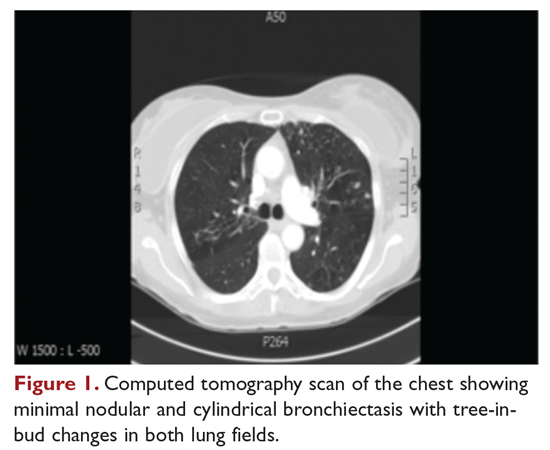
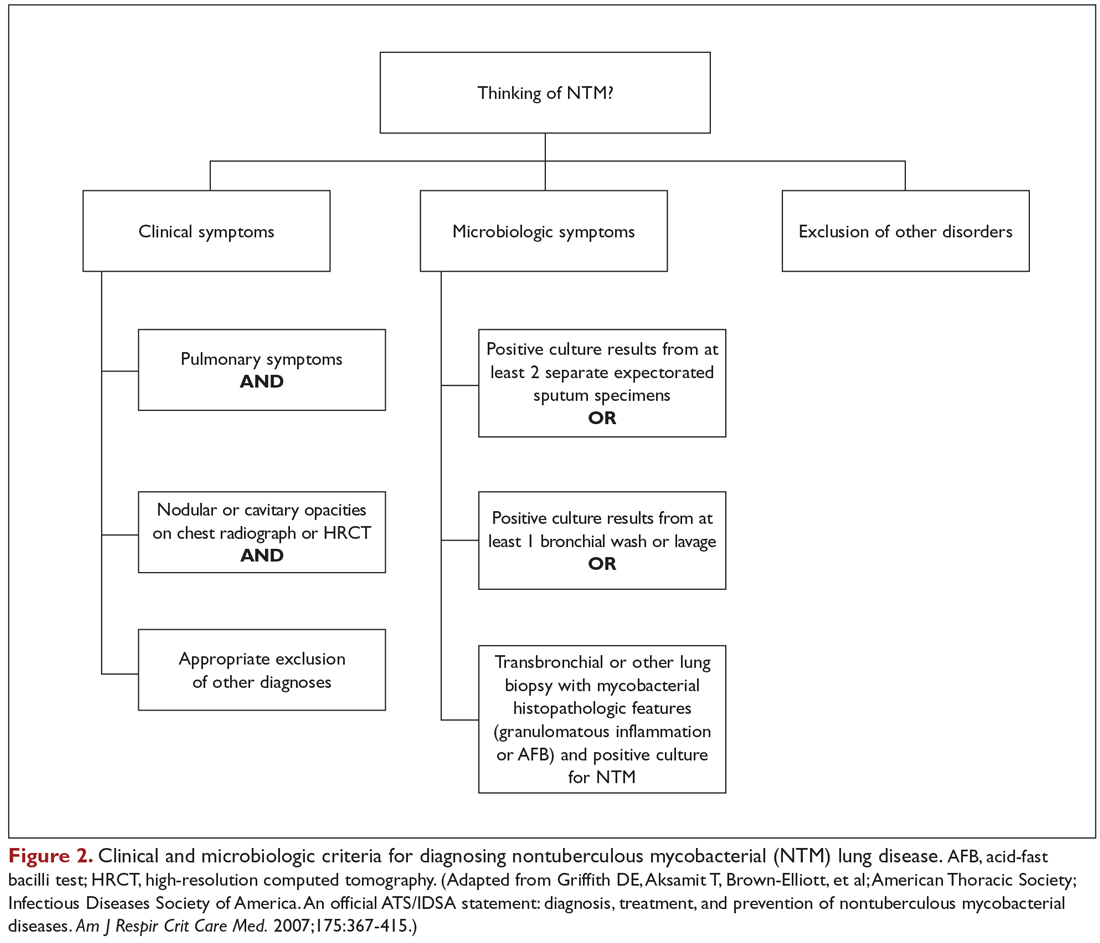
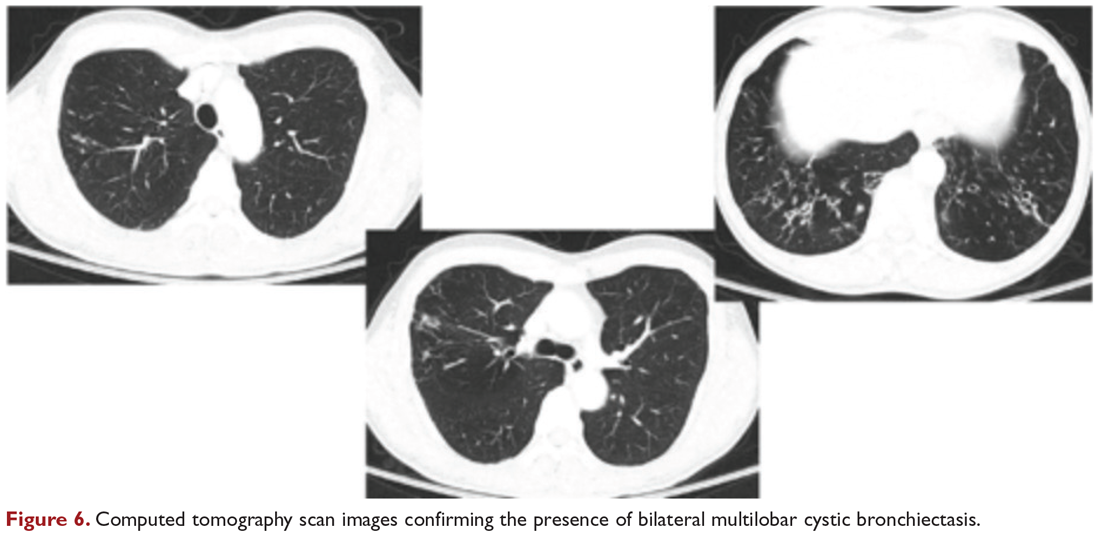
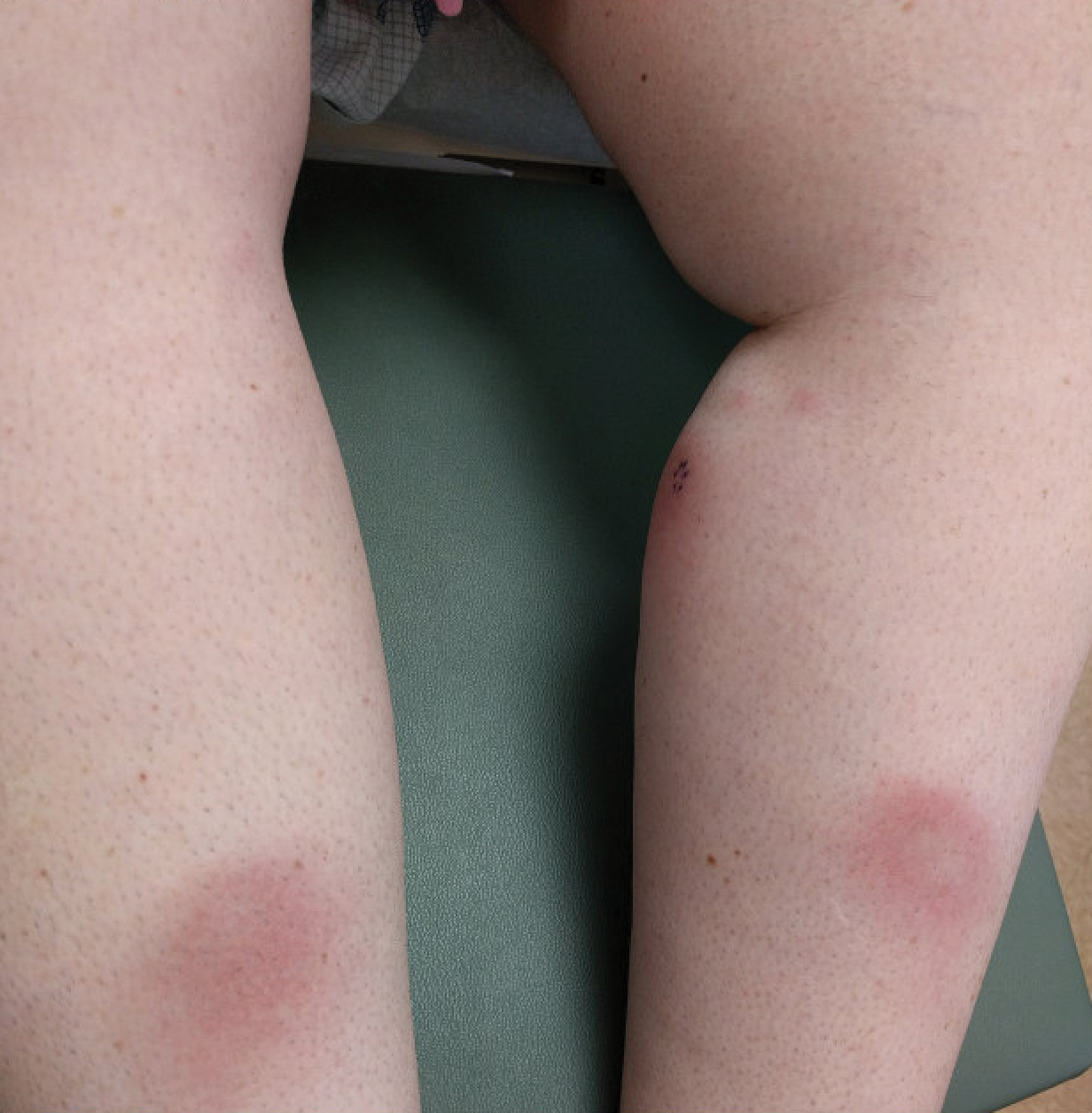
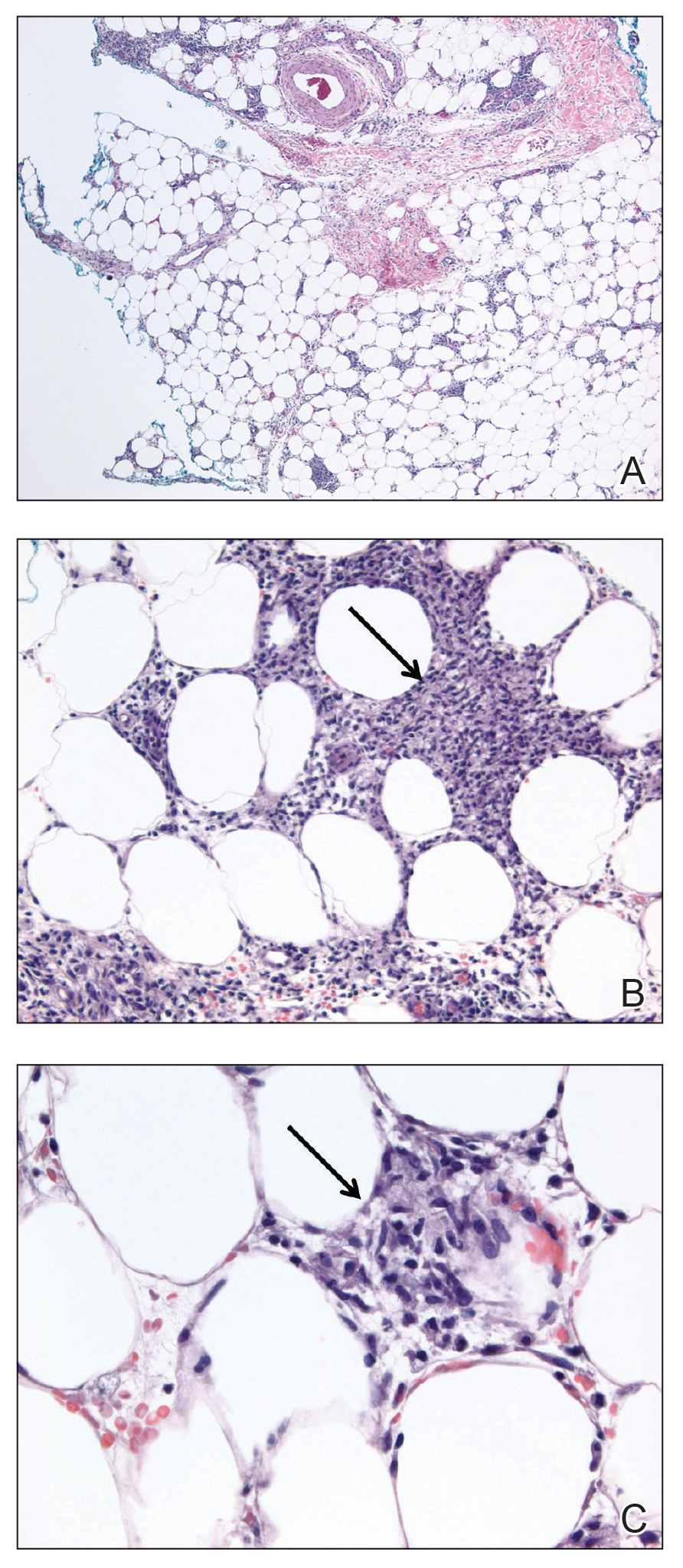
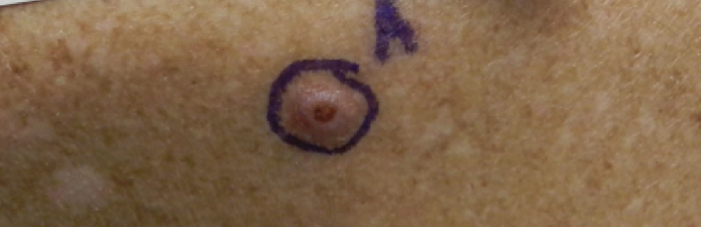
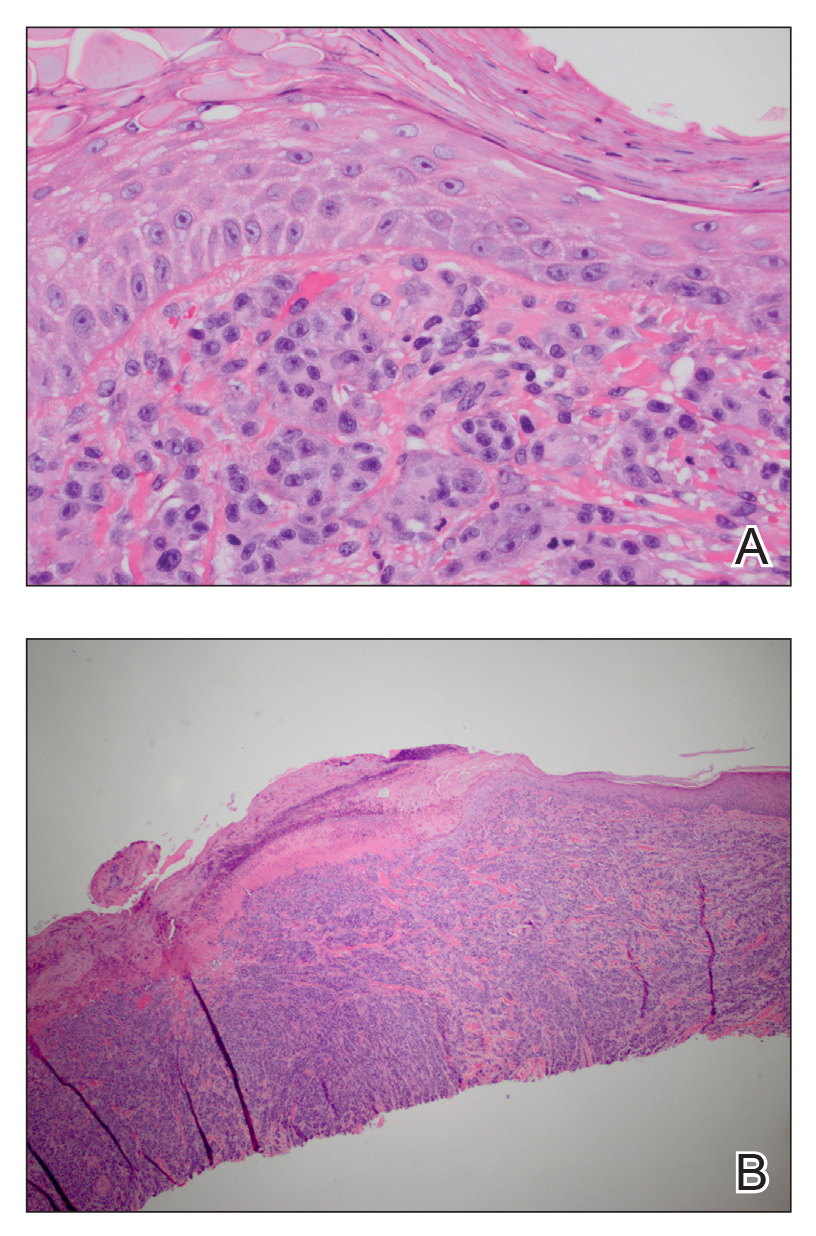
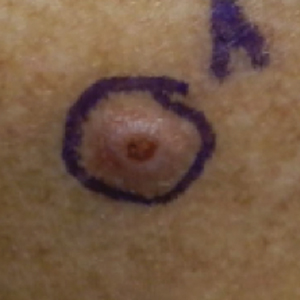
User login
Is primary care relevant?
You probably still remember your pediatrician. Your relationship with her may have influenced your decision to become a physician. She was your parents’ go-to source for pretty much anything to do with your health. You had a primary care physician in large part because your parents felt that children were particularly vulnerable to disease and wanted to avoid any missteps on your road to maturity. On the other hand, while you were growing up your parents probably were much less concerned about their own health. Their peers and friends seemed healthy enough; why would they need annual checkups? Your folks made sure they had life insurance because accidental death and injury felt like more pressing concerns. If they had a primary physician, they may have visited him infrequently. They may have been more likely to visit their dentist, in part because the office put a strong emphasis on the value of preventive care.
A recent survey from Harvard Medical School, Boston, determined that, in 2015, 75% of adult Americans had an established source of primary care. (“Fewer Americans are getting primary care,” Jake Miller, the Harvard Gazette, Dec. 16, 2019). This number sounds pretty good and not unexpected until you learn that in 2002 that number was 77%. While 2% seems like a drop in the bucket, remember we live in a very populous bucket, and that 2% translates to millions fewer Americans who are not receiving primary care than did more than a decade ago.
While the researchers don’t have data to explain the decline in primary care, they suggest raising the pay of primary care physicians, incentivizing rural practice, and making health insurance more available and affordable as solutions. Of course these recommendations are not surprising. We’ve heard them before. More supply might translate into more usage. But could some of the decline in primary care be because it no longer feels relevant to a population that has become accustomed to instant gratification? One click and the thing you didn’t feel like waiting for in line today is on your doorstep tomorrow, or even sooner.
If we want to create meaningful change, we need to learn a thing or two about marketing from the competition and from the successful businesses who are shaping consumer behavior. It’s not surprising that, when people feel healthy (whether they are or not), they will devalue primary care. But if they sprain an ankle or have a cough that is keeping them up at night, they would like some medical attention ... now. And that will drive them away from primary care toward sources of fragmented care – the doc-in-the-box, the walk-in clinic, or even more unfortunately to the local emergency department.
If we want more people to establish relationships with primary care providers, we need to welcome them in the door ... when they feel a need. Once in the door we can establish rapport and show them there is a value to primary care while they are feeling grateful for the prompt attention we gave them. But too many primary care practices are shunting potential patients into fragmented care by appearing unwelcome to minor emergencies and by creating customer-unfriendly communication networks. Most people I know would be happy to go back to the old days of “take two aspirin and call me in the morning” primary care. At least you had talked to a doctor in real time, and you knew that he or she would see you the next day if you still had a problem.
You may think I’ve suddenly gone utopian. But there are ways to run a practice that welcomes patients with minor complaints on short notice. It requires some flexibility, some willingness to work longer on some days, and being more efficient.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
You probably still remember your pediatrician. Your relationship with her may have influenced your decision to become a physician. She was your parents’ go-to source for pretty much anything to do with your health. You had a primary care physician in large part because your parents felt that children were particularly vulnerable to disease and wanted to avoid any missteps on your road to maturity. On the other hand, while you were growing up your parents probably were much less concerned about their own health. Their peers and friends seemed healthy enough; why would they need annual checkups? Your folks made sure they had life insurance because accidental death and injury felt like more pressing concerns. If they had a primary physician, they may have visited him infrequently. They may have been more likely to visit their dentist, in part because the office put a strong emphasis on the value of preventive care.
A recent survey from Harvard Medical School, Boston, determined that, in 2015, 75% of adult Americans had an established source of primary care. (“Fewer Americans are getting primary care,” Jake Miller, the Harvard Gazette, Dec. 16, 2019). This number sounds pretty good and not unexpected until you learn that in 2002 that number was 77%. While 2% seems like a drop in the bucket, remember we live in a very populous bucket, and that 2% translates to millions fewer Americans who are not receiving primary care than did more than a decade ago.
While the researchers don’t have data to explain the decline in primary care, they suggest raising the pay of primary care physicians, incentivizing rural practice, and making health insurance more available and affordable as solutions. Of course these recommendations are not surprising. We’ve heard them before. More supply might translate into more usage. But could some of the decline in primary care be because it no longer feels relevant to a population that has become accustomed to instant gratification? One click and the thing you didn’t feel like waiting for in line today is on your doorstep tomorrow, or even sooner.
If we want to create meaningful change, we need to learn a thing or two about marketing from the competition and from the successful businesses who are shaping consumer behavior. It’s not surprising that, when people feel healthy (whether they are or not), they will devalue primary care. But if they sprain an ankle or have a cough that is keeping them up at night, they would like some medical attention ... now. And that will drive them away from primary care toward sources of fragmented care – the doc-in-the-box, the walk-in clinic, or even more unfortunately to the local emergency department.
If we want more people to establish relationships with primary care providers, we need to welcome them in the door ... when they feel a need. Once in the door we can establish rapport and show them there is a value to primary care while they are feeling grateful for the prompt attention we gave them. But too many primary care practices are shunting potential patients into fragmented care by appearing unwelcome to minor emergencies and by creating customer-unfriendly communication networks. Most people I know would be happy to go back to the old days of “take two aspirin and call me in the morning” primary care. At least you had talked to a doctor in real time, and you knew that he or she would see you the next day if you still had a problem.
You may think I’ve suddenly gone utopian. But there are ways to run a practice that welcomes patients with minor complaints on short notice. It requires some flexibility, some willingness to work longer on some days, and being more efficient.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
You probably still remember your pediatrician. Your relationship with her may have influenced your decision to become a physician. She was your parents’ go-to source for pretty much anything to do with your health. You had a primary care physician in large part because your parents felt that children were particularly vulnerable to disease and wanted to avoid any missteps on your road to maturity. On the other hand, while you were growing up your parents probably were much less concerned about their own health. Their peers and friends seemed healthy enough; why would they need annual checkups? Your folks made sure they had life insurance because accidental death and injury felt like more pressing concerns. If they had a primary physician, they may have visited him infrequently. They may have been more likely to visit their dentist, in part because the office put a strong emphasis on the value of preventive care.
A recent survey from Harvard Medical School, Boston, determined that, in 2015, 75% of adult Americans had an established source of primary care. (“Fewer Americans are getting primary care,” Jake Miller, the Harvard Gazette, Dec. 16, 2019). This number sounds pretty good and not unexpected until you learn that in 2002 that number was 77%. While 2% seems like a drop in the bucket, remember we live in a very populous bucket, and that 2% translates to millions fewer Americans who are not receiving primary care than did more than a decade ago.
While the researchers don’t have data to explain the decline in primary care, they suggest raising the pay of primary care physicians, incentivizing rural practice, and making health insurance more available and affordable as solutions. Of course these recommendations are not surprising. We’ve heard them before. More supply might translate into more usage. But could some of the decline in primary care be because it no longer feels relevant to a population that has become accustomed to instant gratification? One click and the thing you didn’t feel like waiting for in line today is on your doorstep tomorrow, or even sooner.
If we want to create meaningful change, we need to learn a thing or two about marketing from the competition and from the successful businesses who are shaping consumer behavior. It’s not surprising that, when people feel healthy (whether they are or not), they will devalue primary care. But if they sprain an ankle or have a cough that is keeping them up at night, they would like some medical attention ... now. And that will drive them away from primary care toward sources of fragmented care – the doc-in-the-box, the walk-in clinic, or even more unfortunately to the local emergency department.
If we want more people to establish relationships with primary care providers, we need to welcome them in the door ... when they feel a need. Once in the door we can establish rapport and show them there is a value to primary care while they are feeling grateful for the prompt attention we gave them. But too many primary care practices are shunting potential patients into fragmented care by appearing unwelcome to minor emergencies and by creating customer-unfriendly communication networks. Most people I know would be happy to go back to the old days of “take two aspirin and call me in the morning” primary care. At least you had talked to a doctor in real time, and you knew that he or she would see you the next day if you still had a problem.
You may think I’ve suddenly gone utopian. But there are ways to run a practice that welcomes patients with minor complaints on short notice. It requires some flexibility, some willingness to work longer on some days, and being more efficient.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
The 2019 novel coronavirus: Case review IDs clinical characteristics
A group of physicians in Wuhan, China, who are treating patients with the 2019 novel coronavirus have gone the extra mile to share their clinical experiences with colleagues around the world.
Nanshan Chen, MD, of Jinyintan Hospital, Wuhan, and his team conducted a retrospective study on 99 cases and, in very short order, published their initial findings in the Lancet online on Jan. 29. These findings could guide action in other cases and help clinicians all over the world create treatment plans for patients of the 2019-nCoV.
The findings show that and characteristics of those with fatal infections align with the MuLBSTA score – an early warning model for predicting viral pneumonia–related mortality, according to a case review.
Of 99 patients who presented with 2019-nCoV pneumonia at Jinyintan Hospital between Jan. 1 and Jan. 20, 67 were men, the mean age was 55.5 years, and 50 patients had chronic diseases.
“All the data of included cases have been shared with [the World Health Organization]. The study was approved by Jinyintan Hospital Ethics Committee and written informed consent was obtained from patients involved before enrollment when data were collected retrospectively,” the researchers noted.
Nearly half of the patients (49%) lived or worked near a specific seafood market, suggesting disease clustering.
Clinical manifestations affecting the majority of patients included fever and cough in 83% and 82% of patients, respectively. Other symptoms included shortness of breath in 31%, muscle aches in 11%, confusion in 9%, headache in 8%, sore throat in 5%, and rhinorrhea, chest pain, diarrhea, and nausea and vomiting in 1%-4% of patients, the investigators found.
Imaging showed bilateral pneumonia in 75% of cases, multiple mottling and ground-glass opacity in 14%, and pneumothorax in 1%. Organ function damage was present in a third of patients at admission: 17% had acute respiratory distress syndrome (ARDS) – including 11 patients who worsened quickly and died of multiple organ failure. Eight percent had acute respiratory injury, 3% had acute renal injury, 4% had septic shock, and 1% had ventilator-associated pneumonia, they said, noting that all cases were confirmed by real-time polymerase chain reaction.
A notable laboratory finding was reduced absolute lymphocyte counts in most patients, the investigators said.
All patients were treated in isolation and 76% received antiviral treatment with oseltamivir, ganciclovir, lopinavir, or ritonavir for 3-14 days (median, 3 days). Most patients also received antibiotic treatment, including a single antibiotic in 25% of cases and combination therapy in 45%, with most antibiotics used to cover “common pathogens and some atypical pathogens,” they said, adding that “when secondary bacterial infection occurred, medication was administered according to the results of bacterial culture and drug sensitivity.”
Cephalosporins, quinolones, carbapenems, tigecycline against methicillin-resistant Staphylococcus aureus, linezolid, and antifungal drugs were used, and duration ranged from 3 to 17 days (median, 5 days).
Nineteen patients also received steroid treatments.
As of Jan. 25, 31 patients had been discharged and 57 remained hospitalized. Of the 11 who died, the first 2 were a 61-year-old man and a 69-year-old man, each diagnosed with severe pneumonia and ARDS. The first experienced sudden cardiac arrest and died on admission day 11, and the second died of severe pneumonia, septic shock, and respiratory failure on admission day 9. Neither had underlying disease, but both had a long history of smoking, the investigators noted.
“The deaths of these two patients were consistent with the MuLBSTA score,” they wrote, explaining that the scoring system takes into account multilobular infiltration, lymphopenia, bacterial coinfection, smoking history, hypertension, and age.
Eight of the nine other patients who died had lymphopenia, seven had bilateral pneumonia, five were over age 60 years, three had hypertension, and one was a heavy smoker, they added.
Most coronavirus infections cause mild symptoms and have good prognosis, but some patients with the 2019-nCoV, which was identified Jan. 7 following the development of several cases of pneumonia of unknown etiology in Wuhan, develop fatal disease. The paucity of data regarding epidemiology and clinical features of pneumonia associated with 2019-nCoV prompted the current retrospective study at the center where the first cases were admitted, the investigators explained.
They noted that the sequence of 2019-nCoV “is relatively different from the six other coronavirus subtypes, including the highly pathogenic severe acute respiratory syndrome (SARS)-CoV and Middle East Respiratory Syndrome (MERS)-CoV, as well as the human coronaviruses (HCoV)-OC43, -229E, -NL63, and -HKU1 that induce mild upper respiratory disease, but can be classified as a betacoronavirus with evidence of human-to-human transmission.
Mortality associated with SARS-CoV and MERS-CoV have been reported as more than 10% and more than 35%, respectively; at data cutoff for the current study, mortality among the 99 included cases was 11%, which is similar to that in another recent 2019-nCoV report, they said.
The finding of greater risk among older men also has been seen with SARS-CoV and MERS-CoV, and the high rate among individuals with chronic diseases, mainly cerebrovascular disease, cardiovascular disease, and diabetes, also has been reported with MERS-CoV, they added.
“Our results suggest that 2019-nCoV is more likely to infect older adult males with chronic comorbidities as a result of the weaker immune functions of these patients,” they wrote.
Coinfection with bacteria and fungi occurred in some patients, particularly those with severe illness, and cultures most often showed A. baumannii, K. pneumoniae, A. flavus, C. glabrata, and C. albicans, and the findings of reduced absolute lymphocyte values in most patients suggests that “2019-nCoV might mainly act on lymphocytes, especially T lymphocytes, as does SARS-CoV,” they noted.
Given the rapid progression with ARDS and septic shock in some patients in this review, “early identification and timely treatment of critical cases is of crucial importance,” they said.
“Use of intravenous immunoglobulin is recommended to enhance the ability of anti-infection for severely ill patients, and steroids (methylprednisolone 1-2 mg/kg per day) are recommended for patients with ARDS, for as short a duration of treatment as possible,” they added.
Further, since some studies suggest that a substantial decrease in lymphocyte count indicates consumption of many immune cells by coronavirus, thereby inhibiting cellular immune function, damage to T lymphocytes might be “an important factor leading to exacerbations of patients,” they wrote, adding that “[t]he low absolute value of lymphocytes could be used as a reference index in the diagnosis of new coronavirus infections in the clinic.”
The MuLBSTA score also should be investigated to determine its applicability for predicting mortality risk in patients with 2019-nCoV infection, they added.
The current study is limited by its small sample size; additional studies are needed to include “as many patients as possible in Wuhan, in other cities in China, and even in other countries to get a more comprehensive understanding of 2019-nCoV,” they said.
The National Key R&D Program of China funded the study. The authors reported having no conflicts of interest.
SOURCE: Chen N et al. Lancet. 2020 Jan 29. doi: 10.1016/S0140-6736(20)30211-7.
A group of physicians in Wuhan, China, who are treating patients with the 2019 novel coronavirus have gone the extra mile to share their clinical experiences with colleagues around the world.
Nanshan Chen, MD, of Jinyintan Hospital, Wuhan, and his team conducted a retrospective study on 99 cases and, in very short order, published their initial findings in the Lancet online on Jan. 29. These findings could guide action in other cases and help clinicians all over the world create treatment plans for patients of the 2019-nCoV.
The findings show that and characteristics of those with fatal infections align with the MuLBSTA score – an early warning model for predicting viral pneumonia–related mortality, according to a case review.
Of 99 patients who presented with 2019-nCoV pneumonia at Jinyintan Hospital between Jan. 1 and Jan. 20, 67 were men, the mean age was 55.5 years, and 50 patients had chronic diseases.
“All the data of included cases have been shared with [the World Health Organization]. The study was approved by Jinyintan Hospital Ethics Committee and written informed consent was obtained from patients involved before enrollment when data were collected retrospectively,” the researchers noted.
Nearly half of the patients (49%) lived or worked near a specific seafood market, suggesting disease clustering.
Clinical manifestations affecting the majority of patients included fever and cough in 83% and 82% of patients, respectively. Other symptoms included shortness of breath in 31%, muscle aches in 11%, confusion in 9%, headache in 8%, sore throat in 5%, and rhinorrhea, chest pain, diarrhea, and nausea and vomiting in 1%-4% of patients, the investigators found.
Imaging showed bilateral pneumonia in 75% of cases, multiple mottling and ground-glass opacity in 14%, and pneumothorax in 1%. Organ function damage was present in a third of patients at admission: 17% had acute respiratory distress syndrome (ARDS) – including 11 patients who worsened quickly and died of multiple organ failure. Eight percent had acute respiratory injury, 3% had acute renal injury, 4% had septic shock, and 1% had ventilator-associated pneumonia, they said, noting that all cases were confirmed by real-time polymerase chain reaction.
A notable laboratory finding was reduced absolute lymphocyte counts in most patients, the investigators said.
All patients were treated in isolation and 76% received antiviral treatment with oseltamivir, ganciclovir, lopinavir, or ritonavir for 3-14 days (median, 3 days). Most patients also received antibiotic treatment, including a single antibiotic in 25% of cases and combination therapy in 45%, with most antibiotics used to cover “common pathogens and some atypical pathogens,” they said, adding that “when secondary bacterial infection occurred, medication was administered according to the results of bacterial culture and drug sensitivity.”
Cephalosporins, quinolones, carbapenems, tigecycline against methicillin-resistant Staphylococcus aureus, linezolid, and antifungal drugs were used, and duration ranged from 3 to 17 days (median, 5 days).
Nineteen patients also received steroid treatments.
As of Jan. 25, 31 patients had been discharged and 57 remained hospitalized. Of the 11 who died, the first 2 were a 61-year-old man and a 69-year-old man, each diagnosed with severe pneumonia and ARDS. The first experienced sudden cardiac arrest and died on admission day 11, and the second died of severe pneumonia, septic shock, and respiratory failure on admission day 9. Neither had underlying disease, but both had a long history of smoking, the investigators noted.
“The deaths of these two patients were consistent with the MuLBSTA score,” they wrote, explaining that the scoring system takes into account multilobular infiltration, lymphopenia, bacterial coinfection, smoking history, hypertension, and age.
Eight of the nine other patients who died had lymphopenia, seven had bilateral pneumonia, five were over age 60 years, three had hypertension, and one was a heavy smoker, they added.
Most coronavirus infections cause mild symptoms and have good prognosis, but some patients with the 2019-nCoV, which was identified Jan. 7 following the development of several cases of pneumonia of unknown etiology in Wuhan, develop fatal disease. The paucity of data regarding epidemiology and clinical features of pneumonia associated with 2019-nCoV prompted the current retrospective study at the center where the first cases were admitted, the investigators explained.
They noted that the sequence of 2019-nCoV “is relatively different from the six other coronavirus subtypes, including the highly pathogenic severe acute respiratory syndrome (SARS)-CoV and Middle East Respiratory Syndrome (MERS)-CoV, as well as the human coronaviruses (HCoV)-OC43, -229E, -NL63, and -HKU1 that induce mild upper respiratory disease, but can be classified as a betacoronavirus with evidence of human-to-human transmission.
Mortality associated with SARS-CoV and MERS-CoV have been reported as more than 10% and more than 35%, respectively; at data cutoff for the current study, mortality among the 99 included cases was 11%, which is similar to that in another recent 2019-nCoV report, they said.
The finding of greater risk among older men also has been seen with SARS-CoV and MERS-CoV, and the high rate among individuals with chronic diseases, mainly cerebrovascular disease, cardiovascular disease, and diabetes, also has been reported with MERS-CoV, they added.
“Our results suggest that 2019-nCoV is more likely to infect older adult males with chronic comorbidities as a result of the weaker immune functions of these patients,” they wrote.
Coinfection with bacteria and fungi occurred in some patients, particularly those with severe illness, and cultures most often showed A. baumannii, K. pneumoniae, A. flavus, C. glabrata, and C. albicans, and the findings of reduced absolute lymphocyte values in most patients suggests that “2019-nCoV might mainly act on lymphocytes, especially T lymphocytes, as does SARS-CoV,” they noted.
Given the rapid progression with ARDS and septic shock in some patients in this review, “early identification and timely treatment of critical cases is of crucial importance,” they said.
“Use of intravenous immunoglobulin is recommended to enhance the ability of anti-infection for severely ill patients, and steroids (methylprednisolone 1-2 mg/kg per day) are recommended for patients with ARDS, for as short a duration of treatment as possible,” they added.
Further, since some studies suggest that a substantial decrease in lymphocyte count indicates consumption of many immune cells by coronavirus, thereby inhibiting cellular immune function, damage to T lymphocytes might be “an important factor leading to exacerbations of patients,” they wrote, adding that “[t]he low absolute value of lymphocytes could be used as a reference index in the diagnosis of new coronavirus infections in the clinic.”
The MuLBSTA score also should be investigated to determine its applicability for predicting mortality risk in patients with 2019-nCoV infection, they added.
The current study is limited by its small sample size; additional studies are needed to include “as many patients as possible in Wuhan, in other cities in China, and even in other countries to get a more comprehensive understanding of 2019-nCoV,” they said.
The National Key R&D Program of China funded the study. The authors reported having no conflicts of interest.
SOURCE: Chen N et al. Lancet. 2020 Jan 29. doi: 10.1016/S0140-6736(20)30211-7.
A group of physicians in Wuhan, China, who are treating patients with the 2019 novel coronavirus have gone the extra mile to share their clinical experiences with colleagues around the world.
Nanshan Chen, MD, of Jinyintan Hospital, Wuhan, and his team conducted a retrospective study on 99 cases and, in very short order, published their initial findings in the Lancet online on Jan. 29. These findings could guide action in other cases and help clinicians all over the world create treatment plans for patients of the 2019-nCoV.
The findings show that and characteristics of those with fatal infections align with the MuLBSTA score – an early warning model for predicting viral pneumonia–related mortality, according to a case review.
Of 99 patients who presented with 2019-nCoV pneumonia at Jinyintan Hospital between Jan. 1 and Jan. 20, 67 were men, the mean age was 55.5 years, and 50 patients had chronic diseases.
“All the data of included cases have been shared with [the World Health Organization]. The study was approved by Jinyintan Hospital Ethics Committee and written informed consent was obtained from patients involved before enrollment when data were collected retrospectively,” the researchers noted.
Nearly half of the patients (49%) lived or worked near a specific seafood market, suggesting disease clustering.
Clinical manifestations affecting the majority of patients included fever and cough in 83% and 82% of patients, respectively. Other symptoms included shortness of breath in 31%, muscle aches in 11%, confusion in 9%, headache in 8%, sore throat in 5%, and rhinorrhea, chest pain, diarrhea, and nausea and vomiting in 1%-4% of patients, the investigators found.
Imaging showed bilateral pneumonia in 75% of cases, multiple mottling and ground-glass opacity in 14%, and pneumothorax in 1%. Organ function damage was present in a third of patients at admission: 17% had acute respiratory distress syndrome (ARDS) – including 11 patients who worsened quickly and died of multiple organ failure. Eight percent had acute respiratory injury, 3% had acute renal injury, 4% had septic shock, and 1% had ventilator-associated pneumonia, they said, noting that all cases were confirmed by real-time polymerase chain reaction.
A notable laboratory finding was reduced absolute lymphocyte counts in most patients, the investigators said.
All patients were treated in isolation and 76% received antiviral treatment with oseltamivir, ganciclovir, lopinavir, or ritonavir for 3-14 days (median, 3 days). Most patients also received antibiotic treatment, including a single antibiotic in 25% of cases and combination therapy in 45%, with most antibiotics used to cover “common pathogens and some atypical pathogens,” they said, adding that “when secondary bacterial infection occurred, medication was administered according to the results of bacterial culture and drug sensitivity.”
Cephalosporins, quinolones, carbapenems, tigecycline against methicillin-resistant Staphylococcus aureus, linezolid, and antifungal drugs were used, and duration ranged from 3 to 17 days (median, 5 days).
Nineteen patients also received steroid treatments.
As of Jan. 25, 31 patients had been discharged and 57 remained hospitalized. Of the 11 who died, the first 2 were a 61-year-old man and a 69-year-old man, each diagnosed with severe pneumonia and ARDS. The first experienced sudden cardiac arrest and died on admission day 11, and the second died of severe pneumonia, septic shock, and respiratory failure on admission day 9. Neither had underlying disease, but both had a long history of smoking, the investigators noted.
“The deaths of these two patients were consistent with the MuLBSTA score,” they wrote, explaining that the scoring system takes into account multilobular infiltration, lymphopenia, bacterial coinfection, smoking history, hypertension, and age.
Eight of the nine other patients who died had lymphopenia, seven had bilateral pneumonia, five were over age 60 years, three had hypertension, and one was a heavy smoker, they added.
Most coronavirus infections cause mild symptoms and have good prognosis, but some patients with the 2019-nCoV, which was identified Jan. 7 following the development of several cases of pneumonia of unknown etiology in Wuhan, develop fatal disease. The paucity of data regarding epidemiology and clinical features of pneumonia associated with 2019-nCoV prompted the current retrospective study at the center where the first cases were admitted, the investigators explained.
They noted that the sequence of 2019-nCoV “is relatively different from the six other coronavirus subtypes, including the highly pathogenic severe acute respiratory syndrome (SARS)-CoV and Middle East Respiratory Syndrome (MERS)-CoV, as well as the human coronaviruses (HCoV)-OC43, -229E, -NL63, and -HKU1 that induce mild upper respiratory disease, but can be classified as a betacoronavirus with evidence of human-to-human transmission.
Mortality associated with SARS-CoV and MERS-CoV have been reported as more than 10% and more than 35%, respectively; at data cutoff for the current study, mortality among the 99 included cases was 11%, which is similar to that in another recent 2019-nCoV report, they said.
The finding of greater risk among older men also has been seen with SARS-CoV and MERS-CoV, and the high rate among individuals with chronic diseases, mainly cerebrovascular disease, cardiovascular disease, and diabetes, also has been reported with MERS-CoV, they added.
“Our results suggest that 2019-nCoV is more likely to infect older adult males with chronic comorbidities as a result of the weaker immune functions of these patients,” they wrote.
Coinfection with bacteria and fungi occurred in some patients, particularly those with severe illness, and cultures most often showed A. baumannii, K. pneumoniae, A. flavus, C. glabrata, and C. albicans, and the findings of reduced absolute lymphocyte values in most patients suggests that “2019-nCoV might mainly act on lymphocytes, especially T lymphocytes, as does SARS-CoV,” they noted.
Given the rapid progression with ARDS and septic shock in some patients in this review, “early identification and timely treatment of critical cases is of crucial importance,” they said.
“Use of intravenous immunoglobulin is recommended to enhance the ability of anti-infection for severely ill patients, and steroids (methylprednisolone 1-2 mg/kg per day) are recommended for patients with ARDS, for as short a duration of treatment as possible,” they added.
Further, since some studies suggest that a substantial decrease in lymphocyte count indicates consumption of many immune cells by coronavirus, thereby inhibiting cellular immune function, damage to T lymphocytes might be “an important factor leading to exacerbations of patients,” they wrote, adding that “[t]he low absolute value of lymphocytes could be used as a reference index in the diagnosis of new coronavirus infections in the clinic.”
The MuLBSTA score also should be investigated to determine its applicability for predicting mortality risk in patients with 2019-nCoV infection, they added.
The current study is limited by its small sample size; additional studies are needed to include “as many patients as possible in Wuhan, in other cities in China, and even in other countries to get a more comprehensive understanding of 2019-nCoV,” they said.
The National Key R&D Program of China funded the study. The authors reported having no conflicts of interest.
SOURCE: Chen N et al. Lancet. 2020 Jan 29. doi: 10.1016/S0140-6736(20)30211-7.
FROM THE LANCET
World Cancer Day survey exposes ‘glaring inequities’
The first international public survey on cancer perceptions and attitudes in a decade shows that, in spite of progress, low socioeconomic status and lack of education continue to jeopardize the health of the world’s most vulnerable populations.
The survey was commissioned by the Union for International Cancer Control (UICC) to mark the 20th anniversary of World Cancer Day on Feb. 4, 2020.
The survey, which was conducted by Ipsos, was taken by more than 15,000 people in 20 countries. It shows that people of lower socioeconomic status are less likely than those in higher-income households to recognize the risk factors for cancer or to make lifestyle changes. With the exception of tobacco use, people with low educational attainment also showed less cancer awareness and were less likely to engage in preventive behaviors than those with a university degree.
It is “unacceptable that millions of people have a greater chance of developing cancer in their lifetime because they are simply not aware of the cancer risks to avoid and the healthy behaviors to adopt – information that many of us take for granted. And this is true around the world,” Cary Adams, MBA, CEO of the UICC, commented in a statement.
The survey was conducted from Oct. 25 to Nov., 2019, and included 15,427 participants from Australia, Bolivia, Brazil, Canada, China, France, Germany, Great Britain, India, Israel, Japan, Kenya, Mexico, the Philippines, Saudi Arabia, South Africa, Spain, Sweden, Turkey, and the United States.
The vast majority of those surveyed – 87% – said they were aware of the major risk factors for cancer, while only 6% said they were not.
The cancer risk factors that were most recognized were tobacco use (63%), ultraviolet light exposure (54%), and exposure to secondhand tobacco smoke (50%).
The cancer risks that were least recognized included being overweight (29%), a lack of exercise (28%), and exposure to certain viruses or bacteria (28%).
The difference in awareness across the social spectrum was striking. “Emerging from the survey are the apparent and glaring inequities faced by socioeconomically disadvantaged groups,” the authors said.
“Much more must be done to ensure that everyone has an equal chance to reduce their risk of preventable cancer,” commented Sonali Johnson, PhD, head of knowledge, advocacy, and policy at the UICC in Geneva, Switzerland.
“We’ve seen in the results that those surveyed with a lower education and those on lower incomes appear less aware of the main risk factors associated with cancer and thus are less likely to proactively take the steps needed to reduce their cancer risk as compared to those from a high income household or those with a university education,” Dr. Johnson said in an interview.
Does increased cancer awareness translate into behavioral change for the better? This question can only be answered by more research, the survey authors said. They reported that 7 of 10 survey respondents (69%) said they had made a behavioral change to reduce their cancer risk within the past 12 months. Most said they were eating more healthfully.
Slightly fewer than one-quarter reported that they had not taken any preventive measures in the past year.
When it comes to raising cancer awareness, World Cancer Day is “a powerful tool to remind every person that they can play a crucial role in reducing the impact of cancer,” said Dr. Johnson.
Health care providers are “crucial”
Reacting to the findings of the survey, the European Society for Medical Oncology (ESMO) emphasized the key role that physicians play in cancer prevention.
“Research speaks very clearly for prevention,” said ESMO President Solange Peters, MD, PhD. “With the number of cancer cases expected to rise to 29.5 million by 2040, we must act now. ESMO is committed to educating doctors on all aspects of cancer control, which should begin well before a cancer diagnosis.
“In the face of this emergency, which is rendered even more salient by the results of the report, we must work to enlarge the basis of doctors who are properly educated and trained in key prevention measures,” Dr. Peters added. “General practitioners and organ specialists are in the front line to guide and support patients on their quest for healthy lifestyles and reliable ways to detect cancer early.”
In a comment, Dr. Johnson acknowledged the role physicians play in health promotion and informing patients about noncommunicable disease risks, including those related to cancer. However, she emphasized that nurses, pharmacists, community health workers, midwives, and other health care providers who deliver primary care “are crucial around the world to imparting health information and offering services.”
Frontline health care workers can assess patients’ cancer knowledge and health literacy, determine the barriers to health care, and assess “how best to engage with people across the life course,” Dr. Johnson explained. “Rather than just focusing on physicians, we must work with all those involved in primary care, especially as primary care services are scaled up to achieve universal health coverage.”
Call on governments to do more
The authors noted that, although there is wide awareness of the cancer risks from tobacco use, adults younger than 35 years were less likely than those older than 50 to identify tobacco as a cancer risk factor. They described this finding as “most concerning” and said it “underscores the ongoing need to raise awareness about cancer risk factors in every new generation.”
Almost 60% of survey respondents, regardless of age, education, or income, expressed concern about being diagnosed with cancer in the future or having cancer recur.
In Kenya, where the death toll from cancer rose 30% from 2014 to 2018, people appeared to be the most worried about cancer, with four of five survey respondents (82%) expressing concern.
Survey respondents from Saudi Arabia appeared the least concerned, with one of three people saying they were worried.
Notably, 84% of survey respondents said that governments should be doing more to increase cancer prevention and awareness; 33% demanded that governments improve the affordability of cancer care.
“It is understandable that people turn to their governments for support,” Dr. Johnson commented. “Affordability is a big challenge for low-income settings.”
Data from the World Health Organization show that, for every U.S. $1 invested in low- and middle-income countries, the return is U.S. $3.20, Johnson pointed out. “We really need to convince decision makers ... and see the right investments being made. It is important to ensure that the health system strengthening takes place in tandem with prevention services.”
Governments have begun making commitments to tackle noncommunicable diseases and cancer, Dr. Johnson commented. He highlighted the WHO’s Global Action Plan for Healthy Lives and Well-being for All and the updated cancer resolution adopted at the 2017 World Health Assembly.
“Education, training, and awareness-raising efforts need to be backed by strong and progressive health policies that prioritize prevention and help reduce the consumption of known cancer-causing products such as tobacco, sugary food, and beverages,” she said. “Countries should also invest proactively in national cancer control planning and the establishment of population-based registries to ensure the most effective resource allocation that benefits all groups.”
Up-to-date information on cancer risks and cancer prevention must be delivered to the public in ways that are accessible to those in lower socioeconomic groups, Dr. Johnson added. “Awareness needs to be raised continuously with each new generation,” she noted.
The UICC has relationships with Astellas, Daiichi Sankyo, Diaceutics, MSD Inventing for Life, Bristol-Myers Squibb, CUBEBIO, the Icon Group, Roche, and Sanofi.
This article first appeared on Medscape.com.
The first international public survey on cancer perceptions and attitudes in a decade shows that, in spite of progress, low socioeconomic status and lack of education continue to jeopardize the health of the world’s most vulnerable populations.
The survey was commissioned by the Union for International Cancer Control (UICC) to mark the 20th anniversary of World Cancer Day on Feb. 4, 2020.
The survey, which was conducted by Ipsos, was taken by more than 15,000 people in 20 countries. It shows that people of lower socioeconomic status are less likely than those in higher-income households to recognize the risk factors for cancer or to make lifestyle changes. With the exception of tobacco use, people with low educational attainment also showed less cancer awareness and were less likely to engage in preventive behaviors than those with a university degree.
It is “unacceptable that millions of people have a greater chance of developing cancer in their lifetime because they are simply not aware of the cancer risks to avoid and the healthy behaviors to adopt – information that many of us take for granted. And this is true around the world,” Cary Adams, MBA, CEO of the UICC, commented in a statement.
The survey was conducted from Oct. 25 to Nov., 2019, and included 15,427 participants from Australia, Bolivia, Brazil, Canada, China, France, Germany, Great Britain, India, Israel, Japan, Kenya, Mexico, the Philippines, Saudi Arabia, South Africa, Spain, Sweden, Turkey, and the United States.
The vast majority of those surveyed – 87% – said they were aware of the major risk factors for cancer, while only 6% said they were not.
The cancer risk factors that were most recognized were tobacco use (63%), ultraviolet light exposure (54%), and exposure to secondhand tobacco smoke (50%).
The cancer risks that were least recognized included being overweight (29%), a lack of exercise (28%), and exposure to certain viruses or bacteria (28%).
The difference in awareness across the social spectrum was striking. “Emerging from the survey are the apparent and glaring inequities faced by socioeconomically disadvantaged groups,” the authors said.
“Much more must be done to ensure that everyone has an equal chance to reduce their risk of preventable cancer,” commented Sonali Johnson, PhD, head of knowledge, advocacy, and policy at the UICC in Geneva, Switzerland.
“We’ve seen in the results that those surveyed with a lower education and those on lower incomes appear less aware of the main risk factors associated with cancer and thus are less likely to proactively take the steps needed to reduce their cancer risk as compared to those from a high income household or those with a university education,” Dr. Johnson said in an interview.
Does increased cancer awareness translate into behavioral change for the better? This question can only be answered by more research, the survey authors said. They reported that 7 of 10 survey respondents (69%) said they had made a behavioral change to reduce their cancer risk within the past 12 months. Most said they were eating more healthfully.
Slightly fewer than one-quarter reported that they had not taken any preventive measures in the past year.
When it comes to raising cancer awareness, World Cancer Day is “a powerful tool to remind every person that they can play a crucial role in reducing the impact of cancer,” said Dr. Johnson.
Health care providers are “crucial”
Reacting to the findings of the survey, the European Society for Medical Oncology (ESMO) emphasized the key role that physicians play in cancer prevention.
“Research speaks very clearly for prevention,” said ESMO President Solange Peters, MD, PhD. “With the number of cancer cases expected to rise to 29.5 million by 2040, we must act now. ESMO is committed to educating doctors on all aspects of cancer control, which should begin well before a cancer diagnosis.
“In the face of this emergency, which is rendered even more salient by the results of the report, we must work to enlarge the basis of doctors who are properly educated and trained in key prevention measures,” Dr. Peters added. “General practitioners and organ specialists are in the front line to guide and support patients on their quest for healthy lifestyles and reliable ways to detect cancer early.”
In a comment, Dr. Johnson acknowledged the role physicians play in health promotion and informing patients about noncommunicable disease risks, including those related to cancer. However, she emphasized that nurses, pharmacists, community health workers, midwives, and other health care providers who deliver primary care “are crucial around the world to imparting health information and offering services.”
Frontline health care workers can assess patients’ cancer knowledge and health literacy, determine the barriers to health care, and assess “how best to engage with people across the life course,” Dr. Johnson explained. “Rather than just focusing on physicians, we must work with all those involved in primary care, especially as primary care services are scaled up to achieve universal health coverage.”
Call on governments to do more
The authors noted that, although there is wide awareness of the cancer risks from tobacco use, adults younger than 35 years were less likely than those older than 50 to identify tobacco as a cancer risk factor. They described this finding as “most concerning” and said it “underscores the ongoing need to raise awareness about cancer risk factors in every new generation.”
Almost 60% of survey respondents, regardless of age, education, or income, expressed concern about being diagnosed with cancer in the future or having cancer recur.
In Kenya, where the death toll from cancer rose 30% from 2014 to 2018, people appeared to be the most worried about cancer, with four of five survey respondents (82%) expressing concern.
Survey respondents from Saudi Arabia appeared the least concerned, with one of three people saying they were worried.
Notably, 84% of survey respondents said that governments should be doing more to increase cancer prevention and awareness; 33% demanded that governments improve the affordability of cancer care.
“It is understandable that people turn to their governments for support,” Dr. Johnson commented. “Affordability is a big challenge for low-income settings.”
Data from the World Health Organization show that, for every U.S. $1 invested in low- and middle-income countries, the return is U.S. $3.20, Johnson pointed out. “We really need to convince decision makers ... and see the right investments being made. It is important to ensure that the health system strengthening takes place in tandem with prevention services.”
Governments have begun making commitments to tackle noncommunicable diseases and cancer, Dr. Johnson commented. He highlighted the WHO’s Global Action Plan for Healthy Lives and Well-being for All and the updated cancer resolution adopted at the 2017 World Health Assembly.
“Education, training, and awareness-raising efforts need to be backed by strong and progressive health policies that prioritize prevention and help reduce the consumption of known cancer-causing products such as tobacco, sugary food, and beverages,” she said. “Countries should also invest proactively in national cancer control planning and the establishment of population-based registries to ensure the most effective resource allocation that benefits all groups.”
Up-to-date information on cancer risks and cancer prevention must be delivered to the public in ways that are accessible to those in lower socioeconomic groups, Dr. Johnson added. “Awareness needs to be raised continuously with each new generation,” she noted.
The UICC has relationships with Astellas, Daiichi Sankyo, Diaceutics, MSD Inventing for Life, Bristol-Myers Squibb, CUBEBIO, the Icon Group, Roche, and Sanofi.
This article first appeared on Medscape.com.
The first international public survey on cancer perceptions and attitudes in a decade shows that, in spite of progress, low socioeconomic status and lack of education continue to jeopardize the health of the world’s most vulnerable populations.
The survey was commissioned by the Union for International Cancer Control (UICC) to mark the 20th anniversary of World Cancer Day on Feb. 4, 2020.
The survey, which was conducted by Ipsos, was taken by more than 15,000 people in 20 countries. It shows that people of lower socioeconomic status are less likely than those in higher-income households to recognize the risk factors for cancer or to make lifestyle changes. With the exception of tobacco use, people with low educational attainment also showed less cancer awareness and were less likely to engage in preventive behaviors than those with a university degree.
It is “unacceptable that millions of people have a greater chance of developing cancer in their lifetime because they are simply not aware of the cancer risks to avoid and the healthy behaviors to adopt – information that many of us take for granted. And this is true around the world,” Cary Adams, MBA, CEO of the UICC, commented in a statement.
The survey was conducted from Oct. 25 to Nov., 2019, and included 15,427 participants from Australia, Bolivia, Brazil, Canada, China, France, Germany, Great Britain, India, Israel, Japan, Kenya, Mexico, the Philippines, Saudi Arabia, South Africa, Spain, Sweden, Turkey, and the United States.
The vast majority of those surveyed – 87% – said they were aware of the major risk factors for cancer, while only 6% said they were not.
The cancer risk factors that were most recognized were tobacco use (63%), ultraviolet light exposure (54%), and exposure to secondhand tobacco smoke (50%).
The cancer risks that were least recognized included being overweight (29%), a lack of exercise (28%), and exposure to certain viruses or bacteria (28%).
The difference in awareness across the social spectrum was striking. “Emerging from the survey are the apparent and glaring inequities faced by socioeconomically disadvantaged groups,” the authors said.
“Much more must be done to ensure that everyone has an equal chance to reduce their risk of preventable cancer,” commented Sonali Johnson, PhD, head of knowledge, advocacy, and policy at the UICC in Geneva, Switzerland.
“We’ve seen in the results that those surveyed with a lower education and those on lower incomes appear less aware of the main risk factors associated with cancer and thus are less likely to proactively take the steps needed to reduce their cancer risk as compared to those from a high income household or those with a university education,” Dr. Johnson said in an interview.
Does increased cancer awareness translate into behavioral change for the better? This question can only be answered by more research, the survey authors said. They reported that 7 of 10 survey respondents (69%) said they had made a behavioral change to reduce their cancer risk within the past 12 months. Most said they were eating more healthfully.
Slightly fewer than one-quarter reported that they had not taken any preventive measures in the past year.
When it comes to raising cancer awareness, World Cancer Day is “a powerful tool to remind every person that they can play a crucial role in reducing the impact of cancer,” said Dr. Johnson.
Health care providers are “crucial”
Reacting to the findings of the survey, the European Society for Medical Oncology (ESMO) emphasized the key role that physicians play in cancer prevention.
“Research speaks very clearly for prevention,” said ESMO President Solange Peters, MD, PhD. “With the number of cancer cases expected to rise to 29.5 million by 2040, we must act now. ESMO is committed to educating doctors on all aspects of cancer control, which should begin well before a cancer diagnosis.
“In the face of this emergency, which is rendered even more salient by the results of the report, we must work to enlarge the basis of doctors who are properly educated and trained in key prevention measures,” Dr. Peters added. “General practitioners and organ specialists are in the front line to guide and support patients on their quest for healthy lifestyles and reliable ways to detect cancer early.”
In a comment, Dr. Johnson acknowledged the role physicians play in health promotion and informing patients about noncommunicable disease risks, including those related to cancer. However, she emphasized that nurses, pharmacists, community health workers, midwives, and other health care providers who deliver primary care “are crucial around the world to imparting health information and offering services.”
Frontline health care workers can assess patients’ cancer knowledge and health literacy, determine the barriers to health care, and assess “how best to engage with people across the life course,” Dr. Johnson explained. “Rather than just focusing on physicians, we must work with all those involved in primary care, especially as primary care services are scaled up to achieve universal health coverage.”
Call on governments to do more
The authors noted that, although there is wide awareness of the cancer risks from tobacco use, adults younger than 35 years were less likely than those older than 50 to identify tobacco as a cancer risk factor. They described this finding as “most concerning” and said it “underscores the ongoing need to raise awareness about cancer risk factors in every new generation.”
Almost 60% of survey respondents, regardless of age, education, or income, expressed concern about being diagnosed with cancer in the future or having cancer recur.
In Kenya, where the death toll from cancer rose 30% from 2014 to 2018, people appeared to be the most worried about cancer, with four of five survey respondents (82%) expressing concern.
Survey respondents from Saudi Arabia appeared the least concerned, with one of three people saying they were worried.
Notably, 84% of survey respondents said that governments should be doing more to increase cancer prevention and awareness; 33% demanded that governments improve the affordability of cancer care.
“It is understandable that people turn to their governments for support,” Dr. Johnson commented. “Affordability is a big challenge for low-income settings.”
Data from the World Health Organization show that, for every U.S. $1 invested in low- and middle-income countries, the return is U.S. $3.20, Johnson pointed out. “We really need to convince decision makers ... and see the right investments being made. It is important to ensure that the health system strengthening takes place in tandem with prevention services.”
Governments have begun making commitments to tackle noncommunicable diseases and cancer, Dr. Johnson commented. He highlighted the WHO’s Global Action Plan for Healthy Lives and Well-being for All and the updated cancer resolution adopted at the 2017 World Health Assembly.
“Education, training, and awareness-raising efforts need to be backed by strong and progressive health policies that prioritize prevention and help reduce the consumption of known cancer-causing products such as tobacco, sugary food, and beverages,” she said. “Countries should also invest proactively in national cancer control planning and the establishment of population-based registries to ensure the most effective resource allocation that benefits all groups.”
Up-to-date information on cancer risks and cancer prevention must be delivered to the public in ways that are accessible to those in lower socioeconomic groups, Dr. Johnson added. “Awareness needs to be raised continuously with each new generation,” she noted.
The UICC has relationships with Astellas, Daiichi Sankyo, Diaceutics, MSD Inventing for Life, Bristol-Myers Squibb, CUBEBIO, the Icon Group, Roche, and Sanofi.
This article first appeared on Medscape.com.
How much exercise is needed for maximum heart benefit?
SNOWMASS, COLO. – Physical activity is potent medicine, and it doesn’t take all that much of it to derive the maximum cardiovascular benefit: namely, the equivalent of a brisk hour-long walk 5 days/week or jogging at a 10-minute-per-mile pace for half an hour twice weekly, Robert A. Vogel, MD, asserted at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’m not telling you to run marathons. said Dr. Vogel, a cardiologist at the University of Colorado at Denver, Aurora, with a longstanding interest in preventive cardiology.
He presented selected highlights from the massive evidence base underlying the recommendations put forth in the current comprehensive U.S. Department of Health & Human Services Physical Activity Guidelines for Americans.
One particularly compelling chunk of evidence comes from a Taiwanese government–funded prospective cohort study of more than 416,000 individuals followed for an average of 8 years. A key finding: 15 minutes of moderate-intensity physical activity daily was associated with a 14% reduction in the relative risk of all-cause mortality and a 19% reduction in death caused by cardiovascular disease, compared with that of inactive individuals. Moreover, each additional 15 minutes of daily moderate exercise further reduced mortality by 4%. These benefits extended across the full age spectrum of both sexes and applied to patients with cardiovascular disease (Lancet. 2011 Oct 1;378[9798]:1244-53).
“That’s a very impressive result for modest physical activity,” the cardiologist commented.
Data on more than 50,000 adult participants in the Aerobics Center Longitudinal Study based at the Cooper Clinic in Dallas show that vigorous exercise in the form of running at 6 mph for half an hour twice weekly, or a total of 10 metabolic equivalent of task hours (MET-HR) per week, was associated with a roughly 40% reduction in cardiovascular disease mortality. Importantly, 20, 40, or 50 MET-HR/week of vigorous exercise conferred no further survival benefit (J Am Coll Cardiol. 2014 Aug 5;64[5]:472-81). The same group showed that the sweet spot for moderate physical activity in terms of reduced cardiovascular mortality was brisk walking for an hour daily 5 days/week, for a total of 20 MET-HR, which was also associated with roughly a 40% risk reduction compared to inactivity. At that point the benefit plateaued, with no further mortality reduction noted with additional MET-HR of moderate exercise.
“For more than that, we have no evidence of additional cardiovascular benefit. It’s not going to get you to the Tokyo Olympics, but that’s what we need to be doing,” Dr. Vogel observed.
In another report from the Aerobics Center Longitudinal Study, investigators found that moderate-level cardiorespiratory fitness as defined by METs was associated with a 44% reduction in the risk of sudden cardiac death in men and women after adjustment for potential confounders, while high-level cardiorespiratory fitness was associated with a closely similar 48% reduction in risk. This applied to individuals who were hypertensive, overweight, and/or had poor health status, as well as to others (Mayo Clin Proc. 2016 Jul;91[7]:849-57).
All activity counts
Exercise physiologists speak of NEPA – nonexercise physical activity – such as taking out the garbage. Swedish investigators followed more than 4,200 individuals for an average of 12.5 years and found that high NEPA activity was independently associated with a 30% reduction in all-cause mortality and a 27% lower risk of a first cardiovascular disease event, compared with low NEPA. High NEPA in regular exercisers was associated with a lower rate of metabolic syndrome than in low-NEPA regular exercisers (Br J Sports Med. 2014 Feb;48[3]:233-8).
Don’t just sit there – stand!
The current federal physical activity guidelines place a new emphasis on the detrimental effects of sitting. A report on more than 221,000 participants in the Australian 45 and Up Study, with close to 1 million person-years of follow-up, demonstrated a linear inverse relationship between standing time per day and all-cause mortality. In a multivariate analysis adjusted for potential confounders, individuals who stood for 2-5 hours per day had a 10% lower risk of all-cause mortality than did those who stood for less than 2 hours. Standing for 5-8 hours was associated with a 15% relative risk reduction. And standing for more than 8 hours daily was linked to a 24% reduction in risk (Prev Med. 2014 Dec;69:187-91).
And it’s not just total daily sitting time that’s a risk factor. Prolonged, uninterrupted sedentary time was also associated with a dose-dependent increase in all-cause mortality in a prospective cohort study of nearly 8,000 U.S. adults (Ann Intern Med. 2017 Oct 3;167[7]:465-75).
“If you can’t walk around, talk to your patients standing up. That activity of getting out of your chair is lifesaving,” the cardiologist advised.
Get strong
Muscle-strengthening activity on at least 2 days/week is recommended in the federal guidelines because it’s independently associated with decreased all-cause mortality, even in individuals getting sufficient aerobic exercise, as shown in a large national study with 15-years’ follow-up (Prev Med. 2016 Jun;87:121-127).
“As we get older, we tend to forget about muscle. I work with the National Football League. These folks are pretty strong, but we never see diabetes in these very big players, who are often well over 300 lb. They’ve got a lot of muscle. If you want to prevent diabetes, be strong. It’s a very important factor,” Dr. Vogel said.
For the time constrained
Jogging is more time-efficient than brisk walking as a way to attain the maximum cardiovascular benefit of exercise. And the so-called “Weekend Warrior” study of nearly 64,000 U.K. adults showed that it’s okay to cram the full week’s worth of exercise into one or two sessions and be done with it. Compared with the inactive study participants, the weekend warriors had a 40% reduction in cardiovascular disease mortality, while individuals who split their physical activity up into three or more sessions per week had a nearly identical 41% relative risk reduction (JAMA Intern Med. 2017 Mar 1;177[3]:335-42).
Interval training is a standard way for athletes in training to improve their endurance by alternating short, intense exercise with brief recovery periods. It’s also a time saver: In one classic bicycling study, physically active men were randomized to standardized 2-week programs of sprint interval training or high-volume endurance training on the bike. The training time required to pass a rigorous cycling time trial test was 90% lower in the interval training group (J Physiol. 2006 Sep 15;575(Pt 3):901-11).
The same principle is applicable to the nonathlete interested in physical activity for heart health.
“When I run a couple of miles, I walk for 5 minutes, then maybe run for three-quarters of a mile, then walk again, then run. In interval training you get your heart rate up, and you drop it down. It’s a very good form of exercise. As a vascular biologist I know that if you put endothelial cells in a Petri dish and spin them real fast continuously, you will not get as good an improvement in endothelial function as if you spin the dish, stop it, spin it, stop it,” Dr. Vogel said.
High-volume exercise is safe, even with high coronary calcium
A clinically significant coronary artery calcification score of 100 Agatston units or more is no reason not to exercise. A Cooper Clinic report on nearly 22,000 middle-aged men without baseline cardiovascular disease who were followed for a mean of 10.4 years concluded that those in the highest-volume exercise group, many of whom were marathon runners and engaged in the equivalent of running for at least 5-6 hours/week at a pace of 10 minutes per mile, were 11% more likely to have an elevated baseline coronary artery calcification score than those who exercised less. But these highest-volume exercisers with elevated coronary calcium – their mean level was 807 Agatston units – had risks of all-cause and cardiovascular mortality that weren’t significantly different from those of men with elevated coronary calcium who exercised more moderately (JAMA Cardiol. 2019 Feb 1;4[2]:174-81).
Cardiac rehab
Dr. Vogel had harsh words for his physician colleagues with respect to the widespread underprescribing of cardiac rehabilitation programs.
“You guys are doing a crappy job with exercise in our most vulnerable patients: those who’ve had cardiovascular events,” he charged. “Cardiac rehabilitation is a Class I recommendation in our guidelines. And yet utilization in the United States is just 10%-20%. No other Class I recommendation is in that ballpark.”
A meta-analysis of 34 randomized trials totaling more than 6,000 post-MI patients concluded that those randomized to exercise-based cardiac rehabilitation had a 47% reduction in the risk of reinfarction, 36% lower cardiac mortality, and a 26% reduction in all-cause mortality (Am Heart J. 2011 Oct;162[4]:571-584.e2).
“The data show that cardiac rehabilitation is as effective as anything else we do in cardiovascular medicine. I understand that patients live far away, they don’t like to exercise – I’ve heard every excuse. But I am charging you with the responsibility of meeting a Class I recommendation that gets patients to live longer,” he declared.
Medicare now covers an enhanced, 72-session program called Intensive Cardiac Rehabilitation that teaches comprehensive lifestyle change and provides reasonable reimbursement. “It’s a good thing for our patients,” Dr. Vogel commented.
Yoga
For patients who are reluctant to pound the pavement, yoga may provide an alternative form of physical activity with tangible cardiovascular benefits. Dr. Vogel pointed to the Yoga-CaRe trial presented at the 2018 scientific sessions of the American Heart Association. Yoga-CaRe randomized 3,959 post-MI patients at 29 centers in India to a program of 13 supervised in-hospital yoga classes followed by yoga at home, or to a control group with three educational sessions. The rate of major adverse cardiovascular events over 42 months of follow-up was cut in half, compared with controls, in the 27% of participants who attended at least 10 of the 13 yoga classes. Their quality of life scores were higher, too.
Dr. Vogel reported serving as a paid consultant to the National Football League and the Pritikin Longevity Center. He is on the speaker’s bureau for Sanofi and Regeneron.
SNOWMASS, COLO. – Physical activity is potent medicine, and it doesn’t take all that much of it to derive the maximum cardiovascular benefit: namely, the equivalent of a brisk hour-long walk 5 days/week or jogging at a 10-minute-per-mile pace for half an hour twice weekly, Robert A. Vogel, MD, asserted at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’m not telling you to run marathons. said Dr. Vogel, a cardiologist at the University of Colorado at Denver, Aurora, with a longstanding interest in preventive cardiology.
He presented selected highlights from the massive evidence base underlying the recommendations put forth in the current comprehensive U.S. Department of Health & Human Services Physical Activity Guidelines for Americans.
One particularly compelling chunk of evidence comes from a Taiwanese government–funded prospective cohort study of more than 416,000 individuals followed for an average of 8 years. A key finding: 15 minutes of moderate-intensity physical activity daily was associated with a 14% reduction in the relative risk of all-cause mortality and a 19% reduction in death caused by cardiovascular disease, compared with that of inactive individuals. Moreover, each additional 15 minutes of daily moderate exercise further reduced mortality by 4%. These benefits extended across the full age spectrum of both sexes and applied to patients with cardiovascular disease (Lancet. 2011 Oct 1;378[9798]:1244-53).
“That’s a very impressive result for modest physical activity,” the cardiologist commented.
Data on more than 50,000 adult participants in the Aerobics Center Longitudinal Study based at the Cooper Clinic in Dallas show that vigorous exercise in the form of running at 6 mph for half an hour twice weekly, or a total of 10 metabolic equivalent of task hours (MET-HR) per week, was associated with a roughly 40% reduction in cardiovascular disease mortality. Importantly, 20, 40, or 50 MET-HR/week of vigorous exercise conferred no further survival benefit (J Am Coll Cardiol. 2014 Aug 5;64[5]:472-81). The same group showed that the sweet spot for moderate physical activity in terms of reduced cardiovascular mortality was brisk walking for an hour daily 5 days/week, for a total of 20 MET-HR, which was also associated with roughly a 40% risk reduction compared to inactivity. At that point the benefit plateaued, with no further mortality reduction noted with additional MET-HR of moderate exercise.
“For more than that, we have no evidence of additional cardiovascular benefit. It’s not going to get you to the Tokyo Olympics, but that’s what we need to be doing,” Dr. Vogel observed.
In another report from the Aerobics Center Longitudinal Study, investigators found that moderate-level cardiorespiratory fitness as defined by METs was associated with a 44% reduction in the risk of sudden cardiac death in men and women after adjustment for potential confounders, while high-level cardiorespiratory fitness was associated with a closely similar 48% reduction in risk. This applied to individuals who were hypertensive, overweight, and/or had poor health status, as well as to others (Mayo Clin Proc. 2016 Jul;91[7]:849-57).
All activity counts
Exercise physiologists speak of NEPA – nonexercise physical activity – such as taking out the garbage. Swedish investigators followed more than 4,200 individuals for an average of 12.5 years and found that high NEPA activity was independently associated with a 30% reduction in all-cause mortality and a 27% lower risk of a first cardiovascular disease event, compared with low NEPA. High NEPA in regular exercisers was associated with a lower rate of metabolic syndrome than in low-NEPA regular exercisers (Br J Sports Med. 2014 Feb;48[3]:233-8).
Don’t just sit there – stand!
The current federal physical activity guidelines place a new emphasis on the detrimental effects of sitting. A report on more than 221,000 participants in the Australian 45 and Up Study, with close to 1 million person-years of follow-up, demonstrated a linear inverse relationship between standing time per day and all-cause mortality. In a multivariate analysis adjusted for potential confounders, individuals who stood for 2-5 hours per day had a 10% lower risk of all-cause mortality than did those who stood for less than 2 hours. Standing for 5-8 hours was associated with a 15% relative risk reduction. And standing for more than 8 hours daily was linked to a 24% reduction in risk (Prev Med. 2014 Dec;69:187-91).
And it’s not just total daily sitting time that’s a risk factor. Prolonged, uninterrupted sedentary time was also associated with a dose-dependent increase in all-cause mortality in a prospective cohort study of nearly 8,000 U.S. adults (Ann Intern Med. 2017 Oct 3;167[7]:465-75).
“If you can’t walk around, talk to your patients standing up. That activity of getting out of your chair is lifesaving,” the cardiologist advised.
Get strong
Muscle-strengthening activity on at least 2 days/week is recommended in the federal guidelines because it’s independently associated with decreased all-cause mortality, even in individuals getting sufficient aerobic exercise, as shown in a large national study with 15-years’ follow-up (Prev Med. 2016 Jun;87:121-127).
“As we get older, we tend to forget about muscle. I work with the National Football League. These folks are pretty strong, but we never see diabetes in these very big players, who are often well over 300 lb. They’ve got a lot of muscle. If you want to prevent diabetes, be strong. It’s a very important factor,” Dr. Vogel said.
For the time constrained
Jogging is more time-efficient than brisk walking as a way to attain the maximum cardiovascular benefit of exercise. And the so-called “Weekend Warrior” study of nearly 64,000 U.K. adults showed that it’s okay to cram the full week’s worth of exercise into one or two sessions and be done with it. Compared with the inactive study participants, the weekend warriors had a 40% reduction in cardiovascular disease mortality, while individuals who split their physical activity up into three or more sessions per week had a nearly identical 41% relative risk reduction (JAMA Intern Med. 2017 Mar 1;177[3]:335-42).
Interval training is a standard way for athletes in training to improve their endurance by alternating short, intense exercise with brief recovery periods. It’s also a time saver: In one classic bicycling study, physically active men were randomized to standardized 2-week programs of sprint interval training or high-volume endurance training on the bike. The training time required to pass a rigorous cycling time trial test was 90% lower in the interval training group (J Physiol. 2006 Sep 15;575(Pt 3):901-11).
The same principle is applicable to the nonathlete interested in physical activity for heart health.
“When I run a couple of miles, I walk for 5 minutes, then maybe run for three-quarters of a mile, then walk again, then run. In interval training you get your heart rate up, and you drop it down. It’s a very good form of exercise. As a vascular biologist I know that if you put endothelial cells in a Petri dish and spin them real fast continuously, you will not get as good an improvement in endothelial function as if you spin the dish, stop it, spin it, stop it,” Dr. Vogel said.
High-volume exercise is safe, even with high coronary calcium
A clinically significant coronary artery calcification score of 100 Agatston units or more is no reason not to exercise. A Cooper Clinic report on nearly 22,000 middle-aged men without baseline cardiovascular disease who were followed for a mean of 10.4 years concluded that those in the highest-volume exercise group, many of whom were marathon runners and engaged in the equivalent of running for at least 5-6 hours/week at a pace of 10 minutes per mile, were 11% more likely to have an elevated baseline coronary artery calcification score than those who exercised less. But these highest-volume exercisers with elevated coronary calcium – their mean level was 807 Agatston units – had risks of all-cause and cardiovascular mortality that weren’t significantly different from those of men with elevated coronary calcium who exercised more moderately (JAMA Cardiol. 2019 Feb 1;4[2]:174-81).
Cardiac rehab
Dr. Vogel had harsh words for his physician colleagues with respect to the widespread underprescribing of cardiac rehabilitation programs.
“You guys are doing a crappy job with exercise in our most vulnerable patients: those who’ve had cardiovascular events,” he charged. “Cardiac rehabilitation is a Class I recommendation in our guidelines. And yet utilization in the United States is just 10%-20%. No other Class I recommendation is in that ballpark.”
A meta-analysis of 34 randomized trials totaling more than 6,000 post-MI patients concluded that those randomized to exercise-based cardiac rehabilitation had a 47% reduction in the risk of reinfarction, 36% lower cardiac mortality, and a 26% reduction in all-cause mortality (Am Heart J. 2011 Oct;162[4]:571-584.e2).
“The data show that cardiac rehabilitation is as effective as anything else we do in cardiovascular medicine. I understand that patients live far away, they don’t like to exercise – I’ve heard every excuse. But I am charging you with the responsibility of meeting a Class I recommendation that gets patients to live longer,” he declared.
Medicare now covers an enhanced, 72-session program called Intensive Cardiac Rehabilitation that teaches comprehensive lifestyle change and provides reasonable reimbursement. “It’s a good thing for our patients,” Dr. Vogel commented.
Yoga
For patients who are reluctant to pound the pavement, yoga may provide an alternative form of physical activity with tangible cardiovascular benefits. Dr. Vogel pointed to the Yoga-CaRe trial presented at the 2018 scientific sessions of the American Heart Association. Yoga-CaRe randomized 3,959 post-MI patients at 29 centers in India to a program of 13 supervised in-hospital yoga classes followed by yoga at home, or to a control group with three educational sessions. The rate of major adverse cardiovascular events over 42 months of follow-up was cut in half, compared with controls, in the 27% of participants who attended at least 10 of the 13 yoga classes. Their quality of life scores were higher, too.
Dr. Vogel reported serving as a paid consultant to the National Football League and the Pritikin Longevity Center. He is on the speaker’s bureau for Sanofi and Regeneron.
SNOWMASS, COLO. – Physical activity is potent medicine, and it doesn’t take all that much of it to derive the maximum cardiovascular benefit: namely, the equivalent of a brisk hour-long walk 5 days/week or jogging at a 10-minute-per-mile pace for half an hour twice weekly, Robert A. Vogel, MD, asserted at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
“I’m not telling you to run marathons. said Dr. Vogel, a cardiologist at the University of Colorado at Denver, Aurora, with a longstanding interest in preventive cardiology.
He presented selected highlights from the massive evidence base underlying the recommendations put forth in the current comprehensive U.S. Department of Health & Human Services Physical Activity Guidelines for Americans.
One particularly compelling chunk of evidence comes from a Taiwanese government–funded prospective cohort study of more than 416,000 individuals followed for an average of 8 years. A key finding: 15 minutes of moderate-intensity physical activity daily was associated with a 14% reduction in the relative risk of all-cause mortality and a 19% reduction in death caused by cardiovascular disease, compared with that of inactive individuals. Moreover, each additional 15 minutes of daily moderate exercise further reduced mortality by 4%. These benefits extended across the full age spectrum of both sexes and applied to patients with cardiovascular disease (Lancet. 2011 Oct 1;378[9798]:1244-53).
“That’s a very impressive result for modest physical activity,” the cardiologist commented.
Data on more than 50,000 adult participants in the Aerobics Center Longitudinal Study based at the Cooper Clinic in Dallas show that vigorous exercise in the form of running at 6 mph for half an hour twice weekly, or a total of 10 metabolic equivalent of task hours (MET-HR) per week, was associated with a roughly 40% reduction in cardiovascular disease mortality. Importantly, 20, 40, or 50 MET-HR/week of vigorous exercise conferred no further survival benefit (J Am Coll Cardiol. 2014 Aug 5;64[5]:472-81). The same group showed that the sweet spot for moderate physical activity in terms of reduced cardiovascular mortality was brisk walking for an hour daily 5 days/week, for a total of 20 MET-HR, which was also associated with roughly a 40% risk reduction compared to inactivity. At that point the benefit plateaued, with no further mortality reduction noted with additional MET-HR of moderate exercise.
“For more than that, we have no evidence of additional cardiovascular benefit. It’s not going to get you to the Tokyo Olympics, but that’s what we need to be doing,” Dr. Vogel observed.
In another report from the Aerobics Center Longitudinal Study, investigators found that moderate-level cardiorespiratory fitness as defined by METs was associated with a 44% reduction in the risk of sudden cardiac death in men and women after adjustment for potential confounders, while high-level cardiorespiratory fitness was associated with a closely similar 48% reduction in risk. This applied to individuals who were hypertensive, overweight, and/or had poor health status, as well as to others (Mayo Clin Proc. 2016 Jul;91[7]:849-57).
All activity counts
Exercise physiologists speak of NEPA – nonexercise physical activity – such as taking out the garbage. Swedish investigators followed more than 4,200 individuals for an average of 12.5 years and found that high NEPA activity was independently associated with a 30% reduction in all-cause mortality and a 27% lower risk of a first cardiovascular disease event, compared with low NEPA. High NEPA in regular exercisers was associated with a lower rate of metabolic syndrome than in low-NEPA regular exercisers (Br J Sports Med. 2014 Feb;48[3]:233-8).
Don’t just sit there – stand!
The current federal physical activity guidelines place a new emphasis on the detrimental effects of sitting. A report on more than 221,000 participants in the Australian 45 and Up Study, with close to 1 million person-years of follow-up, demonstrated a linear inverse relationship between standing time per day and all-cause mortality. In a multivariate analysis adjusted for potential confounders, individuals who stood for 2-5 hours per day had a 10% lower risk of all-cause mortality than did those who stood for less than 2 hours. Standing for 5-8 hours was associated with a 15% relative risk reduction. And standing for more than 8 hours daily was linked to a 24% reduction in risk (Prev Med. 2014 Dec;69:187-91).
And it’s not just total daily sitting time that’s a risk factor. Prolonged, uninterrupted sedentary time was also associated with a dose-dependent increase in all-cause mortality in a prospective cohort study of nearly 8,000 U.S. adults (Ann Intern Med. 2017 Oct 3;167[7]:465-75).
“If you can’t walk around, talk to your patients standing up. That activity of getting out of your chair is lifesaving,” the cardiologist advised.
Get strong
Muscle-strengthening activity on at least 2 days/week is recommended in the federal guidelines because it’s independently associated with decreased all-cause mortality, even in individuals getting sufficient aerobic exercise, as shown in a large national study with 15-years’ follow-up (Prev Med. 2016 Jun;87:121-127).
“As we get older, we tend to forget about muscle. I work with the National Football League. These folks are pretty strong, but we never see diabetes in these very big players, who are often well over 300 lb. They’ve got a lot of muscle. If you want to prevent diabetes, be strong. It’s a very important factor,” Dr. Vogel said.
For the time constrained
Jogging is more time-efficient than brisk walking as a way to attain the maximum cardiovascular benefit of exercise. And the so-called “Weekend Warrior” study of nearly 64,000 U.K. adults showed that it’s okay to cram the full week’s worth of exercise into one or two sessions and be done with it. Compared with the inactive study participants, the weekend warriors had a 40% reduction in cardiovascular disease mortality, while individuals who split their physical activity up into three or more sessions per week had a nearly identical 41% relative risk reduction (JAMA Intern Med. 2017 Mar 1;177[3]:335-42).
Interval training is a standard way for athletes in training to improve their endurance by alternating short, intense exercise with brief recovery periods. It’s also a time saver: In one classic bicycling study, physically active men were randomized to standardized 2-week programs of sprint interval training or high-volume endurance training on the bike. The training time required to pass a rigorous cycling time trial test was 90% lower in the interval training group (J Physiol. 2006 Sep 15;575(Pt 3):901-11).
The same principle is applicable to the nonathlete interested in physical activity for heart health.
“When I run a couple of miles, I walk for 5 minutes, then maybe run for three-quarters of a mile, then walk again, then run. In interval training you get your heart rate up, and you drop it down. It’s a very good form of exercise. As a vascular biologist I know that if you put endothelial cells in a Petri dish and spin them real fast continuously, you will not get as good an improvement in endothelial function as if you spin the dish, stop it, spin it, stop it,” Dr. Vogel said.
High-volume exercise is safe, even with high coronary calcium
A clinically significant coronary artery calcification score of 100 Agatston units or more is no reason not to exercise. A Cooper Clinic report on nearly 22,000 middle-aged men without baseline cardiovascular disease who were followed for a mean of 10.4 years concluded that those in the highest-volume exercise group, many of whom were marathon runners and engaged in the equivalent of running for at least 5-6 hours/week at a pace of 10 minutes per mile, were 11% more likely to have an elevated baseline coronary artery calcification score than those who exercised less. But these highest-volume exercisers with elevated coronary calcium – their mean level was 807 Agatston units – had risks of all-cause and cardiovascular mortality that weren’t significantly different from those of men with elevated coronary calcium who exercised more moderately (JAMA Cardiol. 2019 Feb 1;4[2]:174-81).
Cardiac rehab
Dr. Vogel had harsh words for his physician colleagues with respect to the widespread underprescribing of cardiac rehabilitation programs.
“You guys are doing a crappy job with exercise in our most vulnerable patients: those who’ve had cardiovascular events,” he charged. “Cardiac rehabilitation is a Class I recommendation in our guidelines. And yet utilization in the United States is just 10%-20%. No other Class I recommendation is in that ballpark.”
A meta-analysis of 34 randomized trials totaling more than 6,000 post-MI patients concluded that those randomized to exercise-based cardiac rehabilitation had a 47% reduction in the risk of reinfarction, 36% lower cardiac mortality, and a 26% reduction in all-cause mortality (Am Heart J. 2011 Oct;162[4]:571-584.e2).
“The data show that cardiac rehabilitation is as effective as anything else we do in cardiovascular medicine. I understand that patients live far away, they don’t like to exercise – I’ve heard every excuse. But I am charging you with the responsibility of meeting a Class I recommendation that gets patients to live longer,” he declared.
Medicare now covers an enhanced, 72-session program called Intensive Cardiac Rehabilitation that teaches comprehensive lifestyle change and provides reasonable reimbursement. “It’s a good thing for our patients,” Dr. Vogel commented.
Yoga
For patients who are reluctant to pound the pavement, yoga may provide an alternative form of physical activity with tangible cardiovascular benefits. Dr. Vogel pointed to the Yoga-CaRe trial presented at the 2018 scientific sessions of the American Heart Association. Yoga-CaRe randomized 3,959 post-MI patients at 29 centers in India to a program of 13 supervised in-hospital yoga classes followed by yoga at home, or to a control group with three educational sessions. The rate of major adverse cardiovascular events over 42 months of follow-up was cut in half, compared with controls, in the 27% of participants who attended at least 10 of the 13 yoga classes. Their quality of life scores were higher, too.
Dr. Vogel reported serving as a paid consultant to the National Football League and the Pritikin Longevity Center. He is on the speaker’s bureau for Sanofi and Regeneron.
EXPERT ANALYSIS FROM ACC SNOWMASS 2020
Nontuberculous Mycobacterial Pulmonary Disease
Nontuberculous mycobacterial pulmonary disease is a broad term for a group of pulmonary disorders caused and characterized by exposure to environmental mycobacteria other than those belonging to the Mycobacterium tuberculosis complex and Mycobacterium leprae. Mycobacteria are aerobic, nonmotile organisms that appear positive with acid-fast alcohol stains. Nontuberculous mycobacteria (NTM) are ubiquitous in the environment and have been recovered from domestic and natural water sources, soil, and food products, and from around livestock, cattle, and wildlife.1-3 To date, no evidence exists of human-to-human or animal-to-human transmission of NTM in the general population. Infections in humans are usually acquired from environmental exposures, although the specific source of infection cannot always be identified. Similarly, the mode of infection with NTM has not been established with certainty, but it is highly likely that the organism is implanted, ingested, aspirated, or inhaled. Aerosolization of droplets associated with use of bathroom showerheads and municipal water sources and soil contamination are some of the factors associated with the transmission of infection. Proven routes of transmission include showerheads and potting soil dust.2,3
NTM pulmonary disease occurs in individuals with or without comorbid conditions such as bronchiectasis, chronic obstructive pulmonary disease, pulmonary fibrosis, or structural lung diseases. Slender, middle-aged or elderly white females with marfanoid body habitus, with or without apparent immune or genetic disorders, showing impaired airway and mucus clearance present with this infection as a form of underlying bronchiectasis (Lady Windermere syndrome). It is unclear why NTM infections and escalation to clinical disease occur in certain individuals. Many risk factors, including inherited and acquired defects of host immune response (eg, cystic fibrosis trait and α1 antitrypsin deficiency), have been associated with increased susceptibility to NTM infections.4
NTM infection can lead to chronic symptoms, frequent exacerbations, progressive functional and structural lung destruction, and impaired quality of life, and is associated with an increased risk of hospitalization and higher 5-year all-cause mortality. As such, NTM disease is drawing increasing attention at the clinical, academic, and research levels.5 This case-based review outlines the clinical features of NTM infection, with a focus on the challenges in diagnosis, treatment, and management of NTM pulmonary disease. The cases use Mycobacterium avium complex (MAC), a slow-growing mycobacteria (SGM), and Mycobacterium abscessus, a rapidly growing mycobacteria (RGM), as prototypes in a non–cystic fibrosis, non-HIV clinical setting.
Epidemiology
Of the almost 200 isolated species of NTM, the most prevalent pathogens for respiratory disease in the United States are MAC, Mycobacterium kansasii, and M. abscessus. MAC accounts for more than 80% of cases of NTM respiratory disease in the United States.6 The prevalence of NTM disease is increasing at a rate of about 8% each year, with 75,000 to 105,000 patients diagnosed with NTM lung disease in the United States annually. NTM infections in the United States are increasing among patients aged 65 years and older, a population that is expected to nearly double by 2030.7,8
Isolation and prevalence of many NTM species are higher in certain geographic areas of the United States, especially in the southeast. The US coastal regions have a higher prevalence of NTM pulmonary disease, and account for 70% of NTM cases in the United States each year. Half of patients diagnosed with NTM lung disease reside in 7 states: Florida, New York, Texas, California, Pennsylvania, New Jersey, and Ohio, with 1 in 7 residing in Florida. Three parishes in Louisiana are among the top 10 counties with the highest prevalence in United States. The prevalence of NTM infection–associated hospitalizations is increasing worldwide as well. Co-infection with tuberculosis and multiple NTMs in individual patients has been observed clinically and documented in patients with and without HIV.9,10
It is not clear why the prevalence of NTM pulmonary disease is increasing, but there may be several contributing factors: (1) an increased awareness and identification of NTM infection sources in the environment; (2) an expanding cohort of immunocompromised individuals with exogenous or endogenous immune deficiencies; (3) availability of improved diagnostic techniques, such as use of high-performance liquid chromatography (HPLC), DNA probes, and gene sequencing; and (4) an increased awareness of the morbidity and mortality associated with NTM pulmonary disease. However, it is important to recognize that to best understand the clinical relevance of epidemiologic studies based on laboratory diagnosis and identification, the findings must be evaluated by correlating them with the microbiological and other clinical criteria established by the American Thoracic Society (ATS)/Infectious Diseases Society of America (IDSA) guidelines.11
Continue to: Mycobacterium avium Complex
Mycobacterium avium Complex
Case Patient 1
A 48-year-old woman who has never smoked and has no past medical problems, except seasonal allergic rhinitis and “colds and flu-like illness” once or twice a year, is evaluated for a chronic lingering cough with occasional sputum production. The patient denies any other chronic symptoms and is otherwise active. Physical examination reveals no specific findings except mild pectus excavatum and mild scoliosis. Body mass index is 22 kg/m2. Chest radiograph shows nonspecific increased markings in the lower zones. Computed tomography (CT) scan of the chest reveals minimal nodular and cylindrical bronchiectasis in both lungs (Figure 1). No previous radiographs are available for comparison. The patient is HIV-negative. Sputum tests reveal normal flora, and both fungus and acid-fast bacilli smear are negative. Culture for mycobacteria shows scanty growth of MAC in 1 specimen.
What is the clinical presentation of MAC pulmonary disease?
Among NTM, MAC is the most common cause of pulmonary disease worldwide.6 MAC primarily includes 2 species: M. avium and Mycobacterium intracellulare. M. avium is the more important pathogen in disseminated disease, whereas M. intracellulare is the more common respiratory pathogen.11 These organisms are genetically similar and generally not differentiated in the clinical microbiology laboratory, although there are isolated reports in the literature suggesting differences in prevalence, presentation, and prognosis in M. avium infection versus M. intracellulare infection.12
Three major disease syndromes are produced by MAC in humans: pulmonary disease, usually in adults whose systemic immunity is intact; disseminated disease, usually in patients with advanced HIV infection; and cervical lymphadenitis.13 Pulmonary disease caused by MAC may take on 1 of several clinically different forms, including asymptomatic “colonization” or persistent minimal infection without obvious clinical significance; endobronchial involvement; progressive pulmonary disease with radiographic and clinical deterioration and nodular bronchiectasis or cavitary lung disease; hypersensitivity pneumonitis; or persistent, overwhelming mycobacterial growth with symptomatic manifestations, often in a lung with underlying damage due to either chronic obstructive lung disease or pulmonary fibrosis (Table 1).14
Cavitary Disease
The traditionally recognized presentation of MAC lung disease has been apical cavitary lung disease in men in their late 40s and early 50s who have a history of cigarette smoking, and frequently, excessive alcohol use. If left untreated, or in the case of erratic treatment or macrolide drug resistance, this form of disease is generally progressive within a relatively short time and can result in extensive cavitary lung destruction and progressive respiratory failure.15
Nodular Bronchiectasis
The more common presentation of MAC lung disease, which is outlined in the case described here, is interstitial nodular infiltrates, frequently involving the right middle lobe or lingula and predominantly occurring in postmenopausal, nonsmoking white women. This is sometimes labelled “Lady Windermere syndrome.” These patients with M. avium infection appear to have similar clinical characteristics and body types, including lean build, scoliosis, pectus excavatum, and mitral valve prolapse.16,17 The mechanism by which this body morphotype predisposes to pulmonary mycobacterial infection is not defined, but ineffective mucociliary clearance is a possible explanation. Evidence suggests that some patients may be predisposed to NTM lung disease because of preexisting bronchiectasis. Some potential etiologies of bronchiectasis in this population include chronic sinusitis, gastroesophageal reflux with chronic aspiration, α1 antitrypsin deficiency, and cystic fibrosis genetic traits and mutations.18 Risk factors for increased morbidity and mortality include the development of cavitary disease, age, weight loss, lower body mass index, and other comorbid conditions.
This form of disease, termed nodular bronchiectasis, tends to have a much slower progression than cavitary disease, such that long-term follow-up (months to years) may be necessary to demonstrate clinical or radiographic changes.11 The radiographic term “tree-in-bud” has been used to describe what may reflect inflammatory changes, including bronchiolitis. High-resolution CT scans of the chest are especially helpful for diagnosing this pattern of MAC lung disease, as bronchiectasis and small nodules may not be easily discernible on plain chest radiograph. The nodular/bronchiectasis radiographic pattern can also be seen with other NTM pathogens, including M. abscessus, Mycobacterium simiae, and M. kansasii. Solitary nodules and dense consolidation have also been described. Pleural effusions are uncommon, but reactive pleural thickening is frequently seen. Co-pathogens may be isolated from culture, including Pseudomonas aeruginosa, Staphylococcus aureus, and, occasionally, other NTM such as M. abscessus or Mycobacterium chelonae.19-21
Hypersensitivity Pneumonitis
Hypersensitivity pneumonitis, initially described in patients who were exposed to hot tubs, mimics allergic hypersensitivity pneumonitis, with respiratory symptoms and culture/tissue identification of MAC or sometimes other NTM. It is unclear whether hypersensitivity pneumonitis is an inflammatory process, an infection, or both, and opinion regarding the need for specific antibiotic treatment is divided.11,22 However, avoidance of exposure is prudent and recommended.
Disseminated Disease
Disseminated NTM disease is associated with very low CD4+ lymphocyte counts and is seen in approximately 5% of patients with HIV infection.23-25 Although disseminated NTM disease is rarely seen in immunosuppressed patients without HIV infection, it has been reported in patients who have undergone renal or cardiac transplant, patients on long-term corticosteroid therapy, and those with leukemia or lymphoma. More than 90% of infections are caused by MAC; other potential pathogens include M. kansasii, M. chelonae, M. abscessus, and Mycobacterium haemophilum. Although seen less frequently since the advent of highly active antiretroviral therapy, disseminated infection can develop progressively from an apparently indolent or localized infection or a respiratory or gastrointestinal source. Signs and symptoms of disseminated infection (specifically MAC-associated disease) are nonspecific and include fever, night sweats, weight loss, and abdominal tenderness. Disseminated MAC disease occurs primarily in patients with more advanced HIV disease (CD4+ count typically < 50 cells/μL). Clinically, disseminated MAC manifests as intermittent or persistent fever, constitutional symptoms with organomegaly and organ-specific abnormalities (eg, anemia, neutropenia from bone marrow involvement, adenopathy, hepatosplenomegaly), and elevations of liver enzymes or lung infiltrates from pulmonary involvement.
Continue to: What are the criteria for diagnosing NTM pulmonary disease?
What are the criteria for diagnosing NTM pulmonary disease?
The diagnosis of NTM disease is based on clinical, radiologic, and mycobacterial correlation with good communication between the experts in this field. The ATS/IDSA criteria for diagnosing NTM lung disease are shown in Figure 2. These criteria best apply to MAC, M. kansasii, and M. abscessus, but are also clinically applied to other NTM respiratory pathogens. The diagnosis of MAC infection is most readily established by culture of blood, bone marrow, respiratory secretions/fluid, or tissue specimens from suspected sites of involvement. Due to erratic shedding of MAC into the respiratory secretions in patients with nodular bronchiectasis, as compared to those with the cavitary form of the disease, sputum may be intermittently positive, with variable colony counts and polyclonal infections.12 Prior to the advent of high-resolution CT, isolation of MAC organisms from the sputum of such patients was frequently dismissed as colonization.
Mycobacterial Testing
Because of the nonspecific symptoms and lack of diagnostic specificity of chest imaging, the diagnosis of NTM lung disease requires microbiologic confirmation. Specimens sent to the laboratory for identification of NTM must be handled with care to prevent contamination and false-positive results. Transport media and preservatives should be avoided, and transportation of the specimens should be prompt. These measures will prevent bacterial overgrowth. Furthermore, the yield of NTM may be affected if the patient has used antibiotics, such as macrolides and fluoroquinolones, prior to obtaining the specimen.
NTM should be identified at the species and subspecies level, although this is not practical in community practice settings. The preferred staining procedure in the laboratory is the fluorochrome method. Some species require special growth conditions and/or lower incubation temperatures, and other identification methods may have to be employed, such as DNA probes, polymerase chain reaction genotyping, nucleic acid sequence determination, and high-performance liquid chromatography. As a gold standard, clinical specimens for mycobacterial cultures should be inoculated onto 1 or more solid media (eg, Middlebrook 7H11 media and/or Lowenstein-Jensen media, the former of which is the preferred medium for NTM) and into a liquid medium (eg, BACTEC 12B broth or Mycobacteria growth indicator tube broth). Growth of visible colonies on solid media typically requires 2 to 4 weeks, but liquid media (eg, the radiometric BACTEC system), used as a supplementary and not as an exclusive test, usually produce results within 10 to 14 days. Furthermore, even after initial growth, identification of specific isolates based on the growth characteristics on solid media requires additional time. Use of specific nucleic acid probes for MAC and M. kansasii and HPLC testing of mycolic acid patterns in acid-fast bacilli smear–positive specimens can reduce the turnaround time of specific identification of a primary culture–positive sample. However, HPLC is not sufficient for definitive identification of many NTM species, including the RGM. Other newer techniques, including 16S ribosomal DNA sequencing and polymerase chain reaction-restriction fragment length polymorphism analysis, also allow NTM to be identified and speciated more reliably and rapidly from clinical specimens.
Cost and other practical considerations limit widespread adoption of these techniques. However, the recognition that M. abscessus can be separated into more than 1 subspecies, and that there are important prognostic implications of that separation, lends urgency to the broader adoption of newer molecular techniques in the mycobacteriology laboratory. Susceptibility testing is based on the broth microdilution method; RGM usually grow within 7 days of subculture, and the laboratory time to culture is a helpful hint, although not necessarily specific. Recognizing the morphology of mycobacterial colony growth may also be helpful in identification.
Are skin tests helpful in diagnosing NTM infection?
Tuberculin skin testing remains a nonspecific marker of mycobacterial infection and does not help in further elucidating NTM infection. However, epidemiologic and laboratory studies with well-characterized antigens have shown that dual skin testing with tuberculosis- versus NTM-derived tuberculin can discriminate between prior NTM and prior tuberculosis disease. Species-specific skin test antigens are not commercially available and are not helpful in the diagnosis of NTM disease because of cross-reactivity of M. tuberculosis and some NTM. However, increased prevalence of NTM sensitization based on purified protein derivative testing has been noted in a recent survey, which is consistent with an observed increase in the rates of NTM infections, specifically MAC, in the United States.26,27
Interferon-gamma release assays (IGRAs) are now being used as an alternative to tuberculin skin testing to diagnose M. tuberculosis infection. Certain NTM species also contain gene sequences that encode for ESAT-6 or CFP-10 antigens used in the IGRAs, and hence, yield a positive IGRA test. These include M. marinum, M. szulgai, and M. kansasii.28,29 However, MAC organisms do not produce positive results on assays that use these antigens.
Continue to: What is the approach to management of NTM pulmonary disease?
What is the approach to management of NTM pulmonary disease?
The correlation of symptoms with radiographic and microbiologic evidence is essential to categorize the disease and determine the need for therapy. Making the diagnosis of NTM lung disease does not necessitate the institution of therapy. The decision to treat should be weighed against potential risks and benefits to the individual patient based on symptomatic, radiographic, and microbiologic criteria, as well as underlying systemic or pulmonary immune status. In the absence of evidence of clinical, radiologic, or mycobacterial progression of disease, pursuing airway clearance therapy and clinical surveillance without initiating specific anti-MAC therapy is a reasonable option.11 Identifying the sustained presence of NTM infection, especially MAC, in a patient with underlying clinical and radiographic evidence of bronchiectasis is of value in determining comprehensive treatment and management strategies. Close observation is indicated if the decision not to treat is made. If treatment is initiated, comprehensive management includes long-term follow-up with periodic bacteriologic surveillance, watching for drug toxicity and drug-drug interactions, ensuring adherence and compliance to treatment, and managing comorbidity.
The Bronchiectasis Severity Index is a useful clinical predictive tool that identifies patients at risk of future mortality, hospitalization, and exacerbations and can be used to evaluate the need for specific treatment.30 The index is based on dyspnea score, lung function tests, colonization of pathogens, and extent of disease.
Case 1 Continued
After approximately 2 months of observation and symptomatic treatment, without specific antibiotic therapy, the patient’s symptoms continue. She now develops intermittent hemoptysis. Repeat sputum studies reveal moderate growth of M. avium. A follow-up CT scan shows progression of disease, with an increase in the tree-in-bud pattern (Figure 3).
What treatment protocols are recommended for MAC pulmonary disease?
As per the ATS/IDSA statement, macrolides are the mainstay of treatment for pulmonary MAC disease.11 Macrolides achieve an increased concentration in the lung, and when used for treatment of pulmonary MAC disease, there is a strong correlation between in vitro susceptibility, in vivo (clinical) response, and the immunomodulating effects of macrolides.31,32 Macrolide-containing regimens have demonstrated efficacy in patients with MAC pulmonary disease33,34; however, macrolide monotherapy should be avoided to prevent the development of resistance.
At the outset, it is critical to establish the objective criteria for determining response and to ensure that the patient understands the goals of the treatment and expectations of the treatment plan. Moreover, experts suggest that due to the possibility of drug intolerance, side effects, and the need for prolonged therapy, a “step ladder” ramping up approach to treatment could be adopted, with gradual introduction of therapy within a short time period; this approach may improve compliance and adherence to treatment.11 If this approach is used, the doses may have to be divided. Patients who are unable to tolerate daily medications, even with dosage adjustment, should be tried on an intermittent treatment regimen. Older female patients frequently require gradual introduction of medications (ie, 1 medication added to the regimen every 1 to 2 weeks) to evaluate tolerance to each medication and medication dose.11 Commonly encountered adverse effects of NTM treatment include intolerance to clarithromycin due to gastrointestinal problems, low body mass index, or age older than 70 years.
After determining that the patient requires therapy, the standard recommended treatment for MAC pulmonary disease includes the following: for most patients with nodular/bronchiectasis disease, a thrice-weekly regimen of clarithromycin (1000 mg) or azithromycin (500 mg), rifampin (600 mg), and ethambutol (25 mg/kg) is recommended. For patients with cavitary MAC pulmonary disease or severe nodular/bronchiectasis disease, the guidelines recommend a daily regimen of clarithromycin (500-1000 mg) or azithromycin (250 mg), rifampin (600 mg) or rifabutin (150–300 mg), and ethambutol (15 mg/kg), with consideration of intravenous (IV) amikacin 3 times/week early in therapy (Table 2).11
The treatment of MAC hypersensitivity-like disease speaks to the controversy of whether this is an inflammatory process, infectious process, or a combination of inflammation and infection. Avoidance of exposure is the mainstay of management. In some cases, steroids are used with or without a short course of anti-MAC therapy (ie, clarithromycin or azithromycin with rifampin and ethambutol).
Prophylaxis for disseminated MAC disease should be given to adults with HIV infection who have a CD4+ count less than 50 cells/μL. Azithromycin 1200 mg/week or clarithromycin 1000 mg/day has proven efficacy, and rifabutin 300 mg/day is also effective but less well tolerated. Rifabutin is more active in vitro against MAC than rifampin, and is used in HIV-positive patients because of drug-drug interaction between antiretroviral drugs and rifampin.
Continue to: Case 1 Continued
Case 1 Continued
The patient is treated with clarithromycin, rifampin, and ethambutol for 1 year, with sputum conversion after 9 months. In the latter part of her treatment, she experiences decreased visual acuity. Treatment is discontinued prematurely after 1 year due to drug toxicity and continued intolerance to drug therapy. The patient remains asymptomatic for 8 months, and then begins to experience mild to moderate hemoptysis, with increasing cough and sputum production associated with postural changes during exercise. Physical examination overall remains unchanged. Three sputum results reveal heavy growth of MAC, and a CT scan of the chest shows a cavitary lesion in the left upper lobe along with the nodular bronchiectasis (Figure 4).
What are the management options at this stage?
Based on this patient’s continued symptoms, progression of radiologic abnormalities, and current culture growth, she requires re-treatment. With the adverse effects associated with ethambutol during the first round of therapy, the drug regimen needs to be modified. Several considerations are relevant at this stage. Relapse rates range from 20% to 30% after treatment with a macrolide-based therapy.11,34 Obtaining a culture-sensitivity profile is imperative in these cases. Of note, treatment should not be discontinued altogether, but instead the toxic agent should be removed from the treatment regimen. Continuing treatment with a 2-drug regimen of clarithromycin and rifampin may be considered in this patient. Re-infection with multiple genotypes may also occur after successful drug therapy, but this is primarily seen in MAC patients with nodular bronchiectasis.34,35 Patients in whom previous therapy has failed, even those with macrolide-susceptible MAC isolates, are less likely to respond to subsequent therapy. Data suggest that intermittent medication dosing is not effective for patients with severe or cavitary disease or in those in whom previous therapy has failed.36 In this case, treatment should include a daily 3-drug therapy, with an injectable thrice-weekly aminoglycoside. Other agents such as linezolid and clofazimine may have to be tried. Cycloserine, ethionamide, and other agents are sometimes used, but their efficacy is unproven and doubtful. Pyrazinamide and isoniazid have no activity against MAC.
Treatment Failure and Drug Resistance
Treatment failure is considered to have occurred if patients have not had a response (microbiologic, clinical, or radiographic) after 6 months of appropriate therapy or had not achieved conversion of sputum to culture-negative after 12 months of appropriate therapy.11 This occurs in about 40% of patients. Multiple factors can interfere with the successful treatment of MAC pulmonary disease, including medication nonadherence, medication side effects or intolerance, lack of response to a medication regimen, or the emergence of a macrolide-resistant or multidrug-resistant strain. Inducible macrolide resistance remains a potential factor.34-36 A number of characteristics of NTM contribute to the poor response to currently used antibiotics: the organisms have a lipid outer membrane and prefer to adhere to surfaces and form biofilms, which makes them relatively impermeable to antibiotics.37 Also, NTM replicate in phagocytic cells, allowing them to subvert normal cellular defense mechanisms. Furthermore, NTM can display colony variants, whereby single colony isolates switch between antibiotic-susceptible and -resistant variants. These factors have also impeded in development of new antibiotics for NTM infection.37
Recent limited approval of amikacin liposomal inhalation suspension (ALIS) for treatment failure and refractory MAC infection in combination with guideline-based antimicrobial therapy (GBT) is a promising addition to the available treatment armamentarium. In a multinational trial, the addition of ALIS to GBT for treatment-refractory MAC lung disease achieved significantly greater culture conversion rates by month 6 than GBT alone, with comparable rates of serious adverse events.38
Is therapeutic drug monitoring recommended during treatment of MAC pulmonary disease?
Treatment failure may also be drug-related, including poor drug penetration into the damaged lung tissue or drug-drug interactions leading to suboptimal drug levels. Peak serum concentrations have been found to be below target ranges in approximately 50% of patients using a macrolide and ethambutol. Concurrent use of rifampin decreases the peak serum concentration of macrolides and quinolones, with acceptable target levels seen in only 18% to 57% of cases. Whether this alters patient outcomes is not clear.39-42 Factors identified as contributing to the poor response to therapy include poor compliance, cavitary disease, previous treatment for MAC pulmonary disease, and a history of chronic obstructive lung disease. Studies by Koh and colleagues40 and van Ingen and colleagues41 with pharmacokinetic and pharmacodynamics data showed that, in patients on MAC treatment with both clarithromycin and rifampicin, plasma levels of clarithromycin were lower than the recommended minimal inhibitory concentrations (MIC) against MAC for that drug. The studies also showed that rifampicin lowered clarithromycin concentrations more than did rifabutin, with the AUC/MIC ratio being suboptimal in nearly half the cases. However, low plasma clarithromycin concentrations did not have any correlation with treatment outcomes, as the peak plasma drug concentrations and the peak plasma drug concentration/MIC ratios did not differ between patients with unfavorable treatment outcomes and those with favorable outcomes. This is further compounded by the fact that macrolides achieve higher levels in lung tissue than in plasma, and hence the significance of low plasma levels is unclear; however, it is postulated that achieving higher drug levels could, in fact, lead to better clinical outcomes. Pending specific well-designed, prospective randomized controlled trials, routine therapeutic drug monitoring is not currently recommended, although some referral centers do this as their practice pattern.
Is surgery an option in this case?
The overall 5-year mortality for MAC pulmonary disease was approximately 28% in a retrospective analysis, with patients with cavitary disease at increased risk for death at 5 years.42 As such, surgery is an option in selected cases as part of adjunctive therapy along with anti-MAC therapy based on mycobacterial sensitivity. Surgery is used as either a curative approach or a “debulking” measure.11 When present, clearly localized disease, especially in the upper lobe, lends itself best to surgical intervention. Surgical resection of a solitary pulmonary nodule due to MAC, in addition to concomitant medical treatment, is recommended. Surgical intervention should be considered early in the course of the disease because it may provide a cure without prolonged treatment and its associated problems, and this approach may lead to early sputum conversion. Surgery should also be considered in patients with macrolide-resistant or multidrug-resistant MAC infection or in those who cannot tolerate the side effects of therapy, provided that the disease is focal and limited. Patients with poor preoperative lung function have poorer outcomes than those with good lung function, and postoperative complications arising from treatment, especially with a right-sided pneumonectomy, tend to occur more frequently in these patients. Thoracic surgery for NTM pulmonary disease must be considered cautiously, as this is associated with significant morbidity and mortality and is best performed at specialized centers that have expertise and experience in this field.43
Continue to: Mycobacterium abscessus Complex
Mycobacterium abscessus Complex
Case Patient 2
A 64-year-old man who is an ex-smoker presents with chronic cough, mild shortness of breath on exertion, low-grade fever, and unintentional weight loss of 10 lb. Physical exam is unremarkable. The patient was diagnosed with immunoglobulin deficiency (low IgM and low IgG4) in 2002, and has been on replacement therapy since then. He also has had multiple episodes of NTM infection, with MAC and M. kansasii infections diagnosed in 2012-2014, which required 18 months of multi-drug antibiotic treatment that resulted in sputum conversion. Pulmonary function testing done on this visit in 2017 shows mild obstructive impairment.
Chest radiograph and CT scan show bilateral bronchiectasis (Figure 5 and Figure 6).
The results of serial sputum microbiology testing performed over the course of 6 months are outlined below:
- 5/2017 (bronchoalveolar lavage): 2+; M. abscessus
- 9/2017 × 2: smear (–); group IV RGM
- 11/2017: smear (–); M. abscessus (> 50 CFU)
- 12/2017: smear (–); M. abscessus (> 50 CFU)
What are the clinical considerations in this patient with multiple NTM infections?
M. abscessus complex was originally described in soft tissue abscesses and skin infections possibly resulting from soil or water contamination. Subspeciation of M. abscessus complex during laboratory testing is critical to facilitate selection of a specific therapeutic approach; treatment decisions are impacted by the presence of an active erm gene and in vitro macrolide sensitivity, which differ between subspecies. The most acceptable classification outlines 3 species in the M. abscessus complex: Mycobacterium abscessus subsp abscessus, Mycobacterium abscessus subsp bolletii (both with an active erm gene responsible for macrolide resistance), and Mycobacterium abscessus subsp massiliense (with an inactive erm gene and therefore susceptible to macrolides).44
RGM typically manifest in skin, soft tissue, and bone, and can cause soft tissue, surgical wound, and catheter-related infections. Although the role of RGM as pulmonary pathogens is unclear, underlying diseases associated with RGM include previously treated mycobacterial disease, coexistent pulmonary diseases with or without MAC, cystic fibrosis, malignancies, and gastroesophageal disorders. M. abscessus is the third most commonly identified respiratory NTM and accounts for the majority (80%) of RGM respiratory isolates. Other NTM reported to cause both lung disease and skin, bone, and joint infections include Mycobacterium simiae, Mycobacterium xenopi, and Mycobacterium malmoense. Ocular granulomatous diseases, such as chorioretinitis and keratitis, have been reported with both RGM and Runyon group III SGM, such as MAC or M. szulgai, following trauma or refractive surgery. These can mimic fungal, herpetic, or amebic keratitis. The pulmonary syndromes associated with multiple culture positivity are seen in elderly women with bronchiectasis or cavitary lung disease and/or associated with gastrointestinal symptoms of acid reflux, with or without achalasia and concomitant lipoid interstitial pneumonia.45
Generally, pulmonary disease progresses slowly, but lung disease attributed to RGM can result in respiratory failure. Thus, RGM should be recognized as a possible cause of chronic mycobacterial lung disease, especially in immunocompromised patients, and respiratory isolates should be assessed carefully. Identification and drug susceptibility testing are essential before initiation of treatment for RGM.
What is the approach to management of M. abscessus pulmonary disease in a patient without cystic fibrosis?
The management of M. abscessus pulmonary infection as a subset of RGM requires a considered step-wise approach. The criteria for diagnosis and threshold for starting treatment are the same as those used in the management of MAC pulmonary disease,11 but the treatment of M. abscessus pulmonary infection is more complex and has lower rates of success and cure. Also, antibiotic treatment presents challenges related to rapid identification of the causative organism, nomenclature, resistance patterns, and tolerance of treatment and side effects. If a source such as catheter, access port, or any surgical site is identified, prompt removal and clearance of the infected site are strongly advised
In the absence of any controlled clinical trials, treatment of RGM is based on in vitro susceptibility testing and expert opinion. As in MAC pulmonary disease, macrolides are the mainstay of treatment, with an induction phase of intravenous antibiotics. Treatment may include a combination of injectable aminoglycosides, imipenem, or cefoxitin and oral drugs such as a macrolide (eg, clarithromycin, azithromycin), doxycycline, fluoroquinolones, trimethoprim/sulfamethoxazole, or linezolid. While antibiotic treatment of M. abscessus pulmonary disease is based on in vitro sensitivity pattern to a greater degree than is treatment of MAC pulmonary disease, this approach has significant practical limitations and hence variable applicability. The final choice of antibiotics is best based on the extended susceptibility results, if available. The presence of an active erm gene on a prolonged growth specimen in M. abscessus subsp abscessus and M. abscessus subsp bolletii precludes the use of a macrolide. In such cases, amikacin, especially in an intravenous form, is the mainstay of treatment based on MIC. Recently, there has been a resurgence in interest in the use of clofazimine in combination with amikacin when treatment is not successful in patients with M. abscessus subsp abscessus or M. bolletii with an active erm gene.45,46 When localized abscess formation is noted, surgery may be the best option, with emphasis on removal of implants and catheters if implicated in RGM infection.
Attention must also be given to confounding pulmonary and associated comorbidities. This includes management of bronchiectasis with appropriately aggressive airway clearance techniques; anti-reflux measures for prevention of micro-aspiration; and management of other comorbid pulmonary conditions, such as chronic obstructive pulmonary disease, pulmonary fibrosis, and sarcoidosis, if applicable. These interventions play a critical role in clearing the M. abscessus infection, preventing progression of disease, and reducing morbidity. The role of immunomodulatory therapy needs to be considered on a regular, ongoing basis. Identification of genetic factors and correction of immune deficiencies may help in managing the infection.
Case Patient 2 Conclusion
The treatment regimen adopted in this case includes a 3-month course of daily intravenous amikacin and imipenem with oral azithromycin, followed by a continuation phase of azithromycin with clofazimine and linezolid. Airway clearance techniques such as Vest/Acapella/CPT are intensified and monthly intravenous immunoglobulin therapy is continued. The patient responds to treatment, with resolution of his clinical symptoms and reduction in the colony count of M. abscessus in the sputum.
Summary
NTM are ubiquitous in the environment, and NTM infection has variable manifestations, especially in patients with no recognizable immune impairments. Underlying comorbid conditions with bronchiectasis complicate its management. Treatment strategies must be individualized based on degree of involvement, associated comorbidities, immune deficiencies, goals of therapy, outcome-based risk-benefit ratio assessment, and patient engagement and expectations. In diffuse pulmonary disease, drug treatment remains difficult due to poor match of in vitro and in vivo culture sensitivity, side effects of medications, and high failure rates. When a localized resectable foci of infection is identified, especially in RGM disease, surgical treatment may be the best approach in selected patients, but it must be performed in centers with expertise and experience in this field.
1. Johnson MM, Odell JA. Nontuberculous mycobacterial pulmonary infections. J Thorac Dis. 2014;6:210-220.
2. Falkinham JO III. Environmental sources of NTM. Clin Chest Med. 2015;36:35-41.
3. Falkinham JO III, Current epidemiological trends in NTM. Curr Environ Health Rep. 2016;3:161-167.
4. Honda JR, Knight V, Chan ED. Pathogenesis and risk factors for nontuberculous mycobacterial lung disease. Clin Chest Med. 2015;36:1-11.
5. Marras TK, Mirsaeidi M, Chou E, et al. Health care utilization and expenditures following diagnosis of nontuberculous mycobacterial lung disease in the United States. Manag Care Spec Pharm. 2018;24:964-974.
6. Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated healthcare delivery systems. Am J Respir Crit Care Med. 2010;182:970-976.
7. Winthrop KL, McNelley E, Kendall B, et al. Pulmonary nontuberculous mycobacterial disease prevalence and clinical features: an emerging public health disease. Am J Respir Crit Care Med. 2010;182:977-982.
8. Adjemian, Olivier KN, Seitz AE, J et al. Prevalence of nontuberculous mycobacterial lung disease in US Medicare beneficiaries. Am J Respir Crit Care Med. 2012;185;881-886.
9. Ringshausen FC, Apel RM, Bange FC, et al. Burden and trends of hospitalizations associated with pulmonary nontuberculous mycobacterial infections in Germany, 2005-2011. BMC Infect Dis. 2013;13:231.
10. Aliyu G, El-Kamary SS, Abimiku A, et al. Prevalence of non-tuberculous mycobacterial infections among tuberculosis suspects in Nigeria. PLoS One. 2013;8:e63170.
11. Griffith DE, Aksamit T, Brown-Elliott, et al; American Thoracic Society; Infectious Diseases Society of America. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367-415.
12. Wallace RJ Jr, Zhang Y, Brown BA, et al. Polyclonal Mycobacterium avium complex infections in patients with nodular bronchiectasis. Am J Respir Crit Care Med. 1998;158:1235-1244.
13. Gordin FM, Horsburgh CR Jr. Mycobacterium avium complex. In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Elsevier; 2015.
14. Chitty S, Ali J. Mycobacterium avium complex pulmonary disease in immune competent patients. South Med J. 2005;98:646-52.
15. Ramirez J, Mason C, Ali J, Lopez FA. MAC pulmonary disease: management options in HIV-negative patients. J La State Med Soc. 2008;160:248-254.
16. Iseman MD, Buschman DL, Ackerson LM. Pectus excavatum and scoliosis. Thoracic anomalies associated with pulmonary disease caused by Mycobacterium avium complex. Am Rev Respir Dis. 1991;144:914-916.
17. Kim RD, Greenburg DE, Ehrmantraut ME, et al. Pulmonary nontuberculous mycobacterial disease: prospective study of a distinct preexisting syndrome. Am J Respir Crit Care Med. 2008;178:1066-1074.
18. Ziedalski TM, Kao PN, Henig NR, et al. Prospective analysis of cystic fibrosis transmembrane regulator mutations in adults with bronchiectasis or pulmonary nontuberculous mycobacterial infection. Chest. 2006;130:995-1002.
19. Koh WJ, Lee KS, Kwon OJ, et al. Bilateral bronchiectasis and bronchiolitis at thin-section CT: diagnostic implications in nontuberculous mycobacterial pulmonary infection. Radiology. 2005;235:282-288.
20. Swensen SJ, Hartman TE, Williams DE. Computed tomographic diagnosis of Mycobacterium avium-intracellulare complex in patients with bronchiectasis. Chest. 1994;105:49-52.
21. Huang JH, Kao PN, Adi V, Ruoss SJ. Mycobacterium avium intracellulare pulmonary infection in HIV-negative patients without preexisting lung disease: diagnostic and management limitations. Chest. 1999;115:1033-1040.
22. Cappelluti E, Fraire AE, Schaefer OP. A case of “hot tub lung” due to Mycobacterium avium complex in an immunocompetent host. Arch Intern Med. 2003;163:845-848.
23. Nightingale SD, Byrd LT, Southern PM, et al. Incidence of Mycobacterium avium-intracellulare complex bacteremia in human immunodeficiency virus-positive patients. J Infect Dis. 1992;165:1082-1085.
24. Horsburgh CR Jr, Selik RM. The epidemiology of disseminated tuberculous mycobacterial infection in the acquired immunodeficiency syndrome (AIDS). Am Rev Respir Dis. 1989;139:4-7.
25. Chin DP, Hopewell PC, Yajko DM, et al. Mycobacterium avium complex in the respiratory or gastrointestinal tract and the risk of M. avium complex bacteremia in patients with human immunodeficiency virus infection. J Infect Dis. 1994;169:289-295.
26. Khan K, Wang J, Marras TK. Nontuberculous mycobacterial sensitization in the United States: national trends over three decades. Am J Respir Crit Care Med. 2007;176:306-313.
27. Lillo M, Orengo S, Cernoch P, Harris RL. Pulmonary and disseminated infection due to Mycobacterium kansasii: a decade of experience. Rev Infect Dis. 1990;12:760-767.
28. Andersen P, Munk ME, Pollock JM, Doherty TM. Specific immune-based diagnosis of tuberculosis. Lancet. 2000;356:1099-1104.
29. Arend SM, van Meijgaarden KE, de Boer K, et al. Tuberculin skin testing and in vitro T cell responses to ESAT-6 and culture filtrate protein 10 after infection with Mycobacterium marinum or M. kansasii. J Infect Dis. 2002;186:1797-1807.
30. James D, Chalmers JD, Goeminne P, et al. The Bronchiectasis Severity Index: an international derivation and validation study. Am J Respir Crit Care Med. 2014;189:576-585.
31. Heifets L. MIC as a quantitative measurement of the susceptibility of Mycobacterium avium strains to seven antituberculosis drugs. Antimicrob Agents Chemother. 1988;32:1131-1136.
32. Horsburgh CR Jr, Mason UG 3rd, Heifits LB, et al. Response to therapy of pulmonary Mycobacterium avium intracellulare infection correlates with results of in vitro susceptibility testing. Am Rev Respir Dis. 1987;135:418-421.
33. Rubin BK, Henke MO. Immunomodulatory activity and effectiveness of macrolides in chronic airway disease. Chest. 2004;125(2 Suppl):70S-78S.
34. Wallace RJ Jr, Brown BA, Griffith DE, et al. Clarithromycin regimens for pulmonary Mycobacterium avium complex. The first 50 patients. Am J Respir Crit Care Med. 1996;153:1766-1772.
35. Griffith DE, Brown-Elliott BA, Langsjoen B, et al. Clinical and molecular analysis of macrolide resistance in Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006;174:928-934.
36. Lam PK, Griffith DE, Aksamit TR, et al. Factors related to response to intermittent treatment of Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006;173:1283-1289.
37. Falkinham J III. Challenges of NTM drug development. Front Microbiol. 2018;9:1613.
38. Griffith DE, Eagle G, Thomson R, et al. Amikacin liposome inhalation suspension for treatment-refractory lung disease caused by Mycobacterium avium complex (CONVERT). A prospective, open-label, randomized study. Am J Respir Crit Care Med. 2018;198:1559-1569.
39. Schluger NW. Treatment of pulmonary Mycobacterium avium complex infections: do drug levels matter? Am J Respir Crit Care Med. 2012;186:710-711.
40. Van Ingen J, Egelund EF, Levin A, et al. The pharmacokinetics and pharmacodynamics of pulmonary Mycobacterium avium complex disease treatment. Am J Respir Crit Care Med. 2012;186:559-565.
41. Koh WJ, Jeong BH, Jeon K, et al. Therapeutic drug monitoring in the treatment of Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2012;186:797-802.
42. Ito Y, Hirai T, Maekawa K, et al. Predictors of 5-year mortality in pulmonary MAC disease. Int J Tuberc Lung Dis. 2012;16:408-414.
43. Yuji S, Yutsuki N, Keiichiso T, et al. Surgery for Mycobacterium avium lung disease in the clarithromycin era. Eur J Cardiothor Surg. 2002;21:314-318.
44. Tortoli E, Kohl TA, Brown-Elliott BA, et al. Emended description of Mycobacterium abscessus, Mycobacterium abscessus subsp. abscessus and Mycobacterium abscessus subsp. bolletii and designation of Mycobacterium abscessus subsp. massiliense comb. Int J Syst Evol Microbiol. 2016; 66:4471-4479.
45. Griffith DE, Girard WM, Wallace RJ Jr. Clinical features of pulmonary disease caused by rapidly growing mycobacteria. An analysis of 154 patients. Am Rev Respir Dis. 1993;147:1271-1278.
46. Koh WJ, Jeong BH, Kim SY, et al. Mycobacterial characteristics and treatment outcomes in Mycobacterium abscessus lung disease. Clin Infect Dis. 2017;64:309-316.
Nontuberculous mycobacterial pulmonary disease is a broad term for a group of pulmonary disorders caused and characterized by exposure to environmental mycobacteria other than those belonging to the Mycobacterium tuberculosis complex and Mycobacterium leprae. Mycobacteria are aerobic, nonmotile organisms that appear positive with acid-fast alcohol stains. Nontuberculous mycobacteria (NTM) are ubiquitous in the environment and have been recovered from domestic and natural water sources, soil, and food products, and from around livestock, cattle, and wildlife.1-3 To date, no evidence exists of human-to-human or animal-to-human transmission of NTM in the general population. Infections in humans are usually acquired from environmental exposures, although the specific source of infection cannot always be identified. Similarly, the mode of infection with NTM has not been established with certainty, but it is highly likely that the organism is implanted, ingested, aspirated, or inhaled. Aerosolization of droplets associated with use of bathroom showerheads and municipal water sources and soil contamination are some of the factors associated with the transmission of infection. Proven routes of transmission include showerheads and potting soil dust.2,3
NTM pulmonary disease occurs in individuals with or without comorbid conditions such as bronchiectasis, chronic obstructive pulmonary disease, pulmonary fibrosis, or structural lung diseases. Slender, middle-aged or elderly white females with marfanoid body habitus, with or without apparent immune or genetic disorders, showing impaired airway and mucus clearance present with this infection as a form of underlying bronchiectasis (Lady Windermere syndrome). It is unclear why NTM infections and escalation to clinical disease occur in certain individuals. Many risk factors, including inherited and acquired defects of host immune response (eg, cystic fibrosis trait and α1 antitrypsin deficiency), have been associated with increased susceptibility to NTM infections.4
NTM infection can lead to chronic symptoms, frequent exacerbations, progressive functional and structural lung destruction, and impaired quality of life, and is associated with an increased risk of hospitalization and higher 5-year all-cause mortality. As such, NTM disease is drawing increasing attention at the clinical, academic, and research levels.5 This case-based review outlines the clinical features of NTM infection, with a focus on the challenges in diagnosis, treatment, and management of NTM pulmonary disease. The cases use Mycobacterium avium complex (MAC), a slow-growing mycobacteria (SGM), and Mycobacterium abscessus, a rapidly growing mycobacteria (RGM), as prototypes in a non–cystic fibrosis, non-HIV clinical setting.
Epidemiology
Of the almost 200 isolated species of NTM, the most prevalent pathogens for respiratory disease in the United States are MAC, Mycobacterium kansasii, and M. abscessus. MAC accounts for more than 80% of cases of NTM respiratory disease in the United States.6 The prevalence of NTM disease is increasing at a rate of about 8% each year, with 75,000 to 105,000 patients diagnosed with NTM lung disease in the United States annually. NTM infections in the United States are increasing among patients aged 65 years and older, a population that is expected to nearly double by 2030.7,8
Isolation and prevalence of many NTM species are higher in certain geographic areas of the United States, especially in the southeast. The US coastal regions have a higher prevalence of NTM pulmonary disease, and account for 70% of NTM cases in the United States each year. Half of patients diagnosed with NTM lung disease reside in 7 states: Florida, New York, Texas, California, Pennsylvania, New Jersey, and Ohio, with 1 in 7 residing in Florida. Three parishes in Louisiana are among the top 10 counties with the highest prevalence in United States. The prevalence of NTM infection–associated hospitalizations is increasing worldwide as well. Co-infection with tuberculosis and multiple NTMs in individual patients has been observed clinically and documented in patients with and without HIV.9,10
It is not clear why the prevalence of NTM pulmonary disease is increasing, but there may be several contributing factors: (1) an increased awareness and identification of NTM infection sources in the environment; (2) an expanding cohort of immunocompromised individuals with exogenous or endogenous immune deficiencies; (3) availability of improved diagnostic techniques, such as use of high-performance liquid chromatography (HPLC), DNA probes, and gene sequencing; and (4) an increased awareness of the morbidity and mortality associated with NTM pulmonary disease. However, it is important to recognize that to best understand the clinical relevance of epidemiologic studies based on laboratory diagnosis and identification, the findings must be evaluated by correlating them with the microbiological and other clinical criteria established by the American Thoracic Society (ATS)/Infectious Diseases Society of America (IDSA) guidelines.11
Continue to: Mycobacterium avium Complex
Mycobacterium avium Complex
Case Patient 1
A 48-year-old woman who has never smoked and has no past medical problems, except seasonal allergic rhinitis and “colds and flu-like illness” once or twice a year, is evaluated for a chronic lingering cough with occasional sputum production. The patient denies any other chronic symptoms and is otherwise active. Physical examination reveals no specific findings except mild pectus excavatum and mild scoliosis. Body mass index is 22 kg/m2. Chest radiograph shows nonspecific increased markings in the lower zones. Computed tomography (CT) scan of the chest reveals minimal nodular and cylindrical bronchiectasis in both lungs (Figure 1). No previous radiographs are available for comparison. The patient is HIV-negative. Sputum tests reveal normal flora, and both fungus and acid-fast bacilli smear are negative. Culture for mycobacteria shows scanty growth of MAC in 1 specimen.
What is the clinical presentation of MAC pulmonary disease?
Among NTM, MAC is the most common cause of pulmonary disease worldwide.6 MAC primarily includes 2 species: M. avium and Mycobacterium intracellulare. M. avium is the more important pathogen in disseminated disease, whereas M. intracellulare is the more common respiratory pathogen.11 These organisms are genetically similar and generally not differentiated in the clinical microbiology laboratory, although there are isolated reports in the literature suggesting differences in prevalence, presentation, and prognosis in M. avium infection versus M. intracellulare infection.12
Three major disease syndromes are produced by MAC in humans: pulmonary disease, usually in adults whose systemic immunity is intact; disseminated disease, usually in patients with advanced HIV infection; and cervical lymphadenitis.13 Pulmonary disease caused by MAC may take on 1 of several clinically different forms, including asymptomatic “colonization” or persistent minimal infection without obvious clinical significance; endobronchial involvement; progressive pulmonary disease with radiographic and clinical deterioration and nodular bronchiectasis or cavitary lung disease; hypersensitivity pneumonitis; or persistent, overwhelming mycobacterial growth with symptomatic manifestations, often in a lung with underlying damage due to either chronic obstructive lung disease or pulmonary fibrosis (Table 1).14
Cavitary Disease
The traditionally recognized presentation of MAC lung disease has been apical cavitary lung disease in men in their late 40s and early 50s who have a history of cigarette smoking, and frequently, excessive alcohol use. If left untreated, or in the case of erratic treatment or macrolide drug resistance, this form of disease is generally progressive within a relatively short time and can result in extensive cavitary lung destruction and progressive respiratory failure.15
Nodular Bronchiectasis
The more common presentation of MAC lung disease, which is outlined in the case described here, is interstitial nodular infiltrates, frequently involving the right middle lobe or lingula and predominantly occurring in postmenopausal, nonsmoking white women. This is sometimes labelled “Lady Windermere syndrome.” These patients with M. avium infection appear to have similar clinical characteristics and body types, including lean build, scoliosis, pectus excavatum, and mitral valve prolapse.16,17 The mechanism by which this body morphotype predisposes to pulmonary mycobacterial infection is not defined, but ineffective mucociliary clearance is a possible explanation. Evidence suggests that some patients may be predisposed to NTM lung disease because of preexisting bronchiectasis. Some potential etiologies of bronchiectasis in this population include chronic sinusitis, gastroesophageal reflux with chronic aspiration, α1 antitrypsin deficiency, and cystic fibrosis genetic traits and mutations.18 Risk factors for increased morbidity and mortality include the development of cavitary disease, age, weight loss, lower body mass index, and other comorbid conditions.
This form of disease, termed nodular bronchiectasis, tends to have a much slower progression than cavitary disease, such that long-term follow-up (months to years) may be necessary to demonstrate clinical or radiographic changes.11 The radiographic term “tree-in-bud” has been used to describe what may reflect inflammatory changes, including bronchiolitis. High-resolution CT scans of the chest are especially helpful for diagnosing this pattern of MAC lung disease, as bronchiectasis and small nodules may not be easily discernible on plain chest radiograph. The nodular/bronchiectasis radiographic pattern can also be seen with other NTM pathogens, including M. abscessus, Mycobacterium simiae, and M. kansasii. Solitary nodules and dense consolidation have also been described. Pleural effusions are uncommon, but reactive pleural thickening is frequently seen. Co-pathogens may be isolated from culture, including Pseudomonas aeruginosa, Staphylococcus aureus, and, occasionally, other NTM such as M. abscessus or Mycobacterium chelonae.19-21
Hypersensitivity Pneumonitis
Hypersensitivity pneumonitis, initially described in patients who were exposed to hot tubs, mimics allergic hypersensitivity pneumonitis, with respiratory symptoms and culture/tissue identification of MAC or sometimes other NTM. It is unclear whether hypersensitivity pneumonitis is an inflammatory process, an infection, or both, and opinion regarding the need for specific antibiotic treatment is divided.11,22 However, avoidance of exposure is prudent and recommended.
Disseminated Disease
Disseminated NTM disease is associated with very low CD4+ lymphocyte counts and is seen in approximately 5% of patients with HIV infection.23-25 Although disseminated NTM disease is rarely seen in immunosuppressed patients without HIV infection, it has been reported in patients who have undergone renal or cardiac transplant, patients on long-term corticosteroid therapy, and those with leukemia or lymphoma. More than 90% of infections are caused by MAC; other potential pathogens include M. kansasii, M. chelonae, M. abscessus, and Mycobacterium haemophilum. Although seen less frequently since the advent of highly active antiretroviral therapy, disseminated infection can develop progressively from an apparently indolent or localized infection or a respiratory or gastrointestinal source. Signs and symptoms of disseminated infection (specifically MAC-associated disease) are nonspecific and include fever, night sweats, weight loss, and abdominal tenderness. Disseminated MAC disease occurs primarily in patients with more advanced HIV disease (CD4+ count typically < 50 cells/μL). Clinically, disseminated MAC manifests as intermittent or persistent fever, constitutional symptoms with organomegaly and organ-specific abnormalities (eg, anemia, neutropenia from bone marrow involvement, adenopathy, hepatosplenomegaly), and elevations of liver enzymes or lung infiltrates from pulmonary involvement.
Continue to: What are the criteria for diagnosing NTM pulmonary disease?
What are the criteria for diagnosing NTM pulmonary disease?
The diagnosis of NTM disease is based on clinical, radiologic, and mycobacterial correlation with good communication between the experts in this field. The ATS/IDSA criteria for diagnosing NTM lung disease are shown in Figure 2. These criteria best apply to MAC, M. kansasii, and M. abscessus, but are also clinically applied to other NTM respiratory pathogens. The diagnosis of MAC infection is most readily established by culture of blood, bone marrow, respiratory secretions/fluid, or tissue specimens from suspected sites of involvement. Due to erratic shedding of MAC into the respiratory secretions in patients with nodular bronchiectasis, as compared to those with the cavitary form of the disease, sputum may be intermittently positive, with variable colony counts and polyclonal infections.12 Prior to the advent of high-resolution CT, isolation of MAC organisms from the sputum of such patients was frequently dismissed as colonization.
Mycobacterial Testing
Because of the nonspecific symptoms and lack of diagnostic specificity of chest imaging, the diagnosis of NTM lung disease requires microbiologic confirmation. Specimens sent to the laboratory for identification of NTM must be handled with care to prevent contamination and false-positive results. Transport media and preservatives should be avoided, and transportation of the specimens should be prompt. These measures will prevent bacterial overgrowth. Furthermore, the yield of NTM may be affected if the patient has used antibiotics, such as macrolides and fluoroquinolones, prior to obtaining the specimen.
NTM should be identified at the species and subspecies level, although this is not practical in community practice settings. The preferred staining procedure in the laboratory is the fluorochrome method. Some species require special growth conditions and/or lower incubation temperatures, and other identification methods may have to be employed, such as DNA probes, polymerase chain reaction genotyping, nucleic acid sequence determination, and high-performance liquid chromatography. As a gold standard, clinical specimens for mycobacterial cultures should be inoculated onto 1 or more solid media (eg, Middlebrook 7H11 media and/or Lowenstein-Jensen media, the former of which is the preferred medium for NTM) and into a liquid medium (eg, BACTEC 12B broth or Mycobacteria growth indicator tube broth). Growth of visible colonies on solid media typically requires 2 to 4 weeks, but liquid media (eg, the radiometric BACTEC system), used as a supplementary and not as an exclusive test, usually produce results within 10 to 14 days. Furthermore, even after initial growth, identification of specific isolates based on the growth characteristics on solid media requires additional time. Use of specific nucleic acid probes for MAC and M. kansasii and HPLC testing of mycolic acid patterns in acid-fast bacilli smear–positive specimens can reduce the turnaround time of specific identification of a primary culture–positive sample. However, HPLC is not sufficient for definitive identification of many NTM species, including the RGM. Other newer techniques, including 16S ribosomal DNA sequencing and polymerase chain reaction-restriction fragment length polymorphism analysis, also allow NTM to be identified and speciated more reliably and rapidly from clinical specimens.
Cost and other practical considerations limit widespread adoption of these techniques. However, the recognition that M. abscessus can be separated into more than 1 subspecies, and that there are important prognostic implications of that separation, lends urgency to the broader adoption of newer molecular techniques in the mycobacteriology laboratory. Susceptibility testing is based on the broth microdilution method; RGM usually grow within 7 days of subculture, and the laboratory time to culture is a helpful hint, although not necessarily specific. Recognizing the morphology of mycobacterial colony growth may also be helpful in identification.
Are skin tests helpful in diagnosing NTM infection?
Tuberculin skin testing remains a nonspecific marker of mycobacterial infection and does not help in further elucidating NTM infection. However, epidemiologic and laboratory studies with well-characterized antigens have shown that dual skin testing with tuberculosis- versus NTM-derived tuberculin can discriminate between prior NTM and prior tuberculosis disease. Species-specific skin test antigens are not commercially available and are not helpful in the diagnosis of NTM disease because of cross-reactivity of M. tuberculosis and some NTM. However, increased prevalence of NTM sensitization based on purified protein derivative testing has been noted in a recent survey, which is consistent with an observed increase in the rates of NTM infections, specifically MAC, in the United States.26,27
Interferon-gamma release assays (IGRAs) are now being used as an alternative to tuberculin skin testing to diagnose M. tuberculosis infection. Certain NTM species also contain gene sequences that encode for ESAT-6 or CFP-10 antigens used in the IGRAs, and hence, yield a positive IGRA test. These include M. marinum, M. szulgai, and M. kansasii.28,29 However, MAC organisms do not produce positive results on assays that use these antigens.
Continue to: What is the approach to management of NTM pulmonary disease?
What is the approach to management of NTM pulmonary disease?
The correlation of symptoms with radiographic and microbiologic evidence is essential to categorize the disease and determine the need for therapy. Making the diagnosis of NTM lung disease does not necessitate the institution of therapy. The decision to treat should be weighed against potential risks and benefits to the individual patient based on symptomatic, radiographic, and microbiologic criteria, as well as underlying systemic or pulmonary immune status. In the absence of evidence of clinical, radiologic, or mycobacterial progression of disease, pursuing airway clearance therapy and clinical surveillance without initiating specific anti-MAC therapy is a reasonable option.11 Identifying the sustained presence of NTM infection, especially MAC, in a patient with underlying clinical and radiographic evidence of bronchiectasis is of value in determining comprehensive treatment and management strategies. Close observation is indicated if the decision not to treat is made. If treatment is initiated, comprehensive management includes long-term follow-up with periodic bacteriologic surveillance, watching for drug toxicity and drug-drug interactions, ensuring adherence and compliance to treatment, and managing comorbidity.
The Bronchiectasis Severity Index is a useful clinical predictive tool that identifies patients at risk of future mortality, hospitalization, and exacerbations and can be used to evaluate the need for specific treatment.30 The index is based on dyspnea score, lung function tests, colonization of pathogens, and extent of disease.
Case 1 Continued
After approximately 2 months of observation and symptomatic treatment, without specific antibiotic therapy, the patient’s symptoms continue. She now develops intermittent hemoptysis. Repeat sputum studies reveal moderate growth of M. avium. A follow-up CT scan shows progression of disease, with an increase in the tree-in-bud pattern (Figure 3).
What treatment protocols are recommended for MAC pulmonary disease?
As per the ATS/IDSA statement, macrolides are the mainstay of treatment for pulmonary MAC disease.11 Macrolides achieve an increased concentration in the lung, and when used for treatment of pulmonary MAC disease, there is a strong correlation between in vitro susceptibility, in vivo (clinical) response, and the immunomodulating effects of macrolides.31,32 Macrolide-containing regimens have demonstrated efficacy in patients with MAC pulmonary disease33,34; however, macrolide monotherapy should be avoided to prevent the development of resistance.
At the outset, it is critical to establish the objective criteria for determining response and to ensure that the patient understands the goals of the treatment and expectations of the treatment plan. Moreover, experts suggest that due to the possibility of drug intolerance, side effects, and the need for prolonged therapy, a “step ladder” ramping up approach to treatment could be adopted, with gradual introduction of therapy within a short time period; this approach may improve compliance and adherence to treatment.11 If this approach is used, the doses may have to be divided. Patients who are unable to tolerate daily medications, even with dosage adjustment, should be tried on an intermittent treatment regimen. Older female patients frequently require gradual introduction of medications (ie, 1 medication added to the regimen every 1 to 2 weeks) to evaluate tolerance to each medication and medication dose.11 Commonly encountered adverse effects of NTM treatment include intolerance to clarithromycin due to gastrointestinal problems, low body mass index, or age older than 70 years.
After determining that the patient requires therapy, the standard recommended treatment for MAC pulmonary disease includes the following: for most patients with nodular/bronchiectasis disease, a thrice-weekly regimen of clarithromycin (1000 mg) or azithromycin (500 mg), rifampin (600 mg), and ethambutol (25 mg/kg) is recommended. For patients with cavitary MAC pulmonary disease or severe nodular/bronchiectasis disease, the guidelines recommend a daily regimen of clarithromycin (500-1000 mg) or azithromycin (250 mg), rifampin (600 mg) or rifabutin (150–300 mg), and ethambutol (15 mg/kg), with consideration of intravenous (IV) amikacin 3 times/week early in therapy (Table 2).11
The treatment of MAC hypersensitivity-like disease speaks to the controversy of whether this is an inflammatory process, infectious process, or a combination of inflammation and infection. Avoidance of exposure is the mainstay of management. In some cases, steroids are used with or without a short course of anti-MAC therapy (ie, clarithromycin or azithromycin with rifampin and ethambutol).
Prophylaxis for disseminated MAC disease should be given to adults with HIV infection who have a CD4+ count less than 50 cells/μL. Azithromycin 1200 mg/week or clarithromycin 1000 mg/day has proven efficacy, and rifabutin 300 mg/day is also effective but less well tolerated. Rifabutin is more active in vitro against MAC than rifampin, and is used in HIV-positive patients because of drug-drug interaction between antiretroviral drugs and rifampin.
Continue to: Case 1 Continued
Case 1 Continued
The patient is treated with clarithromycin, rifampin, and ethambutol for 1 year, with sputum conversion after 9 months. In the latter part of her treatment, she experiences decreased visual acuity. Treatment is discontinued prematurely after 1 year due to drug toxicity and continued intolerance to drug therapy. The patient remains asymptomatic for 8 months, and then begins to experience mild to moderate hemoptysis, with increasing cough and sputum production associated with postural changes during exercise. Physical examination overall remains unchanged. Three sputum results reveal heavy growth of MAC, and a CT scan of the chest shows a cavitary lesion in the left upper lobe along with the nodular bronchiectasis (Figure 4).
What are the management options at this stage?
Based on this patient’s continued symptoms, progression of radiologic abnormalities, and current culture growth, she requires re-treatment. With the adverse effects associated with ethambutol during the first round of therapy, the drug regimen needs to be modified. Several considerations are relevant at this stage. Relapse rates range from 20% to 30% after treatment with a macrolide-based therapy.11,34 Obtaining a culture-sensitivity profile is imperative in these cases. Of note, treatment should not be discontinued altogether, but instead the toxic agent should be removed from the treatment regimen. Continuing treatment with a 2-drug regimen of clarithromycin and rifampin may be considered in this patient. Re-infection with multiple genotypes may also occur after successful drug therapy, but this is primarily seen in MAC patients with nodular bronchiectasis.34,35 Patients in whom previous therapy has failed, even those with macrolide-susceptible MAC isolates, are less likely to respond to subsequent therapy. Data suggest that intermittent medication dosing is not effective for patients with severe or cavitary disease or in those in whom previous therapy has failed.36 In this case, treatment should include a daily 3-drug therapy, with an injectable thrice-weekly aminoglycoside. Other agents such as linezolid and clofazimine may have to be tried. Cycloserine, ethionamide, and other agents are sometimes used, but their efficacy is unproven and doubtful. Pyrazinamide and isoniazid have no activity against MAC.
Treatment Failure and Drug Resistance
Treatment failure is considered to have occurred if patients have not had a response (microbiologic, clinical, or radiographic) after 6 months of appropriate therapy or had not achieved conversion of sputum to culture-negative after 12 months of appropriate therapy.11 This occurs in about 40% of patients. Multiple factors can interfere with the successful treatment of MAC pulmonary disease, including medication nonadherence, medication side effects or intolerance, lack of response to a medication regimen, or the emergence of a macrolide-resistant or multidrug-resistant strain. Inducible macrolide resistance remains a potential factor.34-36 A number of characteristics of NTM contribute to the poor response to currently used antibiotics: the organisms have a lipid outer membrane and prefer to adhere to surfaces and form biofilms, which makes them relatively impermeable to antibiotics.37 Also, NTM replicate in phagocytic cells, allowing them to subvert normal cellular defense mechanisms. Furthermore, NTM can display colony variants, whereby single colony isolates switch between antibiotic-susceptible and -resistant variants. These factors have also impeded in development of new antibiotics for NTM infection.37
Recent limited approval of amikacin liposomal inhalation suspension (ALIS) for treatment failure and refractory MAC infection in combination with guideline-based antimicrobial therapy (GBT) is a promising addition to the available treatment armamentarium. In a multinational trial, the addition of ALIS to GBT for treatment-refractory MAC lung disease achieved significantly greater culture conversion rates by month 6 than GBT alone, with comparable rates of serious adverse events.38
Is therapeutic drug monitoring recommended during treatment of MAC pulmonary disease?
Treatment failure may also be drug-related, including poor drug penetration into the damaged lung tissue or drug-drug interactions leading to suboptimal drug levels. Peak serum concentrations have been found to be below target ranges in approximately 50% of patients using a macrolide and ethambutol. Concurrent use of rifampin decreases the peak serum concentration of macrolides and quinolones, with acceptable target levels seen in only 18% to 57% of cases. Whether this alters patient outcomes is not clear.39-42 Factors identified as contributing to the poor response to therapy include poor compliance, cavitary disease, previous treatment for MAC pulmonary disease, and a history of chronic obstructive lung disease. Studies by Koh and colleagues40 and van Ingen and colleagues41 with pharmacokinetic and pharmacodynamics data showed that, in patients on MAC treatment with both clarithromycin and rifampicin, plasma levels of clarithromycin were lower than the recommended minimal inhibitory concentrations (MIC) against MAC for that drug. The studies also showed that rifampicin lowered clarithromycin concentrations more than did rifabutin, with the AUC/MIC ratio being suboptimal in nearly half the cases. However, low plasma clarithromycin concentrations did not have any correlation with treatment outcomes, as the peak plasma drug concentrations and the peak plasma drug concentration/MIC ratios did not differ between patients with unfavorable treatment outcomes and those with favorable outcomes. This is further compounded by the fact that macrolides achieve higher levels in lung tissue than in plasma, and hence the significance of low plasma levels is unclear; however, it is postulated that achieving higher drug levels could, in fact, lead to better clinical outcomes. Pending specific well-designed, prospective randomized controlled trials, routine therapeutic drug monitoring is not currently recommended, although some referral centers do this as their practice pattern.
Is surgery an option in this case?
The overall 5-year mortality for MAC pulmonary disease was approximately 28% in a retrospective analysis, with patients with cavitary disease at increased risk for death at 5 years.42 As such, surgery is an option in selected cases as part of adjunctive therapy along with anti-MAC therapy based on mycobacterial sensitivity. Surgery is used as either a curative approach or a “debulking” measure.11 When present, clearly localized disease, especially in the upper lobe, lends itself best to surgical intervention. Surgical resection of a solitary pulmonary nodule due to MAC, in addition to concomitant medical treatment, is recommended. Surgical intervention should be considered early in the course of the disease because it may provide a cure without prolonged treatment and its associated problems, and this approach may lead to early sputum conversion. Surgery should also be considered in patients with macrolide-resistant or multidrug-resistant MAC infection or in those who cannot tolerate the side effects of therapy, provided that the disease is focal and limited. Patients with poor preoperative lung function have poorer outcomes than those with good lung function, and postoperative complications arising from treatment, especially with a right-sided pneumonectomy, tend to occur more frequently in these patients. Thoracic surgery for NTM pulmonary disease must be considered cautiously, as this is associated with significant morbidity and mortality and is best performed at specialized centers that have expertise and experience in this field.43
Continue to: Mycobacterium abscessus Complex
Mycobacterium abscessus Complex
Case Patient 2
A 64-year-old man who is an ex-smoker presents with chronic cough, mild shortness of breath on exertion, low-grade fever, and unintentional weight loss of 10 lb. Physical exam is unremarkable. The patient was diagnosed with immunoglobulin deficiency (low IgM and low IgG4) in 2002, and has been on replacement therapy since then. He also has had multiple episodes of NTM infection, with MAC and M. kansasii infections diagnosed in 2012-2014, which required 18 months of multi-drug antibiotic treatment that resulted in sputum conversion. Pulmonary function testing done on this visit in 2017 shows mild obstructive impairment.
Chest radiograph and CT scan show bilateral bronchiectasis (Figure 5 and Figure 6).
The results of serial sputum microbiology testing performed over the course of 6 months are outlined below:
- 5/2017 (bronchoalveolar lavage): 2+; M. abscessus
- 9/2017 × 2: smear (–); group IV RGM
- 11/2017: smear (–); M. abscessus (> 50 CFU)
- 12/2017: smear (–); M. abscessus (> 50 CFU)
What are the clinical considerations in this patient with multiple NTM infections?
M. abscessus complex was originally described in soft tissue abscesses and skin infections possibly resulting from soil or water contamination. Subspeciation of M. abscessus complex during laboratory testing is critical to facilitate selection of a specific therapeutic approach; treatment decisions are impacted by the presence of an active erm gene and in vitro macrolide sensitivity, which differ between subspecies. The most acceptable classification outlines 3 species in the M. abscessus complex: Mycobacterium abscessus subsp abscessus, Mycobacterium abscessus subsp bolletii (both with an active erm gene responsible for macrolide resistance), and Mycobacterium abscessus subsp massiliense (with an inactive erm gene and therefore susceptible to macrolides).44
RGM typically manifest in skin, soft tissue, and bone, and can cause soft tissue, surgical wound, and catheter-related infections. Although the role of RGM as pulmonary pathogens is unclear, underlying diseases associated with RGM include previously treated mycobacterial disease, coexistent pulmonary diseases with or without MAC, cystic fibrosis, malignancies, and gastroesophageal disorders. M. abscessus is the third most commonly identified respiratory NTM and accounts for the majority (80%) of RGM respiratory isolates. Other NTM reported to cause both lung disease and skin, bone, and joint infections include Mycobacterium simiae, Mycobacterium xenopi, and Mycobacterium malmoense. Ocular granulomatous diseases, such as chorioretinitis and keratitis, have been reported with both RGM and Runyon group III SGM, such as MAC or M. szulgai, following trauma or refractive surgery. These can mimic fungal, herpetic, or amebic keratitis. The pulmonary syndromes associated with multiple culture positivity are seen in elderly women with bronchiectasis or cavitary lung disease and/or associated with gastrointestinal symptoms of acid reflux, with or without achalasia and concomitant lipoid interstitial pneumonia.45
Generally, pulmonary disease progresses slowly, but lung disease attributed to RGM can result in respiratory failure. Thus, RGM should be recognized as a possible cause of chronic mycobacterial lung disease, especially in immunocompromised patients, and respiratory isolates should be assessed carefully. Identification and drug susceptibility testing are essential before initiation of treatment for RGM.
What is the approach to management of M. abscessus pulmonary disease in a patient without cystic fibrosis?
The management of M. abscessus pulmonary infection as a subset of RGM requires a considered step-wise approach. The criteria for diagnosis and threshold for starting treatment are the same as those used in the management of MAC pulmonary disease,11 but the treatment of M. abscessus pulmonary infection is more complex and has lower rates of success and cure. Also, antibiotic treatment presents challenges related to rapid identification of the causative organism, nomenclature, resistance patterns, and tolerance of treatment and side effects. If a source such as catheter, access port, or any surgical site is identified, prompt removal and clearance of the infected site are strongly advised
In the absence of any controlled clinical trials, treatment of RGM is based on in vitro susceptibility testing and expert opinion. As in MAC pulmonary disease, macrolides are the mainstay of treatment, with an induction phase of intravenous antibiotics. Treatment may include a combination of injectable aminoglycosides, imipenem, or cefoxitin and oral drugs such as a macrolide (eg, clarithromycin, azithromycin), doxycycline, fluoroquinolones, trimethoprim/sulfamethoxazole, or linezolid. While antibiotic treatment of M. abscessus pulmonary disease is based on in vitro sensitivity pattern to a greater degree than is treatment of MAC pulmonary disease, this approach has significant practical limitations and hence variable applicability. The final choice of antibiotics is best based on the extended susceptibility results, if available. The presence of an active erm gene on a prolonged growth specimen in M. abscessus subsp abscessus and M. abscessus subsp bolletii precludes the use of a macrolide. In such cases, amikacin, especially in an intravenous form, is the mainstay of treatment based on MIC. Recently, there has been a resurgence in interest in the use of clofazimine in combination with amikacin when treatment is not successful in patients with M. abscessus subsp abscessus or M. bolletii with an active erm gene.45,46 When localized abscess formation is noted, surgery may be the best option, with emphasis on removal of implants and catheters if implicated in RGM infection.
Attention must also be given to confounding pulmonary and associated comorbidities. This includes management of bronchiectasis with appropriately aggressive airway clearance techniques; anti-reflux measures for prevention of micro-aspiration; and management of other comorbid pulmonary conditions, such as chronic obstructive pulmonary disease, pulmonary fibrosis, and sarcoidosis, if applicable. These interventions play a critical role in clearing the M. abscessus infection, preventing progression of disease, and reducing morbidity. The role of immunomodulatory therapy needs to be considered on a regular, ongoing basis. Identification of genetic factors and correction of immune deficiencies may help in managing the infection.
Case Patient 2 Conclusion
The treatment regimen adopted in this case includes a 3-month course of daily intravenous amikacin and imipenem with oral azithromycin, followed by a continuation phase of azithromycin with clofazimine and linezolid. Airway clearance techniques such as Vest/Acapella/CPT are intensified and monthly intravenous immunoglobulin therapy is continued. The patient responds to treatment, with resolution of his clinical symptoms and reduction in the colony count of M. abscessus in the sputum.
Summary
NTM are ubiquitous in the environment, and NTM infection has variable manifestations, especially in patients with no recognizable immune impairments. Underlying comorbid conditions with bronchiectasis complicate its management. Treatment strategies must be individualized based on degree of involvement, associated comorbidities, immune deficiencies, goals of therapy, outcome-based risk-benefit ratio assessment, and patient engagement and expectations. In diffuse pulmonary disease, drug treatment remains difficult due to poor match of in vitro and in vivo culture sensitivity, side effects of medications, and high failure rates. When a localized resectable foci of infection is identified, especially in RGM disease, surgical treatment may be the best approach in selected patients, but it must be performed in centers with expertise and experience in this field.
Nontuberculous mycobacterial pulmonary disease is a broad term for a group of pulmonary disorders caused and characterized by exposure to environmental mycobacteria other than those belonging to the Mycobacterium tuberculosis complex and Mycobacterium leprae. Mycobacteria are aerobic, nonmotile organisms that appear positive with acid-fast alcohol stains. Nontuberculous mycobacteria (NTM) are ubiquitous in the environment and have been recovered from domestic and natural water sources, soil, and food products, and from around livestock, cattle, and wildlife.1-3 To date, no evidence exists of human-to-human or animal-to-human transmission of NTM in the general population. Infections in humans are usually acquired from environmental exposures, although the specific source of infection cannot always be identified. Similarly, the mode of infection with NTM has not been established with certainty, but it is highly likely that the organism is implanted, ingested, aspirated, or inhaled. Aerosolization of droplets associated with use of bathroom showerheads and municipal water sources and soil contamination are some of the factors associated with the transmission of infection. Proven routes of transmission include showerheads and potting soil dust.2,3
NTM pulmonary disease occurs in individuals with or without comorbid conditions such as bronchiectasis, chronic obstructive pulmonary disease, pulmonary fibrosis, or structural lung diseases. Slender, middle-aged or elderly white females with marfanoid body habitus, with or without apparent immune or genetic disorders, showing impaired airway and mucus clearance present with this infection as a form of underlying bronchiectasis (Lady Windermere syndrome). It is unclear why NTM infections and escalation to clinical disease occur in certain individuals. Many risk factors, including inherited and acquired defects of host immune response (eg, cystic fibrosis trait and α1 antitrypsin deficiency), have been associated with increased susceptibility to NTM infections.4
NTM infection can lead to chronic symptoms, frequent exacerbations, progressive functional and structural lung destruction, and impaired quality of life, and is associated with an increased risk of hospitalization and higher 5-year all-cause mortality. As such, NTM disease is drawing increasing attention at the clinical, academic, and research levels.5 This case-based review outlines the clinical features of NTM infection, with a focus on the challenges in diagnosis, treatment, and management of NTM pulmonary disease. The cases use Mycobacterium avium complex (MAC), a slow-growing mycobacteria (SGM), and Mycobacterium abscessus, a rapidly growing mycobacteria (RGM), as prototypes in a non–cystic fibrosis, non-HIV clinical setting.
Epidemiology
Of the almost 200 isolated species of NTM, the most prevalent pathogens for respiratory disease in the United States are MAC, Mycobacterium kansasii, and M. abscessus. MAC accounts for more than 80% of cases of NTM respiratory disease in the United States.6 The prevalence of NTM disease is increasing at a rate of about 8% each year, with 75,000 to 105,000 patients diagnosed with NTM lung disease in the United States annually. NTM infections in the United States are increasing among patients aged 65 years and older, a population that is expected to nearly double by 2030.7,8
Isolation and prevalence of many NTM species are higher in certain geographic areas of the United States, especially in the southeast. The US coastal regions have a higher prevalence of NTM pulmonary disease, and account for 70% of NTM cases in the United States each year. Half of patients diagnosed with NTM lung disease reside in 7 states: Florida, New York, Texas, California, Pennsylvania, New Jersey, and Ohio, with 1 in 7 residing in Florida. Three parishes in Louisiana are among the top 10 counties with the highest prevalence in United States. The prevalence of NTM infection–associated hospitalizations is increasing worldwide as well. Co-infection with tuberculosis and multiple NTMs in individual patients has been observed clinically and documented in patients with and without HIV.9,10
It is not clear why the prevalence of NTM pulmonary disease is increasing, but there may be several contributing factors: (1) an increased awareness and identification of NTM infection sources in the environment; (2) an expanding cohort of immunocompromised individuals with exogenous or endogenous immune deficiencies; (3) availability of improved diagnostic techniques, such as use of high-performance liquid chromatography (HPLC), DNA probes, and gene sequencing; and (4) an increased awareness of the morbidity and mortality associated with NTM pulmonary disease. However, it is important to recognize that to best understand the clinical relevance of epidemiologic studies based on laboratory diagnosis and identification, the findings must be evaluated by correlating them with the microbiological and other clinical criteria established by the American Thoracic Society (ATS)/Infectious Diseases Society of America (IDSA) guidelines.11
Continue to: Mycobacterium avium Complex
Mycobacterium avium Complex
Case Patient 1
A 48-year-old woman who has never smoked and has no past medical problems, except seasonal allergic rhinitis and “colds and flu-like illness” once or twice a year, is evaluated for a chronic lingering cough with occasional sputum production. The patient denies any other chronic symptoms and is otherwise active. Physical examination reveals no specific findings except mild pectus excavatum and mild scoliosis. Body mass index is 22 kg/m2. Chest radiograph shows nonspecific increased markings in the lower zones. Computed tomography (CT) scan of the chest reveals minimal nodular and cylindrical bronchiectasis in both lungs (Figure 1). No previous radiographs are available for comparison. The patient is HIV-negative. Sputum tests reveal normal flora, and both fungus and acid-fast bacilli smear are negative. Culture for mycobacteria shows scanty growth of MAC in 1 specimen.
What is the clinical presentation of MAC pulmonary disease?
Among NTM, MAC is the most common cause of pulmonary disease worldwide.6 MAC primarily includes 2 species: M. avium and Mycobacterium intracellulare. M. avium is the more important pathogen in disseminated disease, whereas M. intracellulare is the more common respiratory pathogen.11 These organisms are genetically similar and generally not differentiated in the clinical microbiology laboratory, although there are isolated reports in the literature suggesting differences in prevalence, presentation, and prognosis in M. avium infection versus M. intracellulare infection.12
Three major disease syndromes are produced by MAC in humans: pulmonary disease, usually in adults whose systemic immunity is intact; disseminated disease, usually in patients with advanced HIV infection; and cervical lymphadenitis.13 Pulmonary disease caused by MAC may take on 1 of several clinically different forms, including asymptomatic “colonization” or persistent minimal infection without obvious clinical significance; endobronchial involvement; progressive pulmonary disease with radiographic and clinical deterioration and nodular bronchiectasis or cavitary lung disease; hypersensitivity pneumonitis; or persistent, overwhelming mycobacterial growth with symptomatic manifestations, often in a lung with underlying damage due to either chronic obstructive lung disease or pulmonary fibrosis (Table 1).14
Cavitary Disease
The traditionally recognized presentation of MAC lung disease has been apical cavitary lung disease in men in their late 40s and early 50s who have a history of cigarette smoking, and frequently, excessive alcohol use. If left untreated, or in the case of erratic treatment or macrolide drug resistance, this form of disease is generally progressive within a relatively short time and can result in extensive cavitary lung destruction and progressive respiratory failure.15
Nodular Bronchiectasis
The more common presentation of MAC lung disease, which is outlined in the case described here, is interstitial nodular infiltrates, frequently involving the right middle lobe or lingula and predominantly occurring in postmenopausal, nonsmoking white women. This is sometimes labelled “Lady Windermere syndrome.” These patients with M. avium infection appear to have similar clinical characteristics and body types, including lean build, scoliosis, pectus excavatum, and mitral valve prolapse.16,17 The mechanism by which this body morphotype predisposes to pulmonary mycobacterial infection is not defined, but ineffective mucociliary clearance is a possible explanation. Evidence suggests that some patients may be predisposed to NTM lung disease because of preexisting bronchiectasis. Some potential etiologies of bronchiectasis in this population include chronic sinusitis, gastroesophageal reflux with chronic aspiration, α1 antitrypsin deficiency, and cystic fibrosis genetic traits and mutations.18 Risk factors for increased morbidity and mortality include the development of cavitary disease, age, weight loss, lower body mass index, and other comorbid conditions.
This form of disease, termed nodular bronchiectasis, tends to have a much slower progression than cavitary disease, such that long-term follow-up (months to years) may be necessary to demonstrate clinical or radiographic changes.11 The radiographic term “tree-in-bud” has been used to describe what may reflect inflammatory changes, including bronchiolitis. High-resolution CT scans of the chest are especially helpful for diagnosing this pattern of MAC lung disease, as bronchiectasis and small nodules may not be easily discernible on plain chest radiograph. The nodular/bronchiectasis radiographic pattern can also be seen with other NTM pathogens, including M. abscessus, Mycobacterium simiae, and M. kansasii. Solitary nodules and dense consolidation have also been described. Pleural effusions are uncommon, but reactive pleural thickening is frequently seen. Co-pathogens may be isolated from culture, including Pseudomonas aeruginosa, Staphylococcus aureus, and, occasionally, other NTM such as M. abscessus or Mycobacterium chelonae.19-21
Hypersensitivity Pneumonitis
Hypersensitivity pneumonitis, initially described in patients who were exposed to hot tubs, mimics allergic hypersensitivity pneumonitis, with respiratory symptoms and culture/tissue identification of MAC or sometimes other NTM. It is unclear whether hypersensitivity pneumonitis is an inflammatory process, an infection, or both, and opinion regarding the need for specific antibiotic treatment is divided.11,22 However, avoidance of exposure is prudent and recommended.
Disseminated Disease
Disseminated NTM disease is associated with very low CD4+ lymphocyte counts and is seen in approximately 5% of patients with HIV infection.23-25 Although disseminated NTM disease is rarely seen in immunosuppressed patients without HIV infection, it has been reported in patients who have undergone renal or cardiac transplant, patients on long-term corticosteroid therapy, and those with leukemia or lymphoma. More than 90% of infections are caused by MAC; other potential pathogens include M. kansasii, M. chelonae, M. abscessus, and Mycobacterium haemophilum. Although seen less frequently since the advent of highly active antiretroviral therapy, disseminated infection can develop progressively from an apparently indolent or localized infection or a respiratory or gastrointestinal source. Signs and symptoms of disseminated infection (specifically MAC-associated disease) are nonspecific and include fever, night sweats, weight loss, and abdominal tenderness. Disseminated MAC disease occurs primarily in patients with more advanced HIV disease (CD4+ count typically < 50 cells/μL). Clinically, disseminated MAC manifests as intermittent or persistent fever, constitutional symptoms with organomegaly and organ-specific abnormalities (eg, anemia, neutropenia from bone marrow involvement, adenopathy, hepatosplenomegaly), and elevations of liver enzymes or lung infiltrates from pulmonary involvement.
Continue to: What are the criteria for diagnosing NTM pulmonary disease?
What are the criteria for diagnosing NTM pulmonary disease?
The diagnosis of NTM disease is based on clinical, radiologic, and mycobacterial correlation with good communication between the experts in this field. The ATS/IDSA criteria for diagnosing NTM lung disease are shown in Figure 2. These criteria best apply to MAC, M. kansasii, and M. abscessus, but are also clinically applied to other NTM respiratory pathogens. The diagnosis of MAC infection is most readily established by culture of blood, bone marrow, respiratory secretions/fluid, or tissue specimens from suspected sites of involvement. Due to erratic shedding of MAC into the respiratory secretions in patients with nodular bronchiectasis, as compared to those with the cavitary form of the disease, sputum may be intermittently positive, with variable colony counts and polyclonal infections.12 Prior to the advent of high-resolution CT, isolation of MAC organisms from the sputum of such patients was frequently dismissed as colonization.
Mycobacterial Testing
Because of the nonspecific symptoms and lack of diagnostic specificity of chest imaging, the diagnosis of NTM lung disease requires microbiologic confirmation. Specimens sent to the laboratory for identification of NTM must be handled with care to prevent contamination and false-positive results. Transport media and preservatives should be avoided, and transportation of the specimens should be prompt. These measures will prevent bacterial overgrowth. Furthermore, the yield of NTM may be affected if the patient has used antibiotics, such as macrolides and fluoroquinolones, prior to obtaining the specimen.
NTM should be identified at the species and subspecies level, although this is not practical in community practice settings. The preferred staining procedure in the laboratory is the fluorochrome method. Some species require special growth conditions and/or lower incubation temperatures, and other identification methods may have to be employed, such as DNA probes, polymerase chain reaction genotyping, nucleic acid sequence determination, and high-performance liquid chromatography. As a gold standard, clinical specimens for mycobacterial cultures should be inoculated onto 1 or more solid media (eg, Middlebrook 7H11 media and/or Lowenstein-Jensen media, the former of which is the preferred medium for NTM) and into a liquid medium (eg, BACTEC 12B broth or Mycobacteria growth indicator tube broth). Growth of visible colonies on solid media typically requires 2 to 4 weeks, but liquid media (eg, the radiometric BACTEC system), used as a supplementary and not as an exclusive test, usually produce results within 10 to 14 days. Furthermore, even after initial growth, identification of specific isolates based on the growth characteristics on solid media requires additional time. Use of specific nucleic acid probes for MAC and M. kansasii and HPLC testing of mycolic acid patterns in acid-fast bacilli smear–positive specimens can reduce the turnaround time of specific identification of a primary culture–positive sample. However, HPLC is not sufficient for definitive identification of many NTM species, including the RGM. Other newer techniques, including 16S ribosomal DNA sequencing and polymerase chain reaction-restriction fragment length polymorphism analysis, also allow NTM to be identified and speciated more reliably and rapidly from clinical specimens.
Cost and other practical considerations limit widespread adoption of these techniques. However, the recognition that M. abscessus can be separated into more than 1 subspecies, and that there are important prognostic implications of that separation, lends urgency to the broader adoption of newer molecular techniques in the mycobacteriology laboratory. Susceptibility testing is based on the broth microdilution method; RGM usually grow within 7 days of subculture, and the laboratory time to culture is a helpful hint, although not necessarily specific. Recognizing the morphology of mycobacterial colony growth may also be helpful in identification.
Are skin tests helpful in diagnosing NTM infection?
Tuberculin skin testing remains a nonspecific marker of mycobacterial infection and does not help in further elucidating NTM infection. However, epidemiologic and laboratory studies with well-characterized antigens have shown that dual skin testing with tuberculosis- versus NTM-derived tuberculin can discriminate between prior NTM and prior tuberculosis disease. Species-specific skin test antigens are not commercially available and are not helpful in the diagnosis of NTM disease because of cross-reactivity of M. tuberculosis and some NTM. However, increased prevalence of NTM sensitization based on purified protein derivative testing has been noted in a recent survey, which is consistent with an observed increase in the rates of NTM infections, specifically MAC, in the United States.26,27
Interferon-gamma release assays (IGRAs) are now being used as an alternative to tuberculin skin testing to diagnose M. tuberculosis infection. Certain NTM species also contain gene sequences that encode for ESAT-6 or CFP-10 antigens used in the IGRAs, and hence, yield a positive IGRA test. These include M. marinum, M. szulgai, and M. kansasii.28,29 However, MAC organisms do not produce positive results on assays that use these antigens.
Continue to: What is the approach to management of NTM pulmonary disease?
What is the approach to management of NTM pulmonary disease?
The correlation of symptoms with radiographic and microbiologic evidence is essential to categorize the disease and determine the need for therapy. Making the diagnosis of NTM lung disease does not necessitate the institution of therapy. The decision to treat should be weighed against potential risks and benefits to the individual patient based on symptomatic, radiographic, and microbiologic criteria, as well as underlying systemic or pulmonary immune status. In the absence of evidence of clinical, radiologic, or mycobacterial progression of disease, pursuing airway clearance therapy and clinical surveillance without initiating specific anti-MAC therapy is a reasonable option.11 Identifying the sustained presence of NTM infection, especially MAC, in a patient with underlying clinical and radiographic evidence of bronchiectasis is of value in determining comprehensive treatment and management strategies. Close observation is indicated if the decision not to treat is made. If treatment is initiated, comprehensive management includes long-term follow-up with periodic bacteriologic surveillance, watching for drug toxicity and drug-drug interactions, ensuring adherence and compliance to treatment, and managing comorbidity.
The Bronchiectasis Severity Index is a useful clinical predictive tool that identifies patients at risk of future mortality, hospitalization, and exacerbations and can be used to evaluate the need for specific treatment.30 The index is based on dyspnea score, lung function tests, colonization of pathogens, and extent of disease.
Case 1 Continued
After approximately 2 months of observation and symptomatic treatment, without specific antibiotic therapy, the patient’s symptoms continue. She now develops intermittent hemoptysis. Repeat sputum studies reveal moderate growth of M. avium. A follow-up CT scan shows progression of disease, with an increase in the tree-in-bud pattern (Figure 3).
What treatment protocols are recommended for MAC pulmonary disease?
As per the ATS/IDSA statement, macrolides are the mainstay of treatment for pulmonary MAC disease.11 Macrolides achieve an increased concentration in the lung, and when used for treatment of pulmonary MAC disease, there is a strong correlation between in vitro susceptibility, in vivo (clinical) response, and the immunomodulating effects of macrolides.31,32 Macrolide-containing regimens have demonstrated efficacy in patients with MAC pulmonary disease33,34; however, macrolide monotherapy should be avoided to prevent the development of resistance.
At the outset, it is critical to establish the objective criteria for determining response and to ensure that the patient understands the goals of the treatment and expectations of the treatment plan. Moreover, experts suggest that due to the possibility of drug intolerance, side effects, and the need for prolonged therapy, a “step ladder” ramping up approach to treatment could be adopted, with gradual introduction of therapy within a short time period; this approach may improve compliance and adherence to treatment.11 If this approach is used, the doses may have to be divided. Patients who are unable to tolerate daily medications, even with dosage adjustment, should be tried on an intermittent treatment regimen. Older female patients frequently require gradual introduction of medications (ie, 1 medication added to the regimen every 1 to 2 weeks) to evaluate tolerance to each medication and medication dose.11 Commonly encountered adverse effects of NTM treatment include intolerance to clarithromycin due to gastrointestinal problems, low body mass index, or age older than 70 years.
After determining that the patient requires therapy, the standard recommended treatment for MAC pulmonary disease includes the following: for most patients with nodular/bronchiectasis disease, a thrice-weekly regimen of clarithromycin (1000 mg) or azithromycin (500 mg), rifampin (600 mg), and ethambutol (25 mg/kg) is recommended. For patients with cavitary MAC pulmonary disease or severe nodular/bronchiectasis disease, the guidelines recommend a daily regimen of clarithromycin (500-1000 mg) or azithromycin (250 mg), rifampin (600 mg) or rifabutin (150–300 mg), and ethambutol (15 mg/kg), with consideration of intravenous (IV) amikacin 3 times/week early in therapy (Table 2).11
The treatment of MAC hypersensitivity-like disease speaks to the controversy of whether this is an inflammatory process, infectious process, or a combination of inflammation and infection. Avoidance of exposure is the mainstay of management. In some cases, steroids are used with or without a short course of anti-MAC therapy (ie, clarithromycin or azithromycin with rifampin and ethambutol).
Prophylaxis for disseminated MAC disease should be given to adults with HIV infection who have a CD4+ count less than 50 cells/μL. Azithromycin 1200 mg/week or clarithromycin 1000 mg/day has proven efficacy, and rifabutin 300 mg/day is also effective but less well tolerated. Rifabutin is more active in vitro against MAC than rifampin, and is used in HIV-positive patients because of drug-drug interaction between antiretroviral drugs and rifampin.
Continue to: Case 1 Continued
Case 1 Continued
The patient is treated with clarithromycin, rifampin, and ethambutol for 1 year, with sputum conversion after 9 months. In the latter part of her treatment, she experiences decreased visual acuity. Treatment is discontinued prematurely after 1 year due to drug toxicity and continued intolerance to drug therapy. The patient remains asymptomatic for 8 months, and then begins to experience mild to moderate hemoptysis, with increasing cough and sputum production associated with postural changes during exercise. Physical examination overall remains unchanged. Three sputum results reveal heavy growth of MAC, and a CT scan of the chest shows a cavitary lesion in the left upper lobe along with the nodular bronchiectasis (Figure 4).
What are the management options at this stage?
Based on this patient’s continued symptoms, progression of radiologic abnormalities, and current culture growth, she requires re-treatment. With the adverse effects associated with ethambutol during the first round of therapy, the drug regimen needs to be modified. Several considerations are relevant at this stage. Relapse rates range from 20% to 30% after treatment with a macrolide-based therapy.11,34 Obtaining a culture-sensitivity profile is imperative in these cases. Of note, treatment should not be discontinued altogether, but instead the toxic agent should be removed from the treatment regimen. Continuing treatment with a 2-drug regimen of clarithromycin and rifampin may be considered in this patient. Re-infection with multiple genotypes may also occur after successful drug therapy, but this is primarily seen in MAC patients with nodular bronchiectasis.34,35 Patients in whom previous therapy has failed, even those with macrolide-susceptible MAC isolates, are less likely to respond to subsequent therapy. Data suggest that intermittent medication dosing is not effective for patients with severe or cavitary disease or in those in whom previous therapy has failed.36 In this case, treatment should include a daily 3-drug therapy, with an injectable thrice-weekly aminoglycoside. Other agents such as linezolid and clofazimine may have to be tried. Cycloserine, ethionamide, and other agents are sometimes used, but their efficacy is unproven and doubtful. Pyrazinamide and isoniazid have no activity against MAC.
Treatment Failure and Drug Resistance
Treatment failure is considered to have occurred if patients have not had a response (microbiologic, clinical, or radiographic) after 6 months of appropriate therapy or had not achieved conversion of sputum to culture-negative after 12 months of appropriate therapy.11 This occurs in about 40% of patients. Multiple factors can interfere with the successful treatment of MAC pulmonary disease, including medication nonadherence, medication side effects or intolerance, lack of response to a medication regimen, or the emergence of a macrolide-resistant or multidrug-resistant strain. Inducible macrolide resistance remains a potential factor.34-36 A number of characteristics of NTM contribute to the poor response to currently used antibiotics: the organisms have a lipid outer membrane and prefer to adhere to surfaces and form biofilms, which makes them relatively impermeable to antibiotics.37 Also, NTM replicate in phagocytic cells, allowing them to subvert normal cellular defense mechanisms. Furthermore, NTM can display colony variants, whereby single colony isolates switch between antibiotic-susceptible and -resistant variants. These factors have also impeded in development of new antibiotics for NTM infection.37
Recent limited approval of amikacin liposomal inhalation suspension (ALIS) for treatment failure and refractory MAC infection in combination with guideline-based antimicrobial therapy (GBT) is a promising addition to the available treatment armamentarium. In a multinational trial, the addition of ALIS to GBT for treatment-refractory MAC lung disease achieved significantly greater culture conversion rates by month 6 than GBT alone, with comparable rates of serious adverse events.38
Is therapeutic drug monitoring recommended during treatment of MAC pulmonary disease?
Treatment failure may also be drug-related, including poor drug penetration into the damaged lung tissue or drug-drug interactions leading to suboptimal drug levels. Peak serum concentrations have been found to be below target ranges in approximately 50% of patients using a macrolide and ethambutol. Concurrent use of rifampin decreases the peak serum concentration of macrolides and quinolones, with acceptable target levels seen in only 18% to 57% of cases. Whether this alters patient outcomes is not clear.39-42 Factors identified as contributing to the poor response to therapy include poor compliance, cavitary disease, previous treatment for MAC pulmonary disease, and a history of chronic obstructive lung disease. Studies by Koh and colleagues40 and van Ingen and colleagues41 with pharmacokinetic and pharmacodynamics data showed that, in patients on MAC treatment with both clarithromycin and rifampicin, plasma levels of clarithromycin were lower than the recommended minimal inhibitory concentrations (MIC) against MAC for that drug. The studies also showed that rifampicin lowered clarithromycin concentrations more than did rifabutin, with the AUC/MIC ratio being suboptimal in nearly half the cases. However, low plasma clarithromycin concentrations did not have any correlation with treatment outcomes, as the peak plasma drug concentrations and the peak plasma drug concentration/MIC ratios did not differ between patients with unfavorable treatment outcomes and those with favorable outcomes. This is further compounded by the fact that macrolides achieve higher levels in lung tissue than in plasma, and hence the significance of low plasma levels is unclear; however, it is postulated that achieving higher drug levels could, in fact, lead to better clinical outcomes. Pending specific well-designed, prospective randomized controlled trials, routine therapeutic drug monitoring is not currently recommended, although some referral centers do this as their practice pattern.
Is surgery an option in this case?
The overall 5-year mortality for MAC pulmonary disease was approximately 28% in a retrospective analysis, with patients with cavitary disease at increased risk for death at 5 years.42 As such, surgery is an option in selected cases as part of adjunctive therapy along with anti-MAC therapy based on mycobacterial sensitivity. Surgery is used as either a curative approach or a “debulking” measure.11 When present, clearly localized disease, especially in the upper lobe, lends itself best to surgical intervention. Surgical resection of a solitary pulmonary nodule due to MAC, in addition to concomitant medical treatment, is recommended. Surgical intervention should be considered early in the course of the disease because it may provide a cure without prolonged treatment and its associated problems, and this approach may lead to early sputum conversion. Surgery should also be considered in patients with macrolide-resistant or multidrug-resistant MAC infection or in those who cannot tolerate the side effects of therapy, provided that the disease is focal and limited. Patients with poor preoperative lung function have poorer outcomes than those with good lung function, and postoperative complications arising from treatment, especially with a right-sided pneumonectomy, tend to occur more frequently in these patients. Thoracic surgery for NTM pulmonary disease must be considered cautiously, as this is associated with significant morbidity and mortality and is best performed at specialized centers that have expertise and experience in this field.43
Continue to: Mycobacterium abscessus Complex
Mycobacterium abscessus Complex
Case Patient 2
A 64-year-old man who is an ex-smoker presents with chronic cough, mild shortness of breath on exertion, low-grade fever, and unintentional weight loss of 10 lb. Physical exam is unremarkable. The patient was diagnosed with immunoglobulin deficiency (low IgM and low IgG4) in 2002, and has been on replacement therapy since then. He also has had multiple episodes of NTM infection, with MAC and M. kansasii infections diagnosed in 2012-2014, which required 18 months of multi-drug antibiotic treatment that resulted in sputum conversion. Pulmonary function testing done on this visit in 2017 shows mild obstructive impairment.
Chest radiograph and CT scan show bilateral bronchiectasis (Figure 5 and Figure 6).
The results of serial sputum microbiology testing performed over the course of 6 months are outlined below:
- 5/2017 (bronchoalveolar lavage): 2+; M. abscessus
- 9/2017 × 2: smear (–); group IV RGM
- 11/2017: smear (–); M. abscessus (> 50 CFU)
- 12/2017: smear (–); M. abscessus (> 50 CFU)
What are the clinical considerations in this patient with multiple NTM infections?
M. abscessus complex was originally described in soft tissue abscesses and skin infections possibly resulting from soil or water contamination. Subspeciation of M. abscessus complex during laboratory testing is critical to facilitate selection of a specific therapeutic approach; treatment decisions are impacted by the presence of an active erm gene and in vitro macrolide sensitivity, which differ between subspecies. The most acceptable classification outlines 3 species in the M. abscessus complex: Mycobacterium abscessus subsp abscessus, Mycobacterium abscessus subsp bolletii (both with an active erm gene responsible for macrolide resistance), and Mycobacterium abscessus subsp massiliense (with an inactive erm gene and therefore susceptible to macrolides).44
RGM typically manifest in skin, soft tissue, and bone, and can cause soft tissue, surgical wound, and catheter-related infections. Although the role of RGM as pulmonary pathogens is unclear, underlying diseases associated with RGM include previously treated mycobacterial disease, coexistent pulmonary diseases with or without MAC, cystic fibrosis, malignancies, and gastroesophageal disorders. M. abscessus is the third most commonly identified respiratory NTM and accounts for the majority (80%) of RGM respiratory isolates. Other NTM reported to cause both lung disease and skin, bone, and joint infections include Mycobacterium simiae, Mycobacterium xenopi, and Mycobacterium malmoense. Ocular granulomatous diseases, such as chorioretinitis and keratitis, have been reported with both RGM and Runyon group III SGM, such as MAC or M. szulgai, following trauma or refractive surgery. These can mimic fungal, herpetic, or amebic keratitis. The pulmonary syndromes associated with multiple culture positivity are seen in elderly women with bronchiectasis or cavitary lung disease and/or associated with gastrointestinal symptoms of acid reflux, with or without achalasia and concomitant lipoid interstitial pneumonia.45
Generally, pulmonary disease progresses slowly, but lung disease attributed to RGM can result in respiratory failure. Thus, RGM should be recognized as a possible cause of chronic mycobacterial lung disease, especially in immunocompromised patients, and respiratory isolates should be assessed carefully. Identification and drug susceptibility testing are essential before initiation of treatment for RGM.
What is the approach to management of M. abscessus pulmonary disease in a patient without cystic fibrosis?
The management of M. abscessus pulmonary infection as a subset of RGM requires a considered step-wise approach. The criteria for diagnosis and threshold for starting treatment are the same as those used in the management of MAC pulmonary disease,11 but the treatment of M. abscessus pulmonary infection is more complex and has lower rates of success and cure. Also, antibiotic treatment presents challenges related to rapid identification of the causative organism, nomenclature, resistance patterns, and tolerance of treatment and side effects. If a source such as catheter, access port, or any surgical site is identified, prompt removal and clearance of the infected site are strongly advised
In the absence of any controlled clinical trials, treatment of RGM is based on in vitro susceptibility testing and expert opinion. As in MAC pulmonary disease, macrolides are the mainstay of treatment, with an induction phase of intravenous antibiotics. Treatment may include a combination of injectable aminoglycosides, imipenem, or cefoxitin and oral drugs such as a macrolide (eg, clarithromycin, azithromycin), doxycycline, fluoroquinolones, trimethoprim/sulfamethoxazole, or linezolid. While antibiotic treatment of M. abscessus pulmonary disease is based on in vitro sensitivity pattern to a greater degree than is treatment of MAC pulmonary disease, this approach has significant practical limitations and hence variable applicability. The final choice of antibiotics is best based on the extended susceptibility results, if available. The presence of an active erm gene on a prolonged growth specimen in M. abscessus subsp abscessus and M. abscessus subsp bolletii precludes the use of a macrolide. In such cases, amikacin, especially in an intravenous form, is the mainstay of treatment based on MIC. Recently, there has been a resurgence in interest in the use of clofazimine in combination with amikacin when treatment is not successful in patients with M. abscessus subsp abscessus or M. bolletii with an active erm gene.45,46 When localized abscess formation is noted, surgery may be the best option, with emphasis on removal of implants and catheters if implicated in RGM infection.
Attention must also be given to confounding pulmonary and associated comorbidities. This includes management of bronchiectasis with appropriately aggressive airway clearance techniques; anti-reflux measures for prevention of micro-aspiration; and management of other comorbid pulmonary conditions, such as chronic obstructive pulmonary disease, pulmonary fibrosis, and sarcoidosis, if applicable. These interventions play a critical role in clearing the M. abscessus infection, preventing progression of disease, and reducing morbidity. The role of immunomodulatory therapy needs to be considered on a regular, ongoing basis. Identification of genetic factors and correction of immune deficiencies may help in managing the infection.
Case Patient 2 Conclusion
The treatment regimen adopted in this case includes a 3-month course of daily intravenous amikacin and imipenem with oral azithromycin, followed by a continuation phase of azithromycin with clofazimine and linezolid. Airway clearance techniques such as Vest/Acapella/CPT are intensified and monthly intravenous immunoglobulin therapy is continued. The patient responds to treatment, with resolution of his clinical symptoms and reduction in the colony count of M. abscessus in the sputum.
Summary
NTM are ubiquitous in the environment, and NTM infection has variable manifestations, especially in patients with no recognizable immune impairments. Underlying comorbid conditions with bronchiectasis complicate its management. Treatment strategies must be individualized based on degree of involvement, associated comorbidities, immune deficiencies, goals of therapy, outcome-based risk-benefit ratio assessment, and patient engagement and expectations. In diffuse pulmonary disease, drug treatment remains difficult due to poor match of in vitro and in vivo culture sensitivity, side effects of medications, and high failure rates. When a localized resectable foci of infection is identified, especially in RGM disease, surgical treatment may be the best approach in selected patients, but it must be performed in centers with expertise and experience in this field.
1. Johnson MM, Odell JA. Nontuberculous mycobacterial pulmonary infections. J Thorac Dis. 2014;6:210-220.
2. Falkinham JO III. Environmental sources of NTM. Clin Chest Med. 2015;36:35-41.
3. Falkinham JO III, Current epidemiological trends in NTM. Curr Environ Health Rep. 2016;3:161-167.
4. Honda JR, Knight V, Chan ED. Pathogenesis and risk factors for nontuberculous mycobacterial lung disease. Clin Chest Med. 2015;36:1-11.
5. Marras TK, Mirsaeidi M, Chou E, et al. Health care utilization and expenditures following diagnosis of nontuberculous mycobacterial lung disease in the United States. Manag Care Spec Pharm. 2018;24:964-974.
6. Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated healthcare delivery systems. Am J Respir Crit Care Med. 2010;182:970-976.
7. Winthrop KL, McNelley E, Kendall B, et al. Pulmonary nontuberculous mycobacterial disease prevalence and clinical features: an emerging public health disease. Am J Respir Crit Care Med. 2010;182:977-982.
8. Adjemian, Olivier KN, Seitz AE, J et al. Prevalence of nontuberculous mycobacterial lung disease in US Medicare beneficiaries. Am J Respir Crit Care Med. 2012;185;881-886.
9. Ringshausen FC, Apel RM, Bange FC, et al. Burden and trends of hospitalizations associated with pulmonary nontuberculous mycobacterial infections in Germany, 2005-2011. BMC Infect Dis. 2013;13:231.
10. Aliyu G, El-Kamary SS, Abimiku A, et al. Prevalence of non-tuberculous mycobacterial infections among tuberculosis suspects in Nigeria. PLoS One. 2013;8:e63170.
11. Griffith DE, Aksamit T, Brown-Elliott, et al; American Thoracic Society; Infectious Diseases Society of America. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367-415.
12. Wallace RJ Jr, Zhang Y, Brown BA, et al. Polyclonal Mycobacterium avium complex infections in patients with nodular bronchiectasis. Am J Respir Crit Care Med. 1998;158:1235-1244.
13. Gordin FM, Horsburgh CR Jr. Mycobacterium avium complex. In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Elsevier; 2015.
14. Chitty S, Ali J. Mycobacterium avium complex pulmonary disease in immune competent patients. South Med J. 2005;98:646-52.
15. Ramirez J, Mason C, Ali J, Lopez FA. MAC pulmonary disease: management options in HIV-negative patients. J La State Med Soc. 2008;160:248-254.
16. Iseman MD, Buschman DL, Ackerson LM. Pectus excavatum and scoliosis. Thoracic anomalies associated with pulmonary disease caused by Mycobacterium avium complex. Am Rev Respir Dis. 1991;144:914-916.
17. Kim RD, Greenburg DE, Ehrmantraut ME, et al. Pulmonary nontuberculous mycobacterial disease: prospective study of a distinct preexisting syndrome. Am J Respir Crit Care Med. 2008;178:1066-1074.
18. Ziedalski TM, Kao PN, Henig NR, et al. Prospective analysis of cystic fibrosis transmembrane regulator mutations in adults with bronchiectasis or pulmonary nontuberculous mycobacterial infection. Chest. 2006;130:995-1002.
19. Koh WJ, Lee KS, Kwon OJ, et al. Bilateral bronchiectasis and bronchiolitis at thin-section CT: diagnostic implications in nontuberculous mycobacterial pulmonary infection. Radiology. 2005;235:282-288.
20. Swensen SJ, Hartman TE, Williams DE. Computed tomographic diagnosis of Mycobacterium avium-intracellulare complex in patients with bronchiectasis. Chest. 1994;105:49-52.
21. Huang JH, Kao PN, Adi V, Ruoss SJ. Mycobacterium avium intracellulare pulmonary infection in HIV-negative patients without preexisting lung disease: diagnostic and management limitations. Chest. 1999;115:1033-1040.
22. Cappelluti E, Fraire AE, Schaefer OP. A case of “hot tub lung” due to Mycobacterium avium complex in an immunocompetent host. Arch Intern Med. 2003;163:845-848.
23. Nightingale SD, Byrd LT, Southern PM, et al. Incidence of Mycobacterium avium-intracellulare complex bacteremia in human immunodeficiency virus-positive patients. J Infect Dis. 1992;165:1082-1085.
24. Horsburgh CR Jr, Selik RM. The epidemiology of disseminated tuberculous mycobacterial infection in the acquired immunodeficiency syndrome (AIDS). Am Rev Respir Dis. 1989;139:4-7.
25. Chin DP, Hopewell PC, Yajko DM, et al. Mycobacterium avium complex in the respiratory or gastrointestinal tract and the risk of M. avium complex bacteremia in patients with human immunodeficiency virus infection. J Infect Dis. 1994;169:289-295.
26. Khan K, Wang J, Marras TK. Nontuberculous mycobacterial sensitization in the United States: national trends over three decades. Am J Respir Crit Care Med. 2007;176:306-313.
27. Lillo M, Orengo S, Cernoch P, Harris RL. Pulmonary and disseminated infection due to Mycobacterium kansasii: a decade of experience. Rev Infect Dis. 1990;12:760-767.
28. Andersen P, Munk ME, Pollock JM, Doherty TM. Specific immune-based diagnosis of tuberculosis. Lancet. 2000;356:1099-1104.
29. Arend SM, van Meijgaarden KE, de Boer K, et al. Tuberculin skin testing and in vitro T cell responses to ESAT-6 and culture filtrate protein 10 after infection with Mycobacterium marinum or M. kansasii. J Infect Dis. 2002;186:1797-1807.
30. James D, Chalmers JD, Goeminne P, et al. The Bronchiectasis Severity Index: an international derivation and validation study. Am J Respir Crit Care Med. 2014;189:576-585.
31. Heifets L. MIC as a quantitative measurement of the susceptibility of Mycobacterium avium strains to seven antituberculosis drugs. Antimicrob Agents Chemother. 1988;32:1131-1136.
32. Horsburgh CR Jr, Mason UG 3rd, Heifits LB, et al. Response to therapy of pulmonary Mycobacterium avium intracellulare infection correlates with results of in vitro susceptibility testing. Am Rev Respir Dis. 1987;135:418-421.
33. Rubin BK, Henke MO. Immunomodulatory activity and effectiveness of macrolides in chronic airway disease. Chest. 2004;125(2 Suppl):70S-78S.
34. Wallace RJ Jr, Brown BA, Griffith DE, et al. Clarithromycin regimens for pulmonary Mycobacterium avium complex. The first 50 patients. Am J Respir Crit Care Med. 1996;153:1766-1772.
35. Griffith DE, Brown-Elliott BA, Langsjoen B, et al. Clinical and molecular analysis of macrolide resistance in Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006;174:928-934.
36. Lam PK, Griffith DE, Aksamit TR, et al. Factors related to response to intermittent treatment of Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006;173:1283-1289.
37. Falkinham J III. Challenges of NTM drug development. Front Microbiol. 2018;9:1613.
38. Griffith DE, Eagle G, Thomson R, et al. Amikacin liposome inhalation suspension for treatment-refractory lung disease caused by Mycobacterium avium complex (CONVERT). A prospective, open-label, randomized study. Am J Respir Crit Care Med. 2018;198:1559-1569.
39. Schluger NW. Treatment of pulmonary Mycobacterium avium complex infections: do drug levels matter? Am J Respir Crit Care Med. 2012;186:710-711.
40. Van Ingen J, Egelund EF, Levin A, et al. The pharmacokinetics and pharmacodynamics of pulmonary Mycobacterium avium complex disease treatment. Am J Respir Crit Care Med. 2012;186:559-565.
41. Koh WJ, Jeong BH, Jeon K, et al. Therapeutic drug monitoring in the treatment of Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2012;186:797-802.
42. Ito Y, Hirai T, Maekawa K, et al. Predictors of 5-year mortality in pulmonary MAC disease. Int J Tuberc Lung Dis. 2012;16:408-414.
43. Yuji S, Yutsuki N, Keiichiso T, et al. Surgery for Mycobacterium avium lung disease in the clarithromycin era. Eur J Cardiothor Surg. 2002;21:314-318.
44. Tortoli E, Kohl TA, Brown-Elliott BA, et al. Emended description of Mycobacterium abscessus, Mycobacterium abscessus subsp. abscessus and Mycobacterium abscessus subsp. bolletii and designation of Mycobacterium abscessus subsp. massiliense comb. Int J Syst Evol Microbiol. 2016; 66:4471-4479.
45. Griffith DE, Girard WM, Wallace RJ Jr. Clinical features of pulmonary disease caused by rapidly growing mycobacteria. An analysis of 154 patients. Am Rev Respir Dis. 1993;147:1271-1278.
46. Koh WJ, Jeong BH, Kim SY, et al. Mycobacterial characteristics and treatment outcomes in Mycobacterium abscessus lung disease. Clin Infect Dis. 2017;64:309-316.
1. Johnson MM, Odell JA. Nontuberculous mycobacterial pulmonary infections. J Thorac Dis. 2014;6:210-220.
2. Falkinham JO III. Environmental sources of NTM. Clin Chest Med. 2015;36:35-41.
3. Falkinham JO III, Current epidemiological trends in NTM. Curr Environ Health Rep. 2016;3:161-167.
4. Honda JR, Knight V, Chan ED. Pathogenesis and risk factors for nontuberculous mycobacterial lung disease. Clin Chest Med. 2015;36:1-11.
5. Marras TK, Mirsaeidi M, Chou E, et al. Health care utilization and expenditures following diagnosis of nontuberculous mycobacterial lung disease in the United States. Manag Care Spec Pharm. 2018;24:964-974.
6. Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated healthcare delivery systems. Am J Respir Crit Care Med. 2010;182:970-976.
7. Winthrop KL, McNelley E, Kendall B, et al. Pulmonary nontuberculous mycobacterial disease prevalence and clinical features: an emerging public health disease. Am J Respir Crit Care Med. 2010;182:977-982.
8. Adjemian, Olivier KN, Seitz AE, J et al. Prevalence of nontuberculous mycobacterial lung disease in US Medicare beneficiaries. Am J Respir Crit Care Med. 2012;185;881-886.
9. Ringshausen FC, Apel RM, Bange FC, et al. Burden and trends of hospitalizations associated with pulmonary nontuberculous mycobacterial infections in Germany, 2005-2011. BMC Infect Dis. 2013;13:231.
10. Aliyu G, El-Kamary SS, Abimiku A, et al. Prevalence of non-tuberculous mycobacterial infections among tuberculosis suspects in Nigeria. PLoS One. 2013;8:e63170.
11. Griffith DE, Aksamit T, Brown-Elliott, et al; American Thoracic Society; Infectious Diseases Society of America. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367-415.
12. Wallace RJ Jr, Zhang Y, Brown BA, et al. Polyclonal Mycobacterium avium complex infections in patients with nodular bronchiectasis. Am J Respir Crit Care Med. 1998;158:1235-1244.
13. Gordin FM, Horsburgh CR Jr. Mycobacterium avium complex. In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Elsevier; 2015.
14. Chitty S, Ali J. Mycobacterium avium complex pulmonary disease in immune competent patients. South Med J. 2005;98:646-52.
15. Ramirez J, Mason C, Ali J, Lopez FA. MAC pulmonary disease: management options in HIV-negative patients. J La State Med Soc. 2008;160:248-254.
16. Iseman MD, Buschman DL, Ackerson LM. Pectus excavatum and scoliosis. Thoracic anomalies associated with pulmonary disease caused by Mycobacterium avium complex. Am Rev Respir Dis. 1991;144:914-916.
17. Kim RD, Greenburg DE, Ehrmantraut ME, et al. Pulmonary nontuberculous mycobacterial disease: prospective study of a distinct preexisting syndrome. Am J Respir Crit Care Med. 2008;178:1066-1074.
18. Ziedalski TM, Kao PN, Henig NR, et al. Prospective analysis of cystic fibrosis transmembrane regulator mutations in adults with bronchiectasis or pulmonary nontuberculous mycobacterial infection. Chest. 2006;130:995-1002.
19. Koh WJ, Lee KS, Kwon OJ, et al. Bilateral bronchiectasis and bronchiolitis at thin-section CT: diagnostic implications in nontuberculous mycobacterial pulmonary infection. Radiology. 2005;235:282-288.
20. Swensen SJ, Hartman TE, Williams DE. Computed tomographic diagnosis of Mycobacterium avium-intracellulare complex in patients with bronchiectasis. Chest. 1994;105:49-52.
21. Huang JH, Kao PN, Adi V, Ruoss SJ. Mycobacterium avium intracellulare pulmonary infection in HIV-negative patients without preexisting lung disease: diagnostic and management limitations. Chest. 1999;115:1033-1040.
22. Cappelluti E, Fraire AE, Schaefer OP. A case of “hot tub lung” due to Mycobacterium avium complex in an immunocompetent host. Arch Intern Med. 2003;163:845-848.
23. Nightingale SD, Byrd LT, Southern PM, et al. Incidence of Mycobacterium avium-intracellulare complex bacteremia in human immunodeficiency virus-positive patients. J Infect Dis. 1992;165:1082-1085.
24. Horsburgh CR Jr, Selik RM. The epidemiology of disseminated tuberculous mycobacterial infection in the acquired immunodeficiency syndrome (AIDS). Am Rev Respir Dis. 1989;139:4-7.
25. Chin DP, Hopewell PC, Yajko DM, et al. Mycobacterium avium complex in the respiratory or gastrointestinal tract and the risk of M. avium complex bacteremia in patients with human immunodeficiency virus infection. J Infect Dis. 1994;169:289-295.
26. Khan K, Wang J, Marras TK. Nontuberculous mycobacterial sensitization in the United States: national trends over three decades. Am J Respir Crit Care Med. 2007;176:306-313.
27. Lillo M, Orengo S, Cernoch P, Harris RL. Pulmonary and disseminated infection due to Mycobacterium kansasii: a decade of experience. Rev Infect Dis. 1990;12:760-767.
28. Andersen P, Munk ME, Pollock JM, Doherty TM. Specific immune-based diagnosis of tuberculosis. Lancet. 2000;356:1099-1104.
29. Arend SM, van Meijgaarden KE, de Boer K, et al. Tuberculin skin testing and in vitro T cell responses to ESAT-6 and culture filtrate protein 10 after infection with Mycobacterium marinum or M. kansasii. J Infect Dis. 2002;186:1797-1807.
30. James D, Chalmers JD, Goeminne P, et al. The Bronchiectasis Severity Index: an international derivation and validation study. Am J Respir Crit Care Med. 2014;189:576-585.
31. Heifets L. MIC as a quantitative measurement of the susceptibility of Mycobacterium avium strains to seven antituberculosis drugs. Antimicrob Agents Chemother. 1988;32:1131-1136.
32. Horsburgh CR Jr, Mason UG 3rd, Heifits LB, et al. Response to therapy of pulmonary Mycobacterium avium intracellulare infection correlates with results of in vitro susceptibility testing. Am Rev Respir Dis. 1987;135:418-421.
33. Rubin BK, Henke MO. Immunomodulatory activity and effectiveness of macrolides in chronic airway disease. Chest. 2004;125(2 Suppl):70S-78S.
34. Wallace RJ Jr, Brown BA, Griffith DE, et al. Clarithromycin regimens for pulmonary Mycobacterium avium complex. The first 50 patients. Am J Respir Crit Care Med. 1996;153:1766-1772.
35. Griffith DE, Brown-Elliott BA, Langsjoen B, et al. Clinical and molecular analysis of macrolide resistance in Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006;174:928-934.
36. Lam PK, Griffith DE, Aksamit TR, et al. Factors related to response to intermittent treatment of Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006;173:1283-1289.
37. Falkinham J III. Challenges of NTM drug development. Front Microbiol. 2018;9:1613.
38. Griffith DE, Eagle G, Thomson R, et al. Amikacin liposome inhalation suspension for treatment-refractory lung disease caused by Mycobacterium avium complex (CONVERT). A prospective, open-label, randomized study. Am J Respir Crit Care Med. 2018;198:1559-1569.
39. Schluger NW. Treatment of pulmonary Mycobacterium avium complex infections: do drug levels matter? Am J Respir Crit Care Med. 2012;186:710-711.
40. Van Ingen J, Egelund EF, Levin A, et al. The pharmacokinetics and pharmacodynamics of pulmonary Mycobacterium avium complex disease treatment. Am J Respir Crit Care Med. 2012;186:559-565.
41. Koh WJ, Jeong BH, Jeon K, et al. Therapeutic drug monitoring in the treatment of Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2012;186:797-802.
42. Ito Y, Hirai T, Maekawa K, et al. Predictors of 5-year mortality in pulmonary MAC disease. Int J Tuberc Lung Dis. 2012;16:408-414.
43. Yuji S, Yutsuki N, Keiichiso T, et al. Surgery for Mycobacterium avium lung disease in the clarithromycin era. Eur J Cardiothor Surg. 2002;21:314-318.
44. Tortoli E, Kohl TA, Brown-Elliott BA, et al. Emended description of Mycobacterium abscessus, Mycobacterium abscessus subsp. abscessus and Mycobacterium abscessus subsp. bolletii and designation of Mycobacterium abscessus subsp. massiliense comb. Int J Syst Evol Microbiol. 2016; 66:4471-4479.
45. Griffith DE, Girard WM, Wallace RJ Jr. Clinical features of pulmonary disease caused by rapidly growing mycobacteria. An analysis of 154 patients. Am Rev Respir Dis. 1993;147:1271-1278.
46. Koh WJ, Jeong BH, Kim SY, et al. Mycobacterial characteristics and treatment outcomes in Mycobacterium abscessus lung disease. Clin Infect Dis. 2017;64:309-316.
Measles, scarlet fever among infectious diseases to watch for in 2020
ORLANDO – – leading into 2020, Justin Finch, MD, said at the ODAC Dermatology, Aesthetic, & Surgical Conference.
While group A streptococcus has declined over the past century, there has been “an unprecedented” resurgence in severe, invasive group A streptococcal infections and severe epidemics of scarlet fever worldwide, including in industrialized regions like the United Kingdom. Shedding some light on why this may be occurring, Dr. Finch referred to a recently published population-based molecular epidemiologic study identified a new dominant emm1UK lineage of Streptococcus pyogenes associated with such cases in England (Lancet Infect Dis. 2019 Nov;19(11):1209-18). This new lineage of S. pyogenes was genotypically distinct from other emm1 isolates and had greatly increased expression of the streptococcal pyrogenic exotoxin A, one of the exotoxins responsible for the clinical features of scarlet fever.
“We have not, to my knowledge, seen the strain yet in the United States,” said Dr. Finch, of Central Connecticut Dermatology in Cromwell. “Have it on your radar. With all of the worldwide travel patterns, I expect that you will see this in the United States at some point in the not-too-distant future.”
Also in 2019, promising data on the safety and effectiveness of the recombinant herpes zoster vaccine in immunocompromised patients became available for the first time. A randomized clinical trial published in JAMA of 1,846 patients who were immunosuppressed after autologous hematopoietic stem cell transplantation and received two doses of a recombinant zoster vaccine found that the patients had a reduced incidence of herpes zoster after a median follow-up of 21 months (JAMA. 2019 Jul 9;322[2]:123-33). The study found that the recombinant vaccine was both safe and effective in these immunocompromised patients, “so we can easily generalize this to our dermatology population as well,” Dr. Finch said. In comparing the live attenuated and recombinant vaccines, he noted the recombinant vaccine requires two doses but appears to be slightly more effective. “The number needed to treat to prevent [one case] of zoster is about half as high as that for the live vaccine, and most importantly for us is, it’s safe in immunocompromised patients.”
2019 also saw a record high in the number of measles cases in the United States, the highest since 1993, Dr. Finch pointed out. Most cases were seen in the area in and around New York City, but the percentage of people across the United States who are vaccinated against measles is below the threshold for herd immunity to protect immunocompromised patients. Measles requires a population vaccination rate of 94%, and less than half of U.S. counties in 2014 and 2015 reached that vaccination rate.
“Furthermore, if we look at that over the last 20 years, comparing the domestic measles cases to imported measles cases, we are increasingly breeding these measles epidemics right here at home, whereas they used to be imported from throughout the world,” said Dr. Finch. Patients with measles can be treated with vitamin A, he added, referring to a Cochrane review showing that 200,000 units of vitamin A given daily for 2 days decreased the mortality rate of measles by about 80%. Measles is on the Centers for Disease Control and Prevention’s list of reportable diseases, so should be reported to local health authorities, and will be followed up with confirmatory testing.
In 2019, a study examining herd protection of oral human papillomavirus infection in men and women compared the prevalence of oral HPV infection based on the 4 HPV types present in the quadrivalent HPV vaccine with 33 nonvaccine types from 2009 to 2016. There was no change in the prevalence of nonvaccine type oral HPV infections among men who were unvaccinated, but the prevalence of oral HPV infections because of the four strains in the quadrivalent HPV vaccine declined from 2.7% in 2009-2010 to 1.6% in 2015-2016 (JAMA. 2019 Sep 10;322[10]:977-9). Among unvaccinated women, the prevalence of nonvaccine- and vaccine-type oral HPV infections did not change between the two time periods.
“Notably, this only occurred in men,” Dr. Finch said. Herd immunity is being achieved in men “because we’re vaccinating all women, [but] we’re not seeing that herd immunity in women. Which begs the question: Why are we still vaccinating only half of our population?”
One study published in 2019 (Br J Dermatol. 2019 Nov;181[5]:1093-5) described a patient with CARD9 mutations, which predispose individuals to deep invasive infections – a disseminated Microsporum infection in this case, Dr. Finch said. “You shouldn’t see that,” he added, noting that these mutations are known to predispose individuals to severe Trichophyton infections and familial candidiasis.
“What I think is interesting about this is that, as we look forward to 2020, we’re going to increasingly see studies like this that are identifying specific mutations in our community that underlie a lot of these weird infections,” he added. “I wouldn’t be surprised if within the span of our careers, we find that a lot of those severe treatment-refractory reports that so commonly plague your everyday clinic have some underlying, specific immunity.”
Dr. Finch reported no relevant conflicts of interest.
ORLANDO – – leading into 2020, Justin Finch, MD, said at the ODAC Dermatology, Aesthetic, & Surgical Conference.
While group A streptococcus has declined over the past century, there has been “an unprecedented” resurgence in severe, invasive group A streptococcal infections and severe epidemics of scarlet fever worldwide, including in industrialized regions like the United Kingdom. Shedding some light on why this may be occurring, Dr. Finch referred to a recently published population-based molecular epidemiologic study identified a new dominant emm1UK lineage of Streptococcus pyogenes associated with such cases in England (Lancet Infect Dis. 2019 Nov;19(11):1209-18). This new lineage of S. pyogenes was genotypically distinct from other emm1 isolates and had greatly increased expression of the streptococcal pyrogenic exotoxin A, one of the exotoxins responsible for the clinical features of scarlet fever.
“We have not, to my knowledge, seen the strain yet in the United States,” said Dr. Finch, of Central Connecticut Dermatology in Cromwell. “Have it on your radar. With all of the worldwide travel patterns, I expect that you will see this in the United States at some point in the not-too-distant future.”
Also in 2019, promising data on the safety and effectiveness of the recombinant herpes zoster vaccine in immunocompromised patients became available for the first time. A randomized clinical trial published in JAMA of 1,846 patients who were immunosuppressed after autologous hematopoietic stem cell transplantation and received two doses of a recombinant zoster vaccine found that the patients had a reduced incidence of herpes zoster after a median follow-up of 21 months (JAMA. 2019 Jul 9;322[2]:123-33). The study found that the recombinant vaccine was both safe and effective in these immunocompromised patients, “so we can easily generalize this to our dermatology population as well,” Dr. Finch said. In comparing the live attenuated and recombinant vaccines, he noted the recombinant vaccine requires two doses but appears to be slightly more effective. “The number needed to treat to prevent [one case] of zoster is about half as high as that for the live vaccine, and most importantly for us is, it’s safe in immunocompromised patients.”
2019 also saw a record high in the number of measles cases in the United States, the highest since 1993, Dr. Finch pointed out. Most cases were seen in the area in and around New York City, but the percentage of people across the United States who are vaccinated against measles is below the threshold for herd immunity to protect immunocompromised patients. Measles requires a population vaccination rate of 94%, and less than half of U.S. counties in 2014 and 2015 reached that vaccination rate.
“Furthermore, if we look at that over the last 20 years, comparing the domestic measles cases to imported measles cases, we are increasingly breeding these measles epidemics right here at home, whereas they used to be imported from throughout the world,” said Dr. Finch. Patients with measles can be treated with vitamin A, he added, referring to a Cochrane review showing that 200,000 units of vitamin A given daily for 2 days decreased the mortality rate of measles by about 80%. Measles is on the Centers for Disease Control and Prevention’s list of reportable diseases, so should be reported to local health authorities, and will be followed up with confirmatory testing.
In 2019, a study examining herd protection of oral human papillomavirus infection in men and women compared the prevalence of oral HPV infection based on the 4 HPV types present in the quadrivalent HPV vaccine with 33 nonvaccine types from 2009 to 2016. There was no change in the prevalence of nonvaccine type oral HPV infections among men who were unvaccinated, but the prevalence of oral HPV infections because of the four strains in the quadrivalent HPV vaccine declined from 2.7% in 2009-2010 to 1.6% in 2015-2016 (JAMA. 2019 Sep 10;322[10]:977-9). Among unvaccinated women, the prevalence of nonvaccine- and vaccine-type oral HPV infections did not change between the two time periods.
“Notably, this only occurred in men,” Dr. Finch said. Herd immunity is being achieved in men “because we’re vaccinating all women, [but] we’re not seeing that herd immunity in women. Which begs the question: Why are we still vaccinating only half of our population?”
One study published in 2019 (Br J Dermatol. 2019 Nov;181[5]:1093-5) described a patient with CARD9 mutations, which predispose individuals to deep invasive infections – a disseminated Microsporum infection in this case, Dr. Finch said. “You shouldn’t see that,” he added, noting that these mutations are known to predispose individuals to severe Trichophyton infections and familial candidiasis.
“What I think is interesting about this is that, as we look forward to 2020, we’re going to increasingly see studies like this that are identifying specific mutations in our community that underlie a lot of these weird infections,” he added. “I wouldn’t be surprised if within the span of our careers, we find that a lot of those severe treatment-refractory reports that so commonly plague your everyday clinic have some underlying, specific immunity.”
Dr. Finch reported no relevant conflicts of interest.
ORLANDO – – leading into 2020, Justin Finch, MD, said at the ODAC Dermatology, Aesthetic, & Surgical Conference.
While group A streptococcus has declined over the past century, there has been “an unprecedented” resurgence in severe, invasive group A streptococcal infections and severe epidemics of scarlet fever worldwide, including in industrialized regions like the United Kingdom. Shedding some light on why this may be occurring, Dr. Finch referred to a recently published population-based molecular epidemiologic study identified a new dominant emm1UK lineage of Streptococcus pyogenes associated with such cases in England (Lancet Infect Dis. 2019 Nov;19(11):1209-18). This new lineage of S. pyogenes was genotypically distinct from other emm1 isolates and had greatly increased expression of the streptococcal pyrogenic exotoxin A, one of the exotoxins responsible for the clinical features of scarlet fever.
“We have not, to my knowledge, seen the strain yet in the United States,” said Dr. Finch, of Central Connecticut Dermatology in Cromwell. “Have it on your radar. With all of the worldwide travel patterns, I expect that you will see this in the United States at some point in the not-too-distant future.”
Also in 2019, promising data on the safety and effectiveness of the recombinant herpes zoster vaccine in immunocompromised patients became available for the first time. A randomized clinical trial published in JAMA of 1,846 patients who were immunosuppressed after autologous hematopoietic stem cell transplantation and received two doses of a recombinant zoster vaccine found that the patients had a reduced incidence of herpes zoster after a median follow-up of 21 months (JAMA. 2019 Jul 9;322[2]:123-33). The study found that the recombinant vaccine was both safe and effective in these immunocompromised patients, “so we can easily generalize this to our dermatology population as well,” Dr. Finch said. In comparing the live attenuated and recombinant vaccines, he noted the recombinant vaccine requires two doses but appears to be slightly more effective. “The number needed to treat to prevent [one case] of zoster is about half as high as that for the live vaccine, and most importantly for us is, it’s safe in immunocompromised patients.”
2019 also saw a record high in the number of measles cases in the United States, the highest since 1993, Dr. Finch pointed out. Most cases were seen in the area in and around New York City, but the percentage of people across the United States who are vaccinated against measles is below the threshold for herd immunity to protect immunocompromised patients. Measles requires a population vaccination rate of 94%, and less than half of U.S. counties in 2014 and 2015 reached that vaccination rate.
“Furthermore, if we look at that over the last 20 years, comparing the domestic measles cases to imported measles cases, we are increasingly breeding these measles epidemics right here at home, whereas they used to be imported from throughout the world,” said Dr. Finch. Patients with measles can be treated with vitamin A, he added, referring to a Cochrane review showing that 200,000 units of vitamin A given daily for 2 days decreased the mortality rate of measles by about 80%. Measles is on the Centers for Disease Control and Prevention’s list of reportable diseases, so should be reported to local health authorities, and will be followed up with confirmatory testing.
In 2019, a study examining herd protection of oral human papillomavirus infection in men and women compared the prevalence of oral HPV infection based on the 4 HPV types present in the quadrivalent HPV vaccine with 33 nonvaccine types from 2009 to 2016. There was no change in the prevalence of nonvaccine type oral HPV infections among men who were unvaccinated, but the prevalence of oral HPV infections because of the four strains in the quadrivalent HPV vaccine declined from 2.7% in 2009-2010 to 1.6% in 2015-2016 (JAMA. 2019 Sep 10;322[10]:977-9). Among unvaccinated women, the prevalence of nonvaccine- and vaccine-type oral HPV infections did not change between the two time periods.
“Notably, this only occurred in men,” Dr. Finch said. Herd immunity is being achieved in men “because we’re vaccinating all women, [but] we’re not seeing that herd immunity in women. Which begs the question: Why are we still vaccinating only half of our population?”
One study published in 2019 (Br J Dermatol. 2019 Nov;181[5]:1093-5) described a patient with CARD9 mutations, which predispose individuals to deep invasive infections – a disseminated Microsporum infection in this case, Dr. Finch said. “You shouldn’t see that,” he added, noting that these mutations are known to predispose individuals to severe Trichophyton infections and familial candidiasis.
“What I think is interesting about this is that, as we look forward to 2020, we’re going to increasingly see studies like this that are identifying specific mutations in our community that underlie a lot of these weird infections,” he added. “I wouldn’t be surprised if within the span of our careers, we find that a lot of those severe treatment-refractory reports that so commonly plague your everyday clinic have some underlying, specific immunity.”
Dr. Finch reported no relevant conflicts of interest.
REPORTING FROM ODAC 2020
Trump takes on multiple health topics in State of the Union
President Donald J. Trump took on multiple health care issues in his State of the Union address, imploring Congress to avoid the “socialism” of Medicare-for-all, to pass legislation banning late-term abortions, and to protect insurance coverage for preexisting conditions while joining together to reduce rising drug prices.
Mr. Trump said his administration has already been “taking on the big pharmaceutical companies,” claiming that, in 2019, “for the first time in 51 years, the cost of prescription drugs actually went down.”
That statement was called “misleading” by the New York Times because such efforts have excluded some high-cost drugs, and prices had risen by the end of the year, the publication noted in a fact-check of the president’s speech.
A survey issued in December 2019 found that the United States pays the highest prices in the world for pharmaceuticals, as reported by Medscape Medical News.
But the president did throw down a gauntlet for Congress. “Working together, the Congress can reduce drug prices substantially from current levels,” he said, stating that he had been “speaking to Sen. Chuck Grassley of Iowa and others in the Congress in order to get something on drug pricing done, and done properly.
“Get a bill to my desk, and I will sign it into law without delay,” Mr. Trump said.
A group of House Democrats then stood up in the chamber and loudly chanted, “HR3, HR3,” referring to the Lower Drug Costs Now Act, which the House passed in December 2019.
The bill would give the Department of Health & Human Services the power to negotiate directly with drug companies on up to 250 drugs per year, in particular, the highest-costing and most-utilized drugs.
The Senate has not taken up the legislation, but Sen. Grassley (R) and Sen. Ron Wyden (D-Ore.) introduced a similar bill, the Prescription Drug Pricing Reduction Act. It has been approved by the Senate Finance Committee but has not been moved to the Senate floor.
“I appreciate President Trump recognizing the work we’re doing to lower prescription drug prices,” Sen. Grassley said in a statement after the State of the Union. “Iowans and Americans across the country are demanding reforms that lower sky-high drug costs. A recent poll showed 70% of Americans want Congress to make lowering drug prices its top priority.”
Rep. Greg Walden (R-Ore.), the ranking Republican on the House Energy and Commerce Committee, said he believed Trump was committed to lowering drug costs. “I’ve never seen a president lean in further than President Donald Trump on lowering health care costs,” said Rep. Walden in a statement after the speech.
Trump touted his price transparency rule, which he said would go into effect next January, as a key way to cut health care costs.
Preexisting conditions
The president said that since he’d taken office, insurance had become more affordable and that the quality of health care had improved. He also said that he was making what he called an “iron-clad pledge” to American families.
“We will always protect patients with preexisting conditions – that is a guarantee,” Mr. Trump said.
In a press conference before the speech, Speaker of the House Nancy Pelosi (D-Calif.) took issue with that pledge. “The president swears that he supports protections for people with preexisting conditions, but right now, he is fighting in federal court to eliminate these lifesaving protections and every last protection and benefit of the Affordable Care Act,” she said.
During the speech, Rep. G. K. Butterfield (D-N.C.) tweeted “#FactCheck: Claiming to protect Americans with preexisting conditions, Trump and his administration have repeatedly sought to undermine protections offered by the ACA through executive orders and the courts. He is seeking to strike down the law and its protections entirely.”
Larry Levitt, executive vice president for health policy at the Kaiser Family Foundation, pointed out in a tweet that insurance plans that Trump touted as “affordable alternatives” are in fact missing those protections.
“Ironically, the cheaper health insurance plans that President Trump has expanded are short-term plans that don’t cover preexisting conditions,” Mr. Levitt said.
Socialist takeover
Mr. Trump condemned the Medicare-for-all proposals that have been introduced in Congress and that are being backed in whole or in part by all of the Democratic candidates for president.
“As we work to improve Americans’ health care, there are those who want to take away your health care, take away your doctor, and abolish private insurance entirely,” said Mr. Trump.
He said that 132 members of Congress “have endorsed legislation to impose a socialist takeover of our health care system, wiping out the private health insurance plans of 180 million Americans.”
Added Mr. Trump: “We will never let socialism destroy American health care!”
Medicare-for-all has waxed and waned in popularity among voters, with generally more Democrats than Republicans favoring a single-payer system, with or without a public option.
Preliminary exit polls in Iowa that were conducted during Monday’s caucus found that 57% of Iowa Democratic caucus-goers supported a single-payer plan; 38% opposed such a plan, according to the Washington Post.
Opioids, the coronavirus, and abortion
In some of his final remarks on health care, Mr. Trump cited progress in the opioid crisis, noting that, in 2019, drug overdose deaths declined for the first time in 30 years.
He said that his administration was coordinating with the Chinese government regarding the coronavirus outbreak and noted the launch of initiatives to improve care for people with kidney disease, Alzheimer’s, and mental health problems.
Mr. Trump repeated his 2019 State of the Union claim that the government would help end AIDS in America by the end of the decade.
The president also announced that he was asking Congress for “an additional $50 million” to fund neonatal research. He followed that up with a plea about abortion.
“I am calling upon the members of Congress here tonight to pass legislation finally banning the late-term abortion of babies,” he said.
Insulin costs?
In the days before the speech, some news outlets had reported that Mr. Trump and the HHS were working on a plan to lower insulin prices for Medicare beneficiaries, and there were suggestions it would come up in the speech.
At least 13 members of Congress invited people advocating for lower insulin costs as their guests for the State of the Union, Stat reported. Rep. Pelosi invited twins from San Francisco with type 1 diabetes as her guests.
But Mr. Trump never mentioned insulin in his speech.
This article first appeared on Medscape.com.
President Donald J. Trump took on multiple health care issues in his State of the Union address, imploring Congress to avoid the “socialism” of Medicare-for-all, to pass legislation banning late-term abortions, and to protect insurance coverage for preexisting conditions while joining together to reduce rising drug prices.
Mr. Trump said his administration has already been “taking on the big pharmaceutical companies,” claiming that, in 2019, “for the first time in 51 years, the cost of prescription drugs actually went down.”
That statement was called “misleading” by the New York Times because such efforts have excluded some high-cost drugs, and prices had risen by the end of the year, the publication noted in a fact-check of the president’s speech.
A survey issued in December 2019 found that the United States pays the highest prices in the world for pharmaceuticals, as reported by Medscape Medical News.
But the president did throw down a gauntlet for Congress. “Working together, the Congress can reduce drug prices substantially from current levels,” he said, stating that he had been “speaking to Sen. Chuck Grassley of Iowa and others in the Congress in order to get something on drug pricing done, and done properly.
“Get a bill to my desk, and I will sign it into law without delay,” Mr. Trump said.
A group of House Democrats then stood up in the chamber and loudly chanted, “HR3, HR3,” referring to the Lower Drug Costs Now Act, which the House passed in December 2019.
The bill would give the Department of Health & Human Services the power to negotiate directly with drug companies on up to 250 drugs per year, in particular, the highest-costing and most-utilized drugs.
The Senate has not taken up the legislation, but Sen. Grassley (R) and Sen. Ron Wyden (D-Ore.) introduced a similar bill, the Prescription Drug Pricing Reduction Act. It has been approved by the Senate Finance Committee but has not been moved to the Senate floor.
“I appreciate President Trump recognizing the work we’re doing to lower prescription drug prices,” Sen. Grassley said in a statement after the State of the Union. “Iowans and Americans across the country are demanding reforms that lower sky-high drug costs. A recent poll showed 70% of Americans want Congress to make lowering drug prices its top priority.”
Rep. Greg Walden (R-Ore.), the ranking Republican on the House Energy and Commerce Committee, said he believed Trump was committed to lowering drug costs. “I’ve never seen a president lean in further than President Donald Trump on lowering health care costs,” said Rep. Walden in a statement after the speech.
Trump touted his price transparency rule, which he said would go into effect next January, as a key way to cut health care costs.
Preexisting conditions
The president said that since he’d taken office, insurance had become more affordable and that the quality of health care had improved. He also said that he was making what he called an “iron-clad pledge” to American families.
“We will always protect patients with preexisting conditions – that is a guarantee,” Mr. Trump said.
In a press conference before the speech, Speaker of the House Nancy Pelosi (D-Calif.) took issue with that pledge. “The president swears that he supports protections for people with preexisting conditions, but right now, he is fighting in federal court to eliminate these lifesaving protections and every last protection and benefit of the Affordable Care Act,” she said.
During the speech, Rep. G. K. Butterfield (D-N.C.) tweeted “#FactCheck: Claiming to protect Americans with preexisting conditions, Trump and his administration have repeatedly sought to undermine protections offered by the ACA through executive orders and the courts. He is seeking to strike down the law and its protections entirely.”
Larry Levitt, executive vice president for health policy at the Kaiser Family Foundation, pointed out in a tweet that insurance plans that Trump touted as “affordable alternatives” are in fact missing those protections.
“Ironically, the cheaper health insurance plans that President Trump has expanded are short-term plans that don’t cover preexisting conditions,” Mr. Levitt said.
Socialist takeover
Mr. Trump condemned the Medicare-for-all proposals that have been introduced in Congress and that are being backed in whole or in part by all of the Democratic candidates for president.
“As we work to improve Americans’ health care, there are those who want to take away your health care, take away your doctor, and abolish private insurance entirely,” said Mr. Trump.
He said that 132 members of Congress “have endorsed legislation to impose a socialist takeover of our health care system, wiping out the private health insurance plans of 180 million Americans.”
Added Mr. Trump: “We will never let socialism destroy American health care!”
Medicare-for-all has waxed and waned in popularity among voters, with generally more Democrats than Republicans favoring a single-payer system, with or without a public option.
Preliminary exit polls in Iowa that were conducted during Monday’s caucus found that 57% of Iowa Democratic caucus-goers supported a single-payer plan; 38% opposed such a plan, according to the Washington Post.
Opioids, the coronavirus, and abortion
In some of his final remarks on health care, Mr. Trump cited progress in the opioid crisis, noting that, in 2019, drug overdose deaths declined for the first time in 30 years.
He said that his administration was coordinating with the Chinese government regarding the coronavirus outbreak and noted the launch of initiatives to improve care for people with kidney disease, Alzheimer’s, and mental health problems.
Mr. Trump repeated his 2019 State of the Union claim that the government would help end AIDS in America by the end of the decade.
The president also announced that he was asking Congress for “an additional $50 million” to fund neonatal research. He followed that up with a plea about abortion.
“I am calling upon the members of Congress here tonight to pass legislation finally banning the late-term abortion of babies,” he said.
Insulin costs?
In the days before the speech, some news outlets had reported that Mr. Trump and the HHS were working on a plan to lower insulin prices for Medicare beneficiaries, and there were suggestions it would come up in the speech.
At least 13 members of Congress invited people advocating for lower insulin costs as their guests for the State of the Union, Stat reported. Rep. Pelosi invited twins from San Francisco with type 1 diabetes as her guests.
But Mr. Trump never mentioned insulin in his speech.
This article first appeared on Medscape.com.
President Donald J. Trump took on multiple health care issues in his State of the Union address, imploring Congress to avoid the “socialism” of Medicare-for-all, to pass legislation banning late-term abortions, and to protect insurance coverage for preexisting conditions while joining together to reduce rising drug prices.
Mr. Trump said his administration has already been “taking on the big pharmaceutical companies,” claiming that, in 2019, “for the first time in 51 years, the cost of prescription drugs actually went down.”
That statement was called “misleading” by the New York Times because such efforts have excluded some high-cost drugs, and prices had risen by the end of the year, the publication noted in a fact-check of the president’s speech.
A survey issued in December 2019 found that the United States pays the highest prices in the world for pharmaceuticals, as reported by Medscape Medical News.
But the president did throw down a gauntlet for Congress. “Working together, the Congress can reduce drug prices substantially from current levels,” he said, stating that he had been “speaking to Sen. Chuck Grassley of Iowa and others in the Congress in order to get something on drug pricing done, and done properly.
“Get a bill to my desk, and I will sign it into law without delay,” Mr. Trump said.
A group of House Democrats then stood up in the chamber and loudly chanted, “HR3, HR3,” referring to the Lower Drug Costs Now Act, which the House passed in December 2019.
The bill would give the Department of Health & Human Services the power to negotiate directly with drug companies on up to 250 drugs per year, in particular, the highest-costing and most-utilized drugs.
The Senate has not taken up the legislation, but Sen. Grassley (R) and Sen. Ron Wyden (D-Ore.) introduced a similar bill, the Prescription Drug Pricing Reduction Act. It has been approved by the Senate Finance Committee but has not been moved to the Senate floor.
“I appreciate President Trump recognizing the work we’re doing to lower prescription drug prices,” Sen. Grassley said in a statement after the State of the Union. “Iowans and Americans across the country are demanding reforms that lower sky-high drug costs. A recent poll showed 70% of Americans want Congress to make lowering drug prices its top priority.”
Rep. Greg Walden (R-Ore.), the ranking Republican on the House Energy and Commerce Committee, said he believed Trump was committed to lowering drug costs. “I’ve never seen a president lean in further than President Donald Trump on lowering health care costs,” said Rep. Walden in a statement after the speech.
Trump touted his price transparency rule, which he said would go into effect next January, as a key way to cut health care costs.
Preexisting conditions
The president said that since he’d taken office, insurance had become more affordable and that the quality of health care had improved. He also said that he was making what he called an “iron-clad pledge” to American families.
“We will always protect patients with preexisting conditions – that is a guarantee,” Mr. Trump said.
In a press conference before the speech, Speaker of the House Nancy Pelosi (D-Calif.) took issue with that pledge. “The president swears that he supports protections for people with preexisting conditions, but right now, he is fighting in federal court to eliminate these lifesaving protections and every last protection and benefit of the Affordable Care Act,” she said.
During the speech, Rep. G. K. Butterfield (D-N.C.) tweeted “#FactCheck: Claiming to protect Americans with preexisting conditions, Trump and his administration have repeatedly sought to undermine protections offered by the ACA through executive orders and the courts. He is seeking to strike down the law and its protections entirely.”
Larry Levitt, executive vice president for health policy at the Kaiser Family Foundation, pointed out in a tweet that insurance plans that Trump touted as “affordable alternatives” are in fact missing those protections.
“Ironically, the cheaper health insurance plans that President Trump has expanded are short-term plans that don’t cover preexisting conditions,” Mr. Levitt said.
Socialist takeover
Mr. Trump condemned the Medicare-for-all proposals that have been introduced in Congress and that are being backed in whole or in part by all of the Democratic candidates for president.
“As we work to improve Americans’ health care, there are those who want to take away your health care, take away your doctor, and abolish private insurance entirely,” said Mr. Trump.
He said that 132 members of Congress “have endorsed legislation to impose a socialist takeover of our health care system, wiping out the private health insurance plans of 180 million Americans.”
Added Mr. Trump: “We will never let socialism destroy American health care!”
Medicare-for-all has waxed and waned in popularity among voters, with generally more Democrats than Republicans favoring a single-payer system, with or without a public option.
Preliminary exit polls in Iowa that were conducted during Monday’s caucus found that 57% of Iowa Democratic caucus-goers supported a single-payer plan; 38% opposed such a plan, according to the Washington Post.
Opioids, the coronavirus, and abortion
In some of his final remarks on health care, Mr. Trump cited progress in the opioid crisis, noting that, in 2019, drug overdose deaths declined for the first time in 30 years.
He said that his administration was coordinating with the Chinese government regarding the coronavirus outbreak and noted the launch of initiatives to improve care for people with kidney disease, Alzheimer’s, and mental health problems.
Mr. Trump repeated his 2019 State of the Union claim that the government would help end AIDS in America by the end of the decade.
The president also announced that he was asking Congress for “an additional $50 million” to fund neonatal research. He followed that up with a plea about abortion.
“I am calling upon the members of Congress here tonight to pass legislation finally banning the late-term abortion of babies,” he said.
Insulin costs?
In the days before the speech, some news outlets had reported that Mr. Trump and the HHS were working on a plan to lower insulin prices for Medicare beneficiaries, and there were suggestions it would come up in the speech.
At least 13 members of Congress invited people advocating for lower insulin costs as their guests for the State of the Union, Stat reported. Rep. Pelosi invited twins from San Francisco with type 1 diabetes as her guests.
But Mr. Trump never mentioned insulin in his speech.
This article first appeared on Medscape.com.
Home BP now a class Ia recommendation, with good reason
SNOWMASS, COLO. – The redefinition of hypertension as 130/80 mm Hg or higher introduced in the current American College of Cardiology/American Heart Association hypertension management guidelines has generated considerable controversy. Often overlooked, however, has been another major innovation included in the 2017 guidelines: the rise in the status of out-of-office 24-hour ambulatory blood pressure monitoring and home blood pressure self-measurement to a class I, level of evidence A recommendation, Andrew M. Kates, MD, observed at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
It’s a guideline he strongly endorses.
“We do a lot of this. It can be a challenge to get 24-hour ambulatory blood pressure monitoring covered by payers, so said Dr. Kates, professor of medicine and director of the cardiology fellowship program at Washington University, St. Louis.
He explained that one of the four key questions the guideline committee was tasked with answering at the outset of deliberations was this: What’s the evidence base for self-directed out-of-office blood pressure monitoring? Based on the panel’s systematic review of the literature, this practice wound up receiving the strongest possible class Ia recommendation, specifically for confirming the diagnosis of hypertension and for titration of antihypertensive medications. Moreover, the guidelines also endorsed home blood pressure monitoring for the detection of white-coat hypertension, this time as a Class IIa recommendation, as well as for identification of patients with masked hypertension, with class IIb status (Circulation. 2018 Oct 23;138[17]:e484-594).
The 2017 ACC/AHA guidelines include a detailed checklist for obtaining accurate measurements of office blood pressure. The suggestions include having the patient sit relaxed in a chair with both feet on the floor for at least 5 minutes before taking the measurement, no coffee or exercise for 30 minutes beforehand, empty the bladder, no talking, no clothing over the arm, and other recommendations. Many busy clinicians roll their eyes at the impracticality of doing all this on a routine basis.
“I don’t want to take an audience survey, but I’ll say that even in our office we are not successful in doing this. Patients run up the stairs to the office after dealing with traffic and the parking garage, they’re late for their appointment, in winter they’re wearing a sweater and don’t want to take it off. These are things we don’t do well, and they’re low-hanging fruit where we could do better,” Dr. Kates commented.
The challenges inherent in performing by-the-book office blood pressure measurement reinforce the importance of home self-monitoring of blood pressure in what is hopefully a more stress-free environment.
“We can give patients specific guidance about checking their blood pressure an hour after taking their medications, sitting for 5 minutes, and checking the pressures on a bare arm and not with the sleeve rolled up,” he noted.
The guidelines recommend using home blood pressure monitoring or ambulatory monitoring to detect white-coat hypertension in patients with an office blood pressure of 130/80 mm Hg or more, but less than 160/100 mm Hg, after a 3-month trial of lifestyle modification. If the home blood pressure is less than 130/80 mm Hg, that’s evidence of white-coat hypertension, for which the recommended treatment consists of continued lifestyle modification plus periodic monitoring of out-of-office blood pressures in order to promptly detect progression to hypertension. If, however, the out-of-office blood pressure is not less than 130/80 mm Hg, that’s hypertension, and the guidelines recommend starting dual-agent antihypertensive drug therapy while continuing lifestyle modification.
A confusing array of definitions of hypertension are now in use by various medical societies. While the 2017 ACC/AHA hypertension guidelines define hypertension as office blood pressure of 130/80 mm Hg or more, the 2018 European Society of Cardiology/European Society of Hypertension guidelines use a threshold of 140/90 mm Hg or more. Joint American Academy of Family Physicians/American College of Physicians guidelines recommend a treatment target of less than 150 mm Hg in hypertensive patients aged 60 years or older. And at the other end of the spectrum, the SPRINT trial showed a significant cardiovascular benefit for intensive treatment of hypertension to a target systolic blood pressure below 120 mm Hg, rather than less than 140 mm Hg (N Engl J Med. 2015 Nov 26;373[22]:2103-16).
Dr. Kates believes the debate over the “right” treatment target misses the central point, which is that hypertension is staggeringly undertreated. Indeed, the Centers for Disease Control and Prevention estimates only one in four adults with hypertension have their disease under control. That’s a disconcerting statistic given that hypertension accounts for more cardiovascular deaths than any other modifiable cardiovascular risk factor.
“There’s been some concern raised that maybe too much weight has been put on the SPRINT trial in making the ACC/AHA recommendations, but I think it’s helpful to understand that we vastly undertreat patients with hypertension. So I think that, rather than being so concerned that we’re going to be treating people to too low a target or we’re being overly aggressive, it should give us some pause to think about the fact that we’re ordinarily not being aggressive enough with many of our patients as it is,” the cardiologist said.
Dr. Kates reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – The redefinition of hypertension as 130/80 mm Hg or higher introduced in the current American College of Cardiology/American Heart Association hypertension management guidelines has generated considerable controversy. Often overlooked, however, has been another major innovation included in the 2017 guidelines: the rise in the status of out-of-office 24-hour ambulatory blood pressure monitoring and home blood pressure self-measurement to a class I, level of evidence A recommendation, Andrew M. Kates, MD, observed at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
It’s a guideline he strongly endorses.
“We do a lot of this. It can be a challenge to get 24-hour ambulatory blood pressure monitoring covered by payers, so said Dr. Kates, professor of medicine and director of the cardiology fellowship program at Washington University, St. Louis.
He explained that one of the four key questions the guideline committee was tasked with answering at the outset of deliberations was this: What’s the evidence base for self-directed out-of-office blood pressure monitoring? Based on the panel’s systematic review of the literature, this practice wound up receiving the strongest possible class Ia recommendation, specifically for confirming the diagnosis of hypertension and for titration of antihypertensive medications. Moreover, the guidelines also endorsed home blood pressure monitoring for the detection of white-coat hypertension, this time as a Class IIa recommendation, as well as for identification of patients with masked hypertension, with class IIb status (Circulation. 2018 Oct 23;138[17]:e484-594).
The 2017 ACC/AHA guidelines include a detailed checklist for obtaining accurate measurements of office blood pressure. The suggestions include having the patient sit relaxed in a chair with both feet on the floor for at least 5 minutes before taking the measurement, no coffee or exercise for 30 minutes beforehand, empty the bladder, no talking, no clothing over the arm, and other recommendations. Many busy clinicians roll their eyes at the impracticality of doing all this on a routine basis.
“I don’t want to take an audience survey, but I’ll say that even in our office we are not successful in doing this. Patients run up the stairs to the office after dealing with traffic and the parking garage, they’re late for their appointment, in winter they’re wearing a sweater and don’t want to take it off. These are things we don’t do well, and they’re low-hanging fruit where we could do better,” Dr. Kates commented.
The challenges inherent in performing by-the-book office blood pressure measurement reinforce the importance of home self-monitoring of blood pressure in what is hopefully a more stress-free environment.
“We can give patients specific guidance about checking their blood pressure an hour after taking their medications, sitting for 5 minutes, and checking the pressures on a bare arm and not with the sleeve rolled up,” he noted.
The guidelines recommend using home blood pressure monitoring or ambulatory monitoring to detect white-coat hypertension in patients with an office blood pressure of 130/80 mm Hg or more, but less than 160/100 mm Hg, after a 3-month trial of lifestyle modification. If the home blood pressure is less than 130/80 mm Hg, that’s evidence of white-coat hypertension, for which the recommended treatment consists of continued lifestyle modification plus periodic monitoring of out-of-office blood pressures in order to promptly detect progression to hypertension. If, however, the out-of-office blood pressure is not less than 130/80 mm Hg, that’s hypertension, and the guidelines recommend starting dual-agent antihypertensive drug therapy while continuing lifestyle modification.
A confusing array of definitions of hypertension are now in use by various medical societies. While the 2017 ACC/AHA hypertension guidelines define hypertension as office blood pressure of 130/80 mm Hg or more, the 2018 European Society of Cardiology/European Society of Hypertension guidelines use a threshold of 140/90 mm Hg or more. Joint American Academy of Family Physicians/American College of Physicians guidelines recommend a treatment target of less than 150 mm Hg in hypertensive patients aged 60 years or older. And at the other end of the spectrum, the SPRINT trial showed a significant cardiovascular benefit for intensive treatment of hypertension to a target systolic blood pressure below 120 mm Hg, rather than less than 140 mm Hg (N Engl J Med. 2015 Nov 26;373[22]:2103-16).
Dr. Kates believes the debate over the “right” treatment target misses the central point, which is that hypertension is staggeringly undertreated. Indeed, the Centers for Disease Control and Prevention estimates only one in four adults with hypertension have their disease under control. That’s a disconcerting statistic given that hypertension accounts for more cardiovascular deaths than any other modifiable cardiovascular risk factor.
“There’s been some concern raised that maybe too much weight has been put on the SPRINT trial in making the ACC/AHA recommendations, but I think it’s helpful to understand that we vastly undertreat patients with hypertension. So I think that, rather than being so concerned that we’re going to be treating people to too low a target or we’re being overly aggressive, it should give us some pause to think about the fact that we’re ordinarily not being aggressive enough with many of our patients as it is,” the cardiologist said.
Dr. Kates reported having no financial conflicts regarding his presentation.
SNOWMASS, COLO. – The redefinition of hypertension as 130/80 mm Hg or higher introduced in the current American College of Cardiology/American Heart Association hypertension management guidelines has generated considerable controversy. Often overlooked, however, has been another major innovation included in the 2017 guidelines: the rise in the status of out-of-office 24-hour ambulatory blood pressure monitoring and home blood pressure self-measurement to a class I, level of evidence A recommendation, Andrew M. Kates, MD, observed at the annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
It’s a guideline he strongly endorses.
“We do a lot of this. It can be a challenge to get 24-hour ambulatory blood pressure monitoring covered by payers, so said Dr. Kates, professor of medicine and director of the cardiology fellowship program at Washington University, St. Louis.
He explained that one of the four key questions the guideline committee was tasked with answering at the outset of deliberations was this: What’s the evidence base for self-directed out-of-office blood pressure monitoring? Based on the panel’s systematic review of the literature, this practice wound up receiving the strongest possible class Ia recommendation, specifically for confirming the diagnosis of hypertension and for titration of antihypertensive medications. Moreover, the guidelines also endorsed home blood pressure monitoring for the detection of white-coat hypertension, this time as a Class IIa recommendation, as well as for identification of patients with masked hypertension, with class IIb status (Circulation. 2018 Oct 23;138[17]:e484-594).
The 2017 ACC/AHA guidelines include a detailed checklist for obtaining accurate measurements of office blood pressure. The suggestions include having the patient sit relaxed in a chair with both feet on the floor for at least 5 minutes before taking the measurement, no coffee or exercise for 30 minutes beforehand, empty the bladder, no talking, no clothing over the arm, and other recommendations. Many busy clinicians roll their eyes at the impracticality of doing all this on a routine basis.
“I don’t want to take an audience survey, but I’ll say that even in our office we are not successful in doing this. Patients run up the stairs to the office after dealing with traffic and the parking garage, they’re late for their appointment, in winter they’re wearing a sweater and don’t want to take it off. These are things we don’t do well, and they’re low-hanging fruit where we could do better,” Dr. Kates commented.
The challenges inherent in performing by-the-book office blood pressure measurement reinforce the importance of home self-monitoring of blood pressure in what is hopefully a more stress-free environment.
“We can give patients specific guidance about checking their blood pressure an hour after taking their medications, sitting for 5 minutes, and checking the pressures on a bare arm and not with the sleeve rolled up,” he noted.
The guidelines recommend using home blood pressure monitoring or ambulatory monitoring to detect white-coat hypertension in patients with an office blood pressure of 130/80 mm Hg or more, but less than 160/100 mm Hg, after a 3-month trial of lifestyle modification. If the home blood pressure is less than 130/80 mm Hg, that’s evidence of white-coat hypertension, for which the recommended treatment consists of continued lifestyle modification plus periodic monitoring of out-of-office blood pressures in order to promptly detect progression to hypertension. If, however, the out-of-office blood pressure is not less than 130/80 mm Hg, that’s hypertension, and the guidelines recommend starting dual-agent antihypertensive drug therapy while continuing lifestyle modification.
A confusing array of definitions of hypertension are now in use by various medical societies. While the 2017 ACC/AHA hypertension guidelines define hypertension as office blood pressure of 130/80 mm Hg or more, the 2018 European Society of Cardiology/European Society of Hypertension guidelines use a threshold of 140/90 mm Hg or more. Joint American Academy of Family Physicians/American College of Physicians guidelines recommend a treatment target of less than 150 mm Hg in hypertensive patients aged 60 years or older. And at the other end of the spectrum, the SPRINT trial showed a significant cardiovascular benefit for intensive treatment of hypertension to a target systolic blood pressure below 120 mm Hg, rather than less than 140 mm Hg (N Engl J Med. 2015 Nov 26;373[22]:2103-16).
Dr. Kates believes the debate over the “right” treatment target misses the central point, which is that hypertension is staggeringly undertreated. Indeed, the Centers for Disease Control and Prevention estimates only one in four adults with hypertension have their disease under control. That’s a disconcerting statistic given that hypertension accounts for more cardiovascular deaths than any other modifiable cardiovascular risk factor.
“There’s been some concern raised that maybe too much weight has been put on the SPRINT trial in making the ACC/AHA recommendations, but I think it’s helpful to understand that we vastly undertreat patients with hypertension. So I think that, rather than being so concerned that we’re going to be treating people to too low a target or we’re being overly aggressive, it should give us some pause to think about the fact that we’re ordinarily not being aggressive enough with many of our patients as it is,” the cardiologist said.
Dr. Kates reported having no financial conflicts regarding his presentation.
EXPERT ANALYSIS FROM ACC SNOWMASS 2020
Pembrolizumab-Induced Lobular Panniculitis in the Setting of Metastatic Melanoma
To the Editor:
Pembrolizumab is an anti–programmed death receptor 1 humanized monoclonal antibody used for treating advanced or metastatic melanoma.1 It is associated with several immune-related adverse events because it blocks a T-cell receptor checkpoint.2 The most common dermatologic immune-related adverse event seen with anti–programmed death receptor 1 medications is a nonspecific morbilliform rash, usually seen after the second treatment cycle; however, pruritus, vitiligo, bullous disorders, and lichenoid reactions also have been reported.3 We report a case of pembrolizumab-induced, self-limited lobular panniculitis in a patient with metastatic melanoma.
A 37-year-old woman with malignant melanoma presented with tender, erythematous, subcutaneous nodules on the hips and legs of 2 weeks’ duration (Figure 1). Twelve years prior to the current presentation, she was diagnosed with metastases to the cecum, lung, and brain. A review of systems was otherwise negative. She had been receiving pembrolizumab infusions (2 mg/kg every 3 weeks) for the last 2.7 years as second-line therapy after previously undergoing chemotherapy, radiation, and resection. She was not taking oral contraceptives or other hormone-based medications and did not report any new medications.
Laboratory testing was negative for infectious processes including Lyme disease, tuberculosis, and Streptococcus due to recent upper respiratory infection. Punch biopsy of a left shin lesion revealed a lobular panniculitis with lymphohistiocytic inflammation, a focal lymphocytic vasculitis, and small granulomas (Figure 2). Periodic acid–Schiff, Gram, and acid-fast bacilli stains were negative. After ruling out alternative causes, the etiology of the panniculitis was deemed to be a pembrolizumab side effect. The patient was treated conservatively with ibuprofen; pembrolizumab was not discontinued. Two weeks later, the panniculitis had resolved without additional treatment. She remains on pembrolizumab and is doing well.
Panniculitis is known to be associated with certain BRAF inhibitors used for the treatment of melanoma positive for the BRAF V600E mutation, including vemurafenib and dabrafenib.4,5 Reports of panniculitis in the setting of pembrolizumab are limited and are seen within the larger context of sarcoidosis. One patient on pembrolizumab for metastatic melanoma developed granulomatous lobular panniculitis with oligoarthritis, high fever, and hilar/mediastinal adenopathy, consistent with pembrolizumab-induced sarcoidosis. It developed after her second pembrolizumab infusion and resolved with prednisone and temporary pembrolizumab cessation.6 In another case, pembrolizumab triggered a flare of sarcoidosis with similar granulomatous subcutaneous nodules in a patient with stage IV lymphoma who was previously diagnosed with sarcoidosis but lacked cutaneous manifestations. The lesions resolved with prednisone therapy.7
Chest computed tomography was normal in our patient, and she reported no systemic symptoms. Additional laboratory studies to evaluate for sarcoidosis were not obtained. Furthermore, the lesions quickly resolved despite continued use of pembrolizumab. We report this case to highlight that pembrolizumab may induce an isolated, self-limited lobular panniculitis years after medication initiation.
- Poole RM. Pembrolizumab: first global approval. Drugs. 2014;74:1973-1981.
- Michot JM, Bigenwald C, Champiat S, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer. 2016;54:139-148.
- Naidoo J, Page DB, Li BT, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Ann Oncol. 2016;27:1362.
- Boussemart L, Routier E, Mateus C, et al. Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: a study of 42 patients. Ann Oncol. 2013;24:1691-1697.
- Ramani NS, Curry JL, Kapil J, et al. Panniculitis with necrotizing granulomata in a patient on BRAF inhibitor (dabrafenib) therapy for metastatic melanoma. Am J Dermatopathol. 2015;37:E96-E99.
- Burillo-Martinez S, Morales-Raya C, Prieto-Barrios M, et al. Pembrolizumab-induced extensive panniculitis and nevus regression: two novel cutaneous manifestations of the post-immunotherapy granulomatous reactions spectrum. JAMA Dermatol. 2017;153:721-722.
- Cotliar J, Querfeld C, Boswell WJ, et al. Pembrolizumab-associated sarcoidosis. JAAD Case Rep. 2016;2:290-293.
To the Editor:
Pembrolizumab is an anti–programmed death receptor 1 humanized monoclonal antibody used for treating advanced or metastatic melanoma.1 It is associated with several immune-related adverse events because it blocks a T-cell receptor checkpoint.2 The most common dermatologic immune-related adverse event seen with anti–programmed death receptor 1 medications is a nonspecific morbilliform rash, usually seen after the second treatment cycle; however, pruritus, vitiligo, bullous disorders, and lichenoid reactions also have been reported.3 We report a case of pembrolizumab-induced, self-limited lobular panniculitis in a patient with metastatic melanoma.
A 37-year-old woman with malignant melanoma presented with tender, erythematous, subcutaneous nodules on the hips and legs of 2 weeks’ duration (Figure 1). Twelve years prior to the current presentation, she was diagnosed with metastases to the cecum, lung, and brain. A review of systems was otherwise negative. She had been receiving pembrolizumab infusions (2 mg/kg every 3 weeks) for the last 2.7 years as second-line therapy after previously undergoing chemotherapy, radiation, and resection. She was not taking oral contraceptives or other hormone-based medications and did not report any new medications.
Laboratory testing was negative for infectious processes including Lyme disease, tuberculosis, and Streptococcus due to recent upper respiratory infection. Punch biopsy of a left shin lesion revealed a lobular panniculitis with lymphohistiocytic inflammation, a focal lymphocytic vasculitis, and small granulomas (Figure 2). Periodic acid–Schiff, Gram, and acid-fast bacilli stains were negative. After ruling out alternative causes, the etiology of the panniculitis was deemed to be a pembrolizumab side effect. The patient was treated conservatively with ibuprofen; pembrolizumab was not discontinued. Two weeks later, the panniculitis had resolved without additional treatment. She remains on pembrolizumab and is doing well.
Panniculitis is known to be associated with certain BRAF inhibitors used for the treatment of melanoma positive for the BRAF V600E mutation, including vemurafenib and dabrafenib.4,5 Reports of panniculitis in the setting of pembrolizumab are limited and are seen within the larger context of sarcoidosis. One patient on pembrolizumab for metastatic melanoma developed granulomatous lobular panniculitis with oligoarthritis, high fever, and hilar/mediastinal adenopathy, consistent with pembrolizumab-induced sarcoidosis. It developed after her second pembrolizumab infusion and resolved with prednisone and temporary pembrolizumab cessation.6 In another case, pembrolizumab triggered a flare of sarcoidosis with similar granulomatous subcutaneous nodules in a patient with stage IV lymphoma who was previously diagnosed with sarcoidosis but lacked cutaneous manifestations. The lesions resolved with prednisone therapy.7
Chest computed tomography was normal in our patient, and she reported no systemic symptoms. Additional laboratory studies to evaluate for sarcoidosis were not obtained. Furthermore, the lesions quickly resolved despite continued use of pembrolizumab. We report this case to highlight that pembrolizumab may induce an isolated, self-limited lobular panniculitis years after medication initiation.
To the Editor:
Pembrolizumab is an anti–programmed death receptor 1 humanized monoclonal antibody used for treating advanced or metastatic melanoma.1 It is associated with several immune-related adverse events because it blocks a T-cell receptor checkpoint.2 The most common dermatologic immune-related adverse event seen with anti–programmed death receptor 1 medications is a nonspecific morbilliform rash, usually seen after the second treatment cycle; however, pruritus, vitiligo, bullous disorders, and lichenoid reactions also have been reported.3 We report a case of pembrolizumab-induced, self-limited lobular panniculitis in a patient with metastatic melanoma.
A 37-year-old woman with malignant melanoma presented with tender, erythematous, subcutaneous nodules on the hips and legs of 2 weeks’ duration (Figure 1). Twelve years prior to the current presentation, she was diagnosed with metastases to the cecum, lung, and brain. A review of systems was otherwise negative. She had been receiving pembrolizumab infusions (2 mg/kg every 3 weeks) for the last 2.7 years as second-line therapy after previously undergoing chemotherapy, radiation, and resection. She was not taking oral contraceptives or other hormone-based medications and did not report any new medications.
Laboratory testing was negative for infectious processes including Lyme disease, tuberculosis, and Streptococcus due to recent upper respiratory infection. Punch biopsy of a left shin lesion revealed a lobular panniculitis with lymphohistiocytic inflammation, a focal lymphocytic vasculitis, and small granulomas (Figure 2). Periodic acid–Schiff, Gram, and acid-fast bacilli stains were negative. After ruling out alternative causes, the etiology of the panniculitis was deemed to be a pembrolizumab side effect. The patient was treated conservatively with ibuprofen; pembrolizumab was not discontinued. Two weeks later, the panniculitis had resolved without additional treatment. She remains on pembrolizumab and is doing well.
Panniculitis is known to be associated with certain BRAF inhibitors used for the treatment of melanoma positive for the BRAF V600E mutation, including vemurafenib and dabrafenib.4,5 Reports of panniculitis in the setting of pembrolizumab are limited and are seen within the larger context of sarcoidosis. One patient on pembrolizumab for metastatic melanoma developed granulomatous lobular panniculitis with oligoarthritis, high fever, and hilar/mediastinal adenopathy, consistent with pembrolizumab-induced sarcoidosis. It developed after her second pembrolizumab infusion and resolved with prednisone and temporary pembrolizumab cessation.6 In another case, pembrolizumab triggered a flare of sarcoidosis with similar granulomatous subcutaneous nodules in a patient with stage IV lymphoma who was previously diagnosed with sarcoidosis but lacked cutaneous manifestations. The lesions resolved with prednisone therapy.7
Chest computed tomography was normal in our patient, and she reported no systemic symptoms. Additional laboratory studies to evaluate for sarcoidosis were not obtained. Furthermore, the lesions quickly resolved despite continued use of pembrolizumab. We report this case to highlight that pembrolizumab may induce an isolated, self-limited lobular panniculitis years after medication initiation.
- Poole RM. Pembrolizumab: first global approval. Drugs. 2014;74:1973-1981.
- Michot JM, Bigenwald C, Champiat S, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer. 2016;54:139-148.
- Naidoo J, Page DB, Li BT, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Ann Oncol. 2016;27:1362.
- Boussemart L, Routier E, Mateus C, et al. Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: a study of 42 patients. Ann Oncol. 2013;24:1691-1697.
- Ramani NS, Curry JL, Kapil J, et al. Panniculitis with necrotizing granulomata in a patient on BRAF inhibitor (dabrafenib) therapy for metastatic melanoma. Am J Dermatopathol. 2015;37:E96-E99.
- Burillo-Martinez S, Morales-Raya C, Prieto-Barrios M, et al. Pembrolizumab-induced extensive panniculitis and nevus regression: two novel cutaneous manifestations of the post-immunotherapy granulomatous reactions spectrum. JAMA Dermatol. 2017;153:721-722.
- Cotliar J, Querfeld C, Boswell WJ, et al. Pembrolizumab-associated sarcoidosis. JAAD Case Rep. 2016;2:290-293.
- Poole RM. Pembrolizumab: first global approval. Drugs. 2014;74:1973-1981.
- Michot JM, Bigenwald C, Champiat S, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer. 2016;54:139-148.
- Naidoo J, Page DB, Li BT, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Ann Oncol. 2016;27:1362.
- Boussemart L, Routier E, Mateus C, et al. Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: a study of 42 patients. Ann Oncol. 2013;24:1691-1697.
- Ramani NS, Curry JL, Kapil J, et al. Panniculitis with necrotizing granulomata in a patient on BRAF inhibitor (dabrafenib) therapy for metastatic melanoma. Am J Dermatopathol. 2015;37:E96-E99.
- Burillo-Martinez S, Morales-Raya C, Prieto-Barrios M, et al. Pembrolizumab-induced extensive panniculitis and nevus regression: two novel cutaneous manifestations of the post-immunotherapy granulomatous reactions spectrum. JAMA Dermatol. 2017;153:721-722.
- Cotliar J, Querfeld C, Boswell WJ, et al. Pembrolizumab-associated sarcoidosis. JAAD Case Rep. 2016;2:290-293.
Practice Points
- Pembrolizumab may cause lobular panniculitis years after treatment initiation.
- Pembrolizumab-induced lobular panniculitis may self-resolve without discontinuing the medication.
Metastatic Melanoma Mimicking Eruptive Keratoacanthomas
To the Editor:
Melanoma is the third most common skin cancer. It is estimated that 18% of melanoma patients will develop skin metastases, with skin being the first site of involvement in 56% of cases.1 Of all cancers, it is estimated that 5% will develop skin metastases. It is the presenting sign in nearly 1% of visceral cancers.2 Melanoma and nonmelanoma metastases can have sundry presentations. We present a case of metastatic melanoma with multiple keratoacanthoma (KA)–like skin lesions in a patient with a known history of nonmelanoma skin cancer (NMSC) as well as melanoma.
A 76-year-old man with a history of pT2aNXMX melanoma on the left upper back presented for a routine 3-month follow-up and reported several new asymptomatic bumps on the chest, back, and right upper extremity within the last 2 weeks. The melanoma was removed via wide local excision 2 years prior at an outside facility with a Breslow depth of 1.05 mm and a negative sentinel lymph node biopsy. The mitotic rate or ulceration status was unknown. He also had a history of several NMSCs, as well as a medical history of coronary artery disease, myocardial infarction, and ventricular tachycardia with cardiac defibrillator placement. Physical examination revealed 5 pink, volcano-shaped nodules with central keratotic plugs on the upper back (Figure 1), chest, and right upper extremity, in addition to 1 pink pearly nodule on the right side of the chest. The history and appearance of the lesions were suspicious for eruptive KAs. There was no evidence of cancer recurrence at the prior melanoma and NMSC sites.
A deep shave skin biopsy was performed at all 6 sites. Histopathology showed a diffuse dermal infiltrate of elongated nests of melanocytes and nonnested melanocytes. Marked cytologic atypia and ulceration were present. Minimal connection to the overlying epidermis and a lack of junctional nests was noted. Immunohistochemical studies revealed scattered positivity for Melan-A and negative staining for AE1, AE3, cytokeratin 5, and cytokeratin 6 at all 6 sites (Figure 2). A subsequent metastatic workup showed widespread metastatic disease in the liver, bone, lung, and inferior vena cava. Computed tomography of the head was unremarkable. Magnetic resonance imaging of the brain was not performed due to the cardiac defibrillator. The patient’s lactate dehydrogenase level showed a mild increase compared to 2 months prior to the metastatic melanoma diagnosis (144 U/L vs 207 U/L [reference range, 100–200 U/L]).
The patient had no systemic symptoms at follow-up 5 weeks later. He was already evaluated by an oncologist and received his first dose of ipilimumab. He was BRAF-mutation negative. He had developed 2 new skin metastases. Five of 6 initially biopsied metastases returned and were growing; they were tender and friable with intermittent bleeding. He was subsequently referred to surgical oncology for excision of symptomatic nodules as palliative care.
Although melanoma is well known to metastasize years and even decades later, KA-like lesions have not been reported as manifestations of metastatic melanoma.4,5 Our patient likely had a primary amelanotic melanoma, as the medical records from the outside facility stated that basal cell carcinoma was ruled out via biopsy. The amelanotic nature of the primary melanoma may have influenced the amelanotic appearance of the metastases. Our patient had no signs of immunosuppression that could have contributed to the sudden skin metastases.
Depending on the subtype of cutaneous metastases (eg, satellitosis, in-transit disease, distant cutaneous metastases), the location prevalence of the primary melanoma varies. In a study of 4865 melanoma patients who were diagnosed and followed prospectively over a 30-year period, skin metastases were mostly locoregional and presentation on the leg and foot were disproportionate.1 In contrast, the trunk was overrepresented for distant metastases. Distant metastases also were more associated with concurrent metastases to the viscera.1 Accordingly, a patient’s prognosis and management will differ depending on the subtype of cutaneous metastases.
Eruptive or multiple KAs classically have been associated with the Grzybowski variant, the Ferguson-Smith familial variant, and Muir-Torre syndrome. It was reported as a paraneoplastic syndrome associated with colon cancer, ovarian cancer, and once with myelodysplastic syndrome.3 Keratoacanthomas are being classified as well-differentiated squamous cell carcinomas and have metastatic potential. A biopsy is recommended to diagnose KAs as opposed to historically being monitored for resolution. A skin biopsy is the standard of care in management of KAs.
In addition to being associated with Muir-Torre syndrome and classified as a paraneoplastic syndrome,3 eruptive KAs can occur following skin resurfacing for actinic damage, fractional photothermolysis, cryotherapy, Jessner peels, and trichloroacetic acid peels.6 A couple other uncommon settings include a case report of an arc welder with job-associated radiation and multiple reports of tattoo-induced KAs.7,8 There is the new increasingly common association of squamous cell carcinomas with BRAF inhibitors, such as vemurafenib, for metastatic melanoma.9
In a 2012 review article on cutaneous metastases, Riahi and Cohen10 found 8 cases of cutaneous metastases presenting as KA-like lesions; none were metastatic melanoma. All were solitary lesions, not multiple lesions, as in our patient. The sources were lung (3 cases), breast, esophagus, chondrosarcoma, bronchial, and mesothelioma. The most common location was the upper lip. Additionally, similar to our patient, they behaved clinically as KAs with rapid growth and keratotic plugs and were asymptomatic.10
Metastatic melanoma may mimic many other cutaneous processes that may make the diagnosis more difficult. Our case indicates that cutaneous metastases may mimic KAs. Although multiple KA-like lesions can spontaneously occur, a paraneoplastic syndrome and other underlying etiologies should be considered.
- Savoia P, Fava P, Nardò T, et al. Skin metastases of malignant melanoma: a clinical and prognostic study. Melanoma Res. 2009;19:321-326.
- Lookingbill DP, Spangler N, Sexton FM. Skin involvement as the presenting sign of internal carcinoma. J Am Acad Dermatol. 1990;22:19-26.
- Behzad M, Michl C, Pfützner W. Multiple eruptive keratoacanthomas associated with myelodysplastic syndrome. J Dtsch Dermatol Ges. 2012;10:359-360.
- Cheung WL, Patel RR, Leonard A, et al. Amelanotic melanoma: a detailed morphologic analysis with clinicopathologic correlation of 75 cases. J Cutan Pathol. 2012;39:33-39.
- Ferrari A, Piccolo D, Fargnoli MC, et al. Cutaneous amelanotic melanoma metastasis and dermatofibromas showing a dotted vascular pattern. Acta Dermato Venereologica. 2004;84:164-165.
- Mohr B, Fernandez MP, Krejci-Manwaring J. Eruptive keratoacanthoma after Jessner’s and trichloroacetic acid peel for actinic keratosis. Dermatol Surg. 2013;39:331-333.
- Wolfe CM, Green WH, Cognetta AB, et al. Multiple squamous cell carcinomas and eruptive keratoacanthomas in an arc welder. Dermatol Surg. 2013;39:328-330.
- Kluger N, Phan A, Debarbieux S, et al. Skin cancers arising in tattoos: coincidental or not? Dermatology. 2008;217:219-221.
- Mays R, Curry J, Kim K, et al. Eruptive squamous cell carcinomas after vemurafenib therapy. J Cutan Med Surg. 2013;17:419-422.
- Riahi RR, Cohen PR. Clinical manifestations of cutaneous metastases: a review with special emphasis on cutaneous metastases mimicking keratoacanthoma. Am J Clin Dermatol. 2012;13:103-112.
To the Editor:
Melanoma is the third most common skin cancer. It is estimated that 18% of melanoma patients will develop skin metastases, with skin being the first site of involvement in 56% of cases.1 Of all cancers, it is estimated that 5% will develop skin metastases. It is the presenting sign in nearly 1% of visceral cancers.2 Melanoma and nonmelanoma metastases can have sundry presentations. We present a case of metastatic melanoma with multiple keratoacanthoma (KA)–like skin lesions in a patient with a known history of nonmelanoma skin cancer (NMSC) as well as melanoma.
A 76-year-old man with a history of pT2aNXMX melanoma on the left upper back presented for a routine 3-month follow-up and reported several new asymptomatic bumps on the chest, back, and right upper extremity within the last 2 weeks. The melanoma was removed via wide local excision 2 years prior at an outside facility with a Breslow depth of 1.05 mm and a negative sentinel lymph node biopsy. The mitotic rate or ulceration status was unknown. He also had a history of several NMSCs, as well as a medical history of coronary artery disease, myocardial infarction, and ventricular tachycardia with cardiac defibrillator placement. Physical examination revealed 5 pink, volcano-shaped nodules with central keratotic plugs on the upper back (Figure 1), chest, and right upper extremity, in addition to 1 pink pearly nodule on the right side of the chest. The history and appearance of the lesions were suspicious for eruptive KAs. There was no evidence of cancer recurrence at the prior melanoma and NMSC sites.
A deep shave skin biopsy was performed at all 6 sites. Histopathology showed a diffuse dermal infiltrate of elongated nests of melanocytes and nonnested melanocytes. Marked cytologic atypia and ulceration were present. Minimal connection to the overlying epidermis and a lack of junctional nests was noted. Immunohistochemical studies revealed scattered positivity for Melan-A and negative staining for AE1, AE3, cytokeratin 5, and cytokeratin 6 at all 6 sites (Figure 2). A subsequent metastatic workup showed widespread metastatic disease in the liver, bone, lung, and inferior vena cava. Computed tomography of the head was unremarkable. Magnetic resonance imaging of the brain was not performed due to the cardiac defibrillator. The patient’s lactate dehydrogenase level showed a mild increase compared to 2 months prior to the metastatic melanoma diagnosis (144 U/L vs 207 U/L [reference range, 100–200 U/L]).
The patient had no systemic symptoms at follow-up 5 weeks later. He was already evaluated by an oncologist and received his first dose of ipilimumab. He was BRAF-mutation negative. He had developed 2 new skin metastases. Five of 6 initially biopsied metastases returned and were growing; they were tender and friable with intermittent bleeding. He was subsequently referred to surgical oncology for excision of symptomatic nodules as palliative care.
Although melanoma is well known to metastasize years and even decades later, KA-like lesions have not been reported as manifestations of metastatic melanoma.4,5 Our patient likely had a primary amelanotic melanoma, as the medical records from the outside facility stated that basal cell carcinoma was ruled out via biopsy. The amelanotic nature of the primary melanoma may have influenced the amelanotic appearance of the metastases. Our patient had no signs of immunosuppression that could have contributed to the sudden skin metastases.
Depending on the subtype of cutaneous metastases (eg, satellitosis, in-transit disease, distant cutaneous metastases), the location prevalence of the primary melanoma varies. In a study of 4865 melanoma patients who were diagnosed and followed prospectively over a 30-year period, skin metastases were mostly locoregional and presentation on the leg and foot were disproportionate.1 In contrast, the trunk was overrepresented for distant metastases. Distant metastases also were more associated with concurrent metastases to the viscera.1 Accordingly, a patient’s prognosis and management will differ depending on the subtype of cutaneous metastases.
Eruptive or multiple KAs classically have been associated with the Grzybowski variant, the Ferguson-Smith familial variant, and Muir-Torre syndrome. It was reported as a paraneoplastic syndrome associated with colon cancer, ovarian cancer, and once with myelodysplastic syndrome.3 Keratoacanthomas are being classified as well-differentiated squamous cell carcinomas and have metastatic potential. A biopsy is recommended to diagnose KAs as opposed to historically being monitored for resolution. A skin biopsy is the standard of care in management of KAs.
In addition to being associated with Muir-Torre syndrome and classified as a paraneoplastic syndrome,3 eruptive KAs can occur following skin resurfacing for actinic damage, fractional photothermolysis, cryotherapy, Jessner peels, and trichloroacetic acid peels.6 A couple other uncommon settings include a case report of an arc welder with job-associated radiation and multiple reports of tattoo-induced KAs.7,8 There is the new increasingly common association of squamous cell carcinomas with BRAF inhibitors, such as vemurafenib, for metastatic melanoma.9
In a 2012 review article on cutaneous metastases, Riahi and Cohen10 found 8 cases of cutaneous metastases presenting as KA-like lesions; none were metastatic melanoma. All were solitary lesions, not multiple lesions, as in our patient. The sources were lung (3 cases), breast, esophagus, chondrosarcoma, bronchial, and mesothelioma. The most common location was the upper lip. Additionally, similar to our patient, they behaved clinically as KAs with rapid growth and keratotic plugs and were asymptomatic.10
Metastatic melanoma may mimic many other cutaneous processes that may make the diagnosis more difficult. Our case indicates that cutaneous metastases may mimic KAs. Although multiple KA-like lesions can spontaneously occur, a paraneoplastic syndrome and other underlying etiologies should be considered.
To the Editor:
Melanoma is the third most common skin cancer. It is estimated that 18% of melanoma patients will develop skin metastases, with skin being the first site of involvement in 56% of cases.1 Of all cancers, it is estimated that 5% will develop skin metastases. It is the presenting sign in nearly 1% of visceral cancers.2 Melanoma and nonmelanoma metastases can have sundry presentations. We present a case of metastatic melanoma with multiple keratoacanthoma (KA)–like skin lesions in a patient with a known history of nonmelanoma skin cancer (NMSC) as well as melanoma.
A 76-year-old man with a history of pT2aNXMX melanoma on the left upper back presented for a routine 3-month follow-up and reported several new asymptomatic bumps on the chest, back, and right upper extremity within the last 2 weeks. The melanoma was removed via wide local excision 2 years prior at an outside facility with a Breslow depth of 1.05 mm and a negative sentinel lymph node biopsy. The mitotic rate or ulceration status was unknown. He also had a history of several NMSCs, as well as a medical history of coronary artery disease, myocardial infarction, and ventricular tachycardia with cardiac defibrillator placement. Physical examination revealed 5 pink, volcano-shaped nodules with central keratotic plugs on the upper back (Figure 1), chest, and right upper extremity, in addition to 1 pink pearly nodule on the right side of the chest. The history and appearance of the lesions were suspicious for eruptive KAs. There was no evidence of cancer recurrence at the prior melanoma and NMSC sites.
A deep shave skin biopsy was performed at all 6 sites. Histopathology showed a diffuse dermal infiltrate of elongated nests of melanocytes and nonnested melanocytes. Marked cytologic atypia and ulceration were present. Minimal connection to the overlying epidermis and a lack of junctional nests was noted. Immunohistochemical studies revealed scattered positivity for Melan-A and negative staining for AE1, AE3, cytokeratin 5, and cytokeratin 6 at all 6 sites (Figure 2). A subsequent metastatic workup showed widespread metastatic disease in the liver, bone, lung, and inferior vena cava. Computed tomography of the head was unremarkable. Magnetic resonance imaging of the brain was not performed due to the cardiac defibrillator. The patient’s lactate dehydrogenase level showed a mild increase compared to 2 months prior to the metastatic melanoma diagnosis (144 U/L vs 207 U/L [reference range, 100–200 U/L]).
The patient had no systemic symptoms at follow-up 5 weeks later. He was already evaluated by an oncologist and received his first dose of ipilimumab. He was BRAF-mutation negative. He had developed 2 new skin metastases. Five of 6 initially biopsied metastases returned and were growing; they were tender and friable with intermittent bleeding. He was subsequently referred to surgical oncology for excision of symptomatic nodules as palliative care.
Although melanoma is well known to metastasize years and even decades later, KA-like lesions have not been reported as manifestations of metastatic melanoma.4,5 Our patient likely had a primary amelanotic melanoma, as the medical records from the outside facility stated that basal cell carcinoma was ruled out via biopsy. The amelanotic nature of the primary melanoma may have influenced the amelanotic appearance of the metastases. Our patient had no signs of immunosuppression that could have contributed to the sudden skin metastases.
Depending on the subtype of cutaneous metastases (eg, satellitosis, in-transit disease, distant cutaneous metastases), the location prevalence of the primary melanoma varies. In a study of 4865 melanoma patients who were diagnosed and followed prospectively over a 30-year period, skin metastases were mostly locoregional and presentation on the leg and foot were disproportionate.1 In contrast, the trunk was overrepresented for distant metastases. Distant metastases also were more associated with concurrent metastases to the viscera.1 Accordingly, a patient’s prognosis and management will differ depending on the subtype of cutaneous metastases.
Eruptive or multiple KAs classically have been associated with the Grzybowski variant, the Ferguson-Smith familial variant, and Muir-Torre syndrome. It was reported as a paraneoplastic syndrome associated with colon cancer, ovarian cancer, and once with myelodysplastic syndrome.3 Keratoacanthomas are being classified as well-differentiated squamous cell carcinomas and have metastatic potential. A biopsy is recommended to diagnose KAs as opposed to historically being monitored for resolution. A skin biopsy is the standard of care in management of KAs.
In addition to being associated with Muir-Torre syndrome and classified as a paraneoplastic syndrome,3 eruptive KAs can occur following skin resurfacing for actinic damage, fractional photothermolysis, cryotherapy, Jessner peels, and trichloroacetic acid peels.6 A couple other uncommon settings include a case report of an arc welder with job-associated radiation and multiple reports of tattoo-induced KAs.7,8 There is the new increasingly common association of squamous cell carcinomas with BRAF inhibitors, such as vemurafenib, for metastatic melanoma.9
In a 2012 review article on cutaneous metastases, Riahi and Cohen10 found 8 cases of cutaneous metastases presenting as KA-like lesions; none were metastatic melanoma. All were solitary lesions, not multiple lesions, as in our patient. The sources were lung (3 cases), breast, esophagus, chondrosarcoma, bronchial, and mesothelioma. The most common location was the upper lip. Additionally, similar to our patient, they behaved clinically as KAs with rapid growth and keratotic plugs and were asymptomatic.10
Metastatic melanoma may mimic many other cutaneous processes that may make the diagnosis more difficult. Our case indicates that cutaneous metastases may mimic KAs. Although multiple KA-like lesions can spontaneously occur, a paraneoplastic syndrome and other underlying etiologies should be considered.
- Savoia P, Fava P, Nardò T, et al. Skin metastases of malignant melanoma: a clinical and prognostic study. Melanoma Res. 2009;19:321-326.
- Lookingbill DP, Spangler N, Sexton FM. Skin involvement as the presenting sign of internal carcinoma. J Am Acad Dermatol. 1990;22:19-26.
- Behzad M, Michl C, Pfützner W. Multiple eruptive keratoacanthomas associated with myelodysplastic syndrome. J Dtsch Dermatol Ges. 2012;10:359-360.
- Cheung WL, Patel RR, Leonard A, et al. Amelanotic melanoma: a detailed morphologic analysis with clinicopathologic correlation of 75 cases. J Cutan Pathol. 2012;39:33-39.
- Ferrari A, Piccolo D, Fargnoli MC, et al. Cutaneous amelanotic melanoma metastasis and dermatofibromas showing a dotted vascular pattern. Acta Dermato Venereologica. 2004;84:164-165.
- Mohr B, Fernandez MP, Krejci-Manwaring J. Eruptive keratoacanthoma after Jessner’s and trichloroacetic acid peel for actinic keratosis. Dermatol Surg. 2013;39:331-333.
- Wolfe CM, Green WH, Cognetta AB, et al. Multiple squamous cell carcinomas and eruptive keratoacanthomas in an arc welder. Dermatol Surg. 2013;39:328-330.
- Kluger N, Phan A, Debarbieux S, et al. Skin cancers arising in tattoos: coincidental or not? Dermatology. 2008;217:219-221.
- Mays R, Curry J, Kim K, et al. Eruptive squamous cell carcinomas after vemurafenib therapy. J Cutan Med Surg. 2013;17:419-422.
- Riahi RR, Cohen PR. Clinical manifestations of cutaneous metastases: a review with special emphasis on cutaneous metastases mimicking keratoacanthoma. Am J Clin Dermatol. 2012;13:103-112.
- Savoia P, Fava P, Nardò T, et al. Skin metastases of malignant melanoma: a clinical and prognostic study. Melanoma Res. 2009;19:321-326.
- Lookingbill DP, Spangler N, Sexton FM. Skin involvement as the presenting sign of internal carcinoma. J Am Acad Dermatol. 1990;22:19-26.
- Behzad M, Michl C, Pfützner W. Multiple eruptive keratoacanthomas associated with myelodysplastic syndrome. J Dtsch Dermatol Ges. 2012;10:359-360.
- Cheung WL, Patel RR, Leonard A, et al. Amelanotic melanoma: a detailed morphologic analysis with clinicopathologic correlation of 75 cases. J Cutan Pathol. 2012;39:33-39.
- Ferrari A, Piccolo D, Fargnoli MC, et al. Cutaneous amelanotic melanoma metastasis and dermatofibromas showing a dotted vascular pattern. Acta Dermato Venereologica. 2004;84:164-165.
- Mohr B, Fernandez MP, Krejci-Manwaring J. Eruptive keratoacanthoma after Jessner’s and trichloroacetic acid peel for actinic keratosis. Dermatol Surg. 2013;39:331-333.
- Wolfe CM, Green WH, Cognetta AB, et al. Multiple squamous cell carcinomas and eruptive keratoacanthomas in an arc welder. Dermatol Surg. 2013;39:328-330.
- Kluger N, Phan A, Debarbieux S, et al. Skin cancers arising in tattoos: coincidental or not? Dermatology. 2008;217:219-221.
- Mays R, Curry J, Kim K, et al. Eruptive squamous cell carcinomas after vemurafenib therapy. J Cutan Med Surg. 2013;17:419-422.
- Riahi RR, Cohen PR. Clinical manifestations of cutaneous metastases: a review with special emphasis on cutaneous metastases mimicking keratoacanthoma. Am J Clin Dermatol. 2012;13:103-112.
Practice Points
- Cutaneous metastatic melanoma can have variable clinical presentations.
- Patients with a history of melanoma should be monitored closely with a low threshold for biopsy of new skin lesions.