerated-people-50-state-map. Published July 2016. Accessed December 7, 2018.
20. Binswanger IA, Stern MF, Deyo RA, et al. Release from prison—a high risk of death for former inmates. N Engl J Med. 2007;356(2):157-165.
21. Binswanger IA, Blatchford PJ, Mueller SR, et al. Mortality after prison release: opioid overdose and other causes of death, risk factors, and time trends from 1999 to 2009. Ann Intern Med. 2013;159(9):592-600.
22. Lim S, Seligson AL, Parvez FM, et al. Risks of drug-related death, suicide, and homicide during the immediate post-release period among people released from New York City Jails, 2001-2005. Am J Epidemiol. 2012;175(6):519-526.
23. Cusack KJ, Morrissey JP, Cuddeback GS, et al. Criminal justice involvement, behavioral health service use, and costs of forensic assertive community treatment: a randomized trial. Community Ment Health J. 2010;46(4):356-363.
24. Felthous AR, Stanford MS. A proposed algorithm for the pharmacotherapy of impulsive aggression. J Am Acad Psychiatry Law. 2015:43(4);456-467.
1. Bureau of Justice Statistics. FAQ detail: What is the difference between probation and parole? U.S. Department of Justice. https://www.bjs.gov/index.cfm?ty=qa&iid=324. Accessed November 17, 2018.
2. Kaeble D. Probation and parole in the United States, 2016. U.S. Department of Justice. https://www.bjs.gov/content/pub/pdf/ppus16.pdf. Published April 2018. Accessed April 23, 2019.
3. Kessler RC, Chiu WT, Demler O, et al. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):617-627.
4. Diamond, P.M., et al., The prevalence of mental illness in prison. Adm Policy Ment Health. 2001;29(1):21-40.
5. MacDonald R, Kaba F, Rosner Z, et al. The Rikers Island hot spotters: defining the needs of the most frequently incarcerated. Am J Public Health. 2015;105(11):2262-2268.
6. Trestman RL, Ford J, Zhang W, et al. Current and lifetime psychiatric illness among inmates not identified as acutely mentally ill at intake in Connecticut’s jails. J Am Acad Psychiatry Law. 2007;35(4):490-500.
7. Ditton PM. Bureau of Justice Statistics special report: mental health and treatment of inmates and probationers. U.S. Department of Justice. https://www.bjs.gov/content/pub/pdf/mhtip.pdf. Published July 1999. Accessed April 24, 2019.
8. Crilly JF, Caine ED, Lamberti JS, et al. Mental health services use and symptom prevalence in a cohort of adults on probation. Psychiatr Serv. 2009;60(4):542-544.
9. Herinckx HA, Swart SC, Ama SM, et al. Rearrest and linkage to mental health services among clients of the Clark County mental health court program. Psychiatr Serv. 2005;56(7):853-857.
10. Solomon P, Draine J, Marcus SC. Predicting incarceration of clients of a psychiatric probation and parole service. Psychiatr Serv. 2002;53(1):50-56.
11. Owens GP, Rogers SM, Whitesell AA. Use of mental health services and barriers to care for individuals on probation or parole. J Offender Rehabil. 2011;50(1):35-47.
12. Berman G, Feinblatt J. Problem‐solving courts: a brief primer. Law and Policy. 2001;23(2):126.
13. The Council of State Governments Justice Center. Mental health courts: a guide to research-informed policy and practice. U.S. Department of Justice. https://www.bja.gov/Publications/CSG_MHC_Research.pdf. Published 2009. Accessed November 22, 2018.
14. Landess J, Holoyda B. Mental health courts and forensic assertive community treatment teams as correctional diversion programs. Behav Sci Law. 2017;35(5-6):501-511.
15. Sammon KC. Therapeutic jurisprudence: an examination of problem‐solving justice in New York. Journal of Civil Rights and Economic Development. 2008;23:923.
16. Sarteschi CM, Vaughn MG, Kim, K. Assessing the effectiveness of mental health courts: a quantitative review. Journal of Criminal Justice. 2011;39(1):12-20.
17. Strong SM, Rantala RR. Census of problem-solving courts, 2012. U.S. Department of Justice, Bureau of Justice Assistance. http://www.bjs.gov/content/pub/pdf/cpsc12.pdf. Revised October 12, 2016. Accessed April 24, 2019.
18. McGuire JF, Rosenheck RA. Criminal history as a prognostic indicator in the treatment of homeless people with severe mental illness. Psychiatr Serv. 2004;55(1):42-48.
19. Families USA. Medicaid suspension policies for incarcerated people: a 50-state map. Families USA. https://familiesusa.org/product/medicaid-suspension-policies-incarcerated-people-50-state-map. Published July 2016. Accessed December 7, 2018.
20. Binswanger IA, Stern MF, Deyo RA, et al. Release from prison—a high risk of death for former inmates. N Engl J Med. 2007;356(2):157-165.
21. Binswanger IA, Blatchford PJ, Mueller SR, et al. Mortality after prison release: opioid overdose and other causes of death, risk factors, and time trends from 1999 to 2009. Ann Intern Med. 2013;159(9):592-600.
22. Lim S, Seligson AL, Parvez FM, et al. Risks of drug-related death, suicide, and homicide during the immediate post-release period among people released from New York City Jails, 2001-2005. Am J Epidemiol. 2012;175(6):519-526.
23. Cusack KJ, Morrissey JP, Cuddeback GS, et al. Criminal justice involvement, behavioral health service use, and costs of forensic assertive community treatment: a randomized trial. Community Ment Health J. 2010;46(4):356-363.
24. Felthous AR, Stanford MS. A proposed algorithm for the pharmacotherapy of impulsive aggression. J Am Acad Psychiatry Law. 2015:43(4);456-467.
Agitation in children and adolescents: Diagnostic and treatment considerations
Managing agitation—verbal and/or motor restlessness that often is accompanied by irritability and a predisposition to aggression or violence—can be challenging in any patient, but particularly so in children and adolescents. In the United States, the prevalence of children and adolescents presenting to an emergency department (ED) for treatment of psychiatric symptoms, including agitation, has been on the rise.1,2
Similar to the multitude of causes of fever, agitation among children and adolescents has many possible causes.3 Because agitation can pose a risk for harm to others and/or self, it is important to manage it proactively. Other than studies that focus on agitation in pediatric anesthesia, there is a dearth of studies examining agitation and its treatment in children and adolescents. There is also a scarcity of training in the management of acute agitation in children and adolescents. In a 2017 survey of pediatric hospitalists and consultation-liaison psychiatrists at 38 academic children’s hospitals in North America, approximately 60% of respondents indicated that they had received no training in the evaluation or management of pediatric acute agitation.4 In addition, approximately 54% of participants said they did not screen for risk factors for pediatric agitation, even though 84% encountered the condition at least once a month, and as often as weekly.4
This article reviews evidence on the causes and treatments of agitation in children and adolescents. For the purposes of this review, child refers to a patient age 6 to 12, and adolescent refers to a patient age 13 to 17.
Identifying the cause
Addressing the underlying cause of agitation is essential. It’s also important to manage acute agitation while the underlying cause is being investigated in a way that does not jeopardize the patient’s emotional or physical safety.
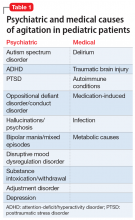
Agitation in children or teens can be due to psychiatric causes such as autism, attention-deficit/hyperactivity disorder (ADHD), or posttraumatic stress disorder (PTSD), or due to medical conditions such as delirium, traumatic brain injury, or other conditions (Table 1).
In a 2005 study of 194 children with agitation in a pediatric post-anesthesia care unit, pain (27%) and anxiety (25%) were found to be the most common causes of agitation.3 Anesthesia-related agitation was a less common cause (11%). Physiologic anomalies were found to be the underlying cause of agitation in only 3 children in this study, but were undiagnosed for a prolonged period in 2 of these 3 children, which highlights the importance of a thorough differential diagnosis in the management of agitation in children.3
Assessment of an agitated child should include a comprehensive history, physical exam, and laboratory testing as indicated. When a pediatric patient comes to the ED with a chief presentation of agitation, a thorough medical and psychiatric assessment should be performed. For patients with a history of psychiatric diagnoses, do not assume that the cause of agitation is psychiatric.
Continue to: Psychiatric causes
Psychiatric causes
Autism spectrum disorder. Children and teens with autism often feel overwhelmed due to transitions, changes, and/or sensory overload. This sensory overload may be in response to relatively subtle sensory stimuli, so it may not always be apparent to parents or others around them.
Research suggests that in general, the ability to cope effectively with emotions is difficult without optimal language development. Due to cognitive/language delays and a related lack of emotional attunement and limited skills in recognizing, expressing, or coping with emotions, difficult emotions in children and adolescents with autism can manifest as agitation.
Attention-deficit/hyperactivity disorder. Children with ADHD may be at a higher risk for agitation, in part due to poor impulse control and limited coping skills. In addition, chronic negative feedback (from parents, teachers, or both) may contribute to low self-esteem, mood symptoms, defiance, and/or other behavioral difficulties. In addition to standard pharmacotherapy for ADHD, treatment involves parent behavior modification training. Setting firm yet empathic limits, “picking battles,” and implementing a developmentally appropriate behavioral plan to manage disruptive behavior in children or adolescents with ADHD can go a long way in helping to prevent the emergence of agitation.
Posttraumatic stress disorder. In some young children, new-onset, unexplained agitation may be the only sign of abuse or trauma. Children who have undergone trauma tend to experience confusion and distress. This may manifest as agitation or aggression, or other symptoms such as increased anxiety or nightmares.5 Trauma may be in the form of witnessing violence (domestic or other); experiencing physical, sexual, and/or emotional abuse; or witnessing/experiencing other significant threats to the safety of self and/or loved ones. Re-establishing (or establishing) a sense of psychological and physical safety is paramount in such patients.6 Psychotherapy is the first-line modality of treatment in children and adolescents with PTSD.6 In general, there is a scarcity of research on medication treatments for PTSD symptoms among children and adolescents.6
Oppositional defiant disorder/conduct disorder. Oppositional defiant disorder (ODD) can be comorbid with ADHD. The diagnosis of ODD requires a pervasive pattern of anger, defiance, vindictiveness, and hostility, particularly towards authority figures. However, these symptoms need to be differentiated from the normal range of childhood behavior. Occasionally, children learn to cope maladaptively through disruptive behavior or agitation. Although a parent or caregiver may see this behavior as intentionally malevolent, in a child with limited coping skills (whether due to young age, developmental/cognitive/language/learning delays, or social communication deficits) or one who has witnessed frequent agitation or aggression in the family environment, agitation and disruptive behavior may be a maladaptive form of coping. Thus, diligence needs to be exercised in the diagnosis of ODD and in understanding the psychosocial factors affecting the child, particularly because impulsiveness and uncooperativeness on their own have been found to be linked to greater likelihood of prescription of psychotropic medications from multiple classes.7 Family-based interventions, particularly parent training, family therapy, and age-appropriate child skills training, are of prime importance in managing this condition.8 Research shows that a shortage of resources, system issues, and cultural roadblocks in implementing family-based psychosocial interventions also can contribute to the increased use of psychotropic medications for aggression in children and teens with ODD, conduct disorder, or ADHD.8 The astute clinician needs to be cognizant of this before prescribing.
Continue to: Hallucinations/psychosis
Hallucinations/psychosis. Hallucinations (whether from psychiatric or medical causes) are significantly associated with agitation.9 In particular, auditory command hallucinations have been linked to agitation. Command hallucinations in children and adolescents may be secondary to early-onset schizophrenia; however, this diagnosis is rare.10 Hallucinations can also be an adverse effect of amphetamine-based stimulant medications in children and adolescents. Visual hallucinations are most often a sign of an underlying medical disorder such as delirium, occipital lobe mass/infection, or drug intoxication or withdrawal. Hallucinations need to be distinguished from the normal, imaginative play of a young child.10
Bipolar mania. In adults, bipolar disorder is a primary psychiatric cause of agitation. In children and adolescents, the diagnosis of bipolar disorder can be complex and requires careful and nuanced history-taking. The risks of agitation are greater with bipolar disorder than with unipolar depression.11,12
Disruptive mood dysregulation disorder. Prior to DSM-5, many children and adolescents with chronic, non-episodic irritability and severe outbursts out of proportion to the situation or stimuli were given a diagnosis of bipolar disorder. These symptoms, in combination with other symptoms, are now considered part of disruptive mood dysregulation disorder when severe outbursts in a child or adolescent occur 3 to 4 times a week consistently, for at least 1 year. The diagnosis of disruptive mood dysregulation disorder requires ruling out other psychiatric and medical conditions, particularly ADHD.13
Substance intoxication/withdrawal. Intoxication or withdrawal from substances such as alcohol, stimulant medications, opioids, methamphetamines, and other agents can lead to agitation. This is more likely to occur among adolescents than children.14
Adjustment disorder. Parental divorce, especially if it is conflictual, or other life stressors, such as experiencing a move or frequent moves, may contribute to the development of agitation in children and adolescents.
Continue to: Depression
Depression. In children and adolescents, depression can manifest as anger or irritability, and occasionally as agitation.
Medical causes
Delirium. Refractory agitation is often a manifestation of delirium in children and adolescents.15 If unrecognized and untreated, delirium can be fatal.16 Therefore, it is imperative that clinicians routinely assess for delirium in any patient who presents with agitation.
Because a patient with delirium often presents with agitation and visual or auditory hallucinations, the medical team may tend to assume these symptoms are secondary to a psychiatric disorder. In this case, the role of the consultation-liaison psychiatrist is critical for guiding the medical team, particularly to continue a thorough exploration of underlying causes while avoiding polypharmacy. Noise, bright lights, frequent changes in nursing staff or caregivers, anticholinergic or benzodiazepine medications, and frequent changes in schedules should be avoided to prevent delirium from occurring or getting worse.17 A multidisciplinary team approach is key in identifying the underlying cause and managing delirium in pediatric patients.
Traumatic brain injury. Agitation may be a presenting symptom in youth with traumatic brain injury (TBI).18 Agitation may present often in the acute recovery phase.19 There is limited evidence on the efficacy and safety of pharmacotherapy for agitation in pediatric patients with TBI.18
Autoimmune conditions. In a study of 27 patients with
Continue to: Medication-induced/iatrogenic
Medication-induced/iatrogenic. Agitation can be an adverse effect of medications such as amantadine (often used for TBI),18 atypical antipsychotics,21 selective serotonin reuptake inhibitors, and serotonin-norepinephrine reuptake inhibitors.
Infection. Agitation can be a result of encephalitis, meningitis, or other infectious processes.22
Metabolic conditions. Hepatic or renal failure, diabetic ketoacidosis, and thyroid toxicosis may cause agitation in children or adolescents.22
Start with nonpharmacologic interventions
Few studies have examined de-escalation techniques in agitated children and adolescents. However, verbal de-escalation is generally viewed as the first-line technique for managing agitation in children and adolescents. When feasible, teaching and modeling developmentally appropriate stress management skills for children and teens can be a beneficial preventative strategy to reduce the incidence and worsening of agitation.23
Clinicians should refrain from using coercion.24 Coercion could harm the therapeutic alliance, thereby impeding assessment of the underlying causes of agitation, and can be particularly harmful for patients who have a history of trauma or abuse. Even in pediatric patients with no such history, coercion is discouraged due to its punitive connotations and potential to adversely impact a vulnerable child or teen.
Continue to: Establishing a therapeutic rapport...
Establishing a therapeutic rapport with the patient, when feasible, can facilitate smoother de-escalation by offering the patient an outlet to air his/her frustrations and emotions, and by helping the patient feel understood.24 To facilitate this, ensure that the patient’s basic comforts and needs are met, such as access to a warm bed, food, and safety.25
The psychiatrist’s role is to help uncover and address the underlying reason for the patient’s agony or distress. Once the child or adolescent has calmed, explore potential triggers or causes of the agitation.
There has been a significant move away from the use of restraints for managing agitation in children and adolescents.26 Restraints have a psychologically traumatizing effect,27 and have been linked to life-threatening injuries and death in children.24
Pharmacotherapy: Proceed with caution
There are no FDA-approved medications for the treatment of agitation in the general pediatric population, and any medication use in this population is off-label. There is also a dearth of research examining the safety and efficacy of using psychotropic medications for agitation in pediatric patients. Because children and adolescents are more susceptible to adverse effects and risks associated with the use of psychotropic medications, special caution is warranted. In general, pharmacologic interventions are not recommended without the use of psychotherapy-based modalities.
In the past, the aim of using medications to treat patients with agitation was to put the patient to sleep.25 This practice did not help clinicians to assess for underlying causes, and was often accompanied by a greater risk of adverse effects and reactions.24 Therefore, the goal of medication treatment for agitation is to help calm the patient instead of inducing sleep.25
Continue to: Pharmacotherapy should...
Pharmacotherapy should be used only when behavioral interventions have been unsuccessful. Key considerations for using psychotropic medications to address agitation in children and adolescents are summarized in Table 2.25
Antipsychotics, particularly second-generation antipsychotics (SGAs), have been commonly used to manage acute agitation in children and adolescents, and there has been an upswing in the use of these medications in the United States in the last several years.28 Research indicates that males, children and adolescents in foster care, and those with Medicaid have been the more frequent youth recipients of SGAs.29 Of particular concern is the prevalence of antipsychotic use among children younger than age 6. In the last few decades, there has been an increase in the prescription of antipsychotics for children younger than age 6, particularly for disruptive behavior and aggression.30 In a study of preschool-age Medicaid patients in Kentucky, 70,777 prescriptions for SGAs were given to 6,915 children <6 years of age; 73% of these prescriptions were for male patients.30 Because there is a lack of controlled studies examining the safety and efficacy of SGAs among children and adolescents, especially with long-term use, further research is needed and caution is warranted.28
The FDA has approved
Externalizing disorders among children and adolescents tend to get treated with antipsychotics.28 A Canadian study examining records of 6,916 children found that most children who had been prescribed risperidone received it for ADHD or conduct disorder, and most patients had not received laboratory testing for monitoring the antipsychotic medication they were taking.31 In a 2018 study examining medical records of 120 pediatric patients who presented to an ED in British Columbia with agitation, antipsychotics were the most commonly used medications for patients with autism spectrum disorder; most patients received at least 1 dose.14
For children and adolescents with agitation or aggression who were admitted to inpatient units, IM
Continue to: In case reports...
In case reports, a combination of olanzapine with CNS-suppressing agents has resulted in death. Therefore, do not combine olanzapine with agents such as benzodiazepines.25 In a patient with a likely medical source of agitation, insufficient evidence exists to support the use of olanzapine, and additional research is needed.25
Low-dose haloperidol has been found to be effective for delirium-related agitation in pediatric studies.15 Before initiating an antipsychotic for any child or adolescent, review the patient’s family history for reports of early cardiac death and the patient’s own history of cardiac symptoms, palpitations, syncope, or prolonged QT interval. Monitor for QT prolongation. Among commonly used antipsychotics, the risk of QT prolongation is higher with IV administration of haloperidol and with ziprasidone. Studies show that compared with oral or IM
A few studies have found risperidone to be efficacious for treating ODD and conduct disorder; however, this use is off-label, and its considerable adverse effect and risk profile needs to be weighed against the potential benefit.8
Antipsychotic polypharmacy should be avoided because of the higher risk of adverse effects and interactions, and a lack of robust, controlled studies evaluating the safety of using antipsychotics for non-FDA-approved indications in children and adolescents.7 All patients who receive antipsychotics require monitoring for extrapyramidal symptoms, tardive dyskinesia, neuroleptic malignant syndrome, orthostatic hypotension, sedation, metabolic syndrome, and other potential adverse effects. Patients receiving risperidone need to have their prolactin levels monitored periodically, and their parents should be made aware of the potential for hyperprolactinemia and other adverse effects. Aripiprazole and quetiapine may increase the risk of suicidality.
Antiepileptics. A meta-analysis of 7 randomized controlled trials examining the use of antiepileptic medications (
Continue to: In a retrospective case series...
In a retrospective case series of 30 pediatric patients with autism spectrum disorder who were given oxcarbazepine, Douglas et al35 found that 47% of participants experienced significant improvement in irritability/agitation. However, 23% of patients reported significant adverse effects leading to discontinuation. Insufficient evidence exists for the safety and efficacy of oxcarbazepine in this population.35
Benzodiazepines. The use of benzodiazepines in pediatric patients has been associated with paradoxical disinhibition reactions, particularly in children with autism and other developmental or cognitive disabilities or delays.21 There is a lack of data on the safety and efficacy of long-term use of benzodiazepines in children, especially in light of these patients’ developing brains, the risk of cognitive impairment, and the potential for dependence with long-term use. Despite this, some studies show that the use of benzodiazepines is fairly common among pediatric patients who present to the ED with agitation.14 In a recent retrospective study, Kendrick et al14 found that among pediatric patients with agitation who were brought to the ED, benzodiazepines were the most commonly prescribed medications.
Other medications. Clonidine and
Diphenhydramine, in both oral and IM forms, has been used to treat agitation in children,32 but has also been associated with a paradoxical disinhibition reaction in pediatric patients21 and therefore should be used only sparingly and with caution. Diphenhydramine has anticholinergic properties, and may worsen delirium.15 Stimulant medications can help aggressive behavior in children and adolescents with ADHD.37
Bottom Line
Agitation among children and adolescents has many possible causes. A combination of a comprehensive assessment and evidence-based, judicious treatment interventions can help prevent and manage agitation in this vulnerable population.
Related Resources
- Baker M, Carlson GA. What do we really know about PRN use in agitated children with mental health conditions: a clinical review. Evid Based Ment Health. 2018;21(4):166-170.
- Gerson R, Malas N, Mroczkowski MM. Crisis in the emergency department: the evaluation and management of acute agitation in children and adolescents. Child Adolesc Psychiatr Clin N Am. 2018;27(3):367-386.
Drug Brand Names
Amantadine • Symmetrel
Aripiprazole • Abilify
Clonidine • Catapres
Guanfacine • Intuniv, Tenex
Haloperidol • Haldol
Lamotrigine • Lamictal
Levetiracetam • Keppra, Spritam
Olanzapine • Zyprexa
Oxcarbazepine • Trileptal
Quetiapine • Seroquel
Topiramate • Topamax
Risperidone • Risperdal
Valproate • Depakene
Ziprasidone • Geodon
1. Frosch E, Kelly P. Issues in pediatric psychiatric emergency care. In: Emergency psychiatry. Cambridge, UK: Cambridge University Press; 2011:185-199.
2. American College of Emergency Physicians. Pediatric mental health emergencies in the emergency department. https://www.acep.org/patient-care/policy-statements/pediatric-mental-health-emergencies-in-the-emergency-medical-services-system/. Revised September 2018. Accessed February 23, 2019.
3. Voepel-Lewis, T, Burke C, Hadden S, et al. Nurses’ diagnoses and treatment decisions regarding care of the agitated child. J Perianesth Nurs. 2005;20(4):239-248.
4. Malas N, Spital L, Fischer J, et al. National survey on pediatric acute agitation and behavioral escalation in academic inpatient pediatric care settings. Psychosomatics. 2017;58(3):299-306.
5. Famularo R, Kinscherff R, Fenton T. Symptom differences in acute and chronic presentation of childhood post-traumatic stress disorder. Child Abuse Negl. 1990;14(3):439-444.
6. Kaminer D, Seedat S, Stein DJ. Post-traumatic stress disorder in children. World Psychiatry. 2005;4(2):121-125.
7. Ninan A, Stewart SL, Theall LA, et al. Adverse effects of psychotropic medications in children: predictive factors. J Can Acad Child Adolesc Psychiatry. 2014;23(3):218-225.
8. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 2: antipsychotics and traditional mood stabilizers. Can J Psychiatry. 2015;60(2):52-61.
9. Vareilles D, Bréhin C, Cortey C, et al. Hallucinations: Etiological analysis of children admitted to a pediatric emergency department. Arch Pediatr. 2017;24(5):445-452.
10. Bartlett J. Childhood-onset schizophrenia: what do we really know? Health Psychol Behav Med. 2014;2(1):735-747.
11. Diler RS, Goldstein TR, Hafeman D, et al. Distinguishing bipolar depression from unipolar depression in youth: Preliminary findings. J Child Adolesc Psychopharmacol. 2017;27(4):310-319.
12. Dervic K, Garcia-Amador M, Sudol K, et al. Bipolar I and II versus unipolar depression: clinical differences and impulsivity/aggression traits. Eur Psychiatry. 2015;30(1):106-113.
13. Masi L, Gignac M ADHD and DMDD comorbidities, similarities and distinctions. J Child Adolesc Behav2016;4:325.
14. Kendrick JG, Goldman RD, Carr RR. Pharmacologic management of agitation and aggression in a pediatric emergency department - a retrospective cohort study. J Pediatr Pharmacol Ther. 2018;23(6):455-459.
15. Schieveld JN, Staal M, Voogd L, et al. Refractory agitation as a marker for pediatric delirium in very young infants at a pediatric intensive care unit. Intensive Care Med. 2010;36(11):1982-1983.
16. Traube C, Silver G, Gerber LM, et al. Delirium and mortality in critically ill children: epidemiology and outcomes of pediatric delirium. Crit Care Med. 2017;45(5):891-898.
17. Bettencourt A, Mullen JE. Delirium in children: identification, prevention, and management. Crit Care Nurse. 2017;37(3):e9-e18.
18. Suskauer SJ, Trovato MK. Update on pharmaceutical intervention for disorders of consciousness and agitation after traumatic brain injury in children. PM R. 2013;5(2):142-147.
19. Nowicki M, Pearlman L, Campbell C, et al. Agitated behavior scale in pediatric traumatic brain injury. Brain Inj. 2019. doi: 10.1080/02699052.2019.1565893.
20. Mohammad SS, Jones H, Hong M, et al. Symptomatic treatment of children with anti-NMDAR encephalitis. Dev Med Child Neurol. 2016;58(4):376-384.
21. Sonnier L, Barzman D. Pharmacologic management of acutely agitated pediatric patients. Pediatr Drugs. 2011;13(1):1-10.
22. Nordstrom K, Zun LS, Wilson MP, et al. Medical evaluation and triage of the agitated patient: consensus statement of the american association for emergency psychiatry project Beta medical evaluation workgroup. West J Emerg Med. 2012;13(1):3-10.
23. Masters KJ, Bellonci C, Bernet W, et al; American Academy of Child and Adolescent Psychiatry. Practice parameter for the prevention and management of aggressive behavior in child and adolescent psychiatric institutions, with special reference to seclusion and restraint. J Am Acad Child Adolesc Psychiatry. 2002;41(2 suppl):4S-25S.
24. Croce ND, Mantovani C. Using de-escalation techniques to prevent violent behavior in pediatric psychiatric emergencies: It is possible. Pediatric Dimensions, 2017;2(1):1-2.
25. Marzullo LR. Pharmacologic management of the agitated child. Pediatr Emerg Care. 2014;30(4):269-275.
26. Caldwell B, Albert C, Azeem MW, et al. Successful seclusion and restraint prevention effort in child and adolescent programs. J Psychosoc Nurs Ment Health Serv. 2014;52(11):30-38.
27. De Hert M, Dirix N, Demunter H, et al. Prevalence and correlates of seclusion and restraint use in children and adolescents: a systematic review. Eur Child Adolesc Psychiatry. 2011;20(5):221-230.
28. Crystal S, Olfson M, Huang C, et al. Broadened use of atypical antipsychotics: safety, effectiveness, and policy challenges. Health Aff (Millwood). 2009;28(5):w770-w781.
29. American Academy of Child and Adolescent Psychiatry. Practice parameters for the use of atypical antipsychotic medication in children and adolescents. https://www.aacap.org/App_Themes/AACAP/docs/practice_parameters/Atypical_Antipsychotic_Medications_Web.pdf. Accessed March 4, 2019.
30. Lohr WD, Chowning RT, Stevenson MD, et al. Trends in atypical antipsychotics prescribed to children six years of age or less on Medicaid in Kentucky. J Child Adolesc Psychopharmacol. 2015;25(5):440-443.
31. Chen W, Cepoiu-Martin M, Stang A, et al. Antipsychotic prescribing and safety monitoring practices in children and youth: a population-based study in Alberta, Canada. Clin Drug Investig. 2018;38(5):449-455.
32. Deshmukh P, Kulkarni G, Barzman D. Recommendations for pharmacological management of inpatient aggression in children and adolescents. Psychiatry (Edgmont). 2010;7(2):32-40.
33. Haldol [package insert]. Beerse, Belgium: Janssen Pharmaceutica NV; 2005.
34. Hirota T, Veenstra-Vanderweele J, Hollander E, et al. Antiepileptic medications in autism spectrum disorder: a systematic review and meta-analysis. J Autism Dev Disord. 2014;44(4):948-957.
35. Douglas JF, Sanders KB, Benneyworth MH, et al. Brief report: retrospective case series of oxcarbazepine for irritability/agitation symptoms in autism spectrum disorder. J Autism Dev Disord. 2013;43(5):1243-1247.
36. Harmon RJ, Riggs PD. Clonidine for posttraumatic stress disorder in preschool children. J Am Acad
37. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 1: Psychostimulants, alpha-2 Agonists, and atomoxetine. Can J Psychiatry. 2015;60(2):42-51
Managing agitation—verbal and/or motor restlessness that often is accompanied by irritability and a predisposition to aggression or violence—can be challenging in any patient, but particularly so in children and adolescents. In the United States, the prevalence of children and adolescents presenting to an emergency department (ED) for treatment of psychiatric symptoms, including agitation, has been on the rise.1,2
Similar to the multitude of causes of fever, agitation among children and adolescents has many possible causes.3 Because agitation can pose a risk for harm to others and/or self, it is important to manage it proactively. Other than studies that focus on agitation in pediatric anesthesia, there is a dearth of studies examining agitation and its treatment in children and adolescents. There is also a scarcity of training in the management of acute agitation in children and adolescents. In a 2017 survey of pediatric hospitalists and consultation-liaison psychiatrists at 38 academic children’s hospitals in North America, approximately 60% of respondents indicated that they had received no training in the evaluation or management of pediatric acute agitation.4 In addition, approximately 54% of participants said they did not screen for risk factors for pediatric agitation, even though 84% encountered the condition at least once a month, and as often as weekly.4
This article reviews evidence on the causes and treatments of agitation in children and adolescents. For the purposes of this review, child refers to a patient age 6 to 12, and adolescent refers to a patient age 13 to 17.
Identifying the cause
Addressing the underlying cause of agitation is essential. It’s also important to manage acute agitation while the underlying cause is being investigated in a way that does not jeopardize the patient’s emotional or physical safety.
Agitation in children or teens can be due to psychiatric causes such as autism, attention-deficit/hyperactivity disorder (ADHD), or posttraumatic stress disorder (PTSD), or due to medical conditions such as delirium, traumatic brain injury, or other conditions (Table 1).
In a 2005 study of 194 children with agitation in a pediatric post-anesthesia care unit, pain (27%) and anxiety (25%) were found to be the most common causes of agitation.3 Anesthesia-related agitation was a less common cause (11%). Physiologic anomalies were found to be the underlying cause of agitation in only 3 children in this study, but were undiagnosed for a prolonged period in 2 of these 3 children, which highlights the importance of a thorough differential diagnosis in the management of agitation in children.3
Assessment of an agitated child should include a comprehensive history, physical exam, and laboratory testing as indicated. When a pediatric patient comes to the ED with a chief presentation of agitation, a thorough medical and psychiatric assessment should be performed. For patients with a history of psychiatric diagnoses, do not assume that the cause of agitation is psychiatric.
Continue to: Psychiatric causes
Psychiatric causes
Autism spectrum disorder. Children and teens with autism often feel overwhelmed due to transitions, changes, and/or sensory overload. This sensory overload may be in response to relatively subtle sensory stimuli, so it may not always be apparent to parents or others around them.
Research suggests that in general, the ability to cope effectively with emotions is difficult without optimal language development. Due to cognitive/language delays and a related lack of emotional attunement and limited skills in recognizing, expressing, or coping with emotions, difficult emotions in children and adolescents with autism can manifest as agitation.
Attention-deficit/hyperactivity disorder. Children with ADHD may be at a higher risk for agitation, in part due to poor impulse control and limited coping skills. In addition, chronic negative feedback (from parents, teachers, or both) may contribute to low self-esteem, mood symptoms, defiance, and/or other behavioral difficulties. In addition to standard pharmacotherapy for ADHD, treatment involves parent behavior modification training. Setting firm yet empathic limits, “picking battles,” and implementing a developmentally appropriate behavioral plan to manage disruptive behavior in children or adolescents with ADHD can go a long way in helping to prevent the emergence of agitation.
Posttraumatic stress disorder. In some young children, new-onset, unexplained agitation may be the only sign of abuse or trauma. Children who have undergone trauma tend to experience confusion and distress. This may manifest as agitation or aggression, or other symptoms such as increased anxiety or nightmares.5 Trauma may be in the form of witnessing violence (domestic or other); experiencing physical, sexual, and/or emotional abuse; or witnessing/experiencing other significant threats to the safety of self and/or loved ones. Re-establishing (or establishing) a sense of psychological and physical safety is paramount in such patients.6 Psychotherapy is the first-line modality of treatment in children and adolescents with PTSD.6 In general, there is a scarcity of research on medication treatments for PTSD symptoms among children and adolescents.6
Oppositional defiant disorder/conduct disorder. Oppositional defiant disorder (ODD) can be comorbid with ADHD. The diagnosis of ODD requires a pervasive pattern of anger, defiance, vindictiveness, and hostility, particularly towards authority figures. However, these symptoms need to be differentiated from the normal range of childhood behavior. Occasionally, children learn to cope maladaptively through disruptive behavior or agitation. Although a parent or caregiver may see this behavior as intentionally malevolent, in a child with limited coping skills (whether due to young age, developmental/cognitive/language/learning delays, or social communication deficits) or one who has witnessed frequent agitation or aggression in the family environment, agitation and disruptive behavior may be a maladaptive form of coping. Thus, diligence needs to be exercised in the diagnosis of ODD and in understanding the psychosocial factors affecting the child, particularly because impulsiveness and uncooperativeness on their own have been found to be linked to greater likelihood of prescription of psychotropic medications from multiple classes.7 Family-based interventions, particularly parent training, family therapy, and age-appropriate child skills training, are of prime importance in managing this condition.8 Research shows that a shortage of resources, system issues, and cultural roadblocks in implementing family-based psychosocial interventions also can contribute to the increased use of psychotropic medications for aggression in children and teens with ODD, conduct disorder, or ADHD.8 The astute clinician needs to be cognizant of this before prescribing.
Continue to: Hallucinations/psychosis
Hallucinations/psychosis. Hallucinations (whether from psychiatric or medical causes) are significantly associated with agitation.9 In particular, auditory command hallucinations have been linked to agitation. Command hallucinations in children and adolescents may be secondary to early-onset schizophrenia; however, this diagnosis is rare.10 Hallucinations can also be an adverse effect of amphetamine-based stimulant medications in children and adolescents. Visual hallucinations are most often a sign of an underlying medical disorder such as delirium, occipital lobe mass/infection, or drug intoxication or withdrawal. Hallucinations need to be distinguished from the normal, imaginative play of a young child.10
Bipolar mania. In adults, bipolar disorder is a primary psychiatric cause of agitation. In children and adolescents, the diagnosis of bipolar disorder can be complex and requires careful and nuanced history-taking. The risks of agitation are greater with bipolar disorder than with unipolar depression.11,12
Disruptive mood dysregulation disorder. Prior to DSM-5, many children and adolescents with chronic, non-episodic irritability and severe outbursts out of proportion to the situation or stimuli were given a diagnosis of bipolar disorder. These symptoms, in combination with other symptoms, are now considered part of disruptive mood dysregulation disorder when severe outbursts in a child or adolescent occur 3 to 4 times a week consistently, for at least 1 year. The diagnosis of disruptive mood dysregulation disorder requires ruling out other psychiatric and medical conditions, particularly ADHD.13
Substance intoxication/withdrawal. Intoxication or withdrawal from substances such as alcohol, stimulant medications, opioids, methamphetamines, and other agents can lead to agitation. This is more likely to occur among adolescents than children.14
Adjustment disorder. Parental divorce, especially if it is conflictual, or other life stressors, such as experiencing a move or frequent moves, may contribute to the development of agitation in children and adolescents.
Continue to: Depression
Depression. In children and adolescents, depression can manifest as anger or irritability, and occasionally as agitation.
Medical causes
Delirium. Refractory agitation is often a manifestation of delirium in children and adolescents.15 If unrecognized and untreated, delirium can be fatal.16 Therefore, it is imperative that clinicians routinely assess for delirium in any patient who presents with agitation.
Because a patient with delirium often presents with agitation and visual or auditory hallucinations, the medical team may tend to assume these symptoms are secondary to a psychiatric disorder. In this case, the role of the consultation-liaison psychiatrist is critical for guiding the medical team, particularly to continue a thorough exploration of underlying causes while avoiding polypharmacy. Noise, bright lights, frequent changes in nursing staff or caregivers, anticholinergic or benzodiazepine medications, and frequent changes in schedules should be avoided to prevent delirium from occurring or getting worse.17 A multidisciplinary team approach is key in identifying the underlying cause and managing delirium in pediatric patients.
Traumatic brain injury. Agitation may be a presenting symptom in youth with traumatic brain injury (TBI).18 Agitation may present often in the acute recovery phase.19 There is limited evidence on the efficacy and safety of pharmacotherapy for agitation in pediatric patients with TBI.18
Autoimmune conditions. In a study of 27 patients with
Continue to: Medication-induced/iatrogenic
Medication-induced/iatrogenic. Agitation can be an adverse effect of medications such as amantadine (often used for TBI),18 atypical antipsychotics,21 selective serotonin reuptake inhibitors, and serotonin-norepinephrine reuptake inhibitors.
Infection. Agitation can be a result of encephalitis, meningitis, or other infectious processes.22
Metabolic conditions. Hepatic or renal failure, diabetic ketoacidosis, and thyroid toxicosis may cause agitation in children or adolescents.22
Start with nonpharmacologic interventions
Few studies have examined de-escalation techniques in agitated children and adolescents. However, verbal de-escalation is generally viewed as the first-line technique for managing agitation in children and adolescents. When feasible, teaching and modeling developmentally appropriate stress management skills for children and teens can be a beneficial preventative strategy to reduce the incidence and worsening of agitation.23
Clinicians should refrain from using coercion.24 Coercion could harm the therapeutic alliance, thereby impeding assessment of the underlying causes of agitation, and can be particularly harmful for patients who have a history of trauma or abuse. Even in pediatric patients with no such history, coercion is discouraged due to its punitive connotations and potential to adversely impact a vulnerable child or teen.
Continue to: Establishing a therapeutic rapport...
Establishing a therapeutic rapport with the patient, when feasible, can facilitate smoother de-escalation by offering the patient an outlet to air his/her frustrations and emotions, and by helping the patient feel understood.24 To facilitate this, ensure that the patient’s basic comforts and needs are met, such as access to a warm bed, food, and safety.25
The psychiatrist’s role is to help uncover and address the underlying reason for the patient’s agony or distress. Once the child or adolescent has calmed, explore potential triggers or causes of the agitation.
There has been a significant move away from the use of restraints for managing agitation in children and adolescents.26 Restraints have a psychologically traumatizing effect,27 and have been linked to life-threatening injuries and death in children.24
Pharmacotherapy: Proceed with caution
There are no FDA-approved medications for the treatment of agitation in the general pediatric population, and any medication use in this population is off-label. There is also a dearth of research examining the safety and efficacy of using psychotropic medications for agitation in pediatric patients. Because children and adolescents are more susceptible to adverse effects and risks associated with the use of psychotropic medications, special caution is warranted. In general, pharmacologic interventions are not recommended without the use of psychotherapy-based modalities.
In the past, the aim of using medications to treat patients with agitation was to put the patient to sleep.25 This practice did not help clinicians to assess for underlying causes, and was often accompanied by a greater risk of adverse effects and reactions.24 Therefore, the goal of medication treatment for agitation is to help calm the patient instead of inducing sleep.25
Continue to: Pharmacotherapy should...
Pharmacotherapy should be used only when behavioral interventions have been unsuccessful. Key considerations for using psychotropic medications to address agitation in children and adolescents are summarized in Table 2.25
Antipsychotics, particularly second-generation antipsychotics (SGAs), have been commonly used to manage acute agitation in children and adolescents, and there has been an upswing in the use of these medications in the United States in the last several years.28 Research indicates that males, children and adolescents in foster care, and those with Medicaid have been the more frequent youth recipients of SGAs.29 Of particular concern is the prevalence of antipsychotic use among children younger than age 6. In the last few decades, there has been an increase in the prescription of antipsychotics for children younger than age 6, particularly for disruptive behavior and aggression.30 In a study of preschool-age Medicaid patients in Kentucky, 70,777 prescriptions for SGAs were given to 6,915 children <6 years of age; 73% of these prescriptions were for male patients.30 Because there is a lack of controlled studies examining the safety and efficacy of SGAs among children and adolescents, especially with long-term use, further research is needed and caution is warranted.28
The FDA has approved
Externalizing disorders among children and adolescents tend to get treated with antipsychotics.28 A Canadian study examining records of 6,916 children found that most children who had been prescribed risperidone received it for ADHD or conduct disorder, and most patients had not received laboratory testing for monitoring the antipsychotic medication they were taking.31 In a 2018 study examining medical records of 120 pediatric patients who presented to an ED in British Columbia with agitation, antipsychotics were the most commonly used medications for patients with autism spectrum disorder; most patients received at least 1 dose.14
For children and adolescents with agitation or aggression who were admitted to inpatient units, IM
Continue to: In case reports...
In case reports, a combination of olanzapine with CNS-suppressing agents has resulted in death. Therefore, do not combine olanzapine with agents such as benzodiazepines.25 In a patient with a likely medical source of agitation, insufficient evidence exists to support the use of olanzapine, and additional research is needed.25
Low-dose haloperidol has been found to be effective for delirium-related agitation in pediatric studies.15 Before initiating an antipsychotic for any child or adolescent, review the patient’s family history for reports of early cardiac death and the patient’s own history of cardiac symptoms, palpitations, syncope, or prolonged QT interval. Monitor for QT prolongation. Among commonly used antipsychotics, the risk of QT prolongation is higher with IV administration of haloperidol and with ziprasidone. Studies show that compared with oral or IM
A few studies have found risperidone to be efficacious for treating ODD and conduct disorder; however, this use is off-label, and its considerable adverse effect and risk profile needs to be weighed against the potential benefit.8
Antipsychotic polypharmacy should be avoided because of the higher risk of adverse effects and interactions, and a lack of robust, controlled studies evaluating the safety of using antipsychotics for non-FDA-approved indications in children and adolescents.7 All patients who receive antipsychotics require monitoring for extrapyramidal symptoms, tardive dyskinesia, neuroleptic malignant syndrome, orthostatic hypotension, sedation, metabolic syndrome, and other potential adverse effects. Patients receiving risperidone need to have their prolactin levels monitored periodically, and their parents should be made aware of the potential for hyperprolactinemia and other adverse effects. Aripiprazole and quetiapine may increase the risk of suicidality.
Antiepileptics. A meta-analysis of 7 randomized controlled trials examining the use of antiepileptic medications (
Continue to: In a retrospective case series...
In a retrospective case series of 30 pediatric patients with autism spectrum disorder who were given oxcarbazepine, Douglas et al35 found that 47% of participants experienced significant improvement in irritability/agitation. However, 23% of patients reported significant adverse effects leading to discontinuation. Insufficient evidence exists for the safety and efficacy of oxcarbazepine in this population.35
Benzodiazepines. The use of benzodiazepines in pediatric patients has been associated with paradoxical disinhibition reactions, particularly in children with autism and other developmental or cognitive disabilities or delays.21 There is a lack of data on the safety and efficacy of long-term use of benzodiazepines in children, especially in light of these patients’ developing brains, the risk of cognitive impairment, and the potential for dependence with long-term use. Despite this, some studies show that the use of benzodiazepines is fairly common among pediatric patients who present to the ED with agitation.14 In a recent retrospective study, Kendrick et al14 found that among pediatric patients with agitation who were brought to the ED, benzodiazepines were the most commonly prescribed medications.
Other medications. Clonidine and
Diphenhydramine, in both oral and IM forms, has been used to treat agitation in children,32 but has also been associated with a paradoxical disinhibition reaction in pediatric patients21 and therefore should be used only sparingly and with caution. Diphenhydramine has anticholinergic properties, and may worsen delirium.15 Stimulant medications can help aggressive behavior in children and adolescents with ADHD.37
Bottom Line
Agitation among children and adolescents has many possible causes. A combination of a comprehensive assessment and evidence-based, judicious treatment interventions can help prevent and manage agitation in this vulnerable population.
Related Resources
- Baker M, Carlson GA. What do we really know about PRN use in agitated children with mental health conditions: a clinical review. Evid Based Ment Health. 2018;21(4):166-170.
- Gerson R, Malas N, Mroczkowski MM. Crisis in the emergency department: the evaluation and management of acute agitation in children and adolescents. Child Adolesc Psychiatr Clin N Am. 2018;27(3):367-386.
Drug Brand Names
Amantadine • Symmetrel
Aripiprazole • Abilify
Clonidine • Catapres
Guanfacine • Intuniv, Tenex
Haloperidol • Haldol
Lamotrigine • Lamictal
Levetiracetam • Keppra, Spritam
Olanzapine • Zyprexa
Oxcarbazepine • Trileptal
Quetiapine • Seroquel
Topiramate • Topamax
Risperidone • Risperdal
Valproate • Depakene
Ziprasidone • Geodon
Managing agitation—verbal and/or motor restlessness that often is accompanied by irritability and a predisposition to aggression or violence—can be challenging in any patient, but particularly so in children and adolescents. In the United States, the prevalence of children and adolescents presenting to an emergency department (ED) for treatment of psychiatric symptoms, including agitation, has been on the rise.1,2
Similar to the multitude of causes of fever, agitation among children and adolescents has many possible causes.3 Because agitation can pose a risk for harm to others and/or self, it is important to manage it proactively. Other than studies that focus on agitation in pediatric anesthesia, there is a dearth of studies examining agitation and its treatment in children and adolescents. There is also a scarcity of training in the management of acute agitation in children and adolescents. In a 2017 survey of pediatric hospitalists and consultation-liaison psychiatrists at 38 academic children’s hospitals in North America, approximately 60% of respondents indicated that they had received no training in the evaluation or management of pediatric acute agitation.4 In addition, approximately 54% of participants said they did not screen for risk factors for pediatric agitation, even though 84% encountered the condition at least once a month, and as often as weekly.4
This article reviews evidence on the causes and treatments of agitation in children and adolescents. For the purposes of this review, child refers to a patient age 6 to 12, and adolescent refers to a patient age 13 to 17.
Identifying the cause
Addressing the underlying cause of agitation is essential. It’s also important to manage acute agitation while the underlying cause is being investigated in a way that does not jeopardize the patient’s emotional or physical safety.
Agitation in children or teens can be due to psychiatric causes such as autism, attention-deficit/hyperactivity disorder (ADHD), or posttraumatic stress disorder (PTSD), or due to medical conditions such as delirium, traumatic brain injury, or other conditions (Table 1).
In a 2005 study of 194 children with agitation in a pediatric post-anesthesia care unit, pain (27%) and anxiety (25%) were found to be the most common causes of agitation.3 Anesthesia-related agitation was a less common cause (11%). Physiologic anomalies were found to be the underlying cause of agitation in only 3 children in this study, but were undiagnosed for a prolonged period in 2 of these 3 children, which highlights the importance of a thorough differential diagnosis in the management of agitation in children.3
Assessment of an agitated child should include a comprehensive history, physical exam, and laboratory testing as indicated. When a pediatric patient comes to the ED with a chief presentation of agitation, a thorough medical and psychiatric assessment should be performed. For patients with a history of psychiatric diagnoses, do not assume that the cause of agitation is psychiatric.
Continue to: Psychiatric causes
Psychiatric causes
Autism spectrum disorder. Children and teens with autism often feel overwhelmed due to transitions, changes, and/or sensory overload. This sensory overload may be in response to relatively subtle sensory stimuli, so it may not always be apparent to parents or others around them.
Research suggests that in general, the ability to cope effectively with emotions is difficult without optimal language development. Due to cognitive/language delays and a related lack of emotional attunement and limited skills in recognizing, expressing, or coping with emotions, difficult emotions in children and adolescents with autism can manifest as agitation.
Attention-deficit/hyperactivity disorder. Children with ADHD may be at a higher risk for agitation, in part due to poor impulse control and limited coping skills. In addition, chronic negative feedback (from parents, teachers, or both) may contribute to low self-esteem, mood symptoms, defiance, and/or other behavioral difficulties. In addition to standard pharmacotherapy for ADHD, treatment involves parent behavior modification training. Setting firm yet empathic limits, “picking battles,” and implementing a developmentally appropriate behavioral plan to manage disruptive behavior in children or adolescents with ADHD can go a long way in helping to prevent the emergence of agitation.
Posttraumatic stress disorder. In some young children, new-onset, unexplained agitation may be the only sign of abuse or trauma. Children who have undergone trauma tend to experience confusion and distress. This may manifest as agitation or aggression, or other symptoms such as increased anxiety or nightmares.5 Trauma may be in the form of witnessing violence (domestic or other); experiencing physical, sexual, and/or emotional abuse; or witnessing/experiencing other significant threats to the safety of self and/or loved ones. Re-establishing (or establishing) a sense of psychological and physical safety is paramount in such patients.6 Psychotherapy is the first-line modality of treatment in children and adolescents with PTSD.6 In general, there is a scarcity of research on medication treatments for PTSD symptoms among children and adolescents.6
Oppositional defiant disorder/conduct disorder. Oppositional defiant disorder (ODD) can be comorbid with ADHD. The diagnosis of ODD requires a pervasive pattern of anger, defiance, vindictiveness, and hostility, particularly towards authority figures. However, these symptoms need to be differentiated from the normal range of childhood behavior. Occasionally, children learn to cope maladaptively through disruptive behavior or agitation. Although a parent or caregiver may see this behavior as intentionally malevolent, in a child with limited coping skills (whether due to young age, developmental/cognitive/language/learning delays, or social communication deficits) or one who has witnessed frequent agitation or aggression in the family environment, agitation and disruptive behavior may be a maladaptive form of coping. Thus, diligence needs to be exercised in the diagnosis of ODD and in understanding the psychosocial factors affecting the child, particularly because impulsiveness and uncooperativeness on their own have been found to be linked to greater likelihood of prescription of psychotropic medications from multiple classes.7 Family-based interventions, particularly parent training, family therapy, and age-appropriate child skills training, are of prime importance in managing this condition.8 Research shows that a shortage of resources, system issues, and cultural roadblocks in implementing family-based psychosocial interventions also can contribute to the increased use of psychotropic medications for aggression in children and teens with ODD, conduct disorder, or ADHD.8 The astute clinician needs to be cognizant of this before prescribing.
Continue to: Hallucinations/psychosis
Hallucinations/psychosis. Hallucinations (whether from psychiatric or medical causes) are significantly associated with agitation.9 In particular, auditory command hallucinations have been linked to agitation. Command hallucinations in children and adolescents may be secondary to early-onset schizophrenia; however, this diagnosis is rare.10 Hallucinations can also be an adverse effect of amphetamine-based stimulant medications in children and adolescents. Visual hallucinations are most often a sign of an underlying medical disorder such as delirium, occipital lobe mass/infection, or drug intoxication or withdrawal. Hallucinations need to be distinguished from the normal, imaginative play of a young child.10
Bipolar mania. In adults, bipolar disorder is a primary psychiatric cause of agitation. In children and adolescents, the diagnosis of bipolar disorder can be complex and requires careful and nuanced history-taking. The risks of agitation are greater with bipolar disorder than with unipolar depression.11,12
Disruptive mood dysregulation disorder. Prior to DSM-5, many children and adolescents with chronic, non-episodic irritability and severe outbursts out of proportion to the situation or stimuli were given a diagnosis of bipolar disorder. These symptoms, in combination with other symptoms, are now considered part of disruptive mood dysregulation disorder when severe outbursts in a child or adolescent occur 3 to 4 times a week consistently, for at least 1 year. The diagnosis of disruptive mood dysregulation disorder requires ruling out other psychiatric and medical conditions, particularly ADHD.13
Substance intoxication/withdrawal. Intoxication or withdrawal from substances such as alcohol, stimulant medications, opioids, methamphetamines, and other agents can lead to agitation. This is more likely to occur among adolescents than children.14
Adjustment disorder. Parental divorce, especially if it is conflictual, or other life stressors, such as experiencing a move or frequent moves, may contribute to the development of agitation in children and adolescents.
Continue to: Depression
Depression. In children and adolescents, depression can manifest as anger or irritability, and occasionally as agitation.
Medical causes
Delirium. Refractory agitation is often a manifestation of delirium in children and adolescents.15 If unrecognized and untreated, delirium can be fatal.16 Therefore, it is imperative that clinicians routinely assess for delirium in any patient who presents with agitation.
Because a patient with delirium often presents with agitation and visual or auditory hallucinations, the medical team may tend to assume these symptoms are secondary to a psychiatric disorder. In this case, the role of the consultation-liaison psychiatrist is critical for guiding the medical team, particularly to continue a thorough exploration of underlying causes while avoiding polypharmacy. Noise, bright lights, frequent changes in nursing staff or caregivers, anticholinergic or benzodiazepine medications, and frequent changes in schedules should be avoided to prevent delirium from occurring or getting worse.17 A multidisciplinary team approach is key in identifying the underlying cause and managing delirium in pediatric patients.
Traumatic brain injury. Agitation may be a presenting symptom in youth with traumatic brain injury (TBI).18 Agitation may present often in the acute recovery phase.19 There is limited evidence on the efficacy and safety of pharmacotherapy for agitation in pediatric patients with TBI.18
Autoimmune conditions. In a study of 27 patients with
Continue to: Medication-induced/iatrogenic
Medication-induced/iatrogenic. Agitation can be an adverse effect of medications such as amantadine (often used for TBI),18 atypical antipsychotics,21 selective serotonin reuptake inhibitors, and serotonin-norepinephrine reuptake inhibitors.
Infection. Agitation can be a result of encephalitis, meningitis, or other infectious processes.22
Metabolic conditions. Hepatic or renal failure, diabetic ketoacidosis, and thyroid toxicosis may cause agitation in children or adolescents.22
Start with nonpharmacologic interventions
Few studies have examined de-escalation techniques in agitated children and adolescents. However, verbal de-escalation is generally viewed as the first-line technique for managing agitation in children and adolescents. When feasible, teaching and modeling developmentally appropriate stress management skills for children and teens can be a beneficial preventative strategy to reduce the incidence and worsening of agitation.23
Clinicians should refrain from using coercion.24 Coercion could harm the therapeutic alliance, thereby impeding assessment of the underlying causes of agitation, and can be particularly harmful for patients who have a history of trauma or abuse. Even in pediatric patients with no such history, coercion is discouraged due to its punitive connotations and potential to adversely impact a vulnerable child or teen.
Continue to: Establishing a therapeutic rapport...
Establishing a therapeutic rapport with the patient, when feasible, can facilitate smoother de-escalation by offering the patient an outlet to air his/her frustrations and emotions, and by helping the patient feel understood.24 To facilitate this, ensure that the patient’s basic comforts and needs are met, such as access to a warm bed, food, and safety.25
The psychiatrist’s role is to help uncover and address the underlying reason for the patient’s agony or distress. Once the child or adolescent has calmed, explore potential triggers or causes of the agitation.
There has been a significant move away from the use of restraints for managing agitation in children and adolescents.26 Restraints have a psychologically traumatizing effect,27 and have been linked to life-threatening injuries and death in children.24
Pharmacotherapy: Proceed with caution
There are no FDA-approved medications for the treatment of agitation in the general pediatric population, and any medication use in this population is off-label. There is also a dearth of research examining the safety and efficacy of using psychotropic medications for agitation in pediatric patients. Because children and adolescents are more susceptible to adverse effects and risks associated with the use of psychotropic medications, special caution is warranted. In general, pharmacologic interventions are not recommended without the use of psychotherapy-based modalities.
In the past, the aim of using medications to treat patients with agitation was to put the patient to sleep.25 This practice did not help clinicians to assess for underlying causes, and was often accompanied by a greater risk of adverse effects and reactions.24 Therefore, the goal of medication treatment for agitation is to help calm the patient instead of inducing sleep.25
Continue to: Pharmacotherapy should...
Pharmacotherapy should be used only when behavioral interventions have been unsuccessful. Key considerations for using psychotropic medications to address agitation in children and adolescents are summarized in Table 2.25
Antipsychotics, particularly second-generation antipsychotics (SGAs), have been commonly used to manage acute agitation in children and adolescents, and there has been an upswing in the use of these medications in the United States in the last several years.28 Research indicates that males, children and adolescents in foster care, and those with Medicaid have been the more frequent youth recipients of SGAs.29 Of particular concern is the prevalence of antipsychotic use among children younger than age 6. In the last few decades, there has been an increase in the prescription of antipsychotics for children younger than age 6, particularly for disruptive behavior and aggression.30 In a study of preschool-age Medicaid patients in Kentucky, 70,777 prescriptions for SGAs were given to 6,915 children <6 years of age; 73% of these prescriptions were for male patients.30 Because there is a lack of controlled studies examining the safety and efficacy of SGAs among children and adolescents, especially with long-term use, further research is needed and caution is warranted.28
The FDA has approved
Externalizing disorders among children and adolescents tend to get treated with antipsychotics.28 A Canadian study examining records of 6,916 children found that most children who had been prescribed risperidone received it for ADHD or conduct disorder, and most patients had not received laboratory testing for monitoring the antipsychotic medication they were taking.31 In a 2018 study examining medical records of 120 pediatric patients who presented to an ED in British Columbia with agitation, antipsychotics were the most commonly used medications for patients with autism spectrum disorder; most patients received at least 1 dose.14
For children and adolescents with agitation or aggression who were admitted to inpatient units, IM
Continue to: In case reports...
In case reports, a combination of olanzapine with CNS-suppressing agents has resulted in death. Therefore, do not combine olanzapine with agents such as benzodiazepines.25 In a patient with a likely medical source of agitation, insufficient evidence exists to support the use of olanzapine, and additional research is needed.25
Low-dose haloperidol has been found to be effective for delirium-related agitation in pediatric studies.15 Before initiating an antipsychotic for any child or adolescent, review the patient’s family history for reports of early cardiac death and the patient’s own history of cardiac symptoms, palpitations, syncope, or prolonged QT interval. Monitor for QT prolongation. Among commonly used antipsychotics, the risk of QT prolongation is higher with IV administration of haloperidol and with ziprasidone. Studies show that compared with oral or IM
A few studies have found risperidone to be efficacious for treating ODD and conduct disorder; however, this use is off-label, and its considerable adverse effect and risk profile needs to be weighed against the potential benefit.8
Antipsychotic polypharmacy should be avoided because of the higher risk of adverse effects and interactions, and a lack of robust, controlled studies evaluating the safety of using antipsychotics for non-FDA-approved indications in children and adolescents.7 All patients who receive antipsychotics require monitoring for extrapyramidal symptoms, tardive dyskinesia, neuroleptic malignant syndrome, orthostatic hypotension, sedation, metabolic syndrome, and other potential adverse effects. Patients receiving risperidone need to have their prolactin levels monitored periodically, and their parents should be made aware of the potential for hyperprolactinemia and other adverse effects. Aripiprazole and quetiapine may increase the risk of suicidality.
Antiepileptics. A meta-analysis of 7 randomized controlled trials examining the use of antiepileptic medications (
Continue to: In a retrospective case series...
In a retrospective case series of 30 pediatric patients with autism spectrum disorder who were given oxcarbazepine, Douglas et al35 found that 47% of participants experienced significant improvement in irritability/agitation. However, 23% of patients reported significant adverse effects leading to discontinuation. Insufficient evidence exists for the safety and efficacy of oxcarbazepine in this population.35
Benzodiazepines. The use of benzodiazepines in pediatric patients has been associated with paradoxical disinhibition reactions, particularly in children with autism and other developmental or cognitive disabilities or delays.21 There is a lack of data on the safety and efficacy of long-term use of benzodiazepines in children, especially in light of these patients’ developing brains, the risk of cognitive impairment, and the potential for dependence with long-term use. Despite this, some studies show that the use of benzodiazepines is fairly common among pediatric patients who present to the ED with agitation.14 In a recent retrospective study, Kendrick et al14 found that among pediatric patients with agitation who were brought to the ED, benzodiazepines were the most commonly prescribed medications.
Other medications. Clonidine and
Diphenhydramine, in both oral and IM forms, has been used to treat agitation in children,32 but has also been associated with a paradoxical disinhibition reaction in pediatric patients21 and therefore should be used only sparingly and with caution. Diphenhydramine has anticholinergic properties, and may worsen delirium.15 Stimulant medications can help aggressive behavior in children and adolescents with ADHD.37
Bottom Line
Agitation among children and adolescents has many possible causes. A combination of a comprehensive assessment and evidence-based, judicious treatment interventions can help prevent and manage agitation in this vulnerable population.
Related Resources
- Baker M, Carlson GA. What do we really know about PRN use in agitated children with mental health conditions: a clinical review. Evid Based Ment Health. 2018;21(4):166-170.
- Gerson R, Malas N, Mroczkowski MM. Crisis in the emergency department: the evaluation and management of acute agitation in children and adolescents. Child Adolesc Psychiatr Clin N Am. 2018;27(3):367-386.
Drug Brand Names
Amantadine • Symmetrel
Aripiprazole • Abilify
Clonidine • Catapres
Guanfacine • Intuniv, Tenex
Haloperidol • Haldol
Lamotrigine • Lamictal
Levetiracetam • Keppra, Spritam
Olanzapine • Zyprexa
Oxcarbazepine • Trileptal
Quetiapine • Seroquel
Topiramate • Topamax
Risperidone • Risperdal
Valproate • Depakene
Ziprasidone • Geodon
1. Frosch E, Kelly P. Issues in pediatric psychiatric emergency care. In: Emergency psychiatry. Cambridge, UK: Cambridge University Press; 2011:185-199.
2. American College of Emergency Physicians. Pediatric mental health emergencies in the emergency department. https://www.acep.org/patient-care/policy-statements/pediatric-mental-health-emergencies-in-the-emergency-medical-services-system/. Revised September 2018. Accessed February 23, 2019.
3. Voepel-Lewis, T, Burke C, Hadden S, et al. Nurses’ diagnoses and treatment decisions regarding care of the agitated child. J Perianesth Nurs. 2005;20(4):239-248.
4. Malas N, Spital L, Fischer J, et al. National survey on pediatric acute agitation and behavioral escalation in academic inpatient pediatric care settings. Psychosomatics. 2017;58(3):299-306.
5. Famularo R, Kinscherff R, Fenton T. Symptom differences in acute and chronic presentation of childhood post-traumatic stress disorder. Child Abuse Negl. 1990;14(3):439-444.
6. Kaminer D, Seedat S, Stein DJ. Post-traumatic stress disorder in children. World Psychiatry. 2005;4(2):121-125.
7. Ninan A, Stewart SL, Theall LA, et al. Adverse effects of psychotropic medications in children: predictive factors. J Can Acad Child Adolesc Psychiatry. 2014;23(3):218-225.
8. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 2: antipsychotics and traditional mood stabilizers. Can J Psychiatry. 2015;60(2):52-61.
9. Vareilles D, Bréhin C, Cortey C, et al. Hallucinations: Etiological analysis of children admitted to a pediatric emergency department. Arch Pediatr. 2017;24(5):445-452.
10. Bartlett J. Childhood-onset schizophrenia: what do we really know? Health Psychol Behav Med. 2014;2(1):735-747.
11. Diler RS, Goldstein TR, Hafeman D, et al. Distinguishing bipolar depression from unipolar depression in youth: Preliminary findings. J Child Adolesc Psychopharmacol. 2017;27(4):310-319.
12. Dervic K, Garcia-Amador M, Sudol K, et al. Bipolar I and II versus unipolar depression: clinical differences and impulsivity/aggression traits. Eur Psychiatry. 2015;30(1):106-113.
13. Masi L, Gignac M ADHD and DMDD comorbidities, similarities and distinctions. J Child Adolesc Behav2016;4:325.
14. Kendrick JG, Goldman RD, Carr RR. Pharmacologic management of agitation and aggression in a pediatric emergency department - a retrospective cohort study. J Pediatr Pharmacol Ther. 2018;23(6):455-459.
15. Schieveld JN, Staal M, Voogd L, et al. Refractory agitation as a marker for pediatric delirium in very young infants at a pediatric intensive care unit. Intensive Care Med. 2010;36(11):1982-1983.
16. Traube C, Silver G, Gerber LM, et al. Delirium and mortality in critically ill children: epidemiology and outcomes of pediatric delirium. Crit Care Med. 2017;45(5):891-898.
17. Bettencourt A, Mullen JE. Delirium in children: identification, prevention, and management. Crit Care Nurse. 2017;37(3):e9-e18.
18. Suskauer SJ, Trovato MK. Update on pharmaceutical intervention for disorders of consciousness and agitation after traumatic brain injury in children. PM R. 2013;5(2):142-147.
19. Nowicki M, Pearlman L, Campbell C, et al. Agitated behavior scale in pediatric traumatic brain injury. Brain Inj. 2019. doi: 10.1080/02699052.2019.1565893.
20. Mohammad SS, Jones H, Hong M, et al. Symptomatic treatment of children with anti-NMDAR encephalitis. Dev Med Child Neurol. 2016;58(4):376-384.
21. Sonnier L, Barzman D. Pharmacologic management of acutely agitated pediatric patients. Pediatr Drugs. 2011;13(1):1-10.
22. Nordstrom K, Zun LS, Wilson MP, et al. Medical evaluation and triage of the agitated patient: consensus statement of the american association for emergency psychiatry project Beta medical evaluation workgroup. West J Emerg Med. 2012;13(1):3-10.
23. Masters KJ, Bellonci C, Bernet W, et al; American Academy of Child and Adolescent Psychiatry. Practice parameter for the prevention and management of aggressive behavior in child and adolescent psychiatric institutions, with special reference to seclusion and restraint. J Am Acad Child Adolesc Psychiatry. 2002;41(2 suppl):4S-25S.
24. Croce ND, Mantovani C. Using de-escalation techniques to prevent violent behavior in pediatric psychiatric emergencies: It is possible. Pediatric Dimensions, 2017;2(1):1-2.
25. Marzullo LR. Pharmacologic management of the agitated child. Pediatr Emerg Care. 2014;30(4):269-275.
26. Caldwell B, Albert C, Azeem MW, et al. Successful seclusion and restraint prevention effort in child and adolescent programs. J Psychosoc Nurs Ment Health Serv. 2014;52(11):30-38.
27. De Hert M, Dirix N, Demunter H, et al. Prevalence and correlates of seclusion and restraint use in children and adolescents: a systematic review. Eur Child Adolesc Psychiatry. 2011;20(5):221-230.
28. Crystal S, Olfson M, Huang C, et al. Broadened use of atypical antipsychotics: safety, effectiveness, and policy challenges. Health Aff (Millwood). 2009;28(5):w770-w781.
29. American Academy of Child and Adolescent Psychiatry. Practice parameters for the use of atypical antipsychotic medication in children and adolescents. https://www.aacap.org/App_Themes/AACAP/docs/practice_parameters/Atypical_Antipsychotic_Medications_Web.pdf. Accessed March 4, 2019.
30. Lohr WD, Chowning RT, Stevenson MD, et al. Trends in atypical antipsychotics prescribed to children six years of age or less on Medicaid in Kentucky. J Child Adolesc Psychopharmacol. 2015;25(5):440-443.
31. Chen W, Cepoiu-Martin M, Stang A, et al. Antipsychotic prescribing and safety monitoring practices in children and youth: a population-based study in Alberta, Canada. Clin Drug Investig. 2018;38(5):449-455.
32. Deshmukh P, Kulkarni G, Barzman D. Recommendations for pharmacological management of inpatient aggression in children and adolescents. Psychiatry (Edgmont). 2010;7(2):32-40.
33. Haldol [package insert]. Beerse, Belgium: Janssen Pharmaceutica NV; 2005.
34. Hirota T, Veenstra-Vanderweele J, Hollander E, et al. Antiepileptic medications in autism spectrum disorder: a systematic review and meta-analysis. J Autism Dev Disord. 2014;44(4):948-957.
35. Douglas JF, Sanders KB, Benneyworth MH, et al. Brief report: retrospective case series of oxcarbazepine for irritability/agitation symptoms in autism spectrum disorder. J Autism Dev Disord. 2013;43(5):1243-1247.
36. Harmon RJ, Riggs PD. Clonidine for posttraumatic stress disorder in preschool children. J Am Acad
37. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 1: Psychostimulants, alpha-2 Agonists, and atomoxetine. Can J Psychiatry. 2015;60(2):42-51
1. Frosch E, Kelly P. Issues in pediatric psychiatric emergency care. In: Emergency psychiatry. Cambridge, UK: Cambridge University Press; 2011:185-199.
2. American College of Emergency Physicians. Pediatric mental health emergencies in the emergency department. https://www.acep.org/patient-care/policy-statements/pediatric-mental-health-emergencies-in-the-emergency-medical-services-system/. Revised September 2018. Accessed February 23, 2019.
3. Voepel-Lewis, T, Burke C, Hadden S, et al. Nurses’ diagnoses and treatment decisions regarding care of the agitated child. J Perianesth Nurs. 2005;20(4):239-248.
4. Malas N, Spital L, Fischer J, et al. National survey on pediatric acute agitation and behavioral escalation in academic inpatient pediatric care settings. Psychosomatics. 2017;58(3):299-306.
5. Famularo R, Kinscherff R, Fenton T. Symptom differences in acute and chronic presentation of childhood post-traumatic stress disorder. Child Abuse Negl. 1990;14(3):439-444.
6. Kaminer D, Seedat S, Stein DJ. Post-traumatic stress disorder in children. World Psychiatry. 2005;4(2):121-125.
7. Ninan A, Stewart SL, Theall LA, et al. Adverse effects of psychotropic medications in children: predictive factors. J Can Acad Child Adolesc Psychiatry. 2014;23(3):218-225.
8. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 2: antipsychotics and traditional mood stabilizers. Can J Psychiatry. 2015;60(2):52-61.
9. Vareilles D, Bréhin C, Cortey C, et al. Hallucinations: Etiological analysis of children admitted to a pediatric emergency department. Arch Pediatr. 2017;24(5):445-452.
10. Bartlett J. Childhood-onset schizophrenia: what do we really know? Health Psychol Behav Med. 2014;2(1):735-747.
11. Diler RS, Goldstein TR, Hafeman D, et al. Distinguishing bipolar depression from unipolar depression in youth: Preliminary findings. J Child Adolesc Psychopharmacol. 2017;27(4):310-319.
12. Dervic K, Garcia-Amador M, Sudol K, et al. Bipolar I and II versus unipolar depression: clinical differences and impulsivity/aggression traits. Eur Psychiatry. 2015;30(1):106-113.
13. Masi L, Gignac M ADHD and DMDD comorbidities, similarities and distinctions. J Child Adolesc Behav2016;4:325.
14. Kendrick JG, Goldman RD, Carr RR. Pharmacologic management of agitation and aggression in a pediatric emergency department - a retrospective cohort study. J Pediatr Pharmacol Ther. 2018;23(6):455-459.
15. Schieveld JN, Staal M, Voogd L, et al. Refractory agitation as a marker for pediatric delirium in very young infants at a pediatric intensive care unit. Intensive Care Med. 2010;36(11):1982-1983.
16. Traube C, Silver G, Gerber LM, et al. Delirium and mortality in critically ill children: epidemiology and outcomes of pediatric delirium. Crit Care Med. 2017;45(5):891-898.
17. Bettencourt A, Mullen JE. Delirium in children: identification, prevention, and management. Crit Care Nurse. 2017;37(3):e9-e18.
18. Suskauer SJ, Trovato MK. Update on pharmaceutical intervention for disorders of consciousness and agitation after traumatic brain injury in children. PM R. 2013;5(2):142-147.
19. Nowicki M, Pearlman L, Campbell C, et al. Agitated behavior scale in pediatric traumatic brain injury. Brain Inj. 2019. doi: 10.1080/02699052.2019.1565893.
20. Mohammad SS, Jones H, Hong M, et al. Symptomatic treatment of children with anti-NMDAR encephalitis. Dev Med Child Neurol. 2016;58(4):376-384.
21. Sonnier L, Barzman D. Pharmacologic management of acutely agitated pediatric patients. Pediatr Drugs. 2011;13(1):1-10.
22. Nordstrom K, Zun LS, Wilson MP, et al. Medical evaluation and triage of the agitated patient: consensus statement of the american association for emergency psychiatry project Beta medical evaluation workgroup. West J Emerg Med. 2012;13(1):3-10.
23. Masters KJ, Bellonci C, Bernet W, et al; American Academy of Child and Adolescent Psychiatry. Practice parameter for the prevention and management of aggressive behavior in child and adolescent psychiatric institutions, with special reference to seclusion and restraint. J Am Acad Child Adolesc Psychiatry. 2002;41(2 suppl):4S-25S.
24. Croce ND, Mantovani C. Using de-escalation techniques to prevent violent behavior in pediatric psychiatric emergencies: It is possible. Pediatric Dimensions, 2017;2(1):1-2.
25. Marzullo LR. Pharmacologic management of the agitated child. Pediatr Emerg Care. 2014;30(4):269-275.
26. Caldwell B, Albert C, Azeem MW, et al. Successful seclusion and restraint prevention effort in child and adolescent programs. J Psychosoc Nurs Ment Health Serv. 2014;52(11):30-38.
27. De Hert M, Dirix N, Demunter H, et al. Prevalence and correlates of seclusion and restraint use in children and adolescents: a systematic review. Eur Child Adolesc Psychiatry. 2011;20(5):221-230.
28. Crystal S, Olfson M, Huang C, et al. Broadened use of atypical antipsychotics: safety, effectiveness, and policy challenges. Health Aff (Millwood). 2009;28(5):w770-w781.
29. American Academy of Child and Adolescent Psychiatry. Practice parameters for the use of atypical antipsychotic medication in children and adolescents. https://www.aacap.org/App_Themes/AACAP/docs/practice_parameters/Atypical_Antipsychotic_Medications_Web.pdf. Accessed March 4, 2019.
30. Lohr WD, Chowning RT, Stevenson MD, et al. Trends in atypical antipsychotics prescribed to children six years of age or less on Medicaid in Kentucky. J Child Adolesc Psychopharmacol. 2015;25(5):440-443.
31. Chen W, Cepoiu-Martin M, Stang A, et al. Antipsychotic prescribing and safety monitoring practices in children and youth: a population-based study in Alberta, Canada. Clin Drug Investig. 2018;38(5):449-455.
32. Deshmukh P, Kulkarni G, Barzman D. Recommendations for pharmacological management of inpatient aggression in children and adolescents. Psychiatry (Edgmont). 2010;7(2):32-40.
33. Haldol [package insert]. Beerse, Belgium: Janssen Pharmaceutica NV; 2005.
34. Hirota T, Veenstra-Vanderweele J, Hollander E, et al. Antiepileptic medications in autism spectrum disorder: a systematic review and meta-analysis. J Autism Dev Disord. 2014;44(4):948-957.
35. Douglas JF, Sanders KB, Benneyworth MH, et al. Brief report: retrospective case series of oxcarbazepine for irritability/agitation symptoms in autism spectrum disorder. J Autism Dev Disord. 2013;43(5):1243-1247.
36. Harmon RJ, Riggs PD. Clonidine for posttraumatic stress disorder in preschool children. J Am Acad
37. Pringsheim T, Hirsch L, Gardner D, et al. The pharmacological management of oppositional behaviour, conduct problems, and aggression in children and adolescents with attention-deficit hyperactivity disorder, oppositional defiant disorder, and conduct disorder: a systematic review and meta-analysis. Part 1: Psychostimulants, alpha-2 Agonists, and atomoxetine. Can J Psychiatry. 2015;60(2):42-51
Unipolar vs bipolar depression: A clinician’s perspective
Mrs. W, age 36, who is married, has a history of military service, and is currently employed as a paralegal, is referred to our practice by her family physician. She complains of severe depression that impairs her ability to function at work. She had seen several other psychiatrists in both military and civilian settings, and had been treated with multiple antidepressants, including fluoxetine, sertraline, bupropion, and paroxetine.
At the time of her initial psychiatric evaluation, she is taking duloxetine, 90 mg/d, but still is experiencing depressive symptoms. She is tearful, sad, lacks energy, spends too much time in bed, and is experiencing thoughts of hopelessness, despair, and escape, verging on thoughts of suicide. As a result, she needs to scale back her work schedule to part-time. When asked about how long she had been suffering from depression, she responds “I’ve been depressed all my life.” She had been briefly hospitalized at age 16, when she made a suicide attempt by overdose. There had been no subsequent suicide attempts or psychiatric hospitalizations, although she acknowledges having intermittent suicidal thoughts.
Mrs. W’s clinical presentation is similar to that of many patients entering our practice—patients who have recurrent depression that began in early life and a history of failure to respond to multiple antidepressants. She and other patients with similar presentations are not suffering from treatment-resistant depression and in need of a trial of electroconvulsive therapy, transcranial magnetic stimulation, direct current stimulation, vagus nerve stimulation, or intranasal esketamine. She has bipolar disorder, and had been repeatedly misdiagnosed and treated inappropriately with antidepressant monotherapy.
In a previous article1 (“Controversies in bipolar disorder: Trust evidence or experience?,”
In this article, based on our more than 25 years of experience in diagnosing and treating psychiatric disorders in patients of all ages, we expand on those observations.
Misdiagnosis is common
Bipolar depression is frequently misdiagnosed as unipolar depression in outpatient2-8 and inpatient9 settings, and in children and adolescents.10 Mrs. W is typical of patients who have what we consider a bipolar spectrum disorder and receive an inaccurate diagnosis and treatment that is ineffective or may worsen the course of their illness.
Reliance on DSM-511 and its predecessor, DSM-IV, is a part of the problem of misdiagnosis because the diagnostic criteria for bipolar disorder fail to capture the clinical features of many patients with “softer” (less obvious manic and hypomanic) variants of the disorder.12,13 For example, DSM-5 criteria for a hypomanic episode (the mild high experienced by patients with a soft bipolar disorder) require that the episode lasts “at least 4 consecutive days” and is “present most of the day, nearly every day.” In our experience, the majority of hypomanic episodes are shorter—ranging from a half-day to 2 days, averaging perhaps 1.5 days.
Continue to: DSM-5 also requires...
DSM-5 also requires severity criteria for hypomania that patients with unequivocal hypomanic episodes often do not meet. For example, they may fail to experience flight of ideas or racing thoughts, or engage in activities such as “unrestrained buying sprees, sexual indiscretions, or foolish business investments.” These patients usually describe these mild highs as feeling normal and report a happier mood, more smiles and laughter, increased energy, less sleepiness, increased talkativeness, increased socialization, and improved motivation to complete tasks left undone and projects left unfinished because of the previous depressive episode. These softer (subthreshold) hypomanic episodes are authentic and, if clinicians do not identify them, may lead to misdiagnosis and inappropriate treatment.
Patients who present with depression often fail to report these brief, subthreshold hypomanic episodes or consider them to be irrelevant to their diagnosis and treatment.12,13 Probing questions can often elicit these unreported highs. For example, a patient with depression should be asked, “Have you had a single good day during the last month?” and “Where were you and what did you do during that day?” Eliciting a history of brief periods of improved mood is the key to differentiating between unipolar and bipolar depression. Screening instruments such as the Mood Disorders Questionnaire14 and the Bipolar Spectrum Diagnostic Scale15 may be helpful in distinguishing between unipolar and bipolar depression. However, we offer our thoughts on making that crucial distinction.
Distinguishing between these 2 types of depression
Although it may be difficult to distinguish between unipolar and bipolar depression, especially in the absence of a history of distinct manic or hypomanic episodes, we find the following criteria to be useful in making that determination.
Age of onset. Bipolar spectrum disorders typically begin earlier in life than unipolar depression.10,16-19 A typical presentation of bipolar disorder in children and adolescents is depression or agitated mixed states with features of both mania and depression, often accompanied by rapid mood cycling.20,21 Unipolar depression usually begins later in life, and patients do not have a history of significant depressive episodes or mood swings in childhood or adolescence. An important question to ask a patient with a chief complaint of depression is, “How old were you when you first experienced an episode of depression?”
Gender differences. Bipolar spectrum disorders with more subtle (softer) presentations, such as subthreshold highs, occur more often in women than men.22 However, overall rates of bipolar disorder may be slightly higher in men than in women.23 Unipolar melancholic depression occurs at approximately the same frequency in men and women.24
Continue to: Rapidity of onset
Rapidity of onset. Bipolar depressive episodes develop more rapidly than unipolar episodes. It is common for a patient with a bipolar spectrum disorder to transition from normal to very depressed virtually overnight, whereas in our clinical experience, unipolar episodes progress more slowly, often over several months.
Deliberate self-harm. Adolescents and young adults with a bipolar spectrum disorder frequently engage in self-injurious behavior, usually cutting with a knife, razor, or even sharp fingernails.25 Although these patients may also have thoughts of suicide and make suicide attempts, the individual usually perceives cutting as a means of gaining relief from tension and distress. These behaviors are often associated with a diagnosis of a personality disorder; in our opinion, however, they are hallmarks of a bipolar spectrum disorder.
ADHD. Bipolar disorder frequently co-occurs with attention-deficit/hyperactivity disorder (ADHD).26,27 Adults with bipolar disorder often have ADHD symptoms, which can complicate their treatment and cause functional impairment even after their mood disorder has been stabilized.28
Substance use disorders. Excessive use of alcohol and drugs is common among people with a wide range of psychiatric disorders, but patients with bipolar disorder have an unusually high rate of co-occurring substance use disorders—40% to 50%.29,30
Appetite and weight differences. Patients with unipolar depression usually experience loss of appetite and weight loss, whereas in our clinical experience, patients with bipolar depression often overeat, crave carbohydrates, and gain weight.
Continue to: Sleep problems
Sleep problems. Patients with bipolar depression have an increased need for sleep (the opposite of what they experience during highs), are sleepy during the day regardless of how many hours they sleep, and have difficulty getting up in the morning. Patients with unipolar depression also have a sleep disturbance: they may fall asleep easily, sleep for a few hours, and then awaken but are unable to fall back to sleep.31 Yet these patients usually do not complain of sleepiness during the day.
Diurnal variation of mood. Patients with unipolar depression often report that their depressive symptoms fluctuate in a circadian manner. For example, they may report that their depression is worse in the morning but improves toward evening.31 This regular alteration of circadian rhythm usually is not evident in patients with bipolar depression, whose mood may vary unpredictably or in response to stressors. Some patients with bipolar disorder, however, exhibit ultradian (ultra-rapid) mood cycling, which may be confused with the diurnal mood variation seen in patients with unipolar depression.
Tendency to recur. Although both unipolar and bipolar depressive episodes recur, a pattern of multiple recurring episodes beginning in early life is characteristic of bipolar spectrum disorders.
Behavioral history. Patients with bipolar depression are more likely than patients with unipolar depression to have a history of multiple marriages, multiple romantic relationships, episodes of promiscuity, legal problems, or financial extravagance.
Response to antidepressants. Patients with bipolar depression exhibit atypical responses to antidepressant monotherapy, such as worsening of depressive symptoms, initial improvement of mood with subsequent loss of effectiveness, premature response to an antidepressant (eg, improvement of mood within 1 to 2 days of beginning the antidepressant), fluctuation of depressive symptoms (mood cycling), or precipitation of a hypomanic or manic episode. We believe that a history of multiple failed antidepressant trials is compelling evidence of misdiagnosis of a bipolar spectrum disorder as unipolar depression.
Continue to: Genetics
Genetics. Bipolar disorder is one of the most heritable of illnesses.32 Family history is important, but affected relatives may have been misdiagnosed with unipolar depression or schizophrenia, or said to have experienced “nervous breakdowns.”
Consequences of misdiagnosis
Misdiagnosis of patients with bipolar disorder is not benign. We see patients who have suffered needlessly for years with severe depression and mood instability. After trying antidepressant after antidepressant without benefit, they begin to feel hopeless, believing they have tried everything and that nothing works for them. Often, these patients have dropped out of high school or college, or lost jobs, friends, and spouses due to their disabling but misdiagnosed psychiatric disorder. Patients with misdiagnosed bipolar disorder have an increased risk of suicide attempts and psychiatric hospitalization.5,8
Misdiagnosis of patients with bipolar disorder is not limited to nonpsychiatric physicians. The majority of patients with bipolar spectrum disorders are misdiagnosed by outpatient psychiatrists as having unipolar depression.2-7 At least 45% of patients hospitalized for depression have bipolar disorder—and most of these patients are treated inappropriately with antidepressants.9 The STAR*D study,33,34 a large randomized clinical trial of antidepressants, concluded that more than one-third of patients had not remitted from their depression after treatment with 3 different antidepressants. In our opinion, many of the nonresponding patients may have undiagnosed bipolar depression, which predictably leads to a failure to respond adequately to antidepressants. We believe that the customary inclusion and exclusion criteria used to select participants for these research studies miss subtle (subthreshold) hypomanic episodes that fall short of meeting DSM criteria for duration and severity. This phenomenon may account for the results of studies that conclude that antidepressants are, at best, minimally more effective than placebo.35
When a patient with a bipolar spectrum disorder is misdiagnosed and treated with an antidepressant, the usual result is mood destabilization. Reports of mood swings, increased crying, and suicidal thoughts and suicidal gestures in children, adolescents, and young adults treated with antidepressants led the FDA to issue a “black-box” warning.36 Because bipolar depression typically begins in youth,10,18,19 the behaviors cited in the warning may reflect misdiagnosis of bipolar depression as unipolar depression, and consequent mood destabilization as a result of treatment with an antidepressant in the absence of a mood stabilizer.
Depression and life stressors
Since many patients who are depressed present with a history of significant stressors, clinicians often face the problem of distinguishing between clinical depression and stress-induced depression. We believe that one typical symptom of depression—increased sensitivity to stressors—may help in making that distinction. A patient who is depressed will often attribute depression to stressors such as marital conflict, divorce, problems with a teenage child, work pressures, financial pressures, or the illness or death of a family member or pet. If clinical depression (unipolar or bipolar) is present, the symptoms are persistent, sometimes antedate the stressor by days or weeks, often outlast the stressor, increase in severity over time, and are disproportional to the stressor. Clinical depression can also cause the patient to become obsessed with traumatic events or losses that occurred many years earlier.
Continue to: Our approach to treatment
Our approach to treatment
Patients with mood disorders often benefit from a combination of pharmacologic management and psychotherapy. Psychotherapy is particularly important in addressing the functional impairment, diminished self-worth, and interpersonal conflicts that often accompany clinical depression. Several styles or systems of psychotherapy have been developed to benefit patients with mood disorders. Their effectiveness may depend on the patient’s ability to gain insight,37 but in our opinion, the most important attribute of helpful psychotherapy is the rapport established between the patient and the therapist, and the therapist’s ability to empathize with the patient and instill in the patient a sense of optimism and hope. We often recommend that patients attend meetings of the Depression and Bipolar Support Alliance (DBSA), a national support group with chapters throughout the country. Patients often find that attending these meetings is both educational and emotionally rewarding.
The foundational pharmacologic treatment for bipolar disorder is a mood stabilizer. The medications we consider to be effective mood stabilizers (some with an FDA indication for bipolar maintenance, some without) are lithium carbonate, divalproex sodium, carbamazepine, oxcarbazepine, and lamotrigine.
Each of these mood stabilizers has its advantages, disadvantages, risks, and adverse effects. For example, although divalproex is a reliable mood stabilizer, it has a significant risk of causing birth defects if taken during pregnancy and can cause increased appetite and weight gain. Carbamazepine has significant drug interactions and the potential to cause neurologic adverse effects, while oxcarbazepine, a derivative of carbamazepine, has fewer drug interactions but is more likely to cause hyponatremia. Lamotrigine must be titrated very slowly to reduce the risk of a potentially fatal skin rash (ie, Stevens-Johnson syndrome or toxic epidermal necrolysis). Lithium is effective but has a significant adverse-effect burden: impairment of renal function with long-term use, nephrogenic diabetes insipidus, hypothyroidism, hyperparathyroidism, acne, and weight gain. Lithium also has potential interactions with multiple commonly prescribed medications, including antihypertensives and diuretics, as well as over-the-counter pain relievers such as ibuprofen and naproxen.
Second-generation antipsychotics (SGAs) have mood stabilizing, antidepressant, and anti-manic properties and are often useful in managing bipolar disorder. In our experience, for patients with bipolar disorder, SGAs are best used in combination with a mood stabilizer. Although virtually all SGAs have demonstrated effectiveness in the treatment of psychosis and some phases of bipolar disorder, the newer agents (aripiprazole, brexpiprazole, lurasidone, and cariprazine) are relatively free of metabolic adverse effects such as weight gain, abnormal cholesterol levels, increased prolactin levels, insulin resistance, and increased risk of diabetes.
Antidepressants may be effective in treating unipolar depression, but when treating bipolar depression, they should be used cautiously and only in combination with a mood stabilizer.
Continue to: As we observed...
As we observed in our previous article,1 thyroid laboratory monitoring and supplementation are critical components of managing mood disorders (Box 138-41).
Box 1
Conventional laboratory reference ranges often indicate that thyroid-stimulating hormone (TSH) levels as high as 4.0, 4.5, or 5.0 mU/L are normal. A recent meta- analysis determined that treatment of subclinical hypothyroidism (elevated TSH with normal free thyroxine) does not benefit patients’ quality of life.38 Patients with mood disorders, however, often fail to respond to mood stabilizers and other psychiatric medications unless their TSH is <3.0 or even <2.5 mU/L.39,40 We typically augment with liothyronine because, unlike levothyroxine, it works quickly, does not require deiodination to be activated, and, contrary to some reports, its elimination and biologic half-life are sufficient for single daily dosing.41
Moving towards better diagnoses
The emergence of a criteria-based psychiatric system in 1980 with the publication of DSM-III, and its subsequent revisions and updates, constituted a major advance in psychiatric diagnosis. As we learn more about the pathophysiology, genetics, and epigenetics of psychiatric symptoms and syndromes, future diagnostic systems will improve problems of validity that have yet to be resolved. While we believe that, for the most part, DSM-5 was an advance over the previous diagnostic iteration, we have 2 issues with DSM-5 in terms of the diagnosis of bipolar disorder (Box 210,12,13,18,19,42).
Box 2
Based on our clinical experience treating thousands of patients over 25 years, we have 2 issues with DSM-5 regarding bipolar disorder:
1. The DSM-5 criteria for hypomania fail to reflect the features of clinical presentations commonly seen in our practice. The majority of patients with authentic bipolar syndromes do not have hypomanias that last for at least 4 days or reach the level of severity required for a DSM-5 diagnosis of hypomania. This results in misdiagnosis of patients with bipolar depression as suffering from unipolar depression, which leads to inappropriate treatment with antidepressant monotherapy.
2. Bipolar disorder frequently makes its first appearance in childhood and adolescence,10,18,19 and increasing numbers of young patients have been receiving this diagnosis.42 In our opinion, this increase reflects clinicians’ improved diagnostic skills. Perhaps alarmed by the increase in young people receiving a diagnosis of bipolar disorder, the authors of DSM-5 created a new diagnosis for children: disruptive mood dysregulation disorder. This diagnostic addition is based on the finding that children with these mood symptoms may not subsequently exhibit classic DSM-5 manic or hypomanic episodes. But the lack of such episodes does not preclude a diagnosis of bipolar disorder, because many adults with unequivocal bipolar spectrum disorders have subthreshold hypomanias and thus fail to exhibit classic manic or hypomanic episodes.12,13
A rose by any other name would smell as sweet. Children who exhibit symptoms of disruptive mood dysregulation disorder— chronic irritability and protracted temper outbursts—usually suffer from depression and mood instability. In our opinion, it is irrational and confusing to clinicians to separate out with a new diagnosis an arbitrarily defined group of children who exhibit substantially the same symptoms as those who receive a diagnosis of bipolar disorder.
Patients with a chief complaint of depression are often given a diagnosis of “major depression, rule out bipolar disorder.” We believe that this formula should be turned on its head. In our opinion, based on our clinical experience, we think that most patients who present to a clinician’s office or psychiatric hospital with depression have bipolar depression, not unipolar depression. We hope that our experience and observations derived from treating thousands of patients over more than 25 years may be helpful to clinicians who sometimes struggle to bring relief to their patients with mood disorders.
CASE CONTINUED
Return to work
Mrs. W is now doing well. She is taking a lower dosage of duloxetine, 60 mg/d, in combination with the mood stabilizer lamotrigine, 200 mg/d. She returns to work full-time as a paralegal and no longer is experiencing depressive episodes.
Bottom Line
Patients with bipolar depression are often misdiagnosed with unipolar depression and treated inappropriately with antidepressant monotherapy, which often results in mood destabilization. Based on our clinical experience, a careful assessment of select criteria, including age of onset, rapidity of onset, comorbidities, diurnal mood variations, and more, can be useful for distinguishing between unipolar and bipolar depression.
Related Resources
- Nasrallah HA. Misdiagnosing bipolar depression as major depressive disorder. Current Psychiatry. 2013;12(10):20-21,A.
- Ghaemi SN. Bipolar spectrum: a review of the concept and a vision for the future. Psychiatry Investig. 2013;10(3):218-224.
Drug Brand Names
Aripiprazole • Abilify
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol, Equetro
Cariprazine • Vraylar
Divalproex • Depakote
Duloxetine • Cymbalta
Esketamine • Spravato
Fluoxetine • Prozac
Lamotrigine • Lamictal
Levothyroxine • Synthroid, Levoxyl
Liothyronine • Cytomel
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Oxcarbazepine • Oxtellar XR, Trileptal
Paroxetine • Paxil
Sertraline • Zoloft
1. Miller GE, Noel RL. Controversies in bipolar disorder: trust evidence or experience? Current Psychiatry. 2009;8(2):27-28,31-33,39.
2. Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry. 2004;6(1):27-33.
3. Ghaemi SN, Sachs GS, Chiou AM, et al. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord. 1999;52(1-3):135-144.
4. Blanco C, Laje G, Olfson M, et al. Trends in the treatment of bipolar disorder by outpatient psychiatrists. Am J Psychiatry. 2002;159(6):1005-1010.
5. Shi L, Thiebaud P, McCombs JS. The impact of unrecognized bipolar disorders for patients treated with antidepressants in the fee-for-services California Medicaid (Medi-Cal) program. J Affect Disord. 2004;82(3):373-383.
6. Sidor MM, MacQueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156-167.
7. Hughes T, Cardno A, West R, et al. Unrecognized bipolar disorder among UK primary care patients prescribed antidepressants: an observational study. Br J Gen Pract. 2016;66(643):e71-e77.
8. Keck PE Jr, Kessler RC, Ross R. Clinical and economic effects of unrecognized or inadequately treated bipolar disorder. J Psychiatric Pract. 2008;14(Suppl 2):31-38.
9. Goldberg JF, Harrow M, Whiteside JF. Risk for bipolar illness in inpatients initially hospitalized for unipolar depression. Am J Psychiatry. 2001:158(8):1265-1270.
10. Chilakamarri JK, Filkowski MM, Ghaemi SN. Misdiagnosis of bipolar disorder in children and adolescents: a comparison with ADHD and major depressive disorder. Ann Clin Psychiatry. 2011;23(1):25-29.
11. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
12. Bowden CL. A different depression: clinical distinctions between bipolar and unipolar depression. J Affect Disord. 2005;84(2-3):117-125.
13. Baldassano C. Distinctions between bipolar I and bipolar II depression. Current Psychiatry. 2017;16(8):S7-S16.
14. Hirschfeld MA, Williams JB, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: The Mood Disorder Questionnaire. Am J Psychiatry. 2000;157(11):1873-1875.
15. Ghaemi SN, Miller CJ, Berv DA, et al. Sensitivity and specificity a new bipolar spectrum diagnostic scale. J Affect Disorder. 2005;84(2-3):273-277
16. Suppes T, Leverich G, Keck P, et al. The Stanley Foundation Continuing Bipolar Treatment Outcome Network. II. Demographics and illness characteristics of the first 261 patients. J Affect Disorder. 2001;67(1-3):45-49.
17. Perlis RH, Miyahara S, Marangell LB. Long-term implications of early onset in bipolar disorder: data from the first 1000 participants in the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP–BD). Biol Psychiatry. 2004;55(9):875-881.
18. Baldessarini RJ, Bolzani L, Kruz N, et al. Onset age of bipolar disorders at six international sites. J Affect Disord. 2010;121(1-2):143-146.
19. Post RM, Altshuler LL, Kupka R, et al. More childhood onset bipolar disorder in the United States than Canada or Europe: implications for treatment and prevention. Neurosci Biobehav Rev. 2017;74(Pt A):204-213.
20. Geller B, Luby J. Child and adolescent bipolar disorder: a review of the past 10 years. J Am Acad Child Adolesc Psychiatry. 1997;36(9):1168-1176.
21. Findling RL, Gracious BL, McNamara NK, et al. Rapid, continuous cycling and psychiatric co-morbidity in pediatric bipolar I disorder. Bipolar Disord. 2001;3(4):202-210.
22. Arnold LM. Gender differences in bipolar disorder. Psychiatr Clin North Am. 2003;26(3):595-620.
23. Deflorio A, Jones I. Is sex important? Gender differences in bipolar disorder. Int Rev Psychiatry. 2010;22(5):437-452.
24. Bogren M, Brådvik L, Holmstrand C, et al. Gender differences in subtypes of depression by first incidence and age of onset: a follow-up of the Lunby population. Eur Arch Psychiatry Clin Neurosci. 2018;268(2):179-189.
25. Singhal A, Ross J, Seminog O, et al. Risks of self-harm and suicide in people with specific psychiatric and physical disorders: comparisons between disorders using English national record linkage. J R Soc Med. 2014;107(5):194-204.
26. Joshi G, Wilens T. Comorbidity in pediatric bipolar disorder. Child Adolesc Psychiatr Clin N Amer. 2009;18(2):291-319.
27. Youngtrom EA, Arnold LE, Frazier TW. Bipolar and ADHD comorbidity: both artifact and outgrowth of shared mechanisms. Clin Psychol (New York). 2010;17(4):350-359.
28. McIntyre RS, Kennedy SH, Soczynska JK, et al. Attention-deficit/hyperactivity disorder in adults with bipolar disorder or major depressive disorder: results from the International Mood Disorders Collaborative Project. Prime Care Companion J Clin Psychiatry. 2010;12(3). doi:10.4088/PCC.09m00861gry.
29. Regier DA, Farmer ME, Rae DS, et al. Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiological Catchment Area (ECA) Study. JAMA. 1990;264(19):2511-2518.
30. Hunt GE, Malhi GS, Cleary M, et al. Prevalence of comorbid bipolar and substance use disorders in clinical settings, 1990-2015: systematic review and meta-analysis. J Affect Disord. 2016;206:331-349.
31. Agargun MY, Besiroglu L, Cilli AS, et al. Nightmares, suicide attempts, and melancholic features in patients with unipolar major depression. J Affect Disord. 2007;98(3):267-270.
32. Birmaher B, Axelson D, Monk K, et al. Lifetime psychiatric disorders in school-aged offspring of parents with bipolar disorder: the Pittsburgh Bipolar Offspring Study. Arch Gen Psychiatry. 2009;66(3):287-296.
33. Rush AJ, Trivedi MH, Wisniewski SR, et al. Buproprion-SR, sertraline, or venlafaxine-XR after failure of SSRIs for depression. N Engl J Med. 2006;354(12):1231-1242.
34. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer term outcomes in depressed outpatients requiring one or several treatment steps: A STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917.
35. Kirsch I. Antidepressants and the placebo effect. Z Psychol. 2014;222(3):128-134.
36. U.S. Food and Drug Administration. Suicidality in children and adolescents being treated with antidepressant medications. https://www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/ucm161679.htm. Published February 5, 2018. Accessed May 10, 2019.
37. Jennissen S, Huber J, Ehrenthal JC, et al. Association between insight and outcome of psychotherapy; systematic review and meta-analysis. Am J Psychiatry. 2018;175(10):961-969.
38. Feller M, Snel M, Moutzouri E, et al. Association of thyroid hormone therapy with quality of life and thyroid-related symptoms in patients with subclinical hypothyroidism. JAMA. 2018;320(13):1349-1359.
39. Cole DP, Thase ME, Mallinger AG, et al. Slower treatment response in bipolar depression predicted by lower pretreatment thyroid function. Am J Psychiatry. 2002;159(1):116-121.
40. Parmentier T, Sienaert P. The use of triiodothyronine (T3) in the treatment of bipolar depression: a review of the literature. J Affect Disord. 2018;229:410-414.
41. Koda-Kimbe MA, Alldredge BK. Koda-Kimble and Young’s applied therapeutics: the clinical use of drugs (10th ed). Baltimore, MD: Walters Klower Health/Lippincott Williams & Wilkins; 2012.
42. Moreno C, Laje G, Blanco C, et al. National trends in the outpatient diagnosis and treatment of bipolar disorder in youth. Arch Gen Psychiatry. 2007;64(9):1032-1039.
Mrs. W, age 36, who is married, has a history of military service, and is currently employed as a paralegal, is referred to our practice by her family physician. She complains of severe depression that impairs her ability to function at work. She had seen several other psychiatrists in both military and civilian settings, and had been treated with multiple antidepressants, including fluoxetine, sertraline, bupropion, and paroxetine.
At the time of her initial psychiatric evaluation, she is taking duloxetine, 90 mg/d, but still is experiencing depressive symptoms. She is tearful, sad, lacks energy, spends too much time in bed, and is experiencing thoughts of hopelessness, despair, and escape, verging on thoughts of suicide. As a result, she needs to scale back her work schedule to part-time. When asked about how long she had been suffering from depression, she responds “I’ve been depressed all my life.” She had been briefly hospitalized at age 16, when she made a suicide attempt by overdose. There had been no subsequent suicide attempts or psychiatric hospitalizations, although she acknowledges having intermittent suicidal thoughts.
Mrs. W’s clinical presentation is similar to that of many patients entering our practice—patients who have recurrent depression that began in early life and a history of failure to respond to multiple antidepressants. She and other patients with similar presentations are not suffering from treatment-resistant depression and in need of a trial of electroconvulsive therapy, transcranial magnetic stimulation, direct current stimulation, vagus nerve stimulation, or intranasal esketamine. She has bipolar disorder, and had been repeatedly misdiagnosed and treated inappropriately with antidepressant monotherapy.
In a previous article1 (“Controversies in bipolar disorder: Trust evidence or experience?,”
In this article, based on our more than 25 years of experience in diagnosing and treating psychiatric disorders in patients of all ages, we expand on those observations.
Misdiagnosis is common
Bipolar depression is frequently misdiagnosed as unipolar depression in outpatient2-8 and inpatient9 settings, and in children and adolescents.10 Mrs. W is typical of patients who have what we consider a bipolar spectrum disorder and receive an inaccurate diagnosis and treatment that is ineffective or may worsen the course of their illness.
Reliance on DSM-511 and its predecessor, DSM-IV, is a part of the problem of misdiagnosis because the diagnostic criteria for bipolar disorder fail to capture the clinical features of many patients with “softer” (less obvious manic and hypomanic) variants of the disorder.12,13 For example, DSM-5 criteria for a hypomanic episode (the mild high experienced by patients with a soft bipolar disorder) require that the episode lasts “at least 4 consecutive days” and is “present most of the day, nearly every day.” In our experience, the majority of hypomanic episodes are shorter—ranging from a half-day to 2 days, averaging perhaps 1.5 days.
Continue to: DSM-5 also requires...
DSM-5 also requires severity criteria for hypomania that patients with unequivocal hypomanic episodes often do not meet. For example, they may fail to experience flight of ideas or racing thoughts, or engage in activities such as “unrestrained buying sprees, sexual indiscretions, or foolish business investments.” These patients usually describe these mild highs as feeling normal and report a happier mood, more smiles and laughter, increased energy, less sleepiness, increased talkativeness, increased socialization, and improved motivation to complete tasks left undone and projects left unfinished because of the previous depressive episode. These softer (subthreshold) hypomanic episodes are authentic and, if clinicians do not identify them, may lead to misdiagnosis and inappropriate treatment.
Patients who present with depression often fail to report these brief, subthreshold hypomanic episodes or consider them to be irrelevant to their diagnosis and treatment.12,13 Probing questions can often elicit these unreported highs. For example, a patient with depression should be asked, “Have you had a single good day during the last month?” and “Where were you and what did you do during that day?” Eliciting a history of brief periods of improved mood is the key to differentiating between unipolar and bipolar depression. Screening instruments such as the Mood Disorders Questionnaire14 and the Bipolar Spectrum Diagnostic Scale15 may be helpful in distinguishing between unipolar and bipolar depression. However, we offer our thoughts on making that crucial distinction.
Distinguishing between these 2 types of depression
Although it may be difficult to distinguish between unipolar and bipolar depression, especially in the absence of a history of distinct manic or hypomanic episodes, we find the following criteria to be useful in making that determination.
Age of onset. Bipolar spectrum disorders typically begin earlier in life than unipolar depression.10,16-19 A typical presentation of bipolar disorder in children and adolescents is depression or agitated mixed states with features of both mania and depression, often accompanied by rapid mood cycling.20,21 Unipolar depression usually begins later in life, and patients do not have a history of significant depressive episodes or mood swings in childhood or adolescence. An important question to ask a patient with a chief complaint of depression is, “How old were you when you first experienced an episode of depression?”
Gender differences. Bipolar spectrum disorders with more subtle (softer) presentations, such as subthreshold highs, occur more often in women than men.22 However, overall rates of bipolar disorder may be slightly higher in men than in women.23 Unipolar melancholic depression occurs at approximately the same frequency in men and women.24
Continue to: Rapidity of onset
Rapidity of onset. Bipolar depressive episodes develop more rapidly than unipolar episodes. It is common for a patient with a bipolar spectrum disorder to transition from normal to very depressed virtually overnight, whereas in our clinical experience, unipolar episodes progress more slowly, often over several months.
Deliberate self-harm. Adolescents and young adults with a bipolar spectrum disorder frequently engage in self-injurious behavior, usually cutting with a knife, razor, or even sharp fingernails.25 Although these patients may also have thoughts of suicide and make suicide attempts, the individual usually perceives cutting as a means of gaining relief from tension and distress. These behaviors are often associated with a diagnosis of a personality disorder; in our opinion, however, they are hallmarks of a bipolar spectrum disorder.
ADHD. Bipolar disorder frequently co-occurs with attention-deficit/hyperactivity disorder (ADHD).26,27 Adults with bipolar disorder often have ADHD symptoms, which can complicate their treatment and cause functional impairment even after their mood disorder has been stabilized.28
Substance use disorders. Excessive use of alcohol and drugs is common among people with a wide range of psychiatric disorders, but patients with bipolar disorder have an unusually high rate of co-occurring substance use disorders—40% to 50%.29,30
Appetite and weight differences. Patients with unipolar depression usually experience loss of appetite and weight loss, whereas in our clinical experience, patients with bipolar depression often overeat, crave carbohydrates, and gain weight.
Continue to: Sleep problems
Sleep problems. Patients with bipolar depression have an increased need for sleep (the opposite of what they experience during highs), are sleepy during the day regardless of how many hours they sleep, and have difficulty getting up in the morning. Patients with unipolar depression also have a sleep disturbance: they may fall asleep easily, sleep for a few hours, and then awaken but are unable to fall back to sleep.31 Yet these patients usually do not complain of sleepiness during the day.
Diurnal variation of mood. Patients with unipolar depression often report that their depressive symptoms fluctuate in a circadian manner. For example, they may report that their depression is worse in the morning but improves toward evening.31 This regular alteration of circadian rhythm usually is not evident in patients with bipolar depression, whose mood may vary unpredictably or in response to stressors. Some patients with bipolar disorder, however, exhibit ultradian (ultra-rapid) mood cycling, which may be confused with the diurnal mood variation seen in patients with unipolar depression.
Tendency to recur. Although both unipolar and bipolar depressive episodes recur, a pattern of multiple recurring episodes beginning in early life is characteristic of bipolar spectrum disorders.
Behavioral history. Patients with bipolar depression are more likely than patients with unipolar depression to have a history of multiple marriages, multiple romantic relationships, episodes of promiscuity, legal problems, or financial extravagance.
Response to antidepressants. Patients with bipolar depression exhibit atypical responses to antidepressant monotherapy, such as worsening of depressive symptoms, initial improvement of mood with subsequent loss of effectiveness, premature response to an antidepressant (eg, improvement of mood within 1 to 2 days of beginning the antidepressant), fluctuation of depressive symptoms (mood cycling), or precipitation of a hypomanic or manic episode. We believe that a history of multiple failed antidepressant trials is compelling evidence of misdiagnosis of a bipolar spectrum disorder as unipolar depression.
Continue to: Genetics
Genetics. Bipolar disorder is one of the most heritable of illnesses.32 Family history is important, but affected relatives may have been misdiagnosed with unipolar depression or schizophrenia, or said to have experienced “nervous breakdowns.”
Consequences of misdiagnosis
Misdiagnosis of patients with bipolar disorder is not benign. We see patients who have suffered needlessly for years with severe depression and mood instability. After trying antidepressant after antidepressant without benefit, they begin to feel hopeless, believing they have tried everything and that nothing works for them. Often, these patients have dropped out of high school or college, or lost jobs, friends, and spouses due to their disabling but misdiagnosed psychiatric disorder. Patients with misdiagnosed bipolar disorder have an increased risk of suicide attempts and psychiatric hospitalization.5,8
Misdiagnosis of patients with bipolar disorder is not limited to nonpsychiatric physicians. The majority of patients with bipolar spectrum disorders are misdiagnosed by outpatient psychiatrists as having unipolar depression.2-7 At least 45% of patients hospitalized for depression have bipolar disorder—and most of these patients are treated inappropriately with antidepressants.9 The STAR*D study,33,34 a large randomized clinical trial of antidepressants, concluded that more than one-third of patients had not remitted from their depression after treatment with 3 different antidepressants. In our opinion, many of the nonresponding patients may have undiagnosed bipolar depression, which predictably leads to a failure to respond adequately to antidepressants. We believe that the customary inclusion and exclusion criteria used to select participants for these research studies miss subtle (subthreshold) hypomanic episodes that fall short of meeting DSM criteria for duration and severity. This phenomenon may account for the results of studies that conclude that antidepressants are, at best, minimally more effective than placebo.35
When a patient with a bipolar spectrum disorder is misdiagnosed and treated with an antidepressant, the usual result is mood destabilization. Reports of mood swings, increased crying, and suicidal thoughts and suicidal gestures in children, adolescents, and young adults treated with antidepressants led the FDA to issue a “black-box” warning.36 Because bipolar depression typically begins in youth,10,18,19 the behaviors cited in the warning may reflect misdiagnosis of bipolar depression as unipolar depression, and consequent mood destabilization as a result of treatment with an antidepressant in the absence of a mood stabilizer.
Depression and life stressors
Since many patients who are depressed present with a history of significant stressors, clinicians often face the problem of distinguishing between clinical depression and stress-induced depression. We believe that one typical symptom of depression—increased sensitivity to stressors—may help in making that distinction. A patient who is depressed will often attribute depression to stressors such as marital conflict, divorce, problems with a teenage child, work pressures, financial pressures, or the illness or death of a family member or pet. If clinical depression (unipolar or bipolar) is present, the symptoms are persistent, sometimes antedate the stressor by days or weeks, often outlast the stressor, increase in severity over time, and are disproportional to the stressor. Clinical depression can also cause the patient to become obsessed with traumatic events or losses that occurred many years earlier.
Continue to: Our approach to treatment
Our approach to treatment
Patients with mood disorders often benefit from a combination of pharmacologic management and psychotherapy. Psychotherapy is particularly important in addressing the functional impairment, diminished self-worth, and interpersonal conflicts that often accompany clinical depression. Several styles or systems of psychotherapy have been developed to benefit patients with mood disorders. Their effectiveness may depend on the patient’s ability to gain insight,37 but in our opinion, the most important attribute of helpful psychotherapy is the rapport established between the patient and the therapist, and the therapist’s ability to empathize with the patient and instill in the patient a sense of optimism and hope. We often recommend that patients attend meetings of the Depression and Bipolar Support Alliance (DBSA), a national support group with chapters throughout the country. Patients often find that attending these meetings is both educational and emotionally rewarding.
The foundational pharmacologic treatment for bipolar disorder is a mood stabilizer. The medications we consider to be effective mood stabilizers (some with an FDA indication for bipolar maintenance, some without) are lithium carbonate, divalproex sodium, carbamazepine, oxcarbazepine, and lamotrigine.
Each of these mood stabilizers has its advantages, disadvantages, risks, and adverse effects. For example, although divalproex is a reliable mood stabilizer, it has a significant risk of causing birth defects if taken during pregnancy and can cause increased appetite and weight gain. Carbamazepine has significant drug interactions and the potential to cause neurologic adverse effects, while oxcarbazepine, a derivative of carbamazepine, has fewer drug interactions but is more likely to cause hyponatremia. Lamotrigine must be titrated very slowly to reduce the risk of a potentially fatal skin rash (ie, Stevens-Johnson syndrome or toxic epidermal necrolysis). Lithium is effective but has a significant adverse-effect burden: impairment of renal function with long-term use, nephrogenic diabetes insipidus, hypothyroidism, hyperparathyroidism, acne, and weight gain. Lithium also has potential interactions with multiple commonly prescribed medications, including antihypertensives and diuretics, as well as over-the-counter pain relievers such as ibuprofen and naproxen.
Second-generation antipsychotics (SGAs) have mood stabilizing, antidepressant, and anti-manic properties and are often useful in managing bipolar disorder. In our experience, for patients with bipolar disorder, SGAs are best used in combination with a mood stabilizer. Although virtually all SGAs have demonstrated effectiveness in the treatment of psychosis and some phases of bipolar disorder, the newer agents (aripiprazole, brexpiprazole, lurasidone, and cariprazine) are relatively free of metabolic adverse effects such as weight gain, abnormal cholesterol levels, increased prolactin levels, insulin resistance, and increased risk of diabetes.
Antidepressants may be effective in treating unipolar depression, but when treating bipolar depression, they should be used cautiously and only in combination with a mood stabilizer.
Continue to: As we observed...
As we observed in our previous article,1 thyroid laboratory monitoring and supplementation are critical components of managing mood disorders (Box 138-41).
Box 1
Conventional laboratory reference ranges often indicate that thyroid-stimulating hormone (TSH) levels as high as 4.0, 4.5, or 5.0 mU/L are normal. A recent meta- analysis determined that treatment of subclinical hypothyroidism (elevated TSH with normal free thyroxine) does not benefit patients’ quality of life.38 Patients with mood disorders, however, often fail to respond to mood stabilizers and other psychiatric medications unless their TSH is <3.0 or even <2.5 mU/L.39,40 We typically augment with liothyronine because, unlike levothyroxine, it works quickly, does not require deiodination to be activated, and, contrary to some reports, its elimination and biologic half-life are sufficient for single daily dosing.41
Moving towards better diagnoses
The emergence of a criteria-based psychiatric system in 1980 with the publication of DSM-III, and its subsequent revisions and updates, constituted a major advance in psychiatric diagnosis. As we learn more about the pathophysiology, genetics, and epigenetics of psychiatric symptoms and syndromes, future diagnostic systems will improve problems of validity that have yet to be resolved. While we believe that, for the most part, DSM-5 was an advance over the previous diagnostic iteration, we have 2 issues with DSM-5 in terms of the diagnosis of bipolar disorder (Box 210,12,13,18,19,42).
Box 2
Based on our clinical experience treating thousands of patients over 25 years, we have 2 issues with DSM-5 regarding bipolar disorder:
1. The DSM-5 criteria for hypomania fail to reflect the features of clinical presentations commonly seen in our practice. The majority of patients with authentic bipolar syndromes do not have hypomanias that last for at least 4 days or reach the level of severity required for a DSM-5 diagnosis of hypomania. This results in misdiagnosis of patients with bipolar depression as suffering from unipolar depression, which leads to inappropriate treatment with antidepressant monotherapy.
2. Bipolar disorder frequently makes its first appearance in childhood and adolescence,10,18,19 and increasing numbers of young patients have been receiving this diagnosis.42 In our opinion, this increase reflects clinicians’ improved diagnostic skills. Perhaps alarmed by the increase in young people receiving a diagnosis of bipolar disorder, the authors of DSM-5 created a new diagnosis for children: disruptive mood dysregulation disorder. This diagnostic addition is based on the finding that children with these mood symptoms may not subsequently exhibit classic DSM-5 manic or hypomanic episodes. But the lack of such episodes does not preclude a diagnosis of bipolar disorder, because many adults with unequivocal bipolar spectrum disorders have subthreshold hypomanias and thus fail to exhibit classic manic or hypomanic episodes.12,13
A rose by any other name would smell as sweet. Children who exhibit symptoms of disruptive mood dysregulation disorder— chronic irritability and protracted temper outbursts—usually suffer from depression and mood instability. In our opinion, it is irrational and confusing to clinicians to separate out with a new diagnosis an arbitrarily defined group of children who exhibit substantially the same symptoms as those who receive a diagnosis of bipolar disorder.
Patients with a chief complaint of depression are often given a diagnosis of “major depression, rule out bipolar disorder.” We believe that this formula should be turned on its head. In our opinion, based on our clinical experience, we think that most patients who present to a clinician’s office or psychiatric hospital with depression have bipolar depression, not unipolar depression. We hope that our experience and observations derived from treating thousands of patients over more than 25 years may be helpful to clinicians who sometimes struggle to bring relief to their patients with mood disorders.
CASE CONTINUED
Return to work
Mrs. W is now doing well. She is taking a lower dosage of duloxetine, 60 mg/d, in combination with the mood stabilizer lamotrigine, 200 mg/d. She returns to work full-time as a paralegal and no longer is experiencing depressive episodes.
Bottom Line
Patients with bipolar depression are often misdiagnosed with unipolar depression and treated inappropriately with antidepressant monotherapy, which often results in mood destabilization. Based on our clinical experience, a careful assessment of select criteria, including age of onset, rapidity of onset, comorbidities, diurnal mood variations, and more, can be useful for distinguishing between unipolar and bipolar depression.
Related Resources
- Nasrallah HA. Misdiagnosing bipolar depression as major depressive disorder. Current Psychiatry. 2013;12(10):20-21,A.
- Ghaemi SN. Bipolar spectrum: a review of the concept and a vision for the future. Psychiatry Investig. 2013;10(3):218-224.
Drug Brand Names
Aripiprazole • Abilify
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol, Equetro
Cariprazine • Vraylar
Divalproex • Depakote
Duloxetine • Cymbalta
Esketamine • Spravato
Fluoxetine • Prozac
Lamotrigine • Lamictal
Levothyroxine • Synthroid, Levoxyl
Liothyronine • Cytomel
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Oxcarbazepine • Oxtellar XR, Trileptal
Paroxetine • Paxil
Sertraline • Zoloft
Mrs. W, age 36, who is married, has a history of military service, and is currently employed as a paralegal, is referred to our practice by her family physician. She complains of severe depression that impairs her ability to function at work. She had seen several other psychiatrists in both military and civilian settings, and had been treated with multiple antidepressants, including fluoxetine, sertraline, bupropion, and paroxetine.
At the time of her initial psychiatric evaluation, she is taking duloxetine, 90 mg/d, but still is experiencing depressive symptoms. She is tearful, sad, lacks energy, spends too much time in bed, and is experiencing thoughts of hopelessness, despair, and escape, verging on thoughts of suicide. As a result, she needs to scale back her work schedule to part-time. When asked about how long she had been suffering from depression, she responds “I’ve been depressed all my life.” She had been briefly hospitalized at age 16, when she made a suicide attempt by overdose. There had been no subsequent suicide attempts or psychiatric hospitalizations, although she acknowledges having intermittent suicidal thoughts.
Mrs. W’s clinical presentation is similar to that of many patients entering our practice—patients who have recurrent depression that began in early life and a history of failure to respond to multiple antidepressants. She and other patients with similar presentations are not suffering from treatment-resistant depression and in need of a trial of electroconvulsive therapy, transcranial magnetic stimulation, direct current stimulation, vagus nerve stimulation, or intranasal esketamine. She has bipolar disorder, and had been repeatedly misdiagnosed and treated inappropriately with antidepressant monotherapy.
In a previous article1 (“Controversies in bipolar disorder: Trust evidence or experience?,”
In this article, based on our more than 25 years of experience in diagnosing and treating psychiatric disorders in patients of all ages, we expand on those observations.
Misdiagnosis is common
Bipolar depression is frequently misdiagnosed as unipolar depression in outpatient2-8 and inpatient9 settings, and in children and adolescents.10 Mrs. W is typical of patients who have what we consider a bipolar spectrum disorder and receive an inaccurate diagnosis and treatment that is ineffective or may worsen the course of their illness.
Reliance on DSM-511 and its predecessor, DSM-IV, is a part of the problem of misdiagnosis because the diagnostic criteria for bipolar disorder fail to capture the clinical features of many patients with “softer” (less obvious manic and hypomanic) variants of the disorder.12,13 For example, DSM-5 criteria for a hypomanic episode (the mild high experienced by patients with a soft bipolar disorder) require that the episode lasts “at least 4 consecutive days” and is “present most of the day, nearly every day.” In our experience, the majority of hypomanic episodes are shorter—ranging from a half-day to 2 days, averaging perhaps 1.5 days.
Continue to: DSM-5 also requires...
DSM-5 also requires severity criteria for hypomania that patients with unequivocal hypomanic episodes often do not meet. For example, they may fail to experience flight of ideas or racing thoughts, or engage in activities such as “unrestrained buying sprees, sexual indiscretions, or foolish business investments.” These patients usually describe these mild highs as feeling normal and report a happier mood, more smiles and laughter, increased energy, less sleepiness, increased talkativeness, increased socialization, and improved motivation to complete tasks left undone and projects left unfinished because of the previous depressive episode. These softer (subthreshold) hypomanic episodes are authentic and, if clinicians do not identify them, may lead to misdiagnosis and inappropriate treatment.
Patients who present with depression often fail to report these brief, subthreshold hypomanic episodes or consider them to be irrelevant to their diagnosis and treatment.12,13 Probing questions can often elicit these unreported highs. For example, a patient with depression should be asked, “Have you had a single good day during the last month?” and “Where were you and what did you do during that day?” Eliciting a history of brief periods of improved mood is the key to differentiating between unipolar and bipolar depression. Screening instruments such as the Mood Disorders Questionnaire14 and the Bipolar Spectrum Diagnostic Scale15 may be helpful in distinguishing between unipolar and bipolar depression. However, we offer our thoughts on making that crucial distinction.
Distinguishing between these 2 types of depression
Although it may be difficult to distinguish between unipolar and bipolar depression, especially in the absence of a history of distinct manic or hypomanic episodes, we find the following criteria to be useful in making that determination.
Age of onset. Bipolar spectrum disorders typically begin earlier in life than unipolar depression.10,16-19 A typical presentation of bipolar disorder in children and adolescents is depression or agitated mixed states with features of both mania and depression, often accompanied by rapid mood cycling.20,21 Unipolar depression usually begins later in life, and patients do not have a history of significant depressive episodes or mood swings in childhood or adolescence. An important question to ask a patient with a chief complaint of depression is, “How old were you when you first experienced an episode of depression?”
Gender differences. Bipolar spectrum disorders with more subtle (softer) presentations, such as subthreshold highs, occur more often in women than men.22 However, overall rates of bipolar disorder may be slightly higher in men than in women.23 Unipolar melancholic depression occurs at approximately the same frequency in men and women.24
Continue to: Rapidity of onset
Rapidity of onset. Bipolar depressive episodes develop more rapidly than unipolar episodes. It is common for a patient with a bipolar spectrum disorder to transition from normal to very depressed virtually overnight, whereas in our clinical experience, unipolar episodes progress more slowly, often over several months.
Deliberate self-harm. Adolescents and young adults with a bipolar spectrum disorder frequently engage in self-injurious behavior, usually cutting with a knife, razor, or even sharp fingernails.25 Although these patients may also have thoughts of suicide and make suicide attempts, the individual usually perceives cutting as a means of gaining relief from tension and distress. These behaviors are often associated with a diagnosis of a personality disorder; in our opinion, however, they are hallmarks of a bipolar spectrum disorder.
ADHD. Bipolar disorder frequently co-occurs with attention-deficit/hyperactivity disorder (ADHD).26,27 Adults with bipolar disorder often have ADHD symptoms, which can complicate their treatment and cause functional impairment even after their mood disorder has been stabilized.28
Substance use disorders. Excessive use of alcohol and drugs is common among people with a wide range of psychiatric disorders, but patients with bipolar disorder have an unusually high rate of co-occurring substance use disorders—40% to 50%.29,30
Appetite and weight differences. Patients with unipolar depression usually experience loss of appetite and weight loss, whereas in our clinical experience, patients with bipolar depression often overeat, crave carbohydrates, and gain weight.
Continue to: Sleep problems
Sleep problems. Patients with bipolar depression have an increased need for sleep (the opposite of what they experience during highs), are sleepy during the day regardless of how many hours they sleep, and have difficulty getting up in the morning. Patients with unipolar depression also have a sleep disturbance: they may fall asleep easily, sleep for a few hours, and then awaken but are unable to fall back to sleep.31 Yet these patients usually do not complain of sleepiness during the day.
Diurnal variation of mood. Patients with unipolar depression often report that their depressive symptoms fluctuate in a circadian manner. For example, they may report that their depression is worse in the morning but improves toward evening.31 This regular alteration of circadian rhythm usually is not evident in patients with bipolar depression, whose mood may vary unpredictably or in response to stressors. Some patients with bipolar disorder, however, exhibit ultradian (ultra-rapid) mood cycling, which may be confused with the diurnal mood variation seen in patients with unipolar depression.
Tendency to recur. Although both unipolar and bipolar depressive episodes recur, a pattern of multiple recurring episodes beginning in early life is characteristic of bipolar spectrum disorders.
Behavioral history. Patients with bipolar depression are more likely than patients with unipolar depression to have a history of multiple marriages, multiple romantic relationships, episodes of promiscuity, legal problems, or financial extravagance.
Response to antidepressants. Patients with bipolar depression exhibit atypical responses to antidepressant monotherapy, such as worsening of depressive symptoms, initial improvement of mood with subsequent loss of effectiveness, premature response to an antidepressant (eg, improvement of mood within 1 to 2 days of beginning the antidepressant), fluctuation of depressive symptoms (mood cycling), or precipitation of a hypomanic or manic episode. We believe that a history of multiple failed antidepressant trials is compelling evidence of misdiagnosis of a bipolar spectrum disorder as unipolar depression.
Continue to: Genetics
Genetics. Bipolar disorder is one of the most heritable of illnesses.32 Family history is important, but affected relatives may have been misdiagnosed with unipolar depression or schizophrenia, or said to have experienced “nervous breakdowns.”
Consequences of misdiagnosis
Misdiagnosis of patients with bipolar disorder is not benign. We see patients who have suffered needlessly for years with severe depression and mood instability. After trying antidepressant after antidepressant without benefit, they begin to feel hopeless, believing they have tried everything and that nothing works for them. Often, these patients have dropped out of high school or college, or lost jobs, friends, and spouses due to their disabling but misdiagnosed psychiatric disorder. Patients with misdiagnosed bipolar disorder have an increased risk of suicide attempts and psychiatric hospitalization.5,8
Misdiagnosis of patients with bipolar disorder is not limited to nonpsychiatric physicians. The majority of patients with bipolar spectrum disorders are misdiagnosed by outpatient psychiatrists as having unipolar depression.2-7 At least 45% of patients hospitalized for depression have bipolar disorder—and most of these patients are treated inappropriately with antidepressants.9 The STAR*D study,33,34 a large randomized clinical trial of antidepressants, concluded that more than one-third of patients had not remitted from their depression after treatment with 3 different antidepressants. In our opinion, many of the nonresponding patients may have undiagnosed bipolar depression, which predictably leads to a failure to respond adequately to antidepressants. We believe that the customary inclusion and exclusion criteria used to select participants for these research studies miss subtle (subthreshold) hypomanic episodes that fall short of meeting DSM criteria for duration and severity. This phenomenon may account for the results of studies that conclude that antidepressants are, at best, minimally more effective than placebo.35
When a patient with a bipolar spectrum disorder is misdiagnosed and treated with an antidepressant, the usual result is mood destabilization. Reports of mood swings, increased crying, and suicidal thoughts and suicidal gestures in children, adolescents, and young adults treated with antidepressants led the FDA to issue a “black-box” warning.36 Because bipolar depression typically begins in youth,10,18,19 the behaviors cited in the warning may reflect misdiagnosis of bipolar depression as unipolar depression, and consequent mood destabilization as a result of treatment with an antidepressant in the absence of a mood stabilizer.
Depression and life stressors
Since many patients who are depressed present with a history of significant stressors, clinicians often face the problem of distinguishing between clinical depression and stress-induced depression. We believe that one typical symptom of depression—increased sensitivity to stressors—may help in making that distinction. A patient who is depressed will often attribute depression to stressors such as marital conflict, divorce, problems with a teenage child, work pressures, financial pressures, or the illness or death of a family member or pet. If clinical depression (unipolar or bipolar) is present, the symptoms are persistent, sometimes antedate the stressor by days or weeks, often outlast the stressor, increase in severity over time, and are disproportional to the stressor. Clinical depression can also cause the patient to become obsessed with traumatic events or losses that occurred many years earlier.
Continue to: Our approach to treatment
Our approach to treatment
Patients with mood disorders often benefit from a combination of pharmacologic management and psychotherapy. Psychotherapy is particularly important in addressing the functional impairment, diminished self-worth, and interpersonal conflicts that often accompany clinical depression. Several styles or systems of psychotherapy have been developed to benefit patients with mood disorders. Their effectiveness may depend on the patient’s ability to gain insight,37 but in our opinion, the most important attribute of helpful psychotherapy is the rapport established between the patient and the therapist, and the therapist’s ability to empathize with the patient and instill in the patient a sense of optimism and hope. We often recommend that patients attend meetings of the Depression and Bipolar Support Alliance (DBSA), a national support group with chapters throughout the country. Patients often find that attending these meetings is both educational and emotionally rewarding.
The foundational pharmacologic treatment for bipolar disorder is a mood stabilizer. The medications we consider to be effective mood stabilizers (some with an FDA indication for bipolar maintenance, some without) are lithium carbonate, divalproex sodium, carbamazepine, oxcarbazepine, and lamotrigine.
Each of these mood stabilizers has its advantages, disadvantages, risks, and adverse effects. For example, although divalproex is a reliable mood stabilizer, it has a significant risk of causing birth defects if taken during pregnancy and can cause increased appetite and weight gain. Carbamazepine has significant drug interactions and the potential to cause neurologic adverse effects, while oxcarbazepine, a derivative of carbamazepine, has fewer drug interactions but is more likely to cause hyponatremia. Lamotrigine must be titrated very slowly to reduce the risk of a potentially fatal skin rash (ie, Stevens-Johnson syndrome or toxic epidermal necrolysis). Lithium is effective but has a significant adverse-effect burden: impairment of renal function with long-term use, nephrogenic diabetes insipidus, hypothyroidism, hyperparathyroidism, acne, and weight gain. Lithium also has potential interactions with multiple commonly prescribed medications, including antihypertensives and diuretics, as well as over-the-counter pain relievers such as ibuprofen and naproxen.
Second-generation antipsychotics (SGAs) have mood stabilizing, antidepressant, and anti-manic properties and are often useful in managing bipolar disorder. In our experience, for patients with bipolar disorder, SGAs are best used in combination with a mood stabilizer. Although virtually all SGAs have demonstrated effectiveness in the treatment of psychosis and some phases of bipolar disorder, the newer agents (aripiprazole, brexpiprazole, lurasidone, and cariprazine) are relatively free of metabolic adverse effects such as weight gain, abnormal cholesterol levels, increased prolactin levels, insulin resistance, and increased risk of diabetes.
Antidepressants may be effective in treating unipolar depression, but when treating bipolar depression, they should be used cautiously and only in combination with a mood stabilizer.
Continue to: As we observed...
As we observed in our previous article,1 thyroid laboratory monitoring and supplementation are critical components of managing mood disorders (Box 138-41).
Box 1
Conventional laboratory reference ranges often indicate that thyroid-stimulating hormone (TSH) levels as high as 4.0, 4.5, or 5.0 mU/L are normal. A recent meta- analysis determined that treatment of subclinical hypothyroidism (elevated TSH with normal free thyroxine) does not benefit patients’ quality of life.38 Patients with mood disorders, however, often fail to respond to mood stabilizers and other psychiatric medications unless their TSH is <3.0 or even <2.5 mU/L.39,40 We typically augment with liothyronine because, unlike levothyroxine, it works quickly, does not require deiodination to be activated, and, contrary to some reports, its elimination and biologic half-life are sufficient for single daily dosing.41
Moving towards better diagnoses
The emergence of a criteria-based psychiatric system in 1980 with the publication of DSM-III, and its subsequent revisions and updates, constituted a major advance in psychiatric diagnosis. As we learn more about the pathophysiology, genetics, and epigenetics of psychiatric symptoms and syndromes, future diagnostic systems will improve problems of validity that have yet to be resolved. While we believe that, for the most part, DSM-5 was an advance over the previous diagnostic iteration, we have 2 issues with DSM-5 in terms of the diagnosis of bipolar disorder (Box 210,12,13,18,19,42).
Box 2
Based on our clinical experience treating thousands of patients over 25 years, we have 2 issues with DSM-5 regarding bipolar disorder:
1. The DSM-5 criteria for hypomania fail to reflect the features of clinical presentations commonly seen in our practice. The majority of patients with authentic bipolar syndromes do not have hypomanias that last for at least 4 days or reach the level of severity required for a DSM-5 diagnosis of hypomania. This results in misdiagnosis of patients with bipolar depression as suffering from unipolar depression, which leads to inappropriate treatment with antidepressant monotherapy.
2. Bipolar disorder frequently makes its first appearance in childhood and adolescence,10,18,19 and increasing numbers of young patients have been receiving this diagnosis.42 In our opinion, this increase reflects clinicians’ improved diagnostic skills. Perhaps alarmed by the increase in young people receiving a diagnosis of bipolar disorder, the authors of DSM-5 created a new diagnosis for children: disruptive mood dysregulation disorder. This diagnostic addition is based on the finding that children with these mood symptoms may not subsequently exhibit classic DSM-5 manic or hypomanic episodes. But the lack of such episodes does not preclude a diagnosis of bipolar disorder, because many adults with unequivocal bipolar spectrum disorders have subthreshold hypomanias and thus fail to exhibit classic manic or hypomanic episodes.12,13
A rose by any other name would smell as sweet. Children who exhibit symptoms of disruptive mood dysregulation disorder— chronic irritability and protracted temper outbursts—usually suffer from depression and mood instability. In our opinion, it is irrational and confusing to clinicians to separate out with a new diagnosis an arbitrarily defined group of children who exhibit substantially the same symptoms as those who receive a diagnosis of bipolar disorder.
Patients with a chief complaint of depression are often given a diagnosis of “major depression, rule out bipolar disorder.” We believe that this formula should be turned on its head. In our opinion, based on our clinical experience, we think that most patients who present to a clinician’s office or psychiatric hospital with depression have bipolar depression, not unipolar depression. We hope that our experience and observations derived from treating thousands of patients over more than 25 years may be helpful to clinicians who sometimes struggle to bring relief to their patients with mood disorders.
CASE CONTINUED
Return to work
Mrs. W is now doing well. She is taking a lower dosage of duloxetine, 60 mg/d, in combination with the mood stabilizer lamotrigine, 200 mg/d. She returns to work full-time as a paralegal and no longer is experiencing depressive episodes.
Bottom Line
Patients with bipolar depression are often misdiagnosed with unipolar depression and treated inappropriately with antidepressant monotherapy, which often results in mood destabilization. Based on our clinical experience, a careful assessment of select criteria, including age of onset, rapidity of onset, comorbidities, diurnal mood variations, and more, can be useful for distinguishing between unipolar and bipolar depression.
Related Resources
- Nasrallah HA. Misdiagnosing bipolar depression as major depressive disorder. Current Psychiatry. 2013;12(10):20-21,A.
- Ghaemi SN. Bipolar spectrum: a review of the concept and a vision for the future. Psychiatry Investig. 2013;10(3):218-224.
Drug Brand Names
Aripiprazole • Abilify
Brexpiprazole • Rexulti
Bupropion • Wellbutrin
Carbamazepine • Tegretol, Equetro
Cariprazine • Vraylar
Divalproex • Depakote
Duloxetine • Cymbalta
Esketamine • Spravato
Fluoxetine • Prozac
Lamotrigine • Lamictal
Levothyroxine • Synthroid, Levoxyl
Liothyronine • Cytomel
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Oxcarbazepine • Oxtellar XR, Trileptal
Paroxetine • Paxil
Sertraline • Zoloft
1. Miller GE, Noel RL. Controversies in bipolar disorder: trust evidence or experience? Current Psychiatry. 2009;8(2):27-28,31-33,39.
2. Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry. 2004;6(1):27-33.
3. Ghaemi SN, Sachs GS, Chiou AM, et al. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord. 1999;52(1-3):135-144.
4. Blanco C, Laje G, Olfson M, et al. Trends in the treatment of bipolar disorder by outpatient psychiatrists. Am J Psychiatry. 2002;159(6):1005-1010.
5. Shi L, Thiebaud P, McCombs JS. The impact of unrecognized bipolar disorders for patients treated with antidepressants in the fee-for-services California Medicaid (Medi-Cal) program. J Affect Disord. 2004;82(3):373-383.
6. Sidor MM, MacQueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156-167.
7. Hughes T, Cardno A, West R, et al. Unrecognized bipolar disorder among UK primary care patients prescribed antidepressants: an observational study. Br J Gen Pract. 2016;66(643):e71-e77.
8. Keck PE Jr, Kessler RC, Ross R. Clinical and economic effects of unrecognized or inadequately treated bipolar disorder. J Psychiatric Pract. 2008;14(Suppl 2):31-38.
9. Goldberg JF, Harrow M, Whiteside JF. Risk for bipolar illness in inpatients initially hospitalized for unipolar depression. Am J Psychiatry. 2001:158(8):1265-1270.
10. Chilakamarri JK, Filkowski MM, Ghaemi SN. Misdiagnosis of bipolar disorder in children and adolescents: a comparison with ADHD and major depressive disorder. Ann Clin Psychiatry. 2011;23(1):25-29.
11. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
12. Bowden CL. A different depression: clinical distinctions between bipolar and unipolar depression. J Affect Disord. 2005;84(2-3):117-125.
13. Baldassano C. Distinctions between bipolar I and bipolar II depression. Current Psychiatry. 2017;16(8):S7-S16.
14. Hirschfeld MA, Williams JB, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: The Mood Disorder Questionnaire. Am J Psychiatry. 2000;157(11):1873-1875.
15. Ghaemi SN, Miller CJ, Berv DA, et al. Sensitivity and specificity a new bipolar spectrum diagnostic scale. J Affect Disorder. 2005;84(2-3):273-277
16. Suppes T, Leverich G, Keck P, et al. The Stanley Foundation Continuing Bipolar Treatment Outcome Network. II. Demographics and illness characteristics of the first 261 patients. J Affect Disorder. 2001;67(1-3):45-49.
17. Perlis RH, Miyahara S, Marangell LB. Long-term implications of early onset in bipolar disorder: data from the first 1000 participants in the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP–BD). Biol Psychiatry. 2004;55(9):875-881.
18. Baldessarini RJ, Bolzani L, Kruz N, et al. Onset age of bipolar disorders at six international sites. J Affect Disord. 2010;121(1-2):143-146.
19. Post RM, Altshuler LL, Kupka R, et al. More childhood onset bipolar disorder in the United States than Canada or Europe: implications for treatment and prevention. Neurosci Biobehav Rev. 2017;74(Pt A):204-213.
20. Geller B, Luby J. Child and adolescent bipolar disorder: a review of the past 10 years. J Am Acad Child Adolesc Psychiatry. 1997;36(9):1168-1176.
21. Findling RL, Gracious BL, McNamara NK, et al. Rapid, continuous cycling and psychiatric co-morbidity in pediatric bipolar I disorder. Bipolar Disord. 2001;3(4):202-210.
22. Arnold LM. Gender differences in bipolar disorder. Psychiatr Clin North Am. 2003;26(3):595-620.
23. Deflorio A, Jones I. Is sex important? Gender differences in bipolar disorder. Int Rev Psychiatry. 2010;22(5):437-452.
24. Bogren M, Brådvik L, Holmstrand C, et al. Gender differences in subtypes of depression by first incidence and age of onset: a follow-up of the Lunby population. Eur Arch Psychiatry Clin Neurosci. 2018;268(2):179-189.
25. Singhal A, Ross J, Seminog O, et al. Risks of self-harm and suicide in people with specific psychiatric and physical disorders: comparisons between disorders using English national record linkage. J R Soc Med. 2014;107(5):194-204.
26. Joshi G, Wilens T. Comorbidity in pediatric bipolar disorder. Child Adolesc Psychiatr Clin N Amer. 2009;18(2):291-319.
27. Youngtrom EA, Arnold LE, Frazier TW. Bipolar and ADHD comorbidity: both artifact and outgrowth of shared mechanisms. Clin Psychol (New York). 2010;17(4):350-359.
28. McIntyre RS, Kennedy SH, Soczynska JK, et al. Attention-deficit/hyperactivity disorder in adults with bipolar disorder or major depressive disorder: results from the International Mood Disorders Collaborative Project. Prime Care Companion J Clin Psychiatry. 2010;12(3). doi:10.4088/PCC.09m00861gry.
29. Regier DA, Farmer ME, Rae DS, et al. Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiological Catchment Area (ECA) Study. JAMA. 1990;264(19):2511-2518.
30. Hunt GE, Malhi GS, Cleary M, et al. Prevalence of comorbid bipolar and substance use disorders in clinical settings, 1990-2015: systematic review and meta-analysis. J Affect Disord. 2016;206:331-349.
31. Agargun MY, Besiroglu L, Cilli AS, et al. Nightmares, suicide attempts, and melancholic features in patients with unipolar major depression. J Affect Disord. 2007;98(3):267-270.
32. Birmaher B, Axelson D, Monk K, et al. Lifetime psychiatric disorders in school-aged offspring of parents with bipolar disorder: the Pittsburgh Bipolar Offspring Study. Arch Gen Psychiatry. 2009;66(3):287-296.
33. Rush AJ, Trivedi MH, Wisniewski SR, et al. Buproprion-SR, sertraline, or venlafaxine-XR after failure of SSRIs for depression. N Engl J Med. 2006;354(12):1231-1242.
34. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer term outcomes in depressed outpatients requiring one or several treatment steps: A STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917.
35. Kirsch I. Antidepressants and the placebo effect. Z Psychol. 2014;222(3):128-134.
36. U.S. Food and Drug Administration. Suicidality in children and adolescents being treated with antidepressant medications. https://www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/ucm161679.htm. Published February 5, 2018. Accessed May 10, 2019.
37. Jennissen S, Huber J, Ehrenthal JC, et al. Association between insight and outcome of psychotherapy; systematic review and meta-analysis. Am J Psychiatry. 2018;175(10):961-969.
38. Feller M, Snel M, Moutzouri E, et al. Association of thyroid hormone therapy with quality of life and thyroid-related symptoms in patients with subclinical hypothyroidism. JAMA. 2018;320(13):1349-1359.
39. Cole DP, Thase ME, Mallinger AG, et al. Slower treatment response in bipolar depression predicted by lower pretreatment thyroid function. Am J Psychiatry. 2002;159(1):116-121.
40. Parmentier T, Sienaert P. The use of triiodothyronine (T3) in the treatment of bipolar depression: a review of the literature. J Affect Disord. 2018;229:410-414.
41. Koda-Kimbe MA, Alldredge BK. Koda-Kimble and Young’s applied therapeutics: the clinical use of drugs (10th ed). Baltimore, MD: Walters Klower Health/Lippincott Williams & Wilkins; 2012.
42. Moreno C, Laje G, Blanco C, et al. National trends in the outpatient diagnosis and treatment of bipolar disorder in youth. Arch Gen Psychiatry. 2007;64(9):1032-1039.
1. Miller GE, Noel RL. Controversies in bipolar disorder: trust evidence or experience? Current Psychiatry. 2009;8(2):27-28,31-33,39.
2. Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry. 2004;6(1):27-33.
3. Ghaemi SN, Sachs GS, Chiou AM, et al. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord. 1999;52(1-3):135-144.
4. Blanco C, Laje G, Olfson M, et al. Trends in the treatment of bipolar disorder by outpatient psychiatrists. Am J Psychiatry. 2002;159(6):1005-1010.
5. Shi L, Thiebaud P, McCombs JS. The impact of unrecognized bipolar disorders for patients treated with antidepressants in the fee-for-services California Medicaid (Medi-Cal) program. J Affect Disord. 2004;82(3):373-383.
6. Sidor MM, MacQueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156-167.
7. Hughes T, Cardno A, West R, et al. Unrecognized bipolar disorder among UK primary care patients prescribed antidepressants: an observational study. Br J Gen Pract. 2016;66(643):e71-e77.
8. Keck PE Jr, Kessler RC, Ross R. Clinical and economic effects of unrecognized or inadequately treated bipolar disorder. J Psychiatric Pract. 2008;14(Suppl 2):31-38.
9. Goldberg JF, Harrow M, Whiteside JF. Risk for bipolar illness in inpatients initially hospitalized for unipolar depression. Am J Psychiatry. 2001:158(8):1265-1270.
10. Chilakamarri JK, Filkowski MM, Ghaemi SN. Misdiagnosis of bipolar disorder in children and adolescents: a comparison with ADHD and major depressive disorder. Ann Clin Psychiatry. 2011;23(1):25-29.
11. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
12. Bowden CL. A different depression: clinical distinctions between bipolar and unipolar depression. J Affect Disord. 2005;84(2-3):117-125.
13. Baldassano C. Distinctions between bipolar I and bipolar II depression. Current Psychiatry. 2017;16(8):S7-S16.
14. Hirschfeld MA, Williams JB, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: The Mood Disorder Questionnaire. Am J Psychiatry. 2000;157(11):1873-1875.
15. Ghaemi SN, Miller CJ, Berv DA, et al. Sensitivity and specificity a new bipolar spectrum diagnostic scale. J Affect Disorder. 2005;84(2-3):273-277
16. Suppes T, Leverich G, Keck P, et al. The Stanley Foundation Continuing Bipolar Treatment Outcome Network. II. Demographics and illness characteristics of the first 261 patients. J Affect Disorder. 2001;67(1-3):45-49.
17. Perlis RH, Miyahara S, Marangell LB. Long-term implications of early onset in bipolar disorder: data from the first 1000 participants in the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP–BD). Biol Psychiatry. 2004;55(9):875-881.
18. Baldessarini RJ, Bolzani L, Kruz N, et al. Onset age of bipolar disorders at six international sites. J Affect Disord. 2010;121(1-2):143-146.
19. Post RM, Altshuler LL, Kupka R, et al. More childhood onset bipolar disorder in the United States than Canada or Europe: implications for treatment and prevention. Neurosci Biobehav Rev. 2017;74(Pt A):204-213.
20. Geller B, Luby J. Child and adolescent bipolar disorder: a review of the past 10 years. J Am Acad Child Adolesc Psychiatry. 1997;36(9):1168-1176.
21. Findling RL, Gracious BL, McNamara NK, et al. Rapid, continuous cycling and psychiatric co-morbidity in pediatric bipolar I disorder. Bipolar Disord. 2001;3(4):202-210.
22. Arnold LM. Gender differences in bipolar disorder. Psychiatr Clin North Am. 2003;26(3):595-620.
23. Deflorio A, Jones I. Is sex important? Gender differences in bipolar disorder. Int Rev Psychiatry. 2010;22(5):437-452.
24. Bogren M, Brådvik L, Holmstrand C, et al. Gender differences in subtypes of depression by first incidence and age of onset: a follow-up of the Lunby population. Eur Arch Psychiatry Clin Neurosci. 2018;268(2):179-189.
25. Singhal A, Ross J, Seminog O, et al. Risks of self-harm and suicide in people with specific psychiatric and physical disorders: comparisons between disorders using English national record linkage. J R Soc Med. 2014;107(5):194-204.
26. Joshi G, Wilens T. Comorbidity in pediatric bipolar disorder. Child Adolesc Psychiatr Clin N Amer. 2009;18(2):291-319.
27. Youngtrom EA, Arnold LE, Frazier TW. Bipolar and ADHD comorbidity: both artifact and outgrowth of shared mechanisms. Clin Psychol (New York). 2010;17(4):350-359.
28. McIntyre RS, Kennedy SH, Soczynska JK, et al. Attention-deficit/hyperactivity disorder in adults with bipolar disorder or major depressive disorder: results from the International Mood Disorders Collaborative Project. Prime Care Companion J Clin Psychiatry. 2010;12(3). doi:10.4088/PCC.09m00861gry.
29. Regier DA, Farmer ME, Rae DS, et al. Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiological Catchment Area (ECA) Study. JAMA. 1990;264(19):2511-2518.
30. Hunt GE, Malhi GS, Cleary M, et al. Prevalence of comorbid bipolar and substance use disorders in clinical settings, 1990-2015: systematic review and meta-analysis. J Affect Disord. 2016;206:331-349.
31. Agargun MY, Besiroglu L, Cilli AS, et al. Nightmares, suicide attempts, and melancholic features in patients with unipolar major depression. J Affect Disord. 2007;98(3):267-270.
32. Birmaher B, Axelson D, Monk K, et al. Lifetime psychiatric disorders in school-aged offspring of parents with bipolar disorder: the Pittsburgh Bipolar Offspring Study. Arch Gen Psychiatry. 2009;66(3):287-296.
33. Rush AJ, Trivedi MH, Wisniewski SR, et al. Buproprion-SR, sertraline, or venlafaxine-XR after failure of SSRIs for depression. N Engl J Med. 2006;354(12):1231-1242.
34. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer term outcomes in depressed outpatients requiring one or several treatment steps: A STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917.
35. Kirsch I. Antidepressants and the placebo effect. Z Psychol. 2014;222(3):128-134.
36. U.S. Food and Drug Administration. Suicidality in children and adolescents being treated with antidepressant medications. https://www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/ucm161679.htm. Published February 5, 2018. Accessed May 10, 2019.
37. Jennissen S, Huber J, Ehrenthal JC, et al. Association between insight and outcome of psychotherapy; systematic review and meta-analysis. Am J Psychiatry. 2018;175(10):961-969.
38. Feller M, Snel M, Moutzouri E, et al. Association of thyroid hormone therapy with quality of life and thyroid-related symptoms in patients with subclinical hypothyroidism. JAMA. 2018;320(13):1349-1359.
39. Cole DP, Thase ME, Mallinger AG, et al. Slower treatment response in bipolar depression predicted by lower pretreatment thyroid function. Am J Psychiatry. 2002;159(1):116-121.
40. Parmentier T, Sienaert P. The use of triiodothyronine (T3) in the treatment of bipolar depression: a review of the literature. J Affect Disord. 2018;229:410-414.
41. Koda-Kimbe MA, Alldredge BK. Koda-Kimble and Young’s applied therapeutics: the clinical use of drugs (10th ed). Baltimore, MD: Walters Klower Health/Lippincott Williams & Wilkins; 2012.
42. Moreno C, Laje G, Blanco C, et al. National trends in the outpatient diagnosis and treatment of bipolar disorder in youth. Arch Gen Psychiatry. 2007;64(9):1032-1039.
Cannabis: Doctors tell FDA to get out of the weeds
The Food and Drug Administration held its first-ever public hearing about products containing cannabis or cannabis-derived compounds – and it got an earful.
Hearing from over 100 individuals, the all-staff FDA panel was asked repeatedly to take the lead in bringing order to a confused morass of state and local cannabis regulations. The regulatory landscape currently contains many voids that slow research and put consumers at risk, many witnesses testified.
The federal Farm Bill of 2018 legalized the cultivation of hemp – cannabis with very low 9-tetrahydrocannabinol (THC) content – with regulatory restrictions.
However, the Farm Bill did not legalize low-THC cannabis products, said FDA Acting Commissioner Norman Sharpless, MD. The agency has concluded that both THC and cannabidiol (CBD) are drugs – not dietary supplements – and any exception to these provisions “would be new terrain for the FDA,” he said.
And although restrictions on CBD sales have generally not been enforced, “under current law, CBD and THC cannot lawfully be added to a food or marketed as a dietary supplement,” said Dr. Sharpless.
Though the FDA could choose to carve out regulatory exceptions, it has not yet done so.
Stakeholders who gave testimony included not just physicians, scientists, consumers, and advocates, but also growers, manufacturers, distributors, and retailers – as well as the legal firms that represent these interests.
Broadly, physicians and scientists encouraged the FDA to move forward with classifying CBD and most CBD-containing products as drugs, rather than dietary supplements. In general, the opposite approach was promoted by agriculture and manufacturing representatives who testified.
However, all were united in asking the FDA for clarity – and alacrity.
Again and again, speakers asked the FDA to move posthaste in tidying up the current clutter of regulations. Ryan Vandrey, PhD, of Johns Hopkins University, Baltimore, explained that today, “Hemp-derived CBD is unscheduled, Epidiolex is Schedule V, and synthetic CBD is Schedule I in the DEA’s current framework.”
Kevin Chapman, MD, of Children’s Hospital Colorado, representing the American Epilepsy Society, called for regulation of CBD as a drug, and an accelerated clinical trial path. He noted that many families of children with epilepsy other than Lennox-Gastaut or Dravet syndrome (the only approved Epidiolex indications) are dosing other CBD products, “making it up as they go along."
Jacqueline French MD, chief scientific officer of the Epilepsy Foundation, agreed that many families of children with epilepsy are doing their best to find consistent and unadulterated CBD products. She said her worry is that “abrupt withdrawal of CBD from the market could lead to seizure worsening, injury, or even death for patients who now rely on non-pharmaceutical CBD for seizure control.”
Dr. French and Dr. Chapman each made the point that without insurance reimbursement, Epidiolex costs families over $30,000 annually, while CBD is a small fraction of that – as little as $1,000, said Dr. Chapman.
The lack of standards for CBD preparations means the content of oils, tinctures, and e-cigarette products can vary widely. Michelle Peace, PhD, a forensic scientist at Virginia Commonwealth University, Richmond, receives funding from the U.S. Department of Justice to study licit and illicit drug use with e-cigarettes. She and her colleagues have found dextromethorphan and the potent synthetic cannabinoid 5F-ADB in vaping supplies advertised as containing only CBD.
In a recent investigation prompted by multiple consumer reports of hallucinations after vaping CBD-labeled products, “17 of 18 samples contained a synthetic cannabinoid. Clinics will not find these kinds of drugs when they just do drug testing,” Dr. Peace said.
Another sampling of CBD products available from retail outlets showed that claims often bore little relation to cannabinoid content, said Bill Gurley, PhD, co-director of the Center for Dietary Supplement Research at the University of Arkansas, Little Rock. While several products that claimed to have CBD actually contained none at all, some had many more CBD than the labeled amount – 228 times more, in one instance. One tested product was actually 45% THC, with little CBD content.
The potential for CBD to interact with many other drugs is cause for concern, noted several presenters. Barry Gidal, PharmD, director of pharmacy practice at the University of Wisconsin, Madison, said that “CBD is a complicated molecule. It has a complicated biotransformation pathway” through at least 9 enzymes within the hepatic cytochrome P450 system.
“It wasn’t until the Epidiolex development program that we began to understand the clinical ramifications of this…. The effect of CBD on other drugs may go beyond the anti-seizure drugs that have been studied so far,” said Dr. Gidal. He pointed to recent published case reports of potential CBD-drug interactions reporting elevated international normalized ratios for a patient on warfarin using CBD, and another report of elevated tacrolimus levels in a patient using CBD.
The way forward
A variety of regulatory pathways were proposed at the hearing. To prevent adulteration and contamination issues, many advocated standardized good manufacturing practices (GMPs), product reporting, and identification or registration, and a centralized reporting registry for adverse events.
Patient advocates, physicians, and scientists called for an easing of access to cannabis for medical research. Currently, cannabis, still classified as a Schedule I substance by the Drug Enforcement Administration, is only legally available for this purpose through a supply maintained by the National Institute on Drug Abuse. A limited number of strains are available, and access requires a lengthy approval process.
Most discussion centered around CBD, though some presenters asked for smoother sailing for THC research as well, particularly as a potential adjunct or alternative to opioids for chronic pain. Cannabidiol has generally been recognized as non-psychoactive, and the FDA assigned it a very low probability of causing dependence or addiction problems in its own review of human data.
However, in his opening remarks, Dr. Sharpless warned that this fact does not make CBD a benign substance, and many questions remain unanswered.
“How much CBD is safe to consume in a day? What if someone applies a topical CBD lotion, consumes a CBD beverage or candy, and also consumes some CBD oil? How much is too much? How will it interact with other drugs the person might be taking? What if she’s pregnant? What if children access CBD products like gummy edibles? What happens when someone chronically uses CBD for prolonged periods? These and many other questions represent important and significant gaps in our knowledge,” said Dr. Sharpless.
The FDA has established a public docket where the public may submit comments and documents until July 2, 2019.
The Food and Drug Administration held its first-ever public hearing about products containing cannabis or cannabis-derived compounds – and it got an earful.
Hearing from over 100 individuals, the all-staff FDA panel was asked repeatedly to take the lead in bringing order to a confused morass of state and local cannabis regulations. The regulatory landscape currently contains many voids that slow research and put consumers at risk, many witnesses testified.
The federal Farm Bill of 2018 legalized the cultivation of hemp – cannabis with very low 9-tetrahydrocannabinol (THC) content – with regulatory restrictions.
However, the Farm Bill did not legalize low-THC cannabis products, said FDA Acting Commissioner Norman Sharpless, MD. The agency has concluded that both THC and cannabidiol (CBD) are drugs – not dietary supplements – and any exception to these provisions “would be new terrain for the FDA,” he said.
And although restrictions on CBD sales have generally not been enforced, “under current law, CBD and THC cannot lawfully be added to a food or marketed as a dietary supplement,” said Dr. Sharpless.
Though the FDA could choose to carve out regulatory exceptions, it has not yet done so.
Stakeholders who gave testimony included not just physicians, scientists, consumers, and advocates, but also growers, manufacturers, distributors, and retailers – as well as the legal firms that represent these interests.
Broadly, physicians and scientists encouraged the FDA to move forward with classifying CBD and most CBD-containing products as drugs, rather than dietary supplements. In general, the opposite approach was promoted by agriculture and manufacturing representatives who testified.
However, all were united in asking the FDA for clarity – and alacrity.
Again and again, speakers asked the FDA to move posthaste in tidying up the current clutter of regulations. Ryan Vandrey, PhD, of Johns Hopkins University, Baltimore, explained that today, “Hemp-derived CBD is unscheduled, Epidiolex is Schedule V, and synthetic CBD is Schedule I in the DEA’s current framework.”
Kevin Chapman, MD, of Children’s Hospital Colorado, representing the American Epilepsy Society, called for regulation of CBD as a drug, and an accelerated clinical trial path. He noted that many families of children with epilepsy other than Lennox-Gastaut or Dravet syndrome (the only approved Epidiolex indications) are dosing other CBD products, “making it up as they go along."
Jacqueline French MD, chief scientific officer of the Epilepsy Foundation, agreed that many families of children with epilepsy are doing their best to find consistent and unadulterated CBD products. She said her worry is that “abrupt withdrawal of CBD from the market could lead to seizure worsening, injury, or even death for patients who now rely on non-pharmaceutical CBD for seizure control.”
Dr. French and Dr. Chapman each made the point that without insurance reimbursement, Epidiolex costs families over $30,000 annually, while CBD is a small fraction of that – as little as $1,000, said Dr. Chapman.
The lack of standards for CBD preparations means the content of oils, tinctures, and e-cigarette products can vary widely. Michelle Peace, PhD, a forensic scientist at Virginia Commonwealth University, Richmond, receives funding from the U.S. Department of Justice to study licit and illicit drug use with e-cigarettes. She and her colleagues have found dextromethorphan and the potent synthetic cannabinoid 5F-ADB in vaping supplies advertised as containing only CBD.
In a recent investigation prompted by multiple consumer reports of hallucinations after vaping CBD-labeled products, “17 of 18 samples contained a synthetic cannabinoid. Clinics will not find these kinds of drugs when they just do drug testing,” Dr. Peace said.
Another sampling of CBD products available from retail outlets showed that claims often bore little relation to cannabinoid content, said Bill Gurley, PhD, co-director of the Center for Dietary Supplement Research at the University of Arkansas, Little Rock. While several products that claimed to have CBD actually contained none at all, some had many more CBD than the labeled amount – 228 times more, in one instance. One tested product was actually 45% THC, with little CBD content.
The potential for CBD to interact with many other drugs is cause for concern, noted several presenters. Barry Gidal, PharmD, director of pharmacy practice at the University of Wisconsin, Madison, said that “CBD is a complicated molecule. It has a complicated biotransformation pathway” through at least 9 enzymes within the hepatic cytochrome P450 system.
“It wasn’t until the Epidiolex development program that we began to understand the clinical ramifications of this…. The effect of CBD on other drugs may go beyond the anti-seizure drugs that have been studied so far,” said Dr. Gidal. He pointed to recent published case reports of potential CBD-drug interactions reporting elevated international normalized ratios for a patient on warfarin using CBD, and another report of elevated tacrolimus levels in a patient using CBD.
The way forward
A variety of regulatory pathways were proposed at the hearing. To prevent adulteration and contamination issues, many advocated standardized good manufacturing practices (GMPs), product reporting, and identification or registration, and a centralized reporting registry for adverse events.
Patient advocates, physicians, and scientists called for an easing of access to cannabis for medical research. Currently, cannabis, still classified as a Schedule I substance by the Drug Enforcement Administration, is only legally available for this purpose through a supply maintained by the National Institute on Drug Abuse. A limited number of strains are available, and access requires a lengthy approval process.
Most discussion centered around CBD, though some presenters asked for smoother sailing for THC research as well, particularly as a potential adjunct or alternative to opioids for chronic pain. Cannabidiol has generally been recognized as non-psychoactive, and the FDA assigned it a very low probability of causing dependence or addiction problems in its own review of human data.
However, in his opening remarks, Dr. Sharpless warned that this fact does not make CBD a benign substance, and many questions remain unanswered.
“How much CBD is safe to consume in a day? What if someone applies a topical CBD lotion, consumes a CBD beverage or candy, and also consumes some CBD oil? How much is too much? How will it interact with other drugs the person might be taking? What if she’s pregnant? What if children access CBD products like gummy edibles? What happens when someone chronically uses CBD for prolonged periods? These and many other questions represent important and significant gaps in our knowledge,” said Dr. Sharpless.
The FDA has established a public docket where the public may submit comments and documents until July 2, 2019.
The Food and Drug Administration held its first-ever public hearing about products containing cannabis or cannabis-derived compounds – and it got an earful.
Hearing from over 100 individuals, the all-staff FDA panel was asked repeatedly to take the lead in bringing order to a confused morass of state and local cannabis regulations. The regulatory landscape currently contains many voids that slow research and put consumers at risk, many witnesses testified.
The federal Farm Bill of 2018 legalized the cultivation of hemp – cannabis with very low 9-tetrahydrocannabinol (THC) content – with regulatory restrictions.
However, the Farm Bill did not legalize low-THC cannabis products, said FDA Acting Commissioner Norman Sharpless, MD. The agency has concluded that both THC and cannabidiol (CBD) are drugs – not dietary supplements – and any exception to these provisions “would be new terrain for the FDA,” he said.
And although restrictions on CBD sales have generally not been enforced, “under current law, CBD and THC cannot lawfully be added to a food or marketed as a dietary supplement,” said Dr. Sharpless.
Though the FDA could choose to carve out regulatory exceptions, it has not yet done so.
Stakeholders who gave testimony included not just physicians, scientists, consumers, and advocates, but also growers, manufacturers, distributors, and retailers – as well as the legal firms that represent these interests.
Broadly, physicians and scientists encouraged the FDA to move forward with classifying CBD and most CBD-containing products as drugs, rather than dietary supplements. In general, the opposite approach was promoted by agriculture and manufacturing representatives who testified.
However, all were united in asking the FDA for clarity – and alacrity.
Again and again, speakers asked the FDA to move posthaste in tidying up the current clutter of regulations. Ryan Vandrey, PhD, of Johns Hopkins University, Baltimore, explained that today, “Hemp-derived CBD is unscheduled, Epidiolex is Schedule V, and synthetic CBD is Schedule I in the DEA’s current framework.”
Kevin Chapman, MD, of Children’s Hospital Colorado, representing the American Epilepsy Society, called for regulation of CBD as a drug, and an accelerated clinical trial path. He noted that many families of children with epilepsy other than Lennox-Gastaut or Dravet syndrome (the only approved Epidiolex indications) are dosing other CBD products, “making it up as they go along."
Jacqueline French MD, chief scientific officer of the Epilepsy Foundation, agreed that many families of children with epilepsy are doing their best to find consistent and unadulterated CBD products. She said her worry is that “abrupt withdrawal of CBD from the market could lead to seizure worsening, injury, or even death for patients who now rely on non-pharmaceutical CBD for seizure control.”
Dr. French and Dr. Chapman each made the point that without insurance reimbursement, Epidiolex costs families over $30,000 annually, while CBD is a small fraction of that – as little as $1,000, said Dr. Chapman.
The lack of standards for CBD preparations means the content of oils, tinctures, and e-cigarette products can vary widely. Michelle Peace, PhD, a forensic scientist at Virginia Commonwealth University, Richmond, receives funding from the U.S. Department of Justice to study licit and illicit drug use with e-cigarettes. She and her colleagues have found dextromethorphan and the potent synthetic cannabinoid 5F-ADB in vaping supplies advertised as containing only CBD.
In a recent investigation prompted by multiple consumer reports of hallucinations after vaping CBD-labeled products, “17 of 18 samples contained a synthetic cannabinoid. Clinics will not find these kinds of drugs when they just do drug testing,” Dr. Peace said.
Another sampling of CBD products available from retail outlets showed that claims often bore little relation to cannabinoid content, said Bill Gurley, PhD, co-director of the Center for Dietary Supplement Research at the University of Arkansas, Little Rock. While several products that claimed to have CBD actually contained none at all, some had many more CBD than the labeled amount – 228 times more, in one instance. One tested product was actually 45% THC, with little CBD content.
The potential for CBD to interact with many other drugs is cause for concern, noted several presenters. Barry Gidal, PharmD, director of pharmacy practice at the University of Wisconsin, Madison, said that “CBD is a complicated molecule. It has a complicated biotransformation pathway” through at least 9 enzymes within the hepatic cytochrome P450 system.
“It wasn’t until the Epidiolex development program that we began to understand the clinical ramifications of this…. The effect of CBD on other drugs may go beyond the anti-seizure drugs that have been studied so far,” said Dr. Gidal. He pointed to recent published case reports of potential CBD-drug interactions reporting elevated international normalized ratios for a patient on warfarin using CBD, and another report of elevated tacrolimus levels in a patient using CBD.
The way forward
A variety of regulatory pathways were proposed at the hearing. To prevent adulteration and contamination issues, many advocated standardized good manufacturing practices (GMPs), product reporting, and identification or registration, and a centralized reporting registry for adverse events.
Patient advocates, physicians, and scientists called for an easing of access to cannabis for medical research. Currently, cannabis, still classified as a Schedule I substance by the Drug Enforcement Administration, is only legally available for this purpose through a supply maintained by the National Institute on Drug Abuse. A limited number of strains are available, and access requires a lengthy approval process.
Most discussion centered around CBD, though some presenters asked for smoother sailing for THC research as well, particularly as a potential adjunct or alternative to opioids for chronic pain. Cannabidiol has generally been recognized as non-psychoactive, and the FDA assigned it a very low probability of causing dependence or addiction problems in its own review of human data.
However, in his opening remarks, Dr. Sharpless warned that this fact does not make CBD a benign substance, and many questions remain unanswered.
“How much CBD is safe to consume in a day? What if someone applies a topical CBD lotion, consumes a CBD beverage or candy, and also consumes some CBD oil? How much is too much? How will it interact with other drugs the person might be taking? What if she’s pregnant? What if children access CBD products like gummy edibles? What happens when someone chronically uses CBD for prolonged periods? These and many other questions represent important and significant gaps in our knowledge,” said Dr. Sharpless.
The FDA has established a public docket where the public may submit comments and documents until July 2, 2019.
FROM AN FDA PUBLIC HEARING
Obesity doesn’t hamper flu vaccine response in pregnancy
LJUBLJANA, SLOVENIA – ; indeed, it might actually improve their seroconversion rate, Michelle Clarke reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
She presented a prospective cohort study of 90 women vaccinated against influenza during pregnancy, 24 of whom had a BMI of 30 kg/m2 or more. The impetus for the study was the investigators’ understanding that influenza in pregnancy carries an increased risk of severe complications, obesity is a known risk factor for more severe episodes of influenza, and vaccine responses could potentially be adversely affected by obesity, either because of the associated inflammatory state and altered cytokine profile or inadequate vaccine delivery via the intramuscular route. Yet the impact of obesity on vaccine responses in pregnancy has been unclear.
Blood samples obtained before and 1 month after vaccination showed similarly high-titer postvaccination seropositivity rates against influenza B, H3N2, and H1N1 regardless of the women’s weight status. Indeed, the seropositivity rate against all three influenza viruses was higher in the obese subgroup, by a margin of 92%-74%. Also, postvaccination geometric mean antibody titers were significantly higher in the obese group. Particularly impressive was the difference in H1N1 seroconversion, defined as a fourfold increase in titer 28 days after vaccination: 79% versus 55%, noted Ms. Clarke of the University of Adelaide.
Of note, influenza vaccination in the first trimester resulted in a significantly lower seropositive antibody rate than vaccination in the second or third trimesters. The implication is that gestational age at vaccination, regardless of BMI, may be an important determinant of optimal vaccine protection for mothers and their newborns. However, this tentative conclusion requires confirmation in an independent larger sample, because the patient numbers in the study were small: Seropositive antibodies to all three vaccine antigens were documented in just 7 of 12 women (58%) vaccinated in the first trimester, compared with 47 of 53 (89%) vaccinated in the second trimester and 18 of 25 (72%) in the third.
Ms. Clarke reported having no financial conflicts regarding the study, which was supported by the Women’s and Children’s Hospital Research Foundation.
LJUBLJANA, SLOVENIA – ; indeed, it might actually improve their seroconversion rate, Michelle Clarke reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
She presented a prospective cohort study of 90 women vaccinated against influenza during pregnancy, 24 of whom had a BMI of 30 kg/m2 or more. The impetus for the study was the investigators’ understanding that influenza in pregnancy carries an increased risk of severe complications, obesity is a known risk factor for more severe episodes of influenza, and vaccine responses could potentially be adversely affected by obesity, either because of the associated inflammatory state and altered cytokine profile or inadequate vaccine delivery via the intramuscular route. Yet the impact of obesity on vaccine responses in pregnancy has been unclear.
Blood samples obtained before and 1 month after vaccination showed similarly high-titer postvaccination seropositivity rates against influenza B, H3N2, and H1N1 regardless of the women’s weight status. Indeed, the seropositivity rate against all three influenza viruses was higher in the obese subgroup, by a margin of 92%-74%. Also, postvaccination geometric mean antibody titers were significantly higher in the obese group. Particularly impressive was the difference in H1N1 seroconversion, defined as a fourfold increase in titer 28 days after vaccination: 79% versus 55%, noted Ms. Clarke of the University of Adelaide.
Of note, influenza vaccination in the first trimester resulted in a significantly lower seropositive antibody rate than vaccination in the second or third trimesters. The implication is that gestational age at vaccination, regardless of BMI, may be an important determinant of optimal vaccine protection for mothers and their newborns. However, this tentative conclusion requires confirmation in an independent larger sample, because the patient numbers in the study were small: Seropositive antibodies to all three vaccine antigens were documented in just 7 of 12 women (58%) vaccinated in the first trimester, compared with 47 of 53 (89%) vaccinated in the second trimester and 18 of 25 (72%) in the third.
Ms. Clarke reported having no financial conflicts regarding the study, which was supported by the Women’s and Children’s Hospital Research Foundation.
LJUBLJANA, SLOVENIA – ; indeed, it might actually improve their seroconversion rate, Michelle Clarke reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
She presented a prospective cohort study of 90 women vaccinated against influenza during pregnancy, 24 of whom had a BMI of 30 kg/m2 or more. The impetus for the study was the investigators’ understanding that influenza in pregnancy carries an increased risk of severe complications, obesity is a known risk factor for more severe episodes of influenza, and vaccine responses could potentially be adversely affected by obesity, either because of the associated inflammatory state and altered cytokine profile or inadequate vaccine delivery via the intramuscular route. Yet the impact of obesity on vaccine responses in pregnancy has been unclear.
Blood samples obtained before and 1 month after vaccination showed similarly high-titer postvaccination seropositivity rates against influenza B, H3N2, and H1N1 regardless of the women’s weight status. Indeed, the seropositivity rate against all three influenza viruses was higher in the obese subgroup, by a margin of 92%-74%. Also, postvaccination geometric mean antibody titers were significantly higher in the obese group. Particularly impressive was the difference in H1N1 seroconversion, defined as a fourfold increase in titer 28 days after vaccination: 79% versus 55%, noted Ms. Clarke of the University of Adelaide.
Of note, influenza vaccination in the first trimester resulted in a significantly lower seropositive antibody rate than vaccination in the second or third trimesters. The implication is that gestational age at vaccination, regardless of BMI, may be an important determinant of optimal vaccine protection for mothers and their newborns. However, this tentative conclusion requires confirmation in an independent larger sample, because the patient numbers in the study were small: Seropositive antibodies to all three vaccine antigens were documented in just 7 of 12 women (58%) vaccinated in the first trimester, compared with 47 of 53 (89%) vaccinated in the second trimester and 18 of 25 (72%) in the third.
Ms. Clarke reported having no financial conflicts regarding the study, which was supported by the Women’s and Children’s Hospital Research Foundation.
REPORTING FROM ESPID 2019
Key clinical point: High BMI doesn’t impair influenza vaccine responses in pregnant women.
Major finding: Protective antibody levels against all three vaccine antigens were documented 1 month post vaccination in 92% of the obese and 74% of the nonobese mothers.
Study details: This was a prospective observational study of 90 women vaccinated against influenza during pregnancy, 24 of whom were obese.
Disclosures: The study was supported by the University of Adelaide Women’s and Children’s Hospital Research Foundation.
Weight-based teasing may mean further weight gain in children
according to Natasha A. Schvey, PhD, of the Uniformed Services University of the Health Sciences, Bethesda, Md., and her associates.
The investigators conducted a longitudinal, observational study of 110 children who were either in the 85th body mass index (BMI) percentile or greater or had two parents with a BMI of at least 25 kg/m2. Children were recruited between July 12, 1996, and July 6, 2009, administered the Perception of Teasing Scale at baseline and during follow-up, and followed for up to 15 years. Children were aged a mean of 12 years at baseline, and attended an average of nine visits.
At baseline, 53% of children were overweight, with overweight being more common in girls and in non-Hispanic whites. A total of 62% of children who were overweight at baseline reported at least one incidence of weight-based teasing (WBT), compared with 21% of children at risk. WBT at baseline was associated with BMI throughout the study (P less than .001). In addition, children who reported more WBT showed a steeper gain in BMI (P = .007). Overall, children who reported high levels of WBT had 33% greater gains in BMI per year than those with no WBT.
Fat mass was associated with WBT in a similar manner, but to an increased extent, as children who reported high levels of WBT gained 91% more fat per year than those with no WBT.
“As adolescence marks a critical period for the study of weight gain, it will be important to further explore the effects of WBT and weight‐related pressures on indices of weight and health throughout development and to identify both risk and protective factors. The present findings ... may provide a foundation upon which to initiate clinical pediatric interventions to determine whether reducing WBT affects weight and fat gain trajectory,” the investigators concluded.
The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The investigators did not report any conflicts of interest.
SOURCE: Schvey NA et al. Pediatr Obes. 2019 May 29. doi: 10.1111/ijpo.12538.
according to Natasha A. Schvey, PhD, of the Uniformed Services University of the Health Sciences, Bethesda, Md., and her associates.
The investigators conducted a longitudinal, observational study of 110 children who were either in the 85th body mass index (BMI) percentile or greater or had two parents with a BMI of at least 25 kg/m2. Children were recruited between July 12, 1996, and July 6, 2009, administered the Perception of Teasing Scale at baseline and during follow-up, and followed for up to 15 years. Children were aged a mean of 12 years at baseline, and attended an average of nine visits.
At baseline, 53% of children were overweight, with overweight being more common in girls and in non-Hispanic whites. A total of 62% of children who were overweight at baseline reported at least one incidence of weight-based teasing (WBT), compared with 21% of children at risk. WBT at baseline was associated with BMI throughout the study (P less than .001). In addition, children who reported more WBT showed a steeper gain in BMI (P = .007). Overall, children who reported high levels of WBT had 33% greater gains in BMI per year than those with no WBT.
Fat mass was associated with WBT in a similar manner, but to an increased extent, as children who reported high levels of WBT gained 91% more fat per year than those with no WBT.
“As adolescence marks a critical period for the study of weight gain, it will be important to further explore the effects of WBT and weight‐related pressures on indices of weight and health throughout development and to identify both risk and protective factors. The present findings ... may provide a foundation upon which to initiate clinical pediatric interventions to determine whether reducing WBT affects weight and fat gain trajectory,” the investigators concluded.
The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The investigators did not report any conflicts of interest.
SOURCE: Schvey NA et al. Pediatr Obes. 2019 May 29. doi: 10.1111/ijpo.12538.
according to Natasha A. Schvey, PhD, of the Uniformed Services University of the Health Sciences, Bethesda, Md., and her associates.
The investigators conducted a longitudinal, observational study of 110 children who were either in the 85th body mass index (BMI) percentile or greater or had two parents with a BMI of at least 25 kg/m2. Children were recruited between July 12, 1996, and July 6, 2009, administered the Perception of Teasing Scale at baseline and during follow-up, and followed for up to 15 years. Children were aged a mean of 12 years at baseline, and attended an average of nine visits.
At baseline, 53% of children were overweight, with overweight being more common in girls and in non-Hispanic whites. A total of 62% of children who were overweight at baseline reported at least one incidence of weight-based teasing (WBT), compared with 21% of children at risk. WBT at baseline was associated with BMI throughout the study (P less than .001). In addition, children who reported more WBT showed a steeper gain in BMI (P = .007). Overall, children who reported high levels of WBT had 33% greater gains in BMI per year than those with no WBT.
Fat mass was associated with WBT in a similar manner, but to an increased extent, as children who reported high levels of WBT gained 91% more fat per year than those with no WBT.
“As adolescence marks a critical period for the study of weight gain, it will be important to further explore the effects of WBT and weight‐related pressures on indices of weight and health throughout development and to identify both risk and protective factors. The present findings ... may provide a foundation upon which to initiate clinical pediatric interventions to determine whether reducing WBT affects weight and fat gain trajectory,” the investigators concluded.
The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The investigators did not report any conflicts of interest.
SOURCE: Schvey NA et al. Pediatr Obes. 2019 May 29. doi: 10.1111/ijpo.12538.
FROM PEDIATRIC OBESITY
Check for complementopathies in lupus pregnancy
SAN FRANCISCO – It’s important to check for complementopathies in pregnant women with lupus, according to Michelle Petri, MD, a professor of rheumatology at Johns Hopkins University, Baltimore.
A new diagnosis being developed at Hopkins and elsewhere, complementopathies involve an inappropriate activation of the alternative pathway of complement (APC), either from a mutation in a complement control protein, or, in the case of lupus, an autoantibody against one. They’ve been implicated as a major cause of hemolysis, elevated liver enzymes, low platelets (HELLP) syndrome, a condition to which women with lupus are particularly prone.
Hopkins has developed a serum test to diagnose inappropriate APC activation in a few hours, the modified Ham test. When HELLP develops in a woman with a complementopathy, the complement inhibitor eculizumab (Soliris) is proving to be a safe alternative to pregnancy termination.
“I urge you to think about using the modified Ham test, because if it is positive, you can treat HELLP without having to stop the pregnancy,” said Dr. Petri, also codirector of the Hopkins Lupus Pregnancy Center.
Lupus management has come a long way from the days when women were counseled to avoid or terminate pregnancy. Risks remain, “but many pregnancies are successful. I think that for every woman with lupus, we do want to offer the possibility of successful pregnancy,” she said.
Disease control is key. Preterm birth, the most common adverse outcome in lupus, correlates closely with disease activity, and disease activity can be controlled with hydroxychloroquine, and, when needed, azathioprine and tacrolimus for renal flairs.
“But these kinds of basic lessons – we need hydroxychloroquine in pregnancy; we must control disease activity – are not heard out in the real world. Claims data have shown that pregnant women with lupus actually take fewer prescribed medications, and they have fewer rheumatology visits.” It’s a problem that needs to be addressed, Dr. Petri said.
Vitamin D is also important. Hopkins has shown that supplementation to hit a level of 40 ng/mL reduces both global and renal disease activity without toxicity; studies in the general population have shown reduced preeclampsia, preterm birth, and low birth weight, all concerns in lupus.
“I haven’t convinced the world of lupus how important vitamin D is,” but “I actually love it just as much as I love hydroxychloroquine,” Dr. Petri said.
Cosupplementation with calcium complicates matters. Together, they seem to reduce the risk of preeclampsia, but increase the risk of preterm birth. More needs to be known, so “for all of us with pregnancy cohorts, it’s time to start to record vitamin D and calcium levels so we can look at this,” she said.
Dr. Petri has worked with numerous companies.
SAN FRANCISCO – It’s important to check for complementopathies in pregnant women with lupus, according to Michelle Petri, MD, a professor of rheumatology at Johns Hopkins University, Baltimore.
A new diagnosis being developed at Hopkins and elsewhere, complementopathies involve an inappropriate activation of the alternative pathway of complement (APC), either from a mutation in a complement control protein, or, in the case of lupus, an autoantibody against one. They’ve been implicated as a major cause of hemolysis, elevated liver enzymes, low platelets (HELLP) syndrome, a condition to which women with lupus are particularly prone.
Hopkins has developed a serum test to diagnose inappropriate APC activation in a few hours, the modified Ham test. When HELLP develops in a woman with a complementopathy, the complement inhibitor eculizumab (Soliris) is proving to be a safe alternative to pregnancy termination.
“I urge you to think about using the modified Ham test, because if it is positive, you can treat HELLP without having to stop the pregnancy,” said Dr. Petri, also codirector of the Hopkins Lupus Pregnancy Center.
Lupus management has come a long way from the days when women were counseled to avoid or terminate pregnancy. Risks remain, “but many pregnancies are successful. I think that for every woman with lupus, we do want to offer the possibility of successful pregnancy,” she said.
Disease control is key. Preterm birth, the most common adverse outcome in lupus, correlates closely with disease activity, and disease activity can be controlled with hydroxychloroquine, and, when needed, azathioprine and tacrolimus for renal flairs.
“But these kinds of basic lessons – we need hydroxychloroquine in pregnancy; we must control disease activity – are not heard out in the real world. Claims data have shown that pregnant women with lupus actually take fewer prescribed medications, and they have fewer rheumatology visits.” It’s a problem that needs to be addressed, Dr. Petri said.
Vitamin D is also important. Hopkins has shown that supplementation to hit a level of 40 ng/mL reduces both global and renal disease activity without toxicity; studies in the general population have shown reduced preeclampsia, preterm birth, and low birth weight, all concerns in lupus.
“I haven’t convinced the world of lupus how important vitamin D is,” but “I actually love it just as much as I love hydroxychloroquine,” Dr. Petri said.
Cosupplementation with calcium complicates matters. Together, they seem to reduce the risk of preeclampsia, but increase the risk of preterm birth. More needs to be known, so “for all of us with pregnancy cohorts, it’s time to start to record vitamin D and calcium levels so we can look at this,” she said.
Dr. Petri has worked with numerous companies.
SAN FRANCISCO – It’s important to check for complementopathies in pregnant women with lupus, according to Michelle Petri, MD, a professor of rheumatology at Johns Hopkins University, Baltimore.
A new diagnosis being developed at Hopkins and elsewhere, complementopathies involve an inappropriate activation of the alternative pathway of complement (APC), either from a mutation in a complement control protein, or, in the case of lupus, an autoantibody against one. They’ve been implicated as a major cause of hemolysis, elevated liver enzymes, low platelets (HELLP) syndrome, a condition to which women with lupus are particularly prone.
Hopkins has developed a serum test to diagnose inappropriate APC activation in a few hours, the modified Ham test. When HELLP develops in a woman with a complementopathy, the complement inhibitor eculizumab (Soliris) is proving to be a safe alternative to pregnancy termination.
“I urge you to think about using the modified Ham test, because if it is positive, you can treat HELLP without having to stop the pregnancy,” said Dr. Petri, also codirector of the Hopkins Lupus Pregnancy Center.
Lupus management has come a long way from the days when women were counseled to avoid or terminate pregnancy. Risks remain, “but many pregnancies are successful. I think that for every woman with lupus, we do want to offer the possibility of successful pregnancy,” she said.
Disease control is key. Preterm birth, the most common adverse outcome in lupus, correlates closely with disease activity, and disease activity can be controlled with hydroxychloroquine, and, when needed, azathioprine and tacrolimus for renal flairs.
“But these kinds of basic lessons – we need hydroxychloroquine in pregnancy; we must control disease activity – are not heard out in the real world. Claims data have shown that pregnant women with lupus actually take fewer prescribed medications, and they have fewer rheumatology visits.” It’s a problem that needs to be addressed, Dr. Petri said.
Vitamin D is also important. Hopkins has shown that supplementation to hit a level of 40 ng/mL reduces both global and renal disease activity without toxicity; studies in the general population have shown reduced preeclampsia, preterm birth, and low birth weight, all concerns in lupus.
“I haven’t convinced the world of lupus how important vitamin D is,” but “I actually love it just as much as I love hydroxychloroquine,” Dr. Petri said.
Cosupplementation with calcium complicates matters. Together, they seem to reduce the risk of preeclampsia, but increase the risk of preterm birth. More needs to be known, so “for all of us with pregnancy cohorts, it’s time to start to record vitamin D and calcium levels so we can look at this,” she said.
Dr. Petri has worked with numerous companies.
EXPERT ANALYSIS FROM LUPUS 2019
Burnout Among Vascular Surgeons – A Report From the SVS Wellness Committee
Physician burnout has been linked to medical errors, decreased patient satisfaction, and reduced career longevity. In light of the increasing prevalence of cardiovascular disease, vascular surgeon burnout presents a legitimate public health concern because of the impact on the adequacy of the vascular surgery workforce. Dawn Coleman, MD, and her colleagues, performed a study on behalf of the Society for Vascular Surgery (SVS) Wellness Task Force to define the prevalence of burnout among practicing vascular surgeons, and to identify the risk factors for burnout. Such information will be used to facilitate future SVS initiatives to avert this crisis.
In Thursday’s von Liebig Forum, Dr. Coleman, of the University of Michigan, Ann Arbor, will present the results of their 2018 anonymous survey of active SVS members. The survey used a validated burnout assessment, Maslach Burnout Index (MBI), embedded in a questionnaire that also captured demographic and practice-related characteristics.
The survey was personalized for the specialty and did allow for free text. according to Dr. Coleman, and “we specifically analyzed emotional exhaustion, one dimension of burnout.”
The accepted threshold of a score of 27 or greater on the MBI Emotional Exhaustion module was used to identify surgeons suffering from burnout. Risk factors for such were identified using bivariate analyses (Chi-square, Kruskal-Wallis), and multivariate logistic regression models were developed to identify independent risk factors for burnout, she added.
Dr. Coleman will present the results from the 960 out of 2,905 active SVS members who responded to the survey (33%). After excluding retired surgeons and incomplete submissions, responses from 872 practicing vascular surgeons were finally analyzed. The mean respondent age was 49.7 years; and the majority of respondents (81%) were men. The primary practice settings were academic (40%), community practice (41%), Veterans Administration hospital (3.3%), active military practice (1.5%), or “other.” Mean years in practice was 15.7.
Overall, 30% of the respondents met criteria for burnout, 37% screened positive for symptoms of depression in the past month and 8% supported thoughts of taking their own life during the last 12 months.
By unadjusted analysis, factors significantly associated with burnout included clinical work hours, on-call frequency, electronic medical record/documentation requirements, perceived conflict between work and personal responsibilities, and physical pain. Multivariate analysis revealed age, work-related physical pain, and conflict between work and personal responsibilities as independent risk factors for burnout, said Dr. Coleman.
“Approximately one-third of practicing vascular surgeons self-report burnout and depression, according to our survey. Advancing age, physical pain, and work-life conflict are each independent predictors for burnout among vascular surgeons. These findings will facilitate SVS efforts to improve vascular surgeon well-being, in an effort to mitigate the personal, economic, and social impact of vascular surgeon burnout,” Dr. Coleman concluded.
See more on the work of the SVS Wellness Task Force in the June issue of Vascular Specialist.
Physician burnout has been linked to medical errors, decreased patient satisfaction, and reduced career longevity. In light of the increasing prevalence of cardiovascular disease, vascular surgeon burnout presents a legitimate public health concern because of the impact on the adequacy of the vascular surgery workforce. Dawn Coleman, MD, and her colleagues, performed a study on behalf of the Society for Vascular Surgery (SVS) Wellness Task Force to define the prevalence of burnout among practicing vascular surgeons, and to identify the risk factors for burnout. Such information will be used to facilitate future SVS initiatives to avert this crisis.
In Thursday’s von Liebig Forum, Dr. Coleman, of the University of Michigan, Ann Arbor, will present the results of their 2018 anonymous survey of active SVS members. The survey used a validated burnout assessment, Maslach Burnout Index (MBI), embedded in a questionnaire that also captured demographic and practice-related characteristics.
The survey was personalized for the specialty and did allow for free text. according to Dr. Coleman, and “we specifically analyzed emotional exhaustion, one dimension of burnout.”
The accepted threshold of a score of 27 or greater on the MBI Emotional Exhaustion module was used to identify surgeons suffering from burnout. Risk factors for such were identified using bivariate analyses (Chi-square, Kruskal-Wallis), and multivariate logistic regression models were developed to identify independent risk factors for burnout, she added.
Dr. Coleman will present the results from the 960 out of 2,905 active SVS members who responded to the survey (33%). After excluding retired surgeons and incomplete submissions, responses from 872 practicing vascular surgeons were finally analyzed. The mean respondent age was 49.7 years; and the majority of respondents (81%) were men. The primary practice settings were academic (40%), community practice (41%), Veterans Administration hospital (3.3%), active military practice (1.5%), or “other.” Mean years in practice was 15.7.
Overall, 30% of the respondents met criteria for burnout, 37% screened positive for symptoms of depression in the past month and 8% supported thoughts of taking their own life during the last 12 months.
By unadjusted analysis, factors significantly associated with burnout included clinical work hours, on-call frequency, electronic medical record/documentation requirements, perceived conflict between work and personal responsibilities, and physical pain. Multivariate analysis revealed age, work-related physical pain, and conflict between work and personal responsibilities as independent risk factors for burnout, said Dr. Coleman.
“Approximately one-third of practicing vascular surgeons self-report burnout and depression, according to our survey. Advancing age, physical pain, and work-life conflict are each independent predictors for burnout among vascular surgeons. These findings will facilitate SVS efforts to improve vascular surgeon well-being, in an effort to mitigate the personal, economic, and social impact of vascular surgeon burnout,” Dr. Coleman concluded.
See more on the work of the SVS Wellness Task Force in the June issue of Vascular Specialist.
Physician burnout has been linked to medical errors, decreased patient satisfaction, and reduced career longevity. In light of the increasing prevalence of cardiovascular disease, vascular surgeon burnout presents a legitimate public health concern because of the impact on the adequacy of the vascular surgery workforce. Dawn Coleman, MD, and her colleagues, performed a study on behalf of the Society for Vascular Surgery (SVS) Wellness Task Force to define the prevalence of burnout among practicing vascular surgeons, and to identify the risk factors for burnout. Such information will be used to facilitate future SVS initiatives to avert this crisis.
In Thursday’s von Liebig Forum, Dr. Coleman, of the University of Michigan, Ann Arbor, will present the results of their 2018 anonymous survey of active SVS members. The survey used a validated burnout assessment, Maslach Burnout Index (MBI), embedded in a questionnaire that also captured demographic and practice-related characteristics.
The survey was personalized for the specialty and did allow for free text. according to Dr. Coleman, and “we specifically analyzed emotional exhaustion, one dimension of burnout.”
The accepted threshold of a score of 27 or greater on the MBI Emotional Exhaustion module was used to identify surgeons suffering from burnout. Risk factors for such were identified using bivariate analyses (Chi-square, Kruskal-Wallis), and multivariate logistic regression models were developed to identify independent risk factors for burnout, she added.
Dr. Coleman will present the results from the 960 out of 2,905 active SVS members who responded to the survey (33%). After excluding retired surgeons and incomplete submissions, responses from 872 practicing vascular surgeons were finally analyzed. The mean respondent age was 49.7 years; and the majority of respondents (81%) were men. The primary practice settings were academic (40%), community practice (41%), Veterans Administration hospital (3.3%), active military practice (1.5%), or “other.” Mean years in practice was 15.7.
Overall, 30% of the respondents met criteria for burnout, 37% screened positive for symptoms of depression in the past month and 8% supported thoughts of taking their own life during the last 12 months.
By unadjusted analysis, factors significantly associated with burnout included clinical work hours, on-call frequency, electronic medical record/documentation requirements, perceived conflict between work and personal responsibilities, and physical pain. Multivariate analysis revealed age, work-related physical pain, and conflict between work and personal responsibilities as independent risk factors for burnout, said Dr. Coleman.
“Approximately one-third of practicing vascular surgeons self-report burnout and depression, according to our survey. Advancing age, physical pain, and work-life conflict are each independent predictors for burnout among vascular surgeons. These findings will facilitate SVS efforts to improve vascular surgeon well-being, in an effort to mitigate the personal, economic, and social impact of vascular surgeon burnout,” Dr. Coleman concluded.
See more on the work of the SVS Wellness Task Force in the June issue of Vascular Specialist.
Gender Disparity and Sexual Harassment in Vascular Surgery Practices
Sexual harassment is known to be more pervasive in male-dominated workplaces and flourishes in a climate of tolerance and culture of silence, according to Bernadette Aulivola, MD, of Loyola University Medical Center, Maywood, Ill.
“We sought to examine the prevalence of sexual harassment in academic vascular surgery practices, identify factors associated with occurrence, determine reporting barriers, and identify any gender bias that exists,” said Dr. Aulivola.
In Wednesday’s VESS session, Dr. Aulivola will present the results of an anonymous survey that she and Matthew R. Smeds, MD, of Saint Louis University, conducted to examine the issue. The survey was emailed to 346 vascular surgery faculty members at 52 training sites in the United States.
“This research stemmed from a similar project done in vascular surgery trainees (residents and trainees) that identified a significant amount of harassment occurring at this level with a concomitant fear of reporting and lack of knowledge of institutional reporting mechanisms. We thought an analysis of harassment at the attending physician level may be relevant. There is significant gender bias in medicine in general and a not insignificant rate of harassment that occurs,” Dr. Smeds said in an interview.
Of the invitations sent, 149 (43%) completed the survey. Among these, 48/149 (32%) thought harassment occurred more commonly in surgical specialties with historical male dominance. In addition, ignoring the behavior, and hierarchy/power dynamics were the most common reasons given for its occurrence. Overall, 61/149 (41%) reported experiencing workplace harassment. Being told unwanted sexually explicit comments/questions/jokes, being called a sexist slur/nickname, or being paid unwanted flirtation were the most commonly described behaviors.
Those harassed were significantly more likely to be female (37% vs. 13%), and on average had experienced 2.6 (of 10) types of harassment. Despite 84% of respondents acknowledging institutional reporting mechanisms, only 7.2% of the harassing behaviors were reported.
The most common reasons for not reporting including feeling the behavior was “harmless” (67%) or “nothing positive would come of it” (28%), although 30% of respondents feared repercussions or felt uncomfortable identifying as a target of sexual harassment and only 59% of respondents reported that they would feel comfortable discussing the issue with departmental/divisional leadership.
“A significant number of vascular surgeons in academic practice have experienced workplace sexual harassment,” Dr. Aulivola said. “While most are aware of institutional reporting mechanisms, very few events are reported and less than 60% of respondents feel comfortable reporting to departmental/divisional leadership. Female vascular surgeons believe gender influences hiring, promotion, compensation, and attainment of life goals. Further work is necessary to identify methods of reducing workplace sexual harassment and optimize gender disparity in academic vascular surgery practice,” she concluded.
Wednesday, June 12
12:30-4:15 p.m.
Gaylord National, Maryland D
V2: VESS Paper Session 2: VESS18
Sexual harassment is known to be more pervasive in male-dominated workplaces and flourishes in a climate of tolerance and culture of silence, according to Bernadette Aulivola, MD, of Loyola University Medical Center, Maywood, Ill.
“We sought to examine the prevalence of sexual harassment in academic vascular surgery practices, identify factors associated with occurrence, determine reporting barriers, and identify any gender bias that exists,” said Dr. Aulivola.
In Wednesday’s VESS session, Dr. Aulivola will present the results of an anonymous survey that she and Matthew R. Smeds, MD, of Saint Louis University, conducted to examine the issue. The survey was emailed to 346 vascular surgery faculty members at 52 training sites in the United States.
“This research stemmed from a similar project done in vascular surgery trainees (residents and trainees) that identified a significant amount of harassment occurring at this level with a concomitant fear of reporting and lack of knowledge of institutional reporting mechanisms. We thought an analysis of harassment at the attending physician level may be relevant. There is significant gender bias in medicine in general and a not insignificant rate of harassment that occurs,” Dr. Smeds said in an interview.
Of the invitations sent, 149 (43%) completed the survey. Among these, 48/149 (32%) thought harassment occurred more commonly in surgical specialties with historical male dominance. In addition, ignoring the behavior, and hierarchy/power dynamics were the most common reasons given for its occurrence. Overall, 61/149 (41%) reported experiencing workplace harassment. Being told unwanted sexually explicit comments/questions/jokes, being called a sexist slur/nickname, or being paid unwanted flirtation were the most commonly described behaviors.
Those harassed were significantly more likely to be female (37% vs. 13%), and on average had experienced 2.6 (of 10) types of harassment. Despite 84% of respondents acknowledging institutional reporting mechanisms, only 7.2% of the harassing behaviors were reported.
The most common reasons for not reporting including feeling the behavior was “harmless” (67%) or “nothing positive would come of it” (28%), although 30% of respondents feared repercussions or felt uncomfortable identifying as a target of sexual harassment and only 59% of respondents reported that they would feel comfortable discussing the issue with departmental/divisional leadership.
“A significant number of vascular surgeons in academic practice have experienced workplace sexual harassment,” Dr. Aulivola said. “While most are aware of institutional reporting mechanisms, very few events are reported and less than 60% of respondents feel comfortable reporting to departmental/divisional leadership. Female vascular surgeons believe gender influences hiring, promotion, compensation, and attainment of life goals. Further work is necessary to identify methods of reducing workplace sexual harassment and optimize gender disparity in academic vascular surgery practice,” she concluded.
Wednesday, June 12
12:30-4:15 p.m.
Gaylord National, Maryland D
V2: VESS Paper Session 2: VESS18
Sexual harassment is known to be more pervasive in male-dominated workplaces and flourishes in a climate of tolerance and culture of silence, according to Bernadette Aulivola, MD, of Loyola University Medical Center, Maywood, Ill.
“We sought to examine the prevalence of sexual harassment in academic vascular surgery practices, identify factors associated with occurrence, determine reporting barriers, and identify any gender bias that exists,” said Dr. Aulivola.
In Wednesday’s VESS session, Dr. Aulivola will present the results of an anonymous survey that she and Matthew R. Smeds, MD, of Saint Louis University, conducted to examine the issue. The survey was emailed to 346 vascular surgery faculty members at 52 training sites in the United States.
“This research stemmed from a similar project done in vascular surgery trainees (residents and trainees) that identified a significant amount of harassment occurring at this level with a concomitant fear of reporting and lack of knowledge of institutional reporting mechanisms. We thought an analysis of harassment at the attending physician level may be relevant. There is significant gender bias in medicine in general and a not insignificant rate of harassment that occurs,” Dr. Smeds said in an interview.
Of the invitations sent, 149 (43%) completed the survey. Among these, 48/149 (32%) thought harassment occurred more commonly in surgical specialties with historical male dominance. In addition, ignoring the behavior, and hierarchy/power dynamics were the most common reasons given for its occurrence. Overall, 61/149 (41%) reported experiencing workplace harassment. Being told unwanted sexually explicit comments/questions/jokes, being called a sexist slur/nickname, or being paid unwanted flirtation were the most commonly described behaviors.
Those harassed were significantly more likely to be female (37% vs. 13%), and on average had experienced 2.6 (of 10) types of harassment. Despite 84% of respondents acknowledging institutional reporting mechanisms, only 7.2% of the harassing behaviors were reported.
The most common reasons for not reporting including feeling the behavior was “harmless” (67%) or “nothing positive would come of it” (28%), although 30% of respondents feared repercussions or felt uncomfortable identifying as a target of sexual harassment and only 59% of respondents reported that they would feel comfortable discussing the issue with departmental/divisional leadership.
“A significant number of vascular surgeons in academic practice have experienced workplace sexual harassment,” Dr. Aulivola said. “While most are aware of institutional reporting mechanisms, very few events are reported and less than 60% of respondents feel comfortable reporting to departmental/divisional leadership. Female vascular surgeons believe gender influences hiring, promotion, compensation, and attainment of life goals. Further work is necessary to identify methods of reducing workplace sexual harassment and optimize gender disparity in academic vascular surgery practice,” she concluded.
Wednesday, June 12
12:30-4:15 p.m.
Gaylord National, Maryland D
V2: VESS Paper Session 2: VESS18
Cultural competence behaviors linked to higher patient satisfaction scores
results from a single-center study showed.
“Cultural competence is valued by patients, and there is a potential for training providers to be more culturally competent, focusing on their behaviors,” lead study author Miquell O. Miller, MD, said at the annual Digestive Disease Week.
While the Society for Surgery of the Alimentary Tract (J Gastrointest Surg 2016;20[5]:879-84) and other medical organizations have recognized the importance of workforce diversity and cultural competence of providers, little is known of the relationship between cultural competence and patient-reported outcomes in surgery, said Dr. Miller, who is a general surgery resident at Stanford (Calif.) University. To investigate this relationship, she and her colleagues recruited surgeons, oncologists, gastroenterologists, and advanced practice providers to complete a validated online survey that measured two domains of cultural competency: awareness and behaviors. They matched these scores with the 10-item Press Ganey provider care scores from 2017 to 2018. Next, the researchers conducted a linear regression analysis with mixed effects to account for clustering of patients within providers. They also adjusted for provider bias by measuring social desirability, “which is the tendency for respondents to put a more socially appropriate answer as opposed to the true answer on the cultural competence survey,” Dr. Miller explained.
A total of 1,322 Press Ganey satisfaction surveys were available for 29 providers. Their mean age was 48 years, 59% were white, 72% were physicians, and 28% were advanced practice providers. They practiced in GI oncology (41%), gastroenterology (31%) and colorectal surgery (28%). Dr. Miller reported that providers who participated in the survey had a mean cultural awareness score of 6.2 out of a possible 7 points, while the mean cultural behavior score was a 4.1 out of a possible 7 points. She and her colleagues observed that providers who had high levels of cultural competence on the behavioral assessment were positively associated with Press Ganey patient satisfaction (P = .039).
“I think we do a poor job of training our providers to be culturally competent, but there are multiple ways to improve behaviors, by teaching people and by having real training for our providers,” Dr. Miller said.
She acknowledged certain limitations of the study, including its single-center design and the fact that not all providers had Press Ganey scores available. The study was funded by the Society for Surgery of the Alimentary Tract and the Black Academic Surgeons Resident Research Award. Dr. Miller reported having no financial disclosures.
results from a single-center study showed.
“Cultural competence is valued by patients, and there is a potential for training providers to be more culturally competent, focusing on their behaviors,” lead study author Miquell O. Miller, MD, said at the annual Digestive Disease Week.
While the Society for Surgery of the Alimentary Tract (J Gastrointest Surg 2016;20[5]:879-84) and other medical organizations have recognized the importance of workforce diversity and cultural competence of providers, little is known of the relationship between cultural competence and patient-reported outcomes in surgery, said Dr. Miller, who is a general surgery resident at Stanford (Calif.) University. To investigate this relationship, she and her colleagues recruited surgeons, oncologists, gastroenterologists, and advanced practice providers to complete a validated online survey that measured two domains of cultural competency: awareness and behaviors. They matched these scores with the 10-item Press Ganey provider care scores from 2017 to 2018. Next, the researchers conducted a linear regression analysis with mixed effects to account for clustering of patients within providers. They also adjusted for provider bias by measuring social desirability, “which is the tendency for respondents to put a more socially appropriate answer as opposed to the true answer on the cultural competence survey,” Dr. Miller explained.
A total of 1,322 Press Ganey satisfaction surveys were available for 29 providers. Their mean age was 48 years, 59% were white, 72% were physicians, and 28% were advanced practice providers. They practiced in GI oncology (41%), gastroenterology (31%) and colorectal surgery (28%). Dr. Miller reported that providers who participated in the survey had a mean cultural awareness score of 6.2 out of a possible 7 points, while the mean cultural behavior score was a 4.1 out of a possible 7 points. She and her colleagues observed that providers who had high levels of cultural competence on the behavioral assessment were positively associated with Press Ganey patient satisfaction (P = .039).
“I think we do a poor job of training our providers to be culturally competent, but there are multiple ways to improve behaviors, by teaching people and by having real training for our providers,” Dr. Miller said.
She acknowledged certain limitations of the study, including its single-center design and the fact that not all providers had Press Ganey scores available. The study was funded by the Society for Surgery of the Alimentary Tract and the Black Academic Surgeons Resident Research Award. Dr. Miller reported having no financial disclosures.
results from a single-center study showed.
“Cultural competence is valued by patients, and there is a potential for training providers to be more culturally competent, focusing on their behaviors,” lead study author Miquell O. Miller, MD, said at the annual Digestive Disease Week.
While the Society for Surgery of the Alimentary Tract (J Gastrointest Surg 2016;20[5]:879-84) and other medical organizations have recognized the importance of workforce diversity and cultural competence of providers, little is known of the relationship between cultural competence and patient-reported outcomes in surgery, said Dr. Miller, who is a general surgery resident at Stanford (Calif.) University. To investigate this relationship, she and her colleagues recruited surgeons, oncologists, gastroenterologists, and advanced practice providers to complete a validated online survey that measured two domains of cultural competency: awareness and behaviors. They matched these scores with the 10-item Press Ganey provider care scores from 2017 to 2018. Next, the researchers conducted a linear regression analysis with mixed effects to account for clustering of patients within providers. They also adjusted for provider bias by measuring social desirability, “which is the tendency for respondents to put a more socially appropriate answer as opposed to the true answer on the cultural competence survey,” Dr. Miller explained.
A total of 1,322 Press Ganey satisfaction surveys were available for 29 providers. Their mean age was 48 years, 59% were white, 72% were physicians, and 28% were advanced practice providers. They practiced in GI oncology (41%), gastroenterology (31%) and colorectal surgery (28%). Dr. Miller reported that providers who participated in the survey had a mean cultural awareness score of 6.2 out of a possible 7 points, while the mean cultural behavior score was a 4.1 out of a possible 7 points. She and her colleagues observed that providers who had high levels of cultural competence on the behavioral assessment were positively associated with Press Ganey patient satisfaction (P = .039).
“I think we do a poor job of training our providers to be culturally competent, but there are multiple ways to improve behaviors, by teaching people and by having real training for our providers,” Dr. Miller said.
She acknowledged certain limitations of the study, including its single-center design and the fact that not all providers had Press Ganey scores available. The study was funded by the Society for Surgery of the Alimentary Tract and the Black Academic Surgeons Resident Research Award. Dr. Miller reported having no financial disclosures.
REPORTING FROM DDW 2019