User login
Updates in Management and Timing of Dialysis in Acute Kidney Injury
Acute kidney injury (AKI) is a common complication in hospitalized patients, affecting one in five inpatients1,2 and more than half of patients in intensive care units (ICU).3 The incidence of AKI appears to be increasing over time.4 Potential contributing factors include an aging population, rising prevalence of comorbid conditions such as heart failure and chronic kidney disease (CKD), using nephrotoxic agents, and increasing complexity of surgical procedures.5,6 AKI during a hospital stay is associated with a two to 10-fold increased risk of inhospital mortality,1,2,7-10 longer hospital length of stay,7,10 higher risk for hospital readmissions,11 and higher healthcare costs.7 Patients who survive an episode of AKI have a higher risk for CKD and dialysis-dependence,9 even after an episode of reversible AKI.12 Despite its clinical importance, several areas of controversy remain regarding the management of AKI and, in particular, the optimal timing of renal replacement therapy (RRT) in patients with AKI. The purpose of this manuscript is to review the approaches to diagnosis and management of AKI in hospitalized patients. We also review recent evidence regarding the timing of dialysis in patients with AKI. This journal recently reviewed the differential diagnosis and diagnostic evaluation of AKI, which is not covered here.13
DEFINITION OF ACUTE KIDNEY INJURY
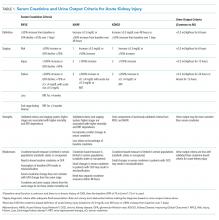
AKI refers to an acute change in kidney function characterized by an increase in serum creatinine and/or a reduction in urine output. It is a clinical syndrome caused by a broad range of etiologies and may be related to primary kidney pathology and/or systemic illness. Until 2004, there was no standard definition for AKI and over 30 different definitions were found in the literature, which resulted in wide variation in the reported incidence and outcomes of AKI and made it challenging to apply an evidence-based approach to patient care. In 2004, the Risk, Injury, Failure, Loss, and End-stage kidney disease (RIFLE)14 criteria for AKI were proposed, which were modified to the Acute Kidney Injury Network (AKIN)15 criteria in 2007 (Table 1). Multiple studies show that the RIFLE and AKIN criteria for AKI are associated with higher mortality1,2,8,10 and increased risk for requiring RRT.1,10
International clinical practice guidelines for AKI were released by Kidney Disease: Improving Global Outcomes (KDIGO) in 2012, which included a standardized definition of AKI that was adapted from the previously validated RIFLE and AKIN definitions.16 Patients are considered to have AKI when the serum creatinine rises by as little as 0.3 mg/dL. It is notable that when the baseline serum creatinine is high, there is more inherent variability in the serum creatinine measurement; thus, patients with CKD have a higher risk of being misclassified as having AKI.17 Although the KDIGO definition for AKI is commonly used in research settings, components of this definition have not been well validated, and it is not widely used in clinical practice. Other renal professional societies still recommend an individualized approach to the diagnosis of AKI, taking into account other factors such as trajectories in kidney function, fluid balance, electrolyte abnormalities, comorbid conditions, and clinical context.18,19 While we endorse the KDIGO approach to the categorization of AKI severity, in practice, a more patient-centered approach is generally required to guide the optimal approach to determining the etiology of AKI and guiding management.
GENERAL MANAGEMENT OF ACUTE KIDNEY INJURY
All patients with AKI should have close monitoring of their serum creatinine and urine output. Noninvasive diagnostic studies (urine microscopy, postvoid residual, and renal ultrasound) should be considered based on the clinical scenario. General management strategies include treatment of the reversible causes of AKI and optimization of volume status, hemodynamics, and nutritional status (Table 2).
Reversible Causes of Acute Kidney Injury
The first step in the treatment of AKI is to identify and treat readily reversible causes of AKI such as volume depletion, hypotension, infection, and urinary obstruction. Nephrotoxins should be avoided and all medications should be reviewed and adjusted for kidney function, particularly those that may affect mental status. Avoid opiates with noxious or active metabolites, including meperidine and morphine. Instead, hydromorphone, fentanyl, and methadone are preferred in patients with AKI. Other commonly used medications that require dose adjustment include gabapentin, baclofen, metoclopramide, H2 antagonists, many commonly prescribed antibiotics (penicillins, most cephalosporins, carbapenems, quinolones, and sulfa drugs), many hypoglycemic agents, and insulin. For patients on RRT, dosing is dependent on dialysis modality. Consultation with a hospital pharmacist is recommended when RRT modalities are initiated or changed.
Intravenous Fluids
Patients with AKI should have their volume status assessed and receive adequate resuscitation with intravenous fluids to promote renal perfusion. However, the optimal type and volume of fluid to give in AKI remains controversial. Colloid-containing solutions are theoretically confined to the intravascular space and should pose a lower risk for pulmonary edema compared with crystalloids. However, these solutions are costly, are not associated with any meaningful benefit,20-22 and may even be associated with potential harm.22-27
The most commonly used colloid worldwide is hydroxyethyl starch (HES). Its potential adverse effects include anaphylactoid reactions, coagulopathy, and AKI. HES is cleared by the kidneys and can cause osmotic nephrosis, a form of AKI characterized by vacuole formation and proximal renal tubular damage.28 Randomized controlled trials have shown an increased risk of AKI, RRT use, and mortality in critically ill patients who were resuscitated with HES.22,26,27 HES is not currently recommended in patients who are critically ill or have impaired kidney function and sepsis guidelines advise against its use.29
In the United States, albumin is the most common colloid-containing solution used for intravascular volume resuscitation. Albumin has been shown to be safe for volume resuscitation in critically ill patients,20 but there is no proven advantage to using albumin over saline with respect to mortality, length of hospital stay, duration of mechanical ventilation, duration of RRT, or number of organ systems failure.20,21 Furthermore, albumin may be harmful in certain patient populations. In patients with traumatic brain injury, albumin resuscitation is associated with higher mean intracranial pressures23 and long-term mortality.24 In a retrospective study of patients undergoing cardiac surgery, albumin administration was associated with more than twice the risk of AKI compared with crystalloids.25 In contrast, in patients with cirrhosis, intravenous albumin lowers the rate of AKI when administered in the setting of a large volume paracentesis30 or spontaneous bacterial peritonitis.31 Outside of these narrow settings, current evidence does not support the use of intravenous albumin to prevent AKI and we would not endorse the use of intravenous albumin as a part of the treatment paradigm for established AKI.
Many renal and critical care guidelines recommend initial fluid resuscitation with isotonic crystalloids except in specific circumstances (ie, hemorrhagic shock), with consideration of albumin in select cases (ie, severe sepsis or cirrhosis).16,18,19,29 That stated, the optimal type of crystalloid solution that should be used in resuscitation remains unclear. Because of its low cost, normal (0.9%) saline is the most commonly used solution, but it can result in hyperchloremic metabolic acidosis, which can cause renal vasoconstriction and may be associated with mortality in critically ill patients.32 A prospective study found that administration of chloride-liberal fluids (including normal saline) to critically ill patients was associated with nearly twice the risk of AKI and RRT use compared with chloride-restrictive fluids,33 but a subsequent trial found no difference in AKI or mortality among patients receiving saline versus a balanced crystalloid (Plasma-Lyte 148).34 A recent pair of large, randomized control trials compared outcomes in patients at a single center who were resuscitated with normal saline versus balanced crystalloid solutions (Lactated Ringer’s or Plasma-Lyte A).35,36 In critically ill patients, the use of balanced crystalloid solutions was associated with a lower risk of the composite outcome of mortality, new RRT, or persistent kidney impairment, but there were no differences in any of the individual components of the composite outcome.35 In noncritically ill patients, there were no differences in the number of hospital-free days based on the type of crystalloid solution used.36 In the absence of compelling evidence for using balanced crystalloid solutions, we continue to use normal saline for initial fluid resuscitation, but to avoid severe hyperchloremia and acidosis, we will consider switching to a balanced solution (Lactated Ringer’s, Plasma-Lyte, or Normosol) for large volume resuscitation (>2 L), particularly in critically ill patients.
Diuretics
As above, volume status is a key component in the management of patients with AKI. In patients with AKI and hypervolemia, loop diuretics are often given prior to the initiation of RRT. Loop diuretics act on the sodium-potassium-chloride cotransporters in the thick ascending limb of the loop of Henle to increase urinary losses of these ions and urine volume. Loop diuretics are dose-dependent, and often, higher doses are needed (eg, furosemide 100 mg intravenous dose) in patients with AKI, since the diuretic effect depends on the proximal tubular secretion of the drug into the urine. The role of diuretics in AKI is controversial and some observational data suggest an increased mortality risk with diuretic use in patients with AKI.37 In critically ill patients with acute lung injury, diuretic use improved survival, which was attributed to better control of volume overload.38 But, a meta-analysis of 11 randomized controlled trials failed to demonstrate that diuretics directly improved survival or recovery of AKI.39 Moreover, randomized controlled trials found that diuretics given to a patient with AKI requiring RRT did not improve recovery of kidney function.40,41 The KDIGO guidelines recommend that diuretics should not be routinely used for AKI except in the management of volume overload.16
Nutritional Targets in Acute Kidney Injury
Critically ill patients have high protein catabolic rates, which put them at increased risk for malnutrition, which in turn is associated with mortality. Patients who receive continuous RRT (CRRT) may lose 5-10 g of protein and 10-15 g of amino acids daily, and these patients may have protein requirements that are twice the usual recommended daily protein intake.16 But excess protein administration can result in high urea generation and azotemia unrelated to the patient’s kidney function. Blood urea nitrogen may also be disproportionately elevated in conditions where tubular reabsorption of urea is increased, such as in volume depletion, diuretic use, corticosteroid use, and gastrointestinal bleeding. Interpretation of blood urea nitrogen results must be made in the appropriate clinical context, with recognition that azotemia alone may not be a good surrogate marker of the patient’s underlying kidney function. We recommend dietary consultation in critically ill patients with AKI to ensure that adequate, but not excessive, protein is administered.
RENAL REPLACEMENT THERAPY IN ACUTE KIDNEY INJURY
In patients with AKI, RRT is initiated for control of volume overload, electrolyte abnormalities, acidemia, or uremic symptoms or complications that are refractory to medical management (Table 3). In a nonoliguric patient, fluid and electrolyte abnormalities can oftentimes be managed medically. Patients with oligoanuria (generally defined as urine output less than 400 mL/day or <20 mL/hour), however, require nephrology evaluation for consideration of RRT. Early nephrology consultation (within 48 hours of AKI diagnosis) may be associated with lower dialysis dependence and mortality in critically ill patients with AKI.42 The decision to initiate dialysis is individualized based on the patient’s comorbid conditions, urine output, and trajectory of kidney function.
Timing of Renal Replacement Therapy
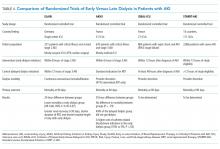
The optimal timing of dialysis initiation in patients with AKI is not known. Theoretically, earlier initiation of dialysis could allow for better volume and electrolyte control and prevent the development of more serious complications of kidney failure such as uremic seizures, encephalopathy, and pericarditis. However, RRT is associated with its own risks and earlier initiation may expose the patient to unnecessary procedures and complications that might delay renal recovery. A meta-analysis of predominantly observational data found that earlier initiation of RRT in AKI was associated with lower 28-day mortality, greater renal recovery, decreased duration of RRT, and decreased ICU length of stay.43 Subsequently, two prospective trials reported conflicting results regarding associations between dialysis timing and outcomes in patients with severe AKI (Table 4).44,45
The Early vs Late Initiation of Renal Replacement Therapy in Critically Ill Patients with Acute Kidney Injury (ELAIN) was a prospective, single-center randomized trial in Germany of 231 critically ill, predominantly surgical ICU patients (about half postcardiac surgery) with at least KDIGO stage 2 AKI.44 Patients were randomized to early (within eight hours of developing KDIGO stage 2 AKI) or delayed (within 12 hours of developing KDIGO stage 3 AKI) RRT initiation; patients in the early RRT group initiated dialysis on average 20 hours earlier than the patients in the late group. All patients were treated with continuous venovenous hemodiafiltration. Early RRT initiation was associated with a 34% lower risk of mortality at 90 days, shorter hospital length of stay, and shorter RRT duration compared with delayed RRT initiation. There was no difference between groups in dialysis dependence at 90 days, but there was a lower risk of dialysis dependence at one year.46The Artificial Kidney Initiation in Kidney Injury Study (AKIKI)45 was a prospective, multicenter randomized trial in France that compared early versus delayed strategies of RRT initiation in 620 critically ill, mostly medical ICU patients with severe AKI (KDIGO stage 3). The median time between randomization and RRT initiation was two hours for the early and 57 hours for the delayed strategy groups. There were no differences between groups in length of hospital or ICU stay, vasopressor use, dialysis dependence, or 60-day survival. The early strategy group had a higher incidence of catheter-related bloodstream infections (10% vs 5%) and hypophosphatemia (22% vs 15%) compared with that of the delayed strategy group. Patients in the delayed strategy group regained normal urine output sooner than in the early strategy group. Approximately half of the patients in the delayed strategy group avoided RRT altogether. The authors of AKIKI concluded that there was no benefit to the early strategy of RRT in critically ill patients with severe AKI, and a delayed strategy of RRT initiation may avoid unnecessary RRT and reduce catheter-related infectious complications.
How can we interpret these discrepant results? Although ELAIN found a benefit to earlier RRT initiation in AKI, it has limited generalizability to medical ICU patients, who have higher mortality and whose outcomes might be less affected by dialysis timing. Patients in ELAIN had a high prevalence of congestive heart failure and CKD; it is possible that select patient populations may derive greater benefit from earlier RRT initiation. Although both ELAIN and AKIKI used the standardized criteria for RRT initiation, neither study could incorporate important clinical factors such as trajectory of kidney function, comorbid conditions, or symptoms, which play a significant role in the decision-making process in real-world clinical practice. Additional large-scale, multicenter trials are needed to guide the timing of RRT in critically ill patients with AKI. The Initiation of Dialysis Early Versus Delayed in the ICU (IDEAL-ICU)47 and Standard versus Accelerated Initiation of RRT in Acute Kidney Injury (STARRT-AKI)48 studies are currently underway and hope to provide clearer guidance regarding the optimal timing of RRT initiation in AKI (Table 4). Until further evidence is available, experts recommend taking into consideration the trajectory of kidney disease, concurrent organ dysfunction, and expected need for fluid and solute control when making decisions regarding RRT initiation in AKI.16
DIALYSIS MODALITIES IN ACUTE KIDNEY INJURY
When RRT is required in patients with AKI, the dialysis modality is often determined by local availability. CRRT and sustained low-efficiency dialysis (SLED) are thought to be better tolerated than intermittent hemodialysis in hemodynamically unstable patients, although a randomized controlled trial could not demonstrate a survival difference between these modalities.49 In general, in settings where CRRT or SLED is available, these modalities are favored for patients with hemodynamic instability, but practice patterns vary widely.
CONCLUSION
Among hospitalized patients, AKI is common and associated with a higher risk of mortality. Although serum creatinine and urine output criteria are used to define AKI, other clinical factors (comorbid conditions, volume status, and trajectory of kidney function decline) can inform the assessment and management of patients with AKI. General strategies for AKI management include treatment of reversible conditions, optimization of volume status, hemodynamics, and nutritional status. The optimal timing of RRT in critically ill patients with AKI is not known, with unclear mortality benefit of earlier dialysis initiation. Two large-scale randomized controlled trials regarding early versus delayed dialysis timing in AKI are currently underway and will hopefully provide clarity in the near future.
Disclosures
Dr. Yu and Dr. Kamal have nothing to disclose. Dr. Chertow is an advisor to DURECT Corporation.
1. Wang HE, Muntner P, Chertow GM, Warnock DG. Acute kidney injury and mortality in hospitalized patients. Am J Nephrol. 2012;35(4):349-355. PubMed
2. Uchino S, Bellomo R, Goldsmith D, Bates S, Ronco C. An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med. 2006;34(7):1913-1917. PubMed
3. Hoste EA, Bagshaw SM, Bellomo R, et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med. 2015;41(8):1411-1423. PubMed
4. Wald R, McArthur E, Adhikari NKJ, et al. Changing incidence and outcomes following dialysis-requiring acute kidney injury among critically ill adults: a population-based cohort study. Am J Kidney Dis. 2015;65(6):870-877. PubMed
5. Siew ED, Davenport A. The growth of acute kidney injury: a rising tide or just closer attention to detail? Kidney Int. 2015;87(1):46-61. PubMed
6. Lenihan CR, Montez-Rath ME, Mora Mangano CT, Chertow GM, Winkelmayer WC. Trends in acute kidney injury, associated use of dialysis, and mortality after cardiac surgery, 1999 to 2008. Ann Thorac Surg. 2013;95(1):20-28. PubMed
7. Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16(11):3365-3370. PubMed
8. Ricci Z, Cruz D, Ronco C. The RIFLE criteria and mortality in acute kidney injury: a systematic review. Kidney Int. 2008;73(5):538-546. PubMed
9. Coca SG, Singanamala S, Parikh CR. Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney Int. 2012;81(5):442-448. PubMed
10. Ali T, Khan I, Simpson W, et al. Incidence and outcomes in acute kidney injury: a comprehensive population-based study. J Am Soc Nephrol. 2007;18(4):1292-1298. PubMed
11. Koulouridis I, Price LL, Madias NE, Jaber BL. Hospital-acquired acute kidney injury and hospital readmission: a cohort study. Am J Kidney Dis. 2015;65(2):275-282. PubMed
12. Bucaloiu ID, Kirchner HL, Norfolk ER, Hartle JE, 2nd, Perkins RM. Increased risk of death and de novo chronic kidney disease following reversible acute kidney injury. Kidney Int. 2012;81(5):477-485. PubMed
13. Cooper CM, Fenves AZ. Before you call renal: acute kidney injury for hospitalists. J Hosp Med. 2015;10(6):403-408. PubMed
14. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Workgroup A. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204-R212. PubMed
15. Mehta RL, Kellum JA, Shah SV, et al. Acute kidney injury network: report of an initiative to improve outcomes in acute kidney injury. Crit Care. 2007;11(2): R31. PubMed
16. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2(1):1-138.
17. Lin J, Fernandez H, Shashaty MG, et al. False-positive rate of AKI using consensus creatinine-based criteria. Clin J Am Soc Nephrol. 2015;10(10):1723-1731. PubMed
18. Palevsky PM, Liu KD, Brophy PD, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013;61(5):649-672. PubMed
19. James M, Bouchard J, Ho J, et al. Canadian Society of Nephrology commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013;61(5):673-685. PubMed
20. Finfer S, Bellomo R, Boyce N, et al. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med. 2004;350(22):2247-2256. PubMed
21. Caironi P, Tognoni G, Masson S, et al. Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med. 2014;370(15):1412-1421. PubMed
22. Myburgh JA, Finfer S, Bellomo R, et al. Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med. 2012;367(20):1901-1911. PubMed
23. Cooper DJ, Myburgh J, Heritier S, et al. Albumin resuscitation for traumatic brain injury: is intracranial hypertension the cause of increased mortality? J Neurotrauma. 2013;30(7):512-518. PubMed
24. Myburgh J, Cooper J, Finfer S, et al. Saline or albumin for fluid resuscitation in patients with traumatic brain injury. N Engl J Med. 2007;357(9):874-884. PubMed
25. Frenette AJ, Bouchard J, Bernier P, et al. Albumin administration is associated with acute kidney injury in cardiac surgery: a propensity score analysis. Crit Care. 2014;18(6):602. PubMed
26. Schortgen F, Lacherade JC, Bruneel F, et al. Effects of hydroxyethyl starch and gelatin on renal function in severe sepsis: a multicentre randomised study. Lancet. 2001;357(9260):911-916. PubMed
27. Perner A, Haase N, Guttormsen AB, et al. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med. 2012;367(2):124-134. PubMed
28. Dickenmann M, Oettl T, Mihatsch MJ. Osmotic nephrosis: acute kidney injury with accumulation of proximal tubular lysosomes due to administration of exogenous solutes. Am J Kidney Dis. 2008;51(3):491-503. PubMed
29. Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580-637. PubMed
30. Bernardi M, Caraceni P, Navickis RJ, Wilkes MM. Albumin infusion in patients undergoing large-volume paracentesis: a meta-analysis of randomized trials. Hepatology. 2012;55(4):1172-1181. PubMed
31. Sort P, Navasa M, Arroyo V, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med. 1999;341(6):403-409. PubMed
32. Boniatti MM, Cardoso PRC, Castilho RK, Vieira SRR. Is hyperchloremia associated with mortality in critically ill patients? A prospective cohort study. J Crit Care. 2011;26(2):175-179. PubMed
33. Yunos NM, Bellomo R, Hegarty C, Story D, Ho L, Bailey M. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. Jama-J Am Med Assoc. 2012;308(15):1566-1572. PubMed
34. Young P, Bailey M, Beasley R, et al. Effect of a buffered crystalloid solution vs saline on acute kidney injury among patients in the intensive care unit: the SPLIT randomized clinical trial. Jama-J Am Med Assoc. 2015;314(16):1701-1710. PubMed
35. Semler MW, Self WH, Wanderer JP, et al. Balanced crystalloids versus saline in critically ill adults. N Engl J Med. 2018;378(9):829-839. PubMed
36. Self WH, Semler MW, Wanderer JP, et al. Balanced crystalloids versus saline in noncritically ill adults. N Engl J Med. 2018;378(9):819-828. PubMed
37. Mehta RL, Pascual MT, Soroko S, Chertow GM, Group PS. Diuretics, mortality, and nonrecovery of renal function in acute renal failure. JAMA. 2002;288(20):2547-2553. PubMed
38. Grams ME, Estrella MM, Coresh J, et al. Fluid balance, diuretic use, and mortality in acute kidney injury. Clin J Am Soc Nephrol. 2011;6(5):966-973. PubMed
39. Ho KM, Power BM. Benefits and risks of furosemide in acute kidney injury. Anaesthesia. 2010;65(3):283-293. PubMed
40. Cantarovich F, Rangoonwala B, Lorenz H, Verho M, Esnault VL, High-Dose Flurosemide in Acute Renal Failure Study Group. High-dose furosemide for established ARF: a prospective, randomized, double-blind, placebo-controlled, multicenter trial. Am J Kidney Dis. 2004;44(3):402-409. PubMed
41. van der Voort PH, Boerma EC, Koopmans M, et al. Furosemide does not improve renal recovery after hemofiltration for acute renal failure in critically ill patients: a double blind randomized controlled trial. Crit Care Med. 2009;37(2):533-538. PubMed
42. Costa e Silva VT, Liano F, Muriel A, Diez R, de Castro I, Yu L. Nephrology referral and outcomes in critically ill acute kidney injury patients. PLoS One. 2013;8(8):e70482. PubMed
43. Karvellas CJ, Farhat MR, Sajjad I, et al. A comparison of early versus late initiation of renal replacement therapy in critically ill patients with acute kidney injury: a systematic review and meta-analysis. Crit Care. 2011;15(1):R72. PubMed
44. Zarbock A, Kellum JA, Schmidt C, et al. Effect of early vs delayed initiation of renal replacement therapy on mortality in critically ill patients with acute kidney injury: the ELAIN randomized clinical trial. JAMA. 2016;315(20):2190-2199. PubMed
45. Gaudry S, Hajage D, Schortgen F, et al. Initiation strategies for renal-replacement therapy in the intensive care unit. N Engl J Med. 2016;375(2):122-133. PubMed
46. Meersch M, Kullmar M, Schmidt C, et al. Long-term clinical outcomes after early initiation of RRT in critically ill patients with AKI. J Am Soc Nephrol. 2018;29(3):1011-1019. PubMed
47. Barbar SD, Binquet C, Monchi M, Bruyere R, Quenot JP. Impact on mortality of the timing of renal replacement therapy in patients with severe acute kidney injury in septic shock: the IDEAL-ICU study (initiation of dialysis early versus delayed in the intensive care unit): study protocol for a randomized controlled trial. Trials. 2014;15:270. PubMed
48. Smith OM, Wald R, Adhikari NK, et al. Standard versus accelerated initiation of renal replacement therapy in acute kidney injury (STARRT-AKI): study protocol for a randomized controlled trial. Trials. 2013;14:320. PubMed
49. Vinsonneau C, Camus C, Combes A, et al. Continuous venovenous haemodiafiltration versus intermittent haemodialysis for acute renal failure in patients with multiple-organ dysfunction syndrome: a multicentre randomised trial. Lancet. 2006;368(9533):379-385. PubMed
Acute kidney injury (AKI) is a common complication in hospitalized patients, affecting one in five inpatients1,2 and more than half of patients in intensive care units (ICU).3 The incidence of AKI appears to be increasing over time.4 Potential contributing factors include an aging population, rising prevalence of comorbid conditions such as heart failure and chronic kidney disease (CKD), using nephrotoxic agents, and increasing complexity of surgical procedures.5,6 AKI during a hospital stay is associated with a two to 10-fold increased risk of inhospital mortality,1,2,7-10 longer hospital length of stay,7,10 higher risk for hospital readmissions,11 and higher healthcare costs.7 Patients who survive an episode of AKI have a higher risk for CKD and dialysis-dependence,9 even after an episode of reversible AKI.12 Despite its clinical importance, several areas of controversy remain regarding the management of AKI and, in particular, the optimal timing of renal replacement therapy (RRT) in patients with AKI. The purpose of this manuscript is to review the approaches to diagnosis and management of AKI in hospitalized patients. We also review recent evidence regarding the timing of dialysis in patients with AKI. This journal recently reviewed the differential diagnosis and diagnostic evaluation of AKI, which is not covered here.13
DEFINITION OF ACUTE KIDNEY INJURY
AKI refers to an acute change in kidney function characterized by an increase in serum creatinine and/or a reduction in urine output. It is a clinical syndrome caused by a broad range of etiologies and may be related to primary kidney pathology and/or systemic illness. Until 2004, there was no standard definition for AKI and over 30 different definitions were found in the literature, which resulted in wide variation in the reported incidence and outcomes of AKI and made it challenging to apply an evidence-based approach to patient care. In 2004, the Risk, Injury, Failure, Loss, and End-stage kidney disease (RIFLE)14 criteria for AKI were proposed, which were modified to the Acute Kidney Injury Network (AKIN)15 criteria in 2007 (Table 1). Multiple studies show that the RIFLE and AKIN criteria for AKI are associated with higher mortality1,2,8,10 and increased risk for requiring RRT.1,10
International clinical practice guidelines for AKI were released by Kidney Disease: Improving Global Outcomes (KDIGO) in 2012, which included a standardized definition of AKI that was adapted from the previously validated RIFLE and AKIN definitions.16 Patients are considered to have AKI when the serum creatinine rises by as little as 0.3 mg/dL. It is notable that when the baseline serum creatinine is high, there is more inherent variability in the serum creatinine measurement; thus, patients with CKD have a higher risk of being misclassified as having AKI.17 Although the KDIGO definition for AKI is commonly used in research settings, components of this definition have not been well validated, and it is not widely used in clinical practice. Other renal professional societies still recommend an individualized approach to the diagnosis of AKI, taking into account other factors such as trajectories in kidney function, fluid balance, electrolyte abnormalities, comorbid conditions, and clinical context.18,19 While we endorse the KDIGO approach to the categorization of AKI severity, in practice, a more patient-centered approach is generally required to guide the optimal approach to determining the etiology of AKI and guiding management.
GENERAL MANAGEMENT OF ACUTE KIDNEY INJURY
All patients with AKI should have close monitoring of their serum creatinine and urine output. Noninvasive diagnostic studies (urine microscopy, postvoid residual, and renal ultrasound) should be considered based on the clinical scenario. General management strategies include treatment of the reversible causes of AKI and optimization of volume status, hemodynamics, and nutritional status (Table 2).
Reversible Causes of Acute Kidney Injury
The first step in the treatment of AKI is to identify and treat readily reversible causes of AKI such as volume depletion, hypotension, infection, and urinary obstruction. Nephrotoxins should be avoided and all medications should be reviewed and adjusted for kidney function, particularly those that may affect mental status. Avoid opiates with noxious or active metabolites, including meperidine and morphine. Instead, hydromorphone, fentanyl, and methadone are preferred in patients with AKI. Other commonly used medications that require dose adjustment include gabapentin, baclofen, metoclopramide, H2 antagonists, many commonly prescribed antibiotics (penicillins, most cephalosporins, carbapenems, quinolones, and sulfa drugs), many hypoglycemic agents, and insulin. For patients on RRT, dosing is dependent on dialysis modality. Consultation with a hospital pharmacist is recommended when RRT modalities are initiated or changed.
Intravenous Fluids
Patients with AKI should have their volume status assessed and receive adequate resuscitation with intravenous fluids to promote renal perfusion. However, the optimal type and volume of fluid to give in AKI remains controversial. Colloid-containing solutions are theoretically confined to the intravascular space and should pose a lower risk for pulmonary edema compared with crystalloids. However, these solutions are costly, are not associated with any meaningful benefit,20-22 and may even be associated with potential harm.22-27
The most commonly used colloid worldwide is hydroxyethyl starch (HES). Its potential adverse effects include anaphylactoid reactions, coagulopathy, and AKI. HES is cleared by the kidneys and can cause osmotic nephrosis, a form of AKI characterized by vacuole formation and proximal renal tubular damage.28 Randomized controlled trials have shown an increased risk of AKI, RRT use, and mortality in critically ill patients who were resuscitated with HES.22,26,27 HES is not currently recommended in patients who are critically ill or have impaired kidney function and sepsis guidelines advise against its use.29
In the United States, albumin is the most common colloid-containing solution used for intravascular volume resuscitation. Albumin has been shown to be safe for volume resuscitation in critically ill patients,20 but there is no proven advantage to using albumin over saline with respect to mortality, length of hospital stay, duration of mechanical ventilation, duration of RRT, or number of organ systems failure.20,21 Furthermore, albumin may be harmful in certain patient populations. In patients with traumatic brain injury, albumin resuscitation is associated with higher mean intracranial pressures23 and long-term mortality.24 In a retrospective study of patients undergoing cardiac surgery, albumin administration was associated with more than twice the risk of AKI compared with crystalloids.25 In contrast, in patients with cirrhosis, intravenous albumin lowers the rate of AKI when administered in the setting of a large volume paracentesis30 or spontaneous bacterial peritonitis.31 Outside of these narrow settings, current evidence does not support the use of intravenous albumin to prevent AKI and we would not endorse the use of intravenous albumin as a part of the treatment paradigm for established AKI.
Many renal and critical care guidelines recommend initial fluid resuscitation with isotonic crystalloids except in specific circumstances (ie, hemorrhagic shock), with consideration of albumin in select cases (ie, severe sepsis or cirrhosis).16,18,19,29 That stated, the optimal type of crystalloid solution that should be used in resuscitation remains unclear. Because of its low cost, normal (0.9%) saline is the most commonly used solution, but it can result in hyperchloremic metabolic acidosis, which can cause renal vasoconstriction and may be associated with mortality in critically ill patients.32 A prospective study found that administration of chloride-liberal fluids (including normal saline) to critically ill patients was associated with nearly twice the risk of AKI and RRT use compared with chloride-restrictive fluids,33 but a subsequent trial found no difference in AKI or mortality among patients receiving saline versus a balanced crystalloid (Plasma-Lyte 148).34 A recent pair of large, randomized control trials compared outcomes in patients at a single center who were resuscitated with normal saline versus balanced crystalloid solutions (Lactated Ringer’s or Plasma-Lyte A).35,36 In critically ill patients, the use of balanced crystalloid solutions was associated with a lower risk of the composite outcome of mortality, new RRT, or persistent kidney impairment, but there were no differences in any of the individual components of the composite outcome.35 In noncritically ill patients, there were no differences in the number of hospital-free days based on the type of crystalloid solution used.36 In the absence of compelling evidence for using balanced crystalloid solutions, we continue to use normal saline for initial fluid resuscitation, but to avoid severe hyperchloremia and acidosis, we will consider switching to a balanced solution (Lactated Ringer’s, Plasma-Lyte, or Normosol) for large volume resuscitation (>2 L), particularly in critically ill patients.
Diuretics
As above, volume status is a key component in the management of patients with AKI. In patients with AKI and hypervolemia, loop diuretics are often given prior to the initiation of RRT. Loop diuretics act on the sodium-potassium-chloride cotransporters in the thick ascending limb of the loop of Henle to increase urinary losses of these ions and urine volume. Loop diuretics are dose-dependent, and often, higher doses are needed (eg, furosemide 100 mg intravenous dose) in patients with AKI, since the diuretic effect depends on the proximal tubular secretion of the drug into the urine. The role of diuretics in AKI is controversial and some observational data suggest an increased mortality risk with diuretic use in patients with AKI.37 In critically ill patients with acute lung injury, diuretic use improved survival, which was attributed to better control of volume overload.38 But, a meta-analysis of 11 randomized controlled trials failed to demonstrate that diuretics directly improved survival or recovery of AKI.39 Moreover, randomized controlled trials found that diuretics given to a patient with AKI requiring RRT did not improve recovery of kidney function.40,41 The KDIGO guidelines recommend that diuretics should not be routinely used for AKI except in the management of volume overload.16
Nutritional Targets in Acute Kidney Injury
Critically ill patients have high protein catabolic rates, which put them at increased risk for malnutrition, which in turn is associated with mortality. Patients who receive continuous RRT (CRRT) may lose 5-10 g of protein and 10-15 g of amino acids daily, and these patients may have protein requirements that are twice the usual recommended daily protein intake.16 But excess protein administration can result in high urea generation and azotemia unrelated to the patient’s kidney function. Blood urea nitrogen may also be disproportionately elevated in conditions where tubular reabsorption of urea is increased, such as in volume depletion, diuretic use, corticosteroid use, and gastrointestinal bleeding. Interpretation of blood urea nitrogen results must be made in the appropriate clinical context, with recognition that azotemia alone may not be a good surrogate marker of the patient’s underlying kidney function. We recommend dietary consultation in critically ill patients with AKI to ensure that adequate, but not excessive, protein is administered.
RENAL REPLACEMENT THERAPY IN ACUTE KIDNEY INJURY
In patients with AKI, RRT is initiated for control of volume overload, electrolyte abnormalities, acidemia, or uremic symptoms or complications that are refractory to medical management (Table 3). In a nonoliguric patient, fluid and electrolyte abnormalities can oftentimes be managed medically. Patients with oligoanuria (generally defined as urine output less than 400 mL/day or <20 mL/hour), however, require nephrology evaluation for consideration of RRT. Early nephrology consultation (within 48 hours of AKI diagnosis) may be associated with lower dialysis dependence and mortality in critically ill patients with AKI.42 The decision to initiate dialysis is individualized based on the patient’s comorbid conditions, urine output, and trajectory of kidney function.
Timing of Renal Replacement Therapy
The optimal timing of dialysis initiation in patients with AKI is not known. Theoretically, earlier initiation of dialysis could allow for better volume and electrolyte control and prevent the development of more serious complications of kidney failure such as uremic seizures, encephalopathy, and pericarditis. However, RRT is associated with its own risks and earlier initiation may expose the patient to unnecessary procedures and complications that might delay renal recovery. A meta-analysis of predominantly observational data found that earlier initiation of RRT in AKI was associated with lower 28-day mortality, greater renal recovery, decreased duration of RRT, and decreased ICU length of stay.43 Subsequently, two prospective trials reported conflicting results regarding associations between dialysis timing and outcomes in patients with severe AKI (Table 4).44,45
The Early vs Late Initiation of Renal Replacement Therapy in Critically Ill Patients with Acute Kidney Injury (ELAIN) was a prospective, single-center randomized trial in Germany of 231 critically ill, predominantly surgical ICU patients (about half postcardiac surgery) with at least KDIGO stage 2 AKI.44 Patients were randomized to early (within eight hours of developing KDIGO stage 2 AKI) or delayed (within 12 hours of developing KDIGO stage 3 AKI) RRT initiation; patients in the early RRT group initiated dialysis on average 20 hours earlier than the patients in the late group. All patients were treated with continuous venovenous hemodiafiltration. Early RRT initiation was associated with a 34% lower risk of mortality at 90 days, shorter hospital length of stay, and shorter RRT duration compared with delayed RRT initiation. There was no difference between groups in dialysis dependence at 90 days, but there was a lower risk of dialysis dependence at one year.46The Artificial Kidney Initiation in Kidney Injury Study (AKIKI)45 was a prospective, multicenter randomized trial in France that compared early versus delayed strategies of RRT initiation in 620 critically ill, mostly medical ICU patients with severe AKI (KDIGO stage 3). The median time between randomization and RRT initiation was two hours for the early and 57 hours for the delayed strategy groups. There were no differences between groups in length of hospital or ICU stay, vasopressor use, dialysis dependence, or 60-day survival. The early strategy group had a higher incidence of catheter-related bloodstream infections (10% vs 5%) and hypophosphatemia (22% vs 15%) compared with that of the delayed strategy group. Patients in the delayed strategy group regained normal urine output sooner than in the early strategy group. Approximately half of the patients in the delayed strategy group avoided RRT altogether. The authors of AKIKI concluded that there was no benefit to the early strategy of RRT in critically ill patients with severe AKI, and a delayed strategy of RRT initiation may avoid unnecessary RRT and reduce catheter-related infectious complications.
How can we interpret these discrepant results? Although ELAIN found a benefit to earlier RRT initiation in AKI, it has limited generalizability to medical ICU patients, who have higher mortality and whose outcomes might be less affected by dialysis timing. Patients in ELAIN had a high prevalence of congestive heart failure and CKD; it is possible that select patient populations may derive greater benefit from earlier RRT initiation. Although both ELAIN and AKIKI used the standardized criteria for RRT initiation, neither study could incorporate important clinical factors such as trajectory of kidney function, comorbid conditions, or symptoms, which play a significant role in the decision-making process in real-world clinical practice. Additional large-scale, multicenter trials are needed to guide the timing of RRT in critically ill patients with AKI. The Initiation of Dialysis Early Versus Delayed in the ICU (IDEAL-ICU)47 and Standard versus Accelerated Initiation of RRT in Acute Kidney Injury (STARRT-AKI)48 studies are currently underway and hope to provide clearer guidance regarding the optimal timing of RRT initiation in AKI (Table 4). Until further evidence is available, experts recommend taking into consideration the trajectory of kidney disease, concurrent organ dysfunction, and expected need for fluid and solute control when making decisions regarding RRT initiation in AKI.16
DIALYSIS MODALITIES IN ACUTE KIDNEY INJURY
When RRT is required in patients with AKI, the dialysis modality is often determined by local availability. CRRT and sustained low-efficiency dialysis (SLED) are thought to be better tolerated than intermittent hemodialysis in hemodynamically unstable patients, although a randomized controlled trial could not demonstrate a survival difference between these modalities.49 In general, in settings where CRRT or SLED is available, these modalities are favored for patients with hemodynamic instability, but practice patterns vary widely.
CONCLUSION
Among hospitalized patients, AKI is common and associated with a higher risk of mortality. Although serum creatinine and urine output criteria are used to define AKI, other clinical factors (comorbid conditions, volume status, and trajectory of kidney function decline) can inform the assessment and management of patients with AKI. General strategies for AKI management include treatment of reversible conditions, optimization of volume status, hemodynamics, and nutritional status. The optimal timing of RRT in critically ill patients with AKI is not known, with unclear mortality benefit of earlier dialysis initiation. Two large-scale randomized controlled trials regarding early versus delayed dialysis timing in AKI are currently underway and will hopefully provide clarity in the near future.
Disclosures
Dr. Yu and Dr. Kamal have nothing to disclose. Dr. Chertow is an advisor to DURECT Corporation.
Acute kidney injury (AKI) is a common complication in hospitalized patients, affecting one in five inpatients1,2 and more than half of patients in intensive care units (ICU).3 The incidence of AKI appears to be increasing over time.4 Potential contributing factors include an aging population, rising prevalence of comorbid conditions such as heart failure and chronic kidney disease (CKD), using nephrotoxic agents, and increasing complexity of surgical procedures.5,6 AKI during a hospital stay is associated with a two to 10-fold increased risk of inhospital mortality,1,2,7-10 longer hospital length of stay,7,10 higher risk for hospital readmissions,11 and higher healthcare costs.7 Patients who survive an episode of AKI have a higher risk for CKD and dialysis-dependence,9 even after an episode of reversible AKI.12 Despite its clinical importance, several areas of controversy remain regarding the management of AKI and, in particular, the optimal timing of renal replacement therapy (RRT) in patients with AKI. The purpose of this manuscript is to review the approaches to diagnosis and management of AKI in hospitalized patients. We also review recent evidence regarding the timing of dialysis in patients with AKI. This journal recently reviewed the differential diagnosis and diagnostic evaluation of AKI, which is not covered here.13
DEFINITION OF ACUTE KIDNEY INJURY
AKI refers to an acute change in kidney function characterized by an increase in serum creatinine and/or a reduction in urine output. It is a clinical syndrome caused by a broad range of etiologies and may be related to primary kidney pathology and/or systemic illness. Until 2004, there was no standard definition for AKI and over 30 different definitions were found in the literature, which resulted in wide variation in the reported incidence and outcomes of AKI and made it challenging to apply an evidence-based approach to patient care. In 2004, the Risk, Injury, Failure, Loss, and End-stage kidney disease (RIFLE)14 criteria for AKI were proposed, which were modified to the Acute Kidney Injury Network (AKIN)15 criteria in 2007 (Table 1). Multiple studies show that the RIFLE and AKIN criteria for AKI are associated with higher mortality1,2,8,10 and increased risk for requiring RRT.1,10
International clinical practice guidelines for AKI were released by Kidney Disease: Improving Global Outcomes (KDIGO) in 2012, which included a standardized definition of AKI that was adapted from the previously validated RIFLE and AKIN definitions.16 Patients are considered to have AKI when the serum creatinine rises by as little as 0.3 mg/dL. It is notable that when the baseline serum creatinine is high, there is more inherent variability in the serum creatinine measurement; thus, patients with CKD have a higher risk of being misclassified as having AKI.17 Although the KDIGO definition for AKI is commonly used in research settings, components of this definition have not been well validated, and it is not widely used in clinical practice. Other renal professional societies still recommend an individualized approach to the diagnosis of AKI, taking into account other factors such as trajectories in kidney function, fluid balance, electrolyte abnormalities, comorbid conditions, and clinical context.18,19 While we endorse the KDIGO approach to the categorization of AKI severity, in practice, a more patient-centered approach is generally required to guide the optimal approach to determining the etiology of AKI and guiding management.
GENERAL MANAGEMENT OF ACUTE KIDNEY INJURY
All patients with AKI should have close monitoring of their serum creatinine and urine output. Noninvasive diagnostic studies (urine microscopy, postvoid residual, and renal ultrasound) should be considered based on the clinical scenario. General management strategies include treatment of the reversible causes of AKI and optimization of volume status, hemodynamics, and nutritional status (Table 2).
Reversible Causes of Acute Kidney Injury
The first step in the treatment of AKI is to identify and treat readily reversible causes of AKI such as volume depletion, hypotension, infection, and urinary obstruction. Nephrotoxins should be avoided and all medications should be reviewed and adjusted for kidney function, particularly those that may affect mental status. Avoid opiates with noxious or active metabolites, including meperidine and morphine. Instead, hydromorphone, fentanyl, and methadone are preferred in patients with AKI. Other commonly used medications that require dose adjustment include gabapentin, baclofen, metoclopramide, H2 antagonists, many commonly prescribed antibiotics (penicillins, most cephalosporins, carbapenems, quinolones, and sulfa drugs), many hypoglycemic agents, and insulin. For patients on RRT, dosing is dependent on dialysis modality. Consultation with a hospital pharmacist is recommended when RRT modalities are initiated or changed.
Intravenous Fluids
Patients with AKI should have their volume status assessed and receive adequate resuscitation with intravenous fluids to promote renal perfusion. However, the optimal type and volume of fluid to give in AKI remains controversial. Colloid-containing solutions are theoretically confined to the intravascular space and should pose a lower risk for pulmonary edema compared with crystalloids. However, these solutions are costly, are not associated with any meaningful benefit,20-22 and may even be associated with potential harm.22-27
The most commonly used colloid worldwide is hydroxyethyl starch (HES). Its potential adverse effects include anaphylactoid reactions, coagulopathy, and AKI. HES is cleared by the kidneys and can cause osmotic nephrosis, a form of AKI characterized by vacuole formation and proximal renal tubular damage.28 Randomized controlled trials have shown an increased risk of AKI, RRT use, and mortality in critically ill patients who were resuscitated with HES.22,26,27 HES is not currently recommended in patients who are critically ill or have impaired kidney function and sepsis guidelines advise against its use.29
In the United States, albumin is the most common colloid-containing solution used for intravascular volume resuscitation. Albumin has been shown to be safe for volume resuscitation in critically ill patients,20 but there is no proven advantage to using albumin over saline with respect to mortality, length of hospital stay, duration of mechanical ventilation, duration of RRT, or number of organ systems failure.20,21 Furthermore, albumin may be harmful in certain patient populations. In patients with traumatic brain injury, albumin resuscitation is associated with higher mean intracranial pressures23 and long-term mortality.24 In a retrospective study of patients undergoing cardiac surgery, albumin administration was associated with more than twice the risk of AKI compared with crystalloids.25 In contrast, in patients with cirrhosis, intravenous albumin lowers the rate of AKI when administered in the setting of a large volume paracentesis30 or spontaneous bacterial peritonitis.31 Outside of these narrow settings, current evidence does not support the use of intravenous albumin to prevent AKI and we would not endorse the use of intravenous albumin as a part of the treatment paradigm for established AKI.
Many renal and critical care guidelines recommend initial fluid resuscitation with isotonic crystalloids except in specific circumstances (ie, hemorrhagic shock), with consideration of albumin in select cases (ie, severe sepsis or cirrhosis).16,18,19,29 That stated, the optimal type of crystalloid solution that should be used in resuscitation remains unclear. Because of its low cost, normal (0.9%) saline is the most commonly used solution, but it can result in hyperchloremic metabolic acidosis, which can cause renal vasoconstriction and may be associated with mortality in critically ill patients.32 A prospective study found that administration of chloride-liberal fluids (including normal saline) to critically ill patients was associated with nearly twice the risk of AKI and RRT use compared with chloride-restrictive fluids,33 but a subsequent trial found no difference in AKI or mortality among patients receiving saline versus a balanced crystalloid (Plasma-Lyte 148).34 A recent pair of large, randomized control trials compared outcomes in patients at a single center who were resuscitated with normal saline versus balanced crystalloid solutions (Lactated Ringer’s or Plasma-Lyte A).35,36 In critically ill patients, the use of balanced crystalloid solutions was associated with a lower risk of the composite outcome of mortality, new RRT, or persistent kidney impairment, but there were no differences in any of the individual components of the composite outcome.35 In noncritically ill patients, there were no differences in the number of hospital-free days based on the type of crystalloid solution used.36 In the absence of compelling evidence for using balanced crystalloid solutions, we continue to use normal saline for initial fluid resuscitation, but to avoid severe hyperchloremia and acidosis, we will consider switching to a balanced solution (Lactated Ringer’s, Plasma-Lyte, or Normosol) for large volume resuscitation (>2 L), particularly in critically ill patients.
Diuretics
As above, volume status is a key component in the management of patients with AKI. In patients with AKI and hypervolemia, loop diuretics are often given prior to the initiation of RRT. Loop diuretics act on the sodium-potassium-chloride cotransporters in the thick ascending limb of the loop of Henle to increase urinary losses of these ions and urine volume. Loop diuretics are dose-dependent, and often, higher doses are needed (eg, furosemide 100 mg intravenous dose) in patients with AKI, since the diuretic effect depends on the proximal tubular secretion of the drug into the urine. The role of diuretics in AKI is controversial and some observational data suggest an increased mortality risk with diuretic use in patients with AKI.37 In critically ill patients with acute lung injury, diuretic use improved survival, which was attributed to better control of volume overload.38 But, a meta-analysis of 11 randomized controlled trials failed to demonstrate that diuretics directly improved survival or recovery of AKI.39 Moreover, randomized controlled trials found that diuretics given to a patient with AKI requiring RRT did not improve recovery of kidney function.40,41 The KDIGO guidelines recommend that diuretics should not be routinely used for AKI except in the management of volume overload.16
Nutritional Targets in Acute Kidney Injury
Critically ill patients have high protein catabolic rates, which put them at increased risk for malnutrition, which in turn is associated with mortality. Patients who receive continuous RRT (CRRT) may lose 5-10 g of protein and 10-15 g of amino acids daily, and these patients may have protein requirements that are twice the usual recommended daily protein intake.16 But excess protein administration can result in high urea generation and azotemia unrelated to the patient’s kidney function. Blood urea nitrogen may also be disproportionately elevated in conditions where tubular reabsorption of urea is increased, such as in volume depletion, diuretic use, corticosteroid use, and gastrointestinal bleeding. Interpretation of blood urea nitrogen results must be made in the appropriate clinical context, with recognition that azotemia alone may not be a good surrogate marker of the patient’s underlying kidney function. We recommend dietary consultation in critically ill patients with AKI to ensure that adequate, but not excessive, protein is administered.
RENAL REPLACEMENT THERAPY IN ACUTE KIDNEY INJURY
In patients with AKI, RRT is initiated for control of volume overload, electrolyte abnormalities, acidemia, or uremic symptoms or complications that are refractory to medical management (Table 3). In a nonoliguric patient, fluid and electrolyte abnormalities can oftentimes be managed medically. Patients with oligoanuria (generally defined as urine output less than 400 mL/day or <20 mL/hour), however, require nephrology evaluation for consideration of RRT. Early nephrology consultation (within 48 hours of AKI diagnosis) may be associated with lower dialysis dependence and mortality in critically ill patients with AKI.42 The decision to initiate dialysis is individualized based on the patient’s comorbid conditions, urine output, and trajectory of kidney function.
Timing of Renal Replacement Therapy
The optimal timing of dialysis initiation in patients with AKI is not known. Theoretically, earlier initiation of dialysis could allow for better volume and electrolyte control and prevent the development of more serious complications of kidney failure such as uremic seizures, encephalopathy, and pericarditis. However, RRT is associated with its own risks and earlier initiation may expose the patient to unnecessary procedures and complications that might delay renal recovery. A meta-analysis of predominantly observational data found that earlier initiation of RRT in AKI was associated with lower 28-day mortality, greater renal recovery, decreased duration of RRT, and decreased ICU length of stay.43 Subsequently, two prospective trials reported conflicting results regarding associations between dialysis timing and outcomes in patients with severe AKI (Table 4).44,45
The Early vs Late Initiation of Renal Replacement Therapy in Critically Ill Patients with Acute Kidney Injury (ELAIN) was a prospective, single-center randomized trial in Germany of 231 critically ill, predominantly surgical ICU patients (about half postcardiac surgery) with at least KDIGO stage 2 AKI.44 Patients were randomized to early (within eight hours of developing KDIGO stage 2 AKI) or delayed (within 12 hours of developing KDIGO stage 3 AKI) RRT initiation; patients in the early RRT group initiated dialysis on average 20 hours earlier than the patients in the late group. All patients were treated with continuous venovenous hemodiafiltration. Early RRT initiation was associated with a 34% lower risk of mortality at 90 days, shorter hospital length of stay, and shorter RRT duration compared with delayed RRT initiation. There was no difference between groups in dialysis dependence at 90 days, but there was a lower risk of dialysis dependence at one year.46The Artificial Kidney Initiation in Kidney Injury Study (AKIKI)45 was a prospective, multicenter randomized trial in France that compared early versus delayed strategies of RRT initiation in 620 critically ill, mostly medical ICU patients with severe AKI (KDIGO stage 3). The median time between randomization and RRT initiation was two hours for the early and 57 hours for the delayed strategy groups. There were no differences between groups in length of hospital or ICU stay, vasopressor use, dialysis dependence, or 60-day survival. The early strategy group had a higher incidence of catheter-related bloodstream infections (10% vs 5%) and hypophosphatemia (22% vs 15%) compared with that of the delayed strategy group. Patients in the delayed strategy group regained normal urine output sooner than in the early strategy group. Approximately half of the patients in the delayed strategy group avoided RRT altogether. The authors of AKIKI concluded that there was no benefit to the early strategy of RRT in critically ill patients with severe AKI, and a delayed strategy of RRT initiation may avoid unnecessary RRT and reduce catheter-related infectious complications.
How can we interpret these discrepant results? Although ELAIN found a benefit to earlier RRT initiation in AKI, it has limited generalizability to medical ICU patients, who have higher mortality and whose outcomes might be less affected by dialysis timing. Patients in ELAIN had a high prevalence of congestive heart failure and CKD; it is possible that select patient populations may derive greater benefit from earlier RRT initiation. Although both ELAIN and AKIKI used the standardized criteria for RRT initiation, neither study could incorporate important clinical factors such as trajectory of kidney function, comorbid conditions, or symptoms, which play a significant role in the decision-making process in real-world clinical practice. Additional large-scale, multicenter trials are needed to guide the timing of RRT in critically ill patients with AKI. The Initiation of Dialysis Early Versus Delayed in the ICU (IDEAL-ICU)47 and Standard versus Accelerated Initiation of RRT in Acute Kidney Injury (STARRT-AKI)48 studies are currently underway and hope to provide clearer guidance regarding the optimal timing of RRT initiation in AKI (Table 4). Until further evidence is available, experts recommend taking into consideration the trajectory of kidney disease, concurrent organ dysfunction, and expected need for fluid and solute control when making decisions regarding RRT initiation in AKI.16
DIALYSIS MODALITIES IN ACUTE KIDNEY INJURY
When RRT is required in patients with AKI, the dialysis modality is often determined by local availability. CRRT and sustained low-efficiency dialysis (SLED) are thought to be better tolerated than intermittent hemodialysis in hemodynamically unstable patients, although a randomized controlled trial could not demonstrate a survival difference between these modalities.49 In general, in settings where CRRT or SLED is available, these modalities are favored for patients with hemodynamic instability, but practice patterns vary widely.
CONCLUSION
Among hospitalized patients, AKI is common and associated with a higher risk of mortality. Although serum creatinine and urine output criteria are used to define AKI, other clinical factors (comorbid conditions, volume status, and trajectory of kidney function decline) can inform the assessment and management of patients with AKI. General strategies for AKI management include treatment of reversible conditions, optimization of volume status, hemodynamics, and nutritional status. The optimal timing of RRT in critically ill patients with AKI is not known, with unclear mortality benefit of earlier dialysis initiation. Two large-scale randomized controlled trials regarding early versus delayed dialysis timing in AKI are currently underway and will hopefully provide clarity in the near future.
Disclosures
Dr. Yu and Dr. Kamal have nothing to disclose. Dr. Chertow is an advisor to DURECT Corporation.
1. Wang HE, Muntner P, Chertow GM, Warnock DG. Acute kidney injury and mortality in hospitalized patients. Am J Nephrol. 2012;35(4):349-355. PubMed
2. Uchino S, Bellomo R, Goldsmith D, Bates S, Ronco C. An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med. 2006;34(7):1913-1917. PubMed
3. Hoste EA, Bagshaw SM, Bellomo R, et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med. 2015;41(8):1411-1423. PubMed
4. Wald R, McArthur E, Adhikari NKJ, et al. Changing incidence and outcomes following dialysis-requiring acute kidney injury among critically ill adults: a population-based cohort study. Am J Kidney Dis. 2015;65(6):870-877. PubMed
5. Siew ED, Davenport A. The growth of acute kidney injury: a rising tide or just closer attention to detail? Kidney Int. 2015;87(1):46-61. PubMed
6. Lenihan CR, Montez-Rath ME, Mora Mangano CT, Chertow GM, Winkelmayer WC. Trends in acute kidney injury, associated use of dialysis, and mortality after cardiac surgery, 1999 to 2008. Ann Thorac Surg. 2013;95(1):20-28. PubMed
7. Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16(11):3365-3370. PubMed
8. Ricci Z, Cruz D, Ronco C. The RIFLE criteria and mortality in acute kidney injury: a systematic review. Kidney Int. 2008;73(5):538-546. PubMed
9. Coca SG, Singanamala S, Parikh CR. Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney Int. 2012;81(5):442-448. PubMed
10. Ali T, Khan I, Simpson W, et al. Incidence and outcomes in acute kidney injury: a comprehensive population-based study. J Am Soc Nephrol. 2007;18(4):1292-1298. PubMed
11. Koulouridis I, Price LL, Madias NE, Jaber BL. Hospital-acquired acute kidney injury and hospital readmission: a cohort study. Am J Kidney Dis. 2015;65(2):275-282. PubMed
12. Bucaloiu ID, Kirchner HL, Norfolk ER, Hartle JE, 2nd, Perkins RM. Increased risk of death and de novo chronic kidney disease following reversible acute kidney injury. Kidney Int. 2012;81(5):477-485. PubMed
13. Cooper CM, Fenves AZ. Before you call renal: acute kidney injury for hospitalists. J Hosp Med. 2015;10(6):403-408. PubMed
14. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Workgroup A. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204-R212. PubMed
15. Mehta RL, Kellum JA, Shah SV, et al. Acute kidney injury network: report of an initiative to improve outcomes in acute kidney injury. Crit Care. 2007;11(2): R31. PubMed
16. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2(1):1-138.
17. Lin J, Fernandez H, Shashaty MG, et al. False-positive rate of AKI using consensus creatinine-based criteria. Clin J Am Soc Nephrol. 2015;10(10):1723-1731. PubMed
18. Palevsky PM, Liu KD, Brophy PD, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013;61(5):649-672. PubMed
19. James M, Bouchard J, Ho J, et al. Canadian Society of Nephrology commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013;61(5):673-685. PubMed
20. Finfer S, Bellomo R, Boyce N, et al. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med. 2004;350(22):2247-2256. PubMed
21. Caironi P, Tognoni G, Masson S, et al. Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med. 2014;370(15):1412-1421. PubMed
22. Myburgh JA, Finfer S, Bellomo R, et al. Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med. 2012;367(20):1901-1911. PubMed
23. Cooper DJ, Myburgh J, Heritier S, et al. Albumin resuscitation for traumatic brain injury: is intracranial hypertension the cause of increased mortality? J Neurotrauma. 2013;30(7):512-518. PubMed
24. Myburgh J, Cooper J, Finfer S, et al. Saline or albumin for fluid resuscitation in patients with traumatic brain injury. N Engl J Med. 2007;357(9):874-884. PubMed
25. Frenette AJ, Bouchard J, Bernier P, et al. Albumin administration is associated with acute kidney injury in cardiac surgery: a propensity score analysis. Crit Care. 2014;18(6):602. PubMed
26. Schortgen F, Lacherade JC, Bruneel F, et al. Effects of hydroxyethyl starch and gelatin on renal function in severe sepsis: a multicentre randomised study. Lancet. 2001;357(9260):911-916. PubMed
27. Perner A, Haase N, Guttormsen AB, et al. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med. 2012;367(2):124-134. PubMed
28. Dickenmann M, Oettl T, Mihatsch MJ. Osmotic nephrosis: acute kidney injury with accumulation of proximal tubular lysosomes due to administration of exogenous solutes. Am J Kidney Dis. 2008;51(3):491-503. PubMed
29. Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580-637. PubMed
30. Bernardi M, Caraceni P, Navickis RJ, Wilkes MM. Albumin infusion in patients undergoing large-volume paracentesis: a meta-analysis of randomized trials. Hepatology. 2012;55(4):1172-1181. PubMed
31. Sort P, Navasa M, Arroyo V, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med. 1999;341(6):403-409. PubMed
32. Boniatti MM, Cardoso PRC, Castilho RK, Vieira SRR. Is hyperchloremia associated with mortality in critically ill patients? A prospective cohort study. J Crit Care. 2011;26(2):175-179. PubMed
33. Yunos NM, Bellomo R, Hegarty C, Story D, Ho L, Bailey M. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. Jama-J Am Med Assoc. 2012;308(15):1566-1572. PubMed
34. Young P, Bailey M, Beasley R, et al. Effect of a buffered crystalloid solution vs saline on acute kidney injury among patients in the intensive care unit: the SPLIT randomized clinical trial. Jama-J Am Med Assoc. 2015;314(16):1701-1710. PubMed
35. Semler MW, Self WH, Wanderer JP, et al. Balanced crystalloids versus saline in critically ill adults. N Engl J Med. 2018;378(9):829-839. PubMed
36. Self WH, Semler MW, Wanderer JP, et al. Balanced crystalloids versus saline in noncritically ill adults. N Engl J Med. 2018;378(9):819-828. PubMed
37. Mehta RL, Pascual MT, Soroko S, Chertow GM, Group PS. Diuretics, mortality, and nonrecovery of renal function in acute renal failure. JAMA. 2002;288(20):2547-2553. PubMed
38. Grams ME, Estrella MM, Coresh J, et al. Fluid balance, diuretic use, and mortality in acute kidney injury. Clin J Am Soc Nephrol. 2011;6(5):966-973. PubMed
39. Ho KM, Power BM. Benefits and risks of furosemide in acute kidney injury. Anaesthesia. 2010;65(3):283-293. PubMed
40. Cantarovich F, Rangoonwala B, Lorenz H, Verho M, Esnault VL, High-Dose Flurosemide in Acute Renal Failure Study Group. High-dose furosemide for established ARF: a prospective, randomized, double-blind, placebo-controlled, multicenter trial. Am J Kidney Dis. 2004;44(3):402-409. PubMed
41. van der Voort PH, Boerma EC, Koopmans M, et al. Furosemide does not improve renal recovery after hemofiltration for acute renal failure in critically ill patients: a double blind randomized controlled trial. Crit Care Med. 2009;37(2):533-538. PubMed
42. Costa e Silva VT, Liano F, Muriel A, Diez R, de Castro I, Yu L. Nephrology referral and outcomes in critically ill acute kidney injury patients. PLoS One. 2013;8(8):e70482. PubMed
43. Karvellas CJ, Farhat MR, Sajjad I, et al. A comparison of early versus late initiation of renal replacement therapy in critically ill patients with acute kidney injury: a systematic review and meta-analysis. Crit Care. 2011;15(1):R72. PubMed
44. Zarbock A, Kellum JA, Schmidt C, et al. Effect of early vs delayed initiation of renal replacement therapy on mortality in critically ill patients with acute kidney injury: the ELAIN randomized clinical trial. JAMA. 2016;315(20):2190-2199. PubMed
45. Gaudry S, Hajage D, Schortgen F, et al. Initiation strategies for renal-replacement therapy in the intensive care unit. N Engl J Med. 2016;375(2):122-133. PubMed
46. Meersch M, Kullmar M, Schmidt C, et al. Long-term clinical outcomes after early initiation of RRT in critically ill patients with AKI. J Am Soc Nephrol. 2018;29(3):1011-1019. PubMed
47. Barbar SD, Binquet C, Monchi M, Bruyere R, Quenot JP. Impact on mortality of the timing of renal replacement therapy in patients with severe acute kidney injury in septic shock: the IDEAL-ICU study (initiation of dialysis early versus delayed in the intensive care unit): study protocol for a randomized controlled trial. Trials. 2014;15:270. PubMed
48. Smith OM, Wald R, Adhikari NK, et al. Standard versus accelerated initiation of renal replacement therapy in acute kidney injury (STARRT-AKI): study protocol for a randomized controlled trial. Trials. 2013;14:320. PubMed
49. Vinsonneau C, Camus C, Combes A, et al. Continuous venovenous haemodiafiltration versus intermittent haemodialysis for acute renal failure in patients with multiple-organ dysfunction syndrome: a multicentre randomised trial. Lancet. 2006;368(9533):379-385. PubMed
1. Wang HE, Muntner P, Chertow GM, Warnock DG. Acute kidney injury and mortality in hospitalized patients. Am J Nephrol. 2012;35(4):349-355. PubMed
2. Uchino S, Bellomo R, Goldsmith D, Bates S, Ronco C. An assessment of the RIFLE criteria for acute renal failure in hospitalized patients. Crit Care Med. 2006;34(7):1913-1917. PubMed
3. Hoste EA, Bagshaw SM, Bellomo R, et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med. 2015;41(8):1411-1423. PubMed
4. Wald R, McArthur E, Adhikari NKJ, et al. Changing incidence and outcomes following dialysis-requiring acute kidney injury among critically ill adults: a population-based cohort study. Am J Kidney Dis. 2015;65(6):870-877. PubMed
5. Siew ED, Davenport A. The growth of acute kidney injury: a rising tide or just closer attention to detail? Kidney Int. 2015;87(1):46-61. PubMed
6. Lenihan CR, Montez-Rath ME, Mora Mangano CT, Chertow GM, Winkelmayer WC. Trends in acute kidney injury, associated use of dialysis, and mortality after cardiac surgery, 1999 to 2008. Ann Thorac Surg. 2013;95(1):20-28. PubMed
7. Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16(11):3365-3370. PubMed
8. Ricci Z, Cruz D, Ronco C. The RIFLE criteria and mortality in acute kidney injury: a systematic review. Kidney Int. 2008;73(5):538-546. PubMed
9. Coca SG, Singanamala S, Parikh CR. Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney Int. 2012;81(5):442-448. PubMed
10. Ali T, Khan I, Simpson W, et al. Incidence and outcomes in acute kidney injury: a comprehensive population-based study. J Am Soc Nephrol. 2007;18(4):1292-1298. PubMed
11. Koulouridis I, Price LL, Madias NE, Jaber BL. Hospital-acquired acute kidney injury and hospital readmission: a cohort study. Am J Kidney Dis. 2015;65(2):275-282. PubMed
12. Bucaloiu ID, Kirchner HL, Norfolk ER, Hartle JE, 2nd, Perkins RM. Increased risk of death and de novo chronic kidney disease following reversible acute kidney injury. Kidney Int. 2012;81(5):477-485. PubMed
13. Cooper CM, Fenves AZ. Before you call renal: acute kidney injury for hospitalists. J Hosp Med. 2015;10(6):403-408. PubMed
14. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Workgroup A. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204-R212. PubMed
15. Mehta RL, Kellum JA, Shah SV, et al. Acute kidney injury network: report of an initiative to improve outcomes in acute kidney injury. Crit Care. 2007;11(2): R31. PubMed
16. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2(1):1-138.
17. Lin J, Fernandez H, Shashaty MG, et al. False-positive rate of AKI using consensus creatinine-based criteria. Clin J Am Soc Nephrol. 2015;10(10):1723-1731. PubMed
18. Palevsky PM, Liu KD, Brophy PD, et al. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013;61(5):649-672. PubMed
19. James M, Bouchard J, Ho J, et al. Canadian Society of Nephrology commentary on the 2012 KDIGO clinical practice guideline for acute kidney injury. Am J Kidney Dis. 2013;61(5):673-685. PubMed
20. Finfer S, Bellomo R, Boyce N, et al. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med. 2004;350(22):2247-2256. PubMed
21. Caironi P, Tognoni G, Masson S, et al. Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med. 2014;370(15):1412-1421. PubMed
22. Myburgh JA, Finfer S, Bellomo R, et al. Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med. 2012;367(20):1901-1911. PubMed
23. Cooper DJ, Myburgh J, Heritier S, et al. Albumin resuscitation for traumatic brain injury: is intracranial hypertension the cause of increased mortality? J Neurotrauma. 2013;30(7):512-518. PubMed
24. Myburgh J, Cooper J, Finfer S, et al. Saline or albumin for fluid resuscitation in patients with traumatic brain injury. N Engl J Med. 2007;357(9):874-884. PubMed
25. Frenette AJ, Bouchard J, Bernier P, et al. Albumin administration is associated with acute kidney injury in cardiac surgery: a propensity score analysis. Crit Care. 2014;18(6):602. PubMed
26. Schortgen F, Lacherade JC, Bruneel F, et al. Effects of hydroxyethyl starch and gelatin on renal function in severe sepsis: a multicentre randomised study. Lancet. 2001;357(9260):911-916. PubMed
27. Perner A, Haase N, Guttormsen AB, et al. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med. 2012;367(2):124-134. PubMed
28. Dickenmann M, Oettl T, Mihatsch MJ. Osmotic nephrosis: acute kidney injury with accumulation of proximal tubular lysosomes due to administration of exogenous solutes. Am J Kidney Dis. 2008;51(3):491-503. PubMed
29. Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580-637. PubMed
30. Bernardi M, Caraceni P, Navickis RJ, Wilkes MM. Albumin infusion in patients undergoing large-volume paracentesis: a meta-analysis of randomized trials. Hepatology. 2012;55(4):1172-1181. PubMed
31. Sort P, Navasa M, Arroyo V, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med. 1999;341(6):403-409. PubMed
32. Boniatti MM, Cardoso PRC, Castilho RK, Vieira SRR. Is hyperchloremia associated with mortality in critically ill patients? A prospective cohort study. J Crit Care. 2011;26(2):175-179. PubMed
33. Yunos NM, Bellomo R, Hegarty C, Story D, Ho L, Bailey M. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. Jama-J Am Med Assoc. 2012;308(15):1566-1572. PubMed
34. Young P, Bailey M, Beasley R, et al. Effect of a buffered crystalloid solution vs saline on acute kidney injury among patients in the intensive care unit: the SPLIT randomized clinical trial. Jama-J Am Med Assoc. 2015;314(16):1701-1710. PubMed
35. Semler MW, Self WH, Wanderer JP, et al. Balanced crystalloids versus saline in critically ill adults. N Engl J Med. 2018;378(9):829-839. PubMed
36. Self WH, Semler MW, Wanderer JP, et al. Balanced crystalloids versus saline in noncritically ill adults. N Engl J Med. 2018;378(9):819-828. PubMed
37. Mehta RL, Pascual MT, Soroko S, Chertow GM, Group PS. Diuretics, mortality, and nonrecovery of renal function in acute renal failure. JAMA. 2002;288(20):2547-2553. PubMed
38. Grams ME, Estrella MM, Coresh J, et al. Fluid balance, diuretic use, and mortality in acute kidney injury. Clin J Am Soc Nephrol. 2011;6(5):966-973. PubMed
39. Ho KM, Power BM. Benefits and risks of furosemide in acute kidney injury. Anaesthesia. 2010;65(3):283-293. PubMed
40. Cantarovich F, Rangoonwala B, Lorenz H, Verho M, Esnault VL, High-Dose Flurosemide in Acute Renal Failure Study Group. High-dose furosemide for established ARF: a prospective, randomized, double-blind, placebo-controlled, multicenter trial. Am J Kidney Dis. 2004;44(3):402-409. PubMed
41. van der Voort PH, Boerma EC, Koopmans M, et al. Furosemide does not improve renal recovery after hemofiltration for acute renal failure in critically ill patients: a double blind randomized controlled trial. Crit Care Med. 2009;37(2):533-538. PubMed
42. Costa e Silva VT, Liano F, Muriel A, Diez R, de Castro I, Yu L. Nephrology referral and outcomes in critically ill acute kidney injury patients. PLoS One. 2013;8(8):e70482. PubMed
43. Karvellas CJ, Farhat MR, Sajjad I, et al. A comparison of early versus late initiation of renal replacement therapy in critically ill patients with acute kidney injury: a systematic review and meta-analysis. Crit Care. 2011;15(1):R72. PubMed
44. Zarbock A, Kellum JA, Schmidt C, et al. Effect of early vs delayed initiation of renal replacement therapy on mortality in critically ill patients with acute kidney injury: the ELAIN randomized clinical trial. JAMA. 2016;315(20):2190-2199. PubMed
45. Gaudry S, Hajage D, Schortgen F, et al. Initiation strategies for renal-replacement therapy in the intensive care unit. N Engl J Med. 2016;375(2):122-133. PubMed
46. Meersch M, Kullmar M, Schmidt C, et al. Long-term clinical outcomes after early initiation of RRT in critically ill patients with AKI. J Am Soc Nephrol. 2018;29(3):1011-1019. PubMed
47. Barbar SD, Binquet C, Monchi M, Bruyere R, Quenot JP. Impact on mortality of the timing of renal replacement therapy in patients with severe acute kidney injury in septic shock: the IDEAL-ICU study (initiation of dialysis early versus delayed in the intensive care unit): study protocol for a randomized controlled trial. Trials. 2014;15:270. PubMed
48. Smith OM, Wald R, Adhikari NK, et al. Standard versus accelerated initiation of renal replacement therapy in acute kidney injury (STARRT-AKI): study protocol for a randomized controlled trial. Trials. 2013;14:320. PubMed
49. Vinsonneau C, Camus C, Combes A, et al. Continuous venovenous haemodiafiltration versus intermittent haemodialysis for acute renal failure in patients with multiple-organ dysfunction syndrome: a multicentre randomised trial. Lancet. 2006;368(9533):379-385. PubMed
© 2019 Society of Hospital Medicine
Things We Do For No Reason: Prealbumin Testing to Diagnose Malnutrition in the Hospitalized Patient
The “Things We Do for No Reason” series reviews practices which have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards, but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion. https://www.choosingwisely.org/

CASE PRESENTATION
A 34-year-old man is admitted for a complicated urinary tract infection related to a chronic in-dwelling Foley catheter. The patient suffered a spinal cord injury at the C4/C5 level as a result of a motor vehicle accident 10 years ago and is confined to a motorized wheelchair. He is an engineer and lives independently but has caregivers. His body mass index (BMI) is 18.5 kg/m2, and he reports his weight has been stable. He has slight muscle atrophy of the biceps, triceps, interosseous muscles, and quadriceps. The patient reports that he eats well, has no chronic conditions, and has not had any gastrointestinal symptoms (eg, anorexia, nausea, diarrhea) over the last six months. You consider whether to order a serum prealbumin test to assess for possible malnutrition.
BACKGROUND
The presence of malnutrition in hospitalized patients is widely recognized as an independent predictor of hospital mortality.1 According to the American Society for Parenteral and Enteral Nutrition (ASPEN), malnutrition is defined as “an acute, subacute or chronic state of nutrition, in which varying degrees of overnutrition or undernutrition with or without inflammatory activity have led to a change in body composition and diminished function.”2 In one large European study, patients screening positive for being at risk of malnutrition had a 12-fold increase in hospital mortality.1
Inpatient malnutrition is remarkably underdocumented. Studies using chart reviews have found a prevalence of malnutrition in hospitalized patients of between 20% and 50%, and only 3% of hospital discharges are associated with a diagnostic code for malnutrition.3–5 Appropriate diagnosis and documentation of malnutrition is important given the profound prognostic and management implications of a malnutrition diagnosis. Appropriate documentation benefits health systems as malnutrition documentation increases expected mortality, thereby improving the observed-to-expected mortality ratio.
Serum prealbumin testing is widely available and frequently ordered in the inpatient setting. In a query we performed of the large aggregate Cerner Electronic Health Record database, HealthFacts, which includes data from inpatient encounters for approximately 700 United States hospitals, prealbumin tests were ordered 129,152 times in 2015. This activity corresponds to estimated total charges of $2,562,375 based on the 2015 clinical laboratory fee schedule.6
WHY YOU MIGHT THINK PREALBUMIN DIAGNOSES MALNUTRITION
Prealbumin is synthesized in the liver and released into circulation prior to excretion by the kidneys and gastrointestinal tract. Prealbumin transports thyroxine, triiodothyronine, and holo-retinol binding protein and, as a result, is also known as transthyretin.7 It was first proposed as a nutritional marker in 1972 with the publication of a study that showed low levels of prealbumin in 40 children with kwashiorkor that improved with intensive dietary supplementation.8 The shorter half-life of prealbumin (2.5 days) as compared with other identified nutritional markers, such as albumin, indicate that it would be suitable for detecting rapid changes in nutritional status.
WHY PREALBUMIN IS NOT HELPFUL FOR DIAGNOSING MALNUTRITION
Prealbumin Is Not Specific
An ideal nutritional marker should be specific enough that changes in this marker reflect changes in nutritional status.9 While there are many systemic factors that affect nutritional markers, such as prealbumin (Table 1), the acute phase response triggered by inflammation is the most significant confounder in the acutely ill hospitalized patient.9 This response to infection, stress, and malignancy leads to an increase in proinflammatory cytokines, increased liver synthesis of inflammatory proteins, such as C-reactive protein (CRP), and increased vascular permeability. Prealbumin is a negative acute phase reactant that decreases in concentration during the stress response due to slowed synthesis and extravasation.9 In a study of 24 patients with severe sepsis and trauma, levels of prealbumin inversely correlated with CRP, a reflection of the stress response, and returned to normal when CRP levels normalized. Neither prealbumin nor CRP, however, correlated with total body protein changes.10 Unfortunately, many studies supporting the use of prealbumin as a nutritional marker do not address the role of the acute phase response in their results. These studies include the original report on prealbumin in kwashiorkor, a condition known to be associated with a high rate of infectious diseases that can trigger the acute phase response.9 A consensus statement from the Academy of Nutrition and Dietetics (AND) and ASPEN noted that prealbumin is an indicator of inflammation and lacks the specificity to diagnose malnutrition.11
Prealbumin Is Not Sensitive
A sensitive laboratory test for malnutrition should allow for detection of malnutrition at an early stage.9 However, patients who demonstrate severe malnutrition without a coexisting inflammatory state do not consistently show low levels of prealbumin. In a systematic review of 20 studies in nondiseased malnourished patients, only two studies, both of which assessed patients with anorexia nervosa, had a mean prealbumin below normal (<20 mg/dL), and this finding corresponded to patient populations with mean BMIs less than 12 kg/m2. More importantly, normal prealbumin levels were seen in groups of patients with a mean BMI as low as 12.9 kg/m2.12 Analysis by AND found insufficient evidence to support a correlation between prealbumin and weight loss in anorexia nervosa, calorie restricted diets, or starvation.13 The data suggest that prealbumin lacks sufficient sensitivity to consistently detect cases of malnutrition easily diagnosed by history and/or physical exam.
Prealbumin Is Not Consistently Responsive to Nutritional Interventions
An accurate marker for malnutrition should improve when nutritional intervention results in adequate nutritional intake.9 While some studies have shown improvements in prealbumin in the setting of a nutritional intervention, many of these works are subject to the same limitations related to specificity and lack of control for concurrent inflammatory processes. In a retrospective study, prealbumin increased significantly in 102 patients receiving TPN for one week. Unfortunately, patients with renal or hepatic disease were excluded, and the role of inflammation was not assessed.14 Institutionalized patients with Alzheimer’s disease and normal CRP levels showed a statistically significant increase in weight gain, arm muscle circumference, and triceps skin-fold thickness following a nutritional program without a notable change in prealbumin.15 In a study assessing the relationship of prealbumin, CRP, and nutritional intake, critically ill populations receiving less than or greater than 60% of their estimated caloric needs showed no significant difference in prealbumin. In fact, prealbumin levels were only correlated with CRP levels.16 This finding argues against the routine use of prealbumin for nutrition monitoring in the acutely ill hospitalized patient.
Prealbumin Is Not Consistently Correlated with Health Outcomes
Even if prealbumin increased consistently in response to nutritional intervention, whether this change corresponds to an improvement in clinical outcomes has yet to be demonstrated.9 In 2005, Koretz reviewed 99 clinical trials and concluded that even when changes in nutritional markers are seen with nutritional support, the “changes in nutritional markers do not predict clinical outcomes.”17
WHAT YOU SHOULD DO INSTEAD: USE NONBIOLOGIC METHODS FOR SCREENING AND DIAGNOSING MALNUTRITION
Given the lack of a suitable biologic assay to identify malnutrition, dieticians and clinicians must rely on other means to assess malnutrition. Professional societies, including ASPEN and the European Society for Clinical Nutrition and Metabolism, have proposed different guidelines for the screening and assessment of malnutrition (Table 2).11,18 In 2016, these organizations, along with the Latin American Federation of Nutritional Therapy, Clinical Nutrition, and Metabolism and the Parenteral and Enteral Nutrition Society of Asia, formed The Global Leadership Initiative on Malnutrition (GLIM). In 2017, the GLIM taskforce agreed on clinically relevant diagnostic variables for the screening and assessment of malnutrition, including reduced food intake (anorexia), nonvolitional weight loss, (reduced) lean mass, status of disease burden and inflammation, and low body mass index or underweight status.19
RECOMMENDATIONS
- Do not use prealbumin to screen for or diagnose malnutrition.
- Consult with local dietitians to ensure that your institutional approach is in agreement with consensus recommendations.
CONCLUSION
In revisiting the case above, the patient does not have clear evidence of malnutrition based on his history (stable weight and good reported nutritional intake), although he does have a low BMI of 18.5 kg/m2. Rather than prealbumin testing, which would likely be low secondary to the acute phase response, he would better benefit from a nutrition-focused history and physical exam.
The uncertainties faced by clinicians in diagnosing malnutrition cannot readily be resolved by relying on a solitary laboratory marker (eg, prealbumin) or a stand-alone assessment protocol. The data obtained reflect the need for multidisciplinary teams of dieticians and clinicians to contextualize each patient’s medical history and ensure that the selected metrics are used appropriately to aid in diagnosis and documentation. We advocate that clinicians not routinely use prealbumin to screen for, confirm the diagnosis of, or assess the severity of malnutrition in the hospitalized patient.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailing TWDFNR@hospitalmedicine.org.
Disclosures
The authors have nothing to disclose.
1. Sorensen J, Kondrup J, Prokopowicz J, et al. EuroOOPS: an international, multicentre study to implement nutritional risk screening and evaluate clinical outcome. Clin Nutr Edinb Scotl. 2008;27(3):340-349. PubMed
2. Mueller C, Compher C, Ellen DM, American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition screening, assessment, and intervention in adults. JPEN J Parenter Enteral Nutr. 2011;35(1):16-24. PubMed
3. Kaiser MJ, Bauer JM, Rämsch C, et al. Frequency of malnutrition in older adults: a multinational perspective using the mini nutritional assessment. J Am Geriatr Soc. 2010;58(9):1734-1738. PubMed
4. Robinson MK, Trujillo EB, Mogensen KM, Rounds J, McManus K, Jacobs DO. Improving nutritional screening of hospitalized patients: the role of prealbumin. JPEN J Parenter Enteral Nutr. 2003;27(6):389-395; quiz 439. PubMed
5. Corkins MR, Guenter P, DiMaria-Ghalili RA, et al. Malnutrition diagnoses in hospitalized patients: United States, 2010. JPEN J Parenter Enteral Nutr. 2014;38(2):186-195. PubMed
6. Clinical Laboratory Fee Schedule Files. cms.org. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/ClinicalLabFeeSched/Clinical-Laboratory-Fee-Schedule-Files.html. Published September 29, 2016. Accessed January 5, 2018.
7. Myron Johnson A, Merlini G, Sheldon J, Ichihara K, Scientific Division Committee on Plasma Proteins (C-PP), International Federation of Clinical Chemistry and Laboratory Medicine (IFCC). Clinical indications for plasma protein assays: transthyretin (prealbumin) in inflammation and malnutrition. Clin Chem Lab Med. 2007;45(3):419-426. PubMed
8. Ingenbleek Y, De Visscher M, De Nayer P. Measurement of prealbumin as index of protein-calorie malnutrition. Lancet. 1972;2(7768):106-109. PubMed
9. Barbosa-Silva MCG. Subjective and objective nutritional assessment methods: what do they really assess? Curr Opin Clin Nutr Metab Care. 2008;11(3):248-254. PubMed
10. Clark MA, Hentzen BTH, Plank LD, Hill GL. Sequential changes in insulin-like growth factor 1, plasma proteins, and total body protein in severe sepsis and multiple injury. J Parenter Enter Nutr. 1996;20(5):363-370. PubMed
11. White JV, Guenter P, Jensen G, et al. Consensus statement of the Academy of Nutrition and Dietetics/American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). J Acad Nutr Diet. 2012;112(5):730-738. PubMed
12. Lee JL, Oh ES, Lee RW, Finucane TE. Serum albumin and prealbumin in calorically restricted, nondiseased individuals: a systematic review. Am J Med. 2015;128(9):1023.e1-22. PubMed
13. Academy of Nutrition and Dietetics Evidence Analysis Library. Nutrition Screening (NSCR) Systematic Review (2009-2010). https://www.andeal.org/tmp/pdf-print-919C51237950859AE3E15F978CEF49D8.pdf. Accessed August 23, 2017.
14. Sawicky CP, Nippo J, Winkler MF, Albina JE. Adequate energy intake and improved prealbumin concentration as indicators of the response to total parenteral nutrition. J Am Diet Assoc. 1992;92(10):1266-1268. PubMed
15. Van Wymelbeke V, Guédon A, Maniere D, Manckoundia P, Pfitzenmeyer P. A 6-month follow-up of nutritional status in institutionalized patients with Alzheimer’s disease. J Nutr Health Aging. 2004;8(6):505-508. PubMed
16. Davis CJ, Sowa D, Keim KS, Kinnare K, Peterson S. The use of prealbumin and C-reactive protein for monitoring nutrition support in adult patients receiving enteral nutrition in an urban medical center. JPEN J Parenter Enteral Nutr. 2012;36(2):197-204. PubMed
17. Koretz RL. Death, morbidity and economics are the only end points for trials. Proc Nutr Soc. 2005;64(3):277-284. PubMed
18. Cederholm T, Bosaeus I, Barazzoni R, et al. Diagnostic criteria for malnutrition - an ESPEN consensus statement. Clin Nutr Edinb Scotl. 2015;34(3):335-340. PubMed
19. Jensen GL, Cederholm T. Global leadership initiative on malnutrition: progress report from ASPEN clinical nutrition week 2017. JPEN J Parenter Enteral Nutr. April 2017:148607117707761. PubMed
The “Things We Do for No Reason” series reviews practices which have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards, but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion. https://www.choosingwisely.org/

CASE PRESENTATION
A 34-year-old man is admitted for a complicated urinary tract infection related to a chronic in-dwelling Foley catheter. The patient suffered a spinal cord injury at the C4/C5 level as a result of a motor vehicle accident 10 years ago and is confined to a motorized wheelchair. He is an engineer and lives independently but has caregivers. His body mass index (BMI) is 18.5 kg/m2, and he reports his weight has been stable. He has slight muscle atrophy of the biceps, triceps, interosseous muscles, and quadriceps. The patient reports that he eats well, has no chronic conditions, and has not had any gastrointestinal symptoms (eg, anorexia, nausea, diarrhea) over the last six months. You consider whether to order a serum prealbumin test to assess for possible malnutrition.
BACKGROUND
The presence of malnutrition in hospitalized patients is widely recognized as an independent predictor of hospital mortality.1 According to the American Society for Parenteral and Enteral Nutrition (ASPEN), malnutrition is defined as “an acute, subacute or chronic state of nutrition, in which varying degrees of overnutrition or undernutrition with or without inflammatory activity have led to a change in body composition and diminished function.”2 In one large European study, patients screening positive for being at risk of malnutrition had a 12-fold increase in hospital mortality.1
Inpatient malnutrition is remarkably underdocumented. Studies using chart reviews have found a prevalence of malnutrition in hospitalized patients of between 20% and 50%, and only 3% of hospital discharges are associated with a diagnostic code for malnutrition.3–5 Appropriate diagnosis and documentation of malnutrition is important given the profound prognostic and management implications of a malnutrition diagnosis. Appropriate documentation benefits health systems as malnutrition documentation increases expected mortality, thereby improving the observed-to-expected mortality ratio.
Serum prealbumin testing is widely available and frequently ordered in the inpatient setting. In a query we performed of the large aggregate Cerner Electronic Health Record database, HealthFacts, which includes data from inpatient encounters for approximately 700 United States hospitals, prealbumin tests were ordered 129,152 times in 2015. This activity corresponds to estimated total charges of $2,562,375 based on the 2015 clinical laboratory fee schedule.6
WHY YOU MIGHT THINK PREALBUMIN DIAGNOSES MALNUTRITION
Prealbumin is synthesized in the liver and released into circulation prior to excretion by the kidneys and gastrointestinal tract. Prealbumin transports thyroxine, triiodothyronine, and holo-retinol binding protein and, as a result, is also known as transthyretin.7 It was first proposed as a nutritional marker in 1972 with the publication of a study that showed low levels of prealbumin in 40 children with kwashiorkor that improved with intensive dietary supplementation.8 The shorter half-life of prealbumin (2.5 days) as compared with other identified nutritional markers, such as albumin, indicate that it would be suitable for detecting rapid changes in nutritional status.
WHY PREALBUMIN IS NOT HELPFUL FOR DIAGNOSING MALNUTRITION
Prealbumin Is Not Specific
An ideal nutritional marker should be specific enough that changes in this marker reflect changes in nutritional status.9 While there are many systemic factors that affect nutritional markers, such as prealbumin (Table 1), the acute phase response triggered by inflammation is the most significant confounder in the acutely ill hospitalized patient.9 This response to infection, stress, and malignancy leads to an increase in proinflammatory cytokines, increased liver synthesis of inflammatory proteins, such as C-reactive protein (CRP), and increased vascular permeability. Prealbumin is a negative acute phase reactant that decreases in concentration during the stress response due to slowed synthesis and extravasation.9 In a study of 24 patients with severe sepsis and trauma, levels of prealbumin inversely correlated with CRP, a reflection of the stress response, and returned to normal when CRP levels normalized. Neither prealbumin nor CRP, however, correlated with total body protein changes.10 Unfortunately, many studies supporting the use of prealbumin as a nutritional marker do not address the role of the acute phase response in their results. These studies include the original report on prealbumin in kwashiorkor, a condition known to be associated with a high rate of infectious diseases that can trigger the acute phase response.9 A consensus statement from the Academy of Nutrition and Dietetics (AND) and ASPEN noted that prealbumin is an indicator of inflammation and lacks the specificity to diagnose malnutrition.11
Prealbumin Is Not Sensitive
A sensitive laboratory test for malnutrition should allow for detection of malnutrition at an early stage.9 However, patients who demonstrate severe malnutrition without a coexisting inflammatory state do not consistently show low levels of prealbumin. In a systematic review of 20 studies in nondiseased malnourished patients, only two studies, both of which assessed patients with anorexia nervosa, had a mean prealbumin below normal (<20 mg/dL), and this finding corresponded to patient populations with mean BMIs less than 12 kg/m2. More importantly, normal prealbumin levels were seen in groups of patients with a mean BMI as low as 12.9 kg/m2.12 Analysis by AND found insufficient evidence to support a correlation between prealbumin and weight loss in anorexia nervosa, calorie restricted diets, or starvation.13 The data suggest that prealbumin lacks sufficient sensitivity to consistently detect cases of malnutrition easily diagnosed by history and/or physical exam.
Prealbumin Is Not Consistently Responsive to Nutritional Interventions
An accurate marker for malnutrition should improve when nutritional intervention results in adequate nutritional intake.9 While some studies have shown improvements in prealbumin in the setting of a nutritional intervention, many of these works are subject to the same limitations related to specificity and lack of control for concurrent inflammatory processes. In a retrospective study, prealbumin increased significantly in 102 patients receiving TPN for one week. Unfortunately, patients with renal or hepatic disease were excluded, and the role of inflammation was not assessed.14 Institutionalized patients with Alzheimer’s disease and normal CRP levels showed a statistically significant increase in weight gain, arm muscle circumference, and triceps skin-fold thickness following a nutritional program without a notable change in prealbumin.15 In a study assessing the relationship of prealbumin, CRP, and nutritional intake, critically ill populations receiving less than or greater than 60% of their estimated caloric needs showed no significant difference in prealbumin. In fact, prealbumin levels were only correlated with CRP levels.16 This finding argues against the routine use of prealbumin for nutrition monitoring in the acutely ill hospitalized patient.
Prealbumin Is Not Consistently Correlated with Health Outcomes
Even if prealbumin increased consistently in response to nutritional intervention, whether this change corresponds to an improvement in clinical outcomes has yet to be demonstrated.9 In 2005, Koretz reviewed 99 clinical trials and concluded that even when changes in nutritional markers are seen with nutritional support, the “changes in nutritional markers do not predict clinical outcomes.”17
WHAT YOU SHOULD DO INSTEAD: USE NONBIOLOGIC METHODS FOR SCREENING AND DIAGNOSING MALNUTRITION
Given the lack of a suitable biologic assay to identify malnutrition, dieticians and clinicians must rely on other means to assess malnutrition. Professional societies, including ASPEN and the European Society for Clinical Nutrition and Metabolism, have proposed different guidelines for the screening and assessment of malnutrition (Table 2).11,18 In 2016, these organizations, along with the Latin American Federation of Nutritional Therapy, Clinical Nutrition, and Metabolism and the Parenteral and Enteral Nutrition Society of Asia, formed The Global Leadership Initiative on Malnutrition (GLIM). In 2017, the GLIM taskforce agreed on clinically relevant diagnostic variables for the screening and assessment of malnutrition, including reduced food intake (anorexia), nonvolitional weight loss, (reduced) lean mass, status of disease burden and inflammation, and low body mass index or underweight status.19
RECOMMENDATIONS
- Do not use prealbumin to screen for or diagnose malnutrition.
- Consult with local dietitians to ensure that your institutional approach is in agreement with consensus recommendations.
CONCLUSION
In revisiting the case above, the patient does not have clear evidence of malnutrition based on his history (stable weight and good reported nutritional intake), although he does have a low BMI of 18.5 kg/m2. Rather than prealbumin testing, which would likely be low secondary to the acute phase response, he would better benefit from a nutrition-focused history and physical exam.
The uncertainties faced by clinicians in diagnosing malnutrition cannot readily be resolved by relying on a solitary laboratory marker (eg, prealbumin) or a stand-alone assessment protocol. The data obtained reflect the need for multidisciplinary teams of dieticians and clinicians to contextualize each patient’s medical history and ensure that the selected metrics are used appropriately to aid in diagnosis and documentation. We advocate that clinicians not routinely use prealbumin to screen for, confirm the diagnosis of, or assess the severity of malnutrition in the hospitalized patient.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailing TWDFNR@hospitalmedicine.org.
Disclosures
The authors have nothing to disclose.
The “Things We Do for No Reason” series reviews practices which have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards, but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion. https://www.choosingwisely.org/

CASE PRESENTATION
A 34-year-old man is admitted for a complicated urinary tract infection related to a chronic in-dwelling Foley catheter. The patient suffered a spinal cord injury at the C4/C5 level as a result of a motor vehicle accident 10 years ago and is confined to a motorized wheelchair. He is an engineer and lives independently but has caregivers. His body mass index (BMI) is 18.5 kg/m2, and he reports his weight has been stable. He has slight muscle atrophy of the biceps, triceps, interosseous muscles, and quadriceps. The patient reports that he eats well, has no chronic conditions, and has not had any gastrointestinal symptoms (eg, anorexia, nausea, diarrhea) over the last six months. You consider whether to order a serum prealbumin test to assess for possible malnutrition.
BACKGROUND
The presence of malnutrition in hospitalized patients is widely recognized as an independent predictor of hospital mortality.1 According to the American Society for Parenteral and Enteral Nutrition (ASPEN), malnutrition is defined as “an acute, subacute or chronic state of nutrition, in which varying degrees of overnutrition or undernutrition with or without inflammatory activity have led to a change in body composition and diminished function.”2 In one large European study, patients screening positive for being at risk of malnutrition had a 12-fold increase in hospital mortality.1
Inpatient malnutrition is remarkably underdocumented. Studies using chart reviews have found a prevalence of malnutrition in hospitalized patients of between 20% and 50%, and only 3% of hospital discharges are associated with a diagnostic code for malnutrition.3–5 Appropriate diagnosis and documentation of malnutrition is important given the profound prognostic and management implications of a malnutrition diagnosis. Appropriate documentation benefits health systems as malnutrition documentation increases expected mortality, thereby improving the observed-to-expected mortality ratio.
Serum prealbumin testing is widely available and frequently ordered in the inpatient setting. In a query we performed of the large aggregate Cerner Electronic Health Record database, HealthFacts, which includes data from inpatient encounters for approximately 700 United States hospitals, prealbumin tests were ordered 129,152 times in 2015. This activity corresponds to estimated total charges of $2,562,375 based on the 2015 clinical laboratory fee schedule.6
WHY YOU MIGHT THINK PREALBUMIN DIAGNOSES MALNUTRITION
Prealbumin is synthesized in the liver and released into circulation prior to excretion by the kidneys and gastrointestinal tract. Prealbumin transports thyroxine, triiodothyronine, and holo-retinol binding protein and, as a result, is also known as transthyretin.7 It was first proposed as a nutritional marker in 1972 with the publication of a study that showed low levels of prealbumin in 40 children with kwashiorkor that improved with intensive dietary supplementation.8 The shorter half-life of prealbumin (2.5 days) as compared with other identified nutritional markers, such as albumin, indicate that it would be suitable for detecting rapid changes in nutritional status.
WHY PREALBUMIN IS NOT HELPFUL FOR DIAGNOSING MALNUTRITION
Prealbumin Is Not Specific
An ideal nutritional marker should be specific enough that changes in this marker reflect changes in nutritional status.9 While there are many systemic factors that affect nutritional markers, such as prealbumin (Table 1), the acute phase response triggered by inflammation is the most significant confounder in the acutely ill hospitalized patient.9 This response to infection, stress, and malignancy leads to an increase in proinflammatory cytokines, increased liver synthesis of inflammatory proteins, such as C-reactive protein (CRP), and increased vascular permeability. Prealbumin is a negative acute phase reactant that decreases in concentration during the stress response due to slowed synthesis and extravasation.9 In a study of 24 patients with severe sepsis and trauma, levels of prealbumin inversely correlated with CRP, a reflection of the stress response, and returned to normal when CRP levels normalized. Neither prealbumin nor CRP, however, correlated with total body protein changes.10 Unfortunately, many studies supporting the use of prealbumin as a nutritional marker do not address the role of the acute phase response in their results. These studies include the original report on prealbumin in kwashiorkor, a condition known to be associated with a high rate of infectious diseases that can trigger the acute phase response.9 A consensus statement from the Academy of Nutrition and Dietetics (AND) and ASPEN noted that prealbumin is an indicator of inflammation and lacks the specificity to diagnose malnutrition.11
Prealbumin Is Not Sensitive
A sensitive laboratory test for malnutrition should allow for detection of malnutrition at an early stage.9 However, patients who demonstrate severe malnutrition without a coexisting inflammatory state do not consistently show low levels of prealbumin. In a systematic review of 20 studies in nondiseased malnourished patients, only two studies, both of which assessed patients with anorexia nervosa, had a mean prealbumin below normal (<20 mg/dL), and this finding corresponded to patient populations with mean BMIs less than 12 kg/m2. More importantly, normal prealbumin levels were seen in groups of patients with a mean BMI as low as 12.9 kg/m2.12 Analysis by AND found insufficient evidence to support a correlation between prealbumin and weight loss in anorexia nervosa, calorie restricted diets, or starvation.13 The data suggest that prealbumin lacks sufficient sensitivity to consistently detect cases of malnutrition easily diagnosed by history and/or physical exam.
Prealbumin Is Not Consistently Responsive to Nutritional Interventions
An accurate marker for malnutrition should improve when nutritional intervention results in adequate nutritional intake.9 While some studies have shown improvements in prealbumin in the setting of a nutritional intervention, many of these works are subject to the same limitations related to specificity and lack of control for concurrent inflammatory processes. In a retrospective study, prealbumin increased significantly in 102 patients receiving TPN for one week. Unfortunately, patients with renal or hepatic disease were excluded, and the role of inflammation was not assessed.14 Institutionalized patients with Alzheimer’s disease and normal CRP levels showed a statistically significant increase in weight gain, arm muscle circumference, and triceps skin-fold thickness following a nutritional program without a notable change in prealbumin.15 In a study assessing the relationship of prealbumin, CRP, and nutritional intake, critically ill populations receiving less than or greater than 60% of their estimated caloric needs showed no significant difference in prealbumin. In fact, prealbumin levels were only correlated with CRP levels.16 This finding argues against the routine use of prealbumin for nutrition monitoring in the acutely ill hospitalized patient.
Prealbumin Is Not Consistently Correlated with Health Outcomes
Even if prealbumin increased consistently in response to nutritional intervention, whether this change corresponds to an improvement in clinical outcomes has yet to be demonstrated.9 In 2005, Koretz reviewed 99 clinical trials and concluded that even when changes in nutritional markers are seen with nutritional support, the “changes in nutritional markers do not predict clinical outcomes.”17
WHAT YOU SHOULD DO INSTEAD: USE NONBIOLOGIC METHODS FOR SCREENING AND DIAGNOSING MALNUTRITION
Given the lack of a suitable biologic assay to identify malnutrition, dieticians and clinicians must rely on other means to assess malnutrition. Professional societies, including ASPEN and the European Society for Clinical Nutrition and Metabolism, have proposed different guidelines for the screening and assessment of malnutrition (Table 2).11,18 In 2016, these organizations, along with the Latin American Federation of Nutritional Therapy, Clinical Nutrition, and Metabolism and the Parenteral and Enteral Nutrition Society of Asia, formed The Global Leadership Initiative on Malnutrition (GLIM). In 2017, the GLIM taskforce agreed on clinically relevant diagnostic variables for the screening and assessment of malnutrition, including reduced food intake (anorexia), nonvolitional weight loss, (reduced) lean mass, status of disease burden and inflammation, and low body mass index or underweight status.19
RECOMMENDATIONS
- Do not use prealbumin to screen for or diagnose malnutrition.
- Consult with local dietitians to ensure that your institutional approach is in agreement with consensus recommendations.
CONCLUSION
In revisiting the case above, the patient does not have clear evidence of malnutrition based on his history (stable weight and good reported nutritional intake), although he does have a low BMI of 18.5 kg/m2. Rather than prealbumin testing, which would likely be low secondary to the acute phase response, he would better benefit from a nutrition-focused history and physical exam.
The uncertainties faced by clinicians in diagnosing malnutrition cannot readily be resolved by relying on a solitary laboratory marker (eg, prealbumin) or a stand-alone assessment protocol. The data obtained reflect the need for multidisciplinary teams of dieticians and clinicians to contextualize each patient’s medical history and ensure that the selected metrics are used appropriately to aid in diagnosis and documentation. We advocate that clinicians not routinely use prealbumin to screen for, confirm the diagnosis of, or assess the severity of malnutrition in the hospitalized patient.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailing TWDFNR@hospitalmedicine.org.
Disclosures
The authors have nothing to disclose.
1. Sorensen J, Kondrup J, Prokopowicz J, et al. EuroOOPS: an international, multicentre study to implement nutritional risk screening and evaluate clinical outcome. Clin Nutr Edinb Scotl. 2008;27(3):340-349. PubMed
2. Mueller C, Compher C, Ellen DM, American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition screening, assessment, and intervention in adults. JPEN J Parenter Enteral Nutr. 2011;35(1):16-24. PubMed
3. Kaiser MJ, Bauer JM, Rämsch C, et al. Frequency of malnutrition in older adults: a multinational perspective using the mini nutritional assessment. J Am Geriatr Soc. 2010;58(9):1734-1738. PubMed
4. Robinson MK, Trujillo EB, Mogensen KM, Rounds J, McManus K, Jacobs DO. Improving nutritional screening of hospitalized patients: the role of prealbumin. JPEN J Parenter Enteral Nutr. 2003;27(6):389-395; quiz 439. PubMed
5. Corkins MR, Guenter P, DiMaria-Ghalili RA, et al. Malnutrition diagnoses in hospitalized patients: United States, 2010. JPEN J Parenter Enteral Nutr. 2014;38(2):186-195. PubMed
6. Clinical Laboratory Fee Schedule Files. cms.org. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/ClinicalLabFeeSched/Clinical-Laboratory-Fee-Schedule-Files.html. Published September 29, 2016. Accessed January 5, 2018.
7. Myron Johnson A, Merlini G, Sheldon J, Ichihara K, Scientific Division Committee on Plasma Proteins (C-PP), International Federation of Clinical Chemistry and Laboratory Medicine (IFCC). Clinical indications for plasma protein assays: transthyretin (prealbumin) in inflammation and malnutrition. Clin Chem Lab Med. 2007;45(3):419-426. PubMed
8. Ingenbleek Y, De Visscher M, De Nayer P. Measurement of prealbumin as index of protein-calorie malnutrition. Lancet. 1972;2(7768):106-109. PubMed
9. Barbosa-Silva MCG. Subjective and objective nutritional assessment methods: what do they really assess? Curr Opin Clin Nutr Metab Care. 2008;11(3):248-254. PubMed
10. Clark MA, Hentzen BTH, Plank LD, Hill GL. Sequential changes in insulin-like growth factor 1, plasma proteins, and total body protein in severe sepsis and multiple injury. J Parenter Enter Nutr. 1996;20(5):363-370. PubMed
11. White JV, Guenter P, Jensen G, et al. Consensus statement of the Academy of Nutrition and Dietetics/American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). J Acad Nutr Diet. 2012;112(5):730-738. PubMed
12. Lee JL, Oh ES, Lee RW, Finucane TE. Serum albumin and prealbumin in calorically restricted, nondiseased individuals: a systematic review. Am J Med. 2015;128(9):1023.e1-22. PubMed
13. Academy of Nutrition and Dietetics Evidence Analysis Library. Nutrition Screening (NSCR) Systematic Review (2009-2010). https://www.andeal.org/tmp/pdf-print-919C51237950859AE3E15F978CEF49D8.pdf. Accessed August 23, 2017.
14. Sawicky CP, Nippo J, Winkler MF, Albina JE. Adequate energy intake and improved prealbumin concentration as indicators of the response to total parenteral nutrition. J Am Diet Assoc. 1992;92(10):1266-1268. PubMed
15. Van Wymelbeke V, Guédon A, Maniere D, Manckoundia P, Pfitzenmeyer P. A 6-month follow-up of nutritional status in institutionalized patients with Alzheimer’s disease. J Nutr Health Aging. 2004;8(6):505-508. PubMed
16. Davis CJ, Sowa D, Keim KS, Kinnare K, Peterson S. The use of prealbumin and C-reactive protein for monitoring nutrition support in adult patients receiving enteral nutrition in an urban medical center. JPEN J Parenter Enteral Nutr. 2012;36(2):197-204. PubMed
17. Koretz RL. Death, morbidity and economics are the only end points for trials. Proc Nutr Soc. 2005;64(3):277-284. PubMed
18. Cederholm T, Bosaeus I, Barazzoni R, et al. Diagnostic criteria for malnutrition - an ESPEN consensus statement. Clin Nutr Edinb Scotl. 2015;34(3):335-340. PubMed
19. Jensen GL, Cederholm T. Global leadership initiative on malnutrition: progress report from ASPEN clinical nutrition week 2017. JPEN J Parenter Enteral Nutr. April 2017:148607117707761. PubMed
1. Sorensen J, Kondrup J, Prokopowicz J, et al. EuroOOPS: an international, multicentre study to implement nutritional risk screening and evaluate clinical outcome. Clin Nutr Edinb Scotl. 2008;27(3):340-349. PubMed
2. Mueller C, Compher C, Ellen DM, American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. A.S.P.E.N. clinical guidelines: nutrition screening, assessment, and intervention in adults. JPEN J Parenter Enteral Nutr. 2011;35(1):16-24. PubMed
3. Kaiser MJ, Bauer JM, Rämsch C, et al. Frequency of malnutrition in older adults: a multinational perspective using the mini nutritional assessment. J Am Geriatr Soc. 2010;58(9):1734-1738. PubMed
4. Robinson MK, Trujillo EB, Mogensen KM, Rounds J, McManus K, Jacobs DO. Improving nutritional screening of hospitalized patients: the role of prealbumin. JPEN J Parenter Enteral Nutr. 2003;27(6):389-395; quiz 439. PubMed
5. Corkins MR, Guenter P, DiMaria-Ghalili RA, et al. Malnutrition diagnoses in hospitalized patients: United States, 2010. JPEN J Parenter Enteral Nutr. 2014;38(2):186-195. PubMed
6. Clinical Laboratory Fee Schedule Files. cms.org. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/ClinicalLabFeeSched/Clinical-Laboratory-Fee-Schedule-Files.html. Published September 29, 2016. Accessed January 5, 2018.
7. Myron Johnson A, Merlini G, Sheldon J, Ichihara K, Scientific Division Committee on Plasma Proteins (C-PP), International Federation of Clinical Chemistry and Laboratory Medicine (IFCC). Clinical indications for plasma protein assays: transthyretin (prealbumin) in inflammation and malnutrition. Clin Chem Lab Med. 2007;45(3):419-426. PubMed
8. Ingenbleek Y, De Visscher M, De Nayer P. Measurement of prealbumin as index of protein-calorie malnutrition. Lancet. 1972;2(7768):106-109. PubMed
9. Barbosa-Silva MCG. Subjective and objective nutritional assessment methods: what do they really assess? Curr Opin Clin Nutr Metab Care. 2008;11(3):248-254. PubMed
10. Clark MA, Hentzen BTH, Plank LD, Hill GL. Sequential changes in insulin-like growth factor 1, plasma proteins, and total body protein in severe sepsis and multiple injury. J Parenter Enter Nutr. 1996;20(5):363-370. PubMed
11. White JV, Guenter P, Jensen G, et al. Consensus statement of the Academy of Nutrition and Dietetics/American Society for Parenteral and Enteral Nutrition: characteristics recommended for the identification and documentation of adult malnutrition (undernutrition). J Acad Nutr Diet. 2012;112(5):730-738. PubMed
12. Lee JL, Oh ES, Lee RW, Finucane TE. Serum albumin and prealbumin in calorically restricted, nondiseased individuals: a systematic review. Am J Med. 2015;128(9):1023.e1-22. PubMed
13. Academy of Nutrition and Dietetics Evidence Analysis Library. Nutrition Screening (NSCR) Systematic Review (2009-2010). https://www.andeal.org/tmp/pdf-print-919C51237950859AE3E15F978CEF49D8.pdf. Accessed August 23, 2017.
14. Sawicky CP, Nippo J, Winkler MF, Albina JE. Adequate energy intake and improved prealbumin concentration as indicators of the response to total parenteral nutrition. J Am Diet Assoc. 1992;92(10):1266-1268. PubMed
15. Van Wymelbeke V, Guédon A, Maniere D, Manckoundia P, Pfitzenmeyer P. A 6-month follow-up of nutritional status in institutionalized patients with Alzheimer’s disease. J Nutr Health Aging. 2004;8(6):505-508. PubMed
16. Davis CJ, Sowa D, Keim KS, Kinnare K, Peterson S. The use of prealbumin and C-reactive protein for monitoring nutrition support in adult patients receiving enteral nutrition in an urban medical center. JPEN J Parenter Enteral Nutr. 2012;36(2):197-204. PubMed
17. Koretz RL. Death, morbidity and economics are the only end points for trials. Proc Nutr Soc. 2005;64(3):277-284. PubMed
18. Cederholm T, Bosaeus I, Barazzoni R, et al. Diagnostic criteria for malnutrition - an ESPEN consensus statement. Clin Nutr Edinb Scotl. 2015;34(3):335-340. PubMed
19. Jensen GL, Cederholm T. Global leadership initiative on malnutrition: progress report from ASPEN clinical nutrition week 2017. JPEN J Parenter Enteral Nutr. April 2017:148607117707761. PubMed
© 2018 Society of Hospital Medicine
Transthyretin (Prealbumin) and the Ambiguous Nature of Malnutrition
Lacy and colleagues identify an important “Thing We Do For No Reason”—prealbumin testing to diagnose malnutrition in hospitalized patients.1 They highlight the frequency and costs of ordering prealbumin tests although prealbumin is neither specific nor sensitive as a “marker of nutritional status,” shows
The term is also used to mean a condition where evidence shows better patient outcomes when improved nutrition is provided. Distinguishing between these two meanings is essential, as numerous patients with inflammatory illness will present abnormal “markers” when good evidence shows that they cannot benefit from nutritional support.
For example, a patient with advanced untreated human immunodeficiency virus (HIV) is likely to be considered malnourished because all of her “markers of nutritional status” are abnormal. She barely eats, has lost weight, and has low anthropometric, immunologic, and serologic measures, poor functional status, extreme vulnerability, and very poor prognosis. In this way she resembles a person in a famine situation. However, the patient is not malnourished in the sense that improved nutrient intake will lead to better patient outcomes. A Cochrane review of “nutritional interventions for reducing morbidity and mortality in people with HIV” found “no evidence that such supplementation translates into reductions in disease progression or HIV‐related complications, such as opportunistic infections or death.”2 The patient is dying of an inflammatory, cachectic illness. The same is true in managing patients with advanced cancer or several other serious illnesses.
Low prealbumin measures are associated with poor outcomes, which are then attributed to “malnutrition.” However, as Lacy and colleagues argue, prealbumin is a negative acute phase reactant and is thus a marker of the inflammatory effects of sickness/injury; it also responds variably to nutritional support. Citing Koretz, they note that “even when changes in nutritional markers are seen with nutritional support, the ‘changes in nutritional markers do not predict clinical outcomes.’”1,3 We know of no evidence from randomized controlled trials that prealbumin measurements help identify patients who can benefit from nutrition support.
By contrast, we and our colleagues have shown that in people who barely eat but show no inflammatory disease, eg, prison hunger-strikers and patients with anorexia nervosa, prealbumin level remains normal down to a body mass index below 13. The same is generally true for albumin.4 These measures fail to identify “malnutrition” in people who are starving.
Despite the complete lack of clinical trial evidence of benefit, prealbumin is widely used as an indicator of malnutrition. The National Institutes of Health’s Medline Plus website for the general public lists low prealbumin levels as a possible sign of malnutrition, for example, and advises that the prealbumin test may be used to “find out if you are getting enough nutrients, especially protein, in your diet” and to “check to see if you are getting enough nutrition if you are in the hospital.”5 Unjustified assertions such as these contribute to the dramatic overuse of nutritional interventions.
However, as a rule, things do occur for a reason. Using the term “prealbumin” conjures a certain relationship, perhaps as a precursor, to albumin, a venerable (but valueless) “marker of nutrition status.” In fact, the term refers only to a difference in electrophoretic mobility (prealbumin migrates faster). If prealbumin were called it by its proper name, transthyretin, it would probably have languished in obscurity among serum proteins until, in recent years, drug suppression of transthyretin synthesis has been shown to benefit patients with hereditary transthyretin amyloidosis.6 Using a name that references albumin, this protein has found the limelight as a marker of nutritional status.
The close similarity in appearance between starvation and wasting illness enables the strong, largely evidence-free7 emphasis on nutrition support. Many families and individuals suffer when a loved one loses weight. As a prominent reminder of serious illness, this wasted appearance can be painful to bear. Several caregivers may fear that they will be judged as neglectful by outside observers. Other individuals also wish to maintain their body weight for social reasons (as weight loss may be interpreted as a sign of illness, especially HIV). Nutrition maintains a special status in various contexts during the care of sick patients, and the drive to provide food to individuals who appear undernourished seems fundamental in humans.
A third reason for the frivolous, widespread overdiagnosis of “malnutrition” is that it leads directly to favorable consequences for the multibillion-dollar nutritional support industry. A consistent rational approach to the use of nutritional support products for sick people would lead to multibillion-dollar harm for that industry. For now, however, no self-respecting clinician could fail to provide nutritional support to a patient diagnosed as “malnourished” regardless of evidence.
The consistent rational approach in caring for patients is to search for good evidence of benefit before initiating a treatment course. Although sending blood tests for “nutritional markers” to diagnose nutritional needs may be easier and more popular, we caution against such over-simplification. Using prealbumin as a marker for malnutrition could lead to overlooking potentially treatable inflammatory or infectious illness. On the other hand, the use of prealbumin could also lead to unnecessary and potentially dangerous treatments, such as feeding tube placement and/or total parental nutrition. Thus, with one small amendment, we fully support Lacy and colleagues’ conclusion that prealbumin testing to identify malnutrition in hospitalized patients is a “Thing We Do For No (good) Reason.”
Disclosures
Drs. Lee and Finucane declare no financial conflicts of interest. Dr. Finucane discloses that he serves the pharmacy committee of an insurance company.
1. Lacy M, Roesch J, Langsjoen J. Things we do for no reason: prealbumin testing to diagnose malnutrition in the hospitalized patient. J Hosp Med. 2019;14(4):239-241. PubMed
2. Grobler L, Siegfried N, Visser ME, Mahlungulu SSN, Volmink J. Nutritional interventions for reducing morbidity and mortality in people with HIV. Cochrane Database Syst Rev. 2013;28(2):CD004536. PubMed
3. Koretz RL. Death, morbidity and economics are the only end points for trials. Proc Nutr Soc. 2005;64(3):277-284. PubMed
4. Lee JL, Oh ES, Lee RW, Finucane TE. Serum albumin and prealbumin in calorically restricted, nondiseased individuals: a systematic review. Am J Med. 2015;128(9):1023.e1-e22. PubMed
5. Prealbumin Blood Test. https://medlineplus.gov/lab-tests/prealbumin-blood-test/, updated June 14, 2018. Accessed November 12, 2018.
6. Benson MD, Waddington-Cruz M, Berk JL, et al. Inotersen treatment for patients with hereditary transthyretin amyloidosis. N Engl J Med. 2018;379(1):22-31. PubMed
7. U.S. dietary guidelines: an evidence-free zone. Ann Intern Med. 2016;164(8):558-559. PubMed
Lacy and colleagues identify an important “Thing We Do For No Reason”—prealbumin testing to diagnose malnutrition in hospitalized patients.1 They highlight the frequency and costs of ordering prealbumin tests although prealbumin is neither specific nor sensitive as a “marker of nutritional status,” shows
The term is also used to mean a condition where evidence shows better patient outcomes when improved nutrition is provided. Distinguishing between these two meanings is essential, as numerous patients with inflammatory illness will present abnormal “markers” when good evidence shows that they cannot benefit from nutritional support.
For example, a patient with advanced untreated human immunodeficiency virus (HIV) is likely to be considered malnourished because all of her “markers of nutritional status” are abnormal. She barely eats, has lost weight, and has low anthropometric, immunologic, and serologic measures, poor functional status, extreme vulnerability, and very poor prognosis. In this way she resembles a person in a famine situation. However, the patient is not malnourished in the sense that improved nutrient intake will lead to better patient outcomes. A Cochrane review of “nutritional interventions for reducing morbidity and mortality in people with HIV” found “no evidence that such supplementation translates into reductions in disease progression or HIV‐related complications, such as opportunistic infections or death.”2 The patient is dying of an inflammatory, cachectic illness. The same is true in managing patients with advanced cancer or several other serious illnesses.
Low prealbumin measures are associated with poor outcomes, which are then attributed to “malnutrition.” However, as Lacy and colleagues argue, prealbumin is a negative acute phase reactant and is thus a marker of the inflammatory effects of sickness/injury; it also responds variably to nutritional support. Citing Koretz, they note that “even when changes in nutritional markers are seen with nutritional support, the ‘changes in nutritional markers do not predict clinical outcomes.’”1,3 We know of no evidence from randomized controlled trials that prealbumin measurements help identify patients who can benefit from nutrition support.
By contrast, we and our colleagues have shown that in people who barely eat but show no inflammatory disease, eg, prison hunger-strikers and patients with anorexia nervosa, prealbumin level remains normal down to a body mass index below 13. The same is generally true for albumin.4 These measures fail to identify “malnutrition” in people who are starving.
Despite the complete lack of clinical trial evidence of benefit, prealbumin is widely used as an indicator of malnutrition. The National Institutes of Health’s Medline Plus website for the general public lists low prealbumin levels as a possible sign of malnutrition, for example, and advises that the prealbumin test may be used to “find out if you are getting enough nutrients, especially protein, in your diet” and to “check to see if you are getting enough nutrition if you are in the hospital.”5 Unjustified assertions such as these contribute to the dramatic overuse of nutritional interventions.
However, as a rule, things do occur for a reason. Using the term “prealbumin” conjures a certain relationship, perhaps as a precursor, to albumin, a venerable (but valueless) “marker of nutrition status.” In fact, the term refers only to a difference in electrophoretic mobility (prealbumin migrates faster). If prealbumin were called it by its proper name, transthyretin, it would probably have languished in obscurity among serum proteins until, in recent years, drug suppression of transthyretin synthesis has been shown to benefit patients with hereditary transthyretin amyloidosis.6 Using a name that references albumin, this protein has found the limelight as a marker of nutritional status.
The close similarity in appearance between starvation and wasting illness enables the strong, largely evidence-free7 emphasis on nutrition support. Many families and individuals suffer when a loved one loses weight. As a prominent reminder of serious illness, this wasted appearance can be painful to bear. Several caregivers may fear that they will be judged as neglectful by outside observers. Other individuals also wish to maintain their body weight for social reasons (as weight loss may be interpreted as a sign of illness, especially HIV). Nutrition maintains a special status in various contexts during the care of sick patients, and the drive to provide food to individuals who appear undernourished seems fundamental in humans.
A third reason for the frivolous, widespread overdiagnosis of “malnutrition” is that it leads directly to favorable consequences for the multibillion-dollar nutritional support industry. A consistent rational approach to the use of nutritional support products for sick people would lead to multibillion-dollar harm for that industry. For now, however, no self-respecting clinician could fail to provide nutritional support to a patient diagnosed as “malnourished” regardless of evidence.
The consistent rational approach in caring for patients is to search for good evidence of benefit before initiating a treatment course. Although sending blood tests for “nutritional markers” to diagnose nutritional needs may be easier and more popular, we caution against such over-simplification. Using prealbumin as a marker for malnutrition could lead to overlooking potentially treatable inflammatory or infectious illness. On the other hand, the use of prealbumin could also lead to unnecessary and potentially dangerous treatments, such as feeding tube placement and/or total parental nutrition. Thus, with one small amendment, we fully support Lacy and colleagues’ conclusion that prealbumin testing to identify malnutrition in hospitalized patients is a “Thing We Do For No (good) Reason.”
Disclosures
Drs. Lee and Finucane declare no financial conflicts of interest. Dr. Finucane discloses that he serves the pharmacy committee of an insurance company.
Lacy and colleagues identify an important “Thing We Do For No Reason”—prealbumin testing to diagnose malnutrition in hospitalized patients.1 They highlight the frequency and costs of ordering prealbumin tests although prealbumin is neither specific nor sensitive as a “marker of nutritional status,” shows
The term is also used to mean a condition where evidence shows better patient outcomes when improved nutrition is provided. Distinguishing between these two meanings is essential, as numerous patients with inflammatory illness will present abnormal “markers” when good evidence shows that they cannot benefit from nutritional support.
For example, a patient with advanced untreated human immunodeficiency virus (HIV) is likely to be considered malnourished because all of her “markers of nutritional status” are abnormal. She barely eats, has lost weight, and has low anthropometric, immunologic, and serologic measures, poor functional status, extreme vulnerability, and very poor prognosis. In this way she resembles a person in a famine situation. However, the patient is not malnourished in the sense that improved nutrient intake will lead to better patient outcomes. A Cochrane review of “nutritional interventions for reducing morbidity and mortality in people with HIV” found “no evidence that such supplementation translates into reductions in disease progression or HIV‐related complications, such as opportunistic infections or death.”2 The patient is dying of an inflammatory, cachectic illness. The same is true in managing patients with advanced cancer or several other serious illnesses.
Low prealbumin measures are associated with poor outcomes, which are then attributed to “malnutrition.” However, as Lacy and colleagues argue, prealbumin is a negative acute phase reactant and is thus a marker of the inflammatory effects of sickness/injury; it also responds variably to nutritional support. Citing Koretz, they note that “even when changes in nutritional markers are seen with nutritional support, the ‘changes in nutritional markers do not predict clinical outcomes.’”1,3 We know of no evidence from randomized controlled trials that prealbumin measurements help identify patients who can benefit from nutrition support.
By contrast, we and our colleagues have shown that in people who barely eat but show no inflammatory disease, eg, prison hunger-strikers and patients with anorexia nervosa, prealbumin level remains normal down to a body mass index below 13. The same is generally true for albumin.4 These measures fail to identify “malnutrition” in people who are starving.
Despite the complete lack of clinical trial evidence of benefit, prealbumin is widely used as an indicator of malnutrition. The National Institutes of Health’s Medline Plus website for the general public lists low prealbumin levels as a possible sign of malnutrition, for example, and advises that the prealbumin test may be used to “find out if you are getting enough nutrients, especially protein, in your diet” and to “check to see if you are getting enough nutrition if you are in the hospital.”5 Unjustified assertions such as these contribute to the dramatic overuse of nutritional interventions.
However, as a rule, things do occur for a reason. Using the term “prealbumin” conjures a certain relationship, perhaps as a precursor, to albumin, a venerable (but valueless) “marker of nutrition status.” In fact, the term refers only to a difference in electrophoretic mobility (prealbumin migrates faster). If prealbumin were called it by its proper name, transthyretin, it would probably have languished in obscurity among serum proteins until, in recent years, drug suppression of transthyretin synthesis has been shown to benefit patients with hereditary transthyretin amyloidosis.6 Using a name that references albumin, this protein has found the limelight as a marker of nutritional status.
The close similarity in appearance between starvation and wasting illness enables the strong, largely evidence-free7 emphasis on nutrition support. Many families and individuals suffer when a loved one loses weight. As a prominent reminder of serious illness, this wasted appearance can be painful to bear. Several caregivers may fear that they will be judged as neglectful by outside observers. Other individuals also wish to maintain their body weight for social reasons (as weight loss may be interpreted as a sign of illness, especially HIV). Nutrition maintains a special status in various contexts during the care of sick patients, and the drive to provide food to individuals who appear undernourished seems fundamental in humans.
A third reason for the frivolous, widespread overdiagnosis of “malnutrition” is that it leads directly to favorable consequences for the multibillion-dollar nutritional support industry. A consistent rational approach to the use of nutritional support products for sick people would lead to multibillion-dollar harm for that industry. For now, however, no self-respecting clinician could fail to provide nutritional support to a patient diagnosed as “malnourished” regardless of evidence.
The consistent rational approach in caring for patients is to search for good evidence of benefit before initiating a treatment course. Although sending blood tests for “nutritional markers” to diagnose nutritional needs may be easier and more popular, we caution against such over-simplification. Using prealbumin as a marker for malnutrition could lead to overlooking potentially treatable inflammatory or infectious illness. On the other hand, the use of prealbumin could also lead to unnecessary and potentially dangerous treatments, such as feeding tube placement and/or total parental nutrition. Thus, with one small amendment, we fully support Lacy and colleagues’ conclusion that prealbumin testing to identify malnutrition in hospitalized patients is a “Thing We Do For No (good) Reason.”
Disclosures
Drs. Lee and Finucane declare no financial conflicts of interest. Dr. Finucane discloses that he serves the pharmacy committee of an insurance company.
1. Lacy M, Roesch J, Langsjoen J. Things we do for no reason: prealbumin testing to diagnose malnutrition in the hospitalized patient. J Hosp Med. 2019;14(4):239-241. PubMed
2. Grobler L, Siegfried N, Visser ME, Mahlungulu SSN, Volmink J. Nutritional interventions for reducing morbidity and mortality in people with HIV. Cochrane Database Syst Rev. 2013;28(2):CD004536. PubMed
3. Koretz RL. Death, morbidity and economics are the only end points for trials. Proc Nutr Soc. 2005;64(3):277-284. PubMed
4. Lee JL, Oh ES, Lee RW, Finucane TE. Serum albumin and prealbumin in calorically restricted, nondiseased individuals: a systematic review. Am J Med. 2015;128(9):1023.e1-e22. PubMed
5. Prealbumin Blood Test. https://medlineplus.gov/lab-tests/prealbumin-blood-test/, updated June 14, 2018. Accessed November 12, 2018.
6. Benson MD, Waddington-Cruz M, Berk JL, et al. Inotersen treatment for patients with hereditary transthyretin amyloidosis. N Engl J Med. 2018;379(1):22-31. PubMed
7. U.S. dietary guidelines: an evidence-free zone. Ann Intern Med. 2016;164(8):558-559. PubMed
1. Lacy M, Roesch J, Langsjoen J. Things we do for no reason: prealbumin testing to diagnose malnutrition in the hospitalized patient. J Hosp Med. 2019;14(4):239-241. PubMed
2. Grobler L, Siegfried N, Visser ME, Mahlungulu SSN, Volmink J. Nutritional interventions for reducing morbidity and mortality in people with HIV. Cochrane Database Syst Rev. 2013;28(2):CD004536. PubMed
3. Koretz RL. Death, morbidity and economics are the only end points for trials. Proc Nutr Soc. 2005;64(3):277-284. PubMed
4. Lee JL, Oh ES, Lee RW, Finucane TE. Serum albumin and prealbumin in calorically restricted, nondiseased individuals: a systematic review. Am J Med. 2015;128(9):1023.e1-e22. PubMed
5. Prealbumin Blood Test. https://medlineplus.gov/lab-tests/prealbumin-blood-test/, updated June 14, 2018. Accessed November 12, 2018.
6. Benson MD, Waddington-Cruz M, Berk JL, et al. Inotersen treatment for patients with hereditary transthyretin amyloidosis. N Engl J Med. 2018;379(1):22-31. PubMed
7. U.S. dietary guidelines: an evidence-free zone. Ann Intern Med. 2016;164(8):558-559. PubMed
© 2019 Society of Hospital Medicine
Things We Do for No Reason: Routine Echocardiography in Hemodynamically Stable Patients with Acute Pulmonary Embolism
Inspired by the ABIM Foundation’s Choosing Wisely® campaign, the “Things We Do for No Reason” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A 28 year-old woman presents to the emergency department with acute onset bilateral chest pain and dyspnea. She has a respiratory rate of 28, a heart rate of 106, blood pressure of 110/65 mm Hg, and pulse oximetry of 92% saturation on room air. She has no history of cardiac or pulmonary disease and no personal history of venous thromboembolism. She takes an estrogen-containing oral contraceptive. On examination, she has no jugular venous distention, normal cardiac tones without murmur, and no lower extremity swelling. D-dimer is elevated at 3.4 mg/L (normal < 0.5 mg/L), and she undergoes computed tomography (CT) of the chest, which demonstrates acute segmental pulmonary emboli (PE) in the right upper and middle lobes as well as multiple bilateral subsegmental PEs. The CT suggests right ventricular dysfunction (RVD), and her troponin T is 0.06 ng/mL (normal < 0.01 ng/mL). Bilateral lower extremity venous Doppler ultrasonography demonstrates no acute thrombus.
BACKGROUND
Acute pulmonary embolism (PE) accounts for more than 300,000 inpatient admissions annually in the United States.1 The vast majority of patients with acute PE who receive adequate anticoagulation will have favorable outcomes.2,3 In the past two decades, for example, mortality has decreased significantly among patients admitted with acute PE,2 with 30-day all-cause mortality falling to approximately 5%.3 The risk-adjusted rate of recurrent venous thromboembolism (VTE) within 30 days has concomitantly dropped below 1%.3
Acute PE severity was previously classified as massive or high risk, submassive or intermediate risk, and low risk.4 Massive PE was defined by RVD and persistent hypotension or shock requiring vasopressors. 4 Intermediate-risk or submassive PE typically referred to normotensive patients with RVD and/or myocardial necrosis (eg, elevated troponin).4,5 Low-risk PEs had neither hemodynamic instability nor RVD. This classification scheme, however, has fallen out of favor as PE severity exists on a risk spectrum.6 Instead, recent guidelines from the European Society of Cardiology and the American College of Chest Physicians recommend first parsing PE severity by the presence or absence of hypotension (Figure 1).6,7 Risk assessment can be subsequently enhanced by validated clinical risk prediction scores, imaging-based assessment of RVD, and cardiac biomarker testing.6
In acute PE, hypotension and/or shock are associated with a 12%-35% risk of short-term mortality.2,3,8 Accordingly, patients with high-risk PE, who comprise 3%-12% of hospitalizations for PE,2,3,8 typically receive more intensive monitoring and treatment.2,8,9 In addition to systemic anticoagulation, thrombolysis is generally recommended for hypotensive patients with PE and no contraindications.6,7
Between 7% and 59% of patients with acute PE are hemodynamically stable but have objective evidence of myocardial necrosis and/or RVD.8,10,11 Among these patients, fewer than 10% will have a complicated course as defined by all-cause death, hemodynamic collapse, or recurrent PE in the first month after diagnosis,11 and short-term PE-related mortality rates range from approximately 2%-5%.5,8,11
WHY YOU MIGHT THINK ECHOCARDIOGRAPHY IS HELPFUL IN HEMODYNAMICALLY STABLE ACUTE PE
Echocardiography is a common method for evaluating RVD, and echocardiographic RVD confers an increased risk of adverse outcomes in PE.10-12 In the earliest meta-analysis to evaluate this association, Sanchez et al. combined data from five studies that included 623 patients from emergency room and inpatient settings. They found that echocardiographic RVD conferred an unadjusted relative risk for short-term mortality of 2.53 (95%CI 1.17-5.50).12 A subsequent meta-analysis by Cho et al. pooled data from both prospective and retrospective cohorts to examine short-term mortality in a total of 3,283 hemodynamically stable patients with PE, of whom 1,223 (37.3%) had RVD diagnosed by echocardiogram.10 In this population, RVD was associated with an odds ratio of 2.29 (95%CI 1.61-3.26) for short-term death. Thus, echocardiography could be viewed as a risk stratification tool, even in hemodynamically stable PE.
WHY ECHOCARDIOGRAPHY IN HEMODYNAMICALLY
STABLE ACUTE PE IS NOT AS HELPFUL AS YOU THINK
For most hemodynamically stable patients, echocardiographic findings will not enhance prognostication and/or have a therapeutic impact. The following four reasons explain why echocardiography adds little value to the care of these patients.
First, phenotypic expression of RVD varies from asymptomatic, despite abnormalities on diagnostic testing, to obstructive shock. Unfortunately, available prognostic models classify echocardiographic RVD in a binary fashion (present/absent)4,7,10 whereas RVD exists on a continuum. Consequently, RVD is commonly found in acute PE8,10,11 and has been identified in more than half of patients hospitalized with PE referred for echocardiography.8 Existing data do not allow clinicians to judge the clinical impact of the severity of echocardiographic RVD,8 and only the phenotypic expression of refractory hypotension has clear therapeutic implications.6,7
Second, while echocardiographic RVD is associated with short-term mortality,10-12 absolute rates of adverse outcomes are quite low when RVD is identified. For example, in a study merging multiple prospective cohorts, Becattini et al. demonstrated that RVD diagnosed by echocardiography or CT occurred in 41% of hospitalized patients stratified to low-risk PE by the simplified Pulmonary Embolism Severity Index (sPESI).8 For these patients, the 30-day mortality was 1.2%,8 which approximates the expected mortality from a low-risk sPESI score alone (1.1%).13 Even among intermediate-risk acute PE patients with RVD and/or elevated troponin enrolled in thrombolysis trials, the overall risk of death at 30 days was approximately 2%-3%, irrespective of the treatment arm.5,14,15
Third, RVD identified by echocardiography does not inform or enhance prognostication as compared with cardiac biomarker testing. In a meta-analysis by Sanchez et al., echocardiographic RVD predicted death with a risk ratio of 2.53 (95% CI 1.17-5.50).12 However, both elevated cardiac troponin and brain natriuretic peptide indicated a significantly worse outcome than imaging findings, with risk ratios of 8.3 (95% CI 3.6-19.3) and 9.5 (95% CI 3.2-28.6), respectively.13 More recently, Jiménez derived and validated a multivariable risk prediction model for stable PE.11 In their data, echocardiographic RVD had an unadjusted odds ratio of 2.62 (95% CI 1.54-4.45) for predicting a 30-day complicated course. After multivariable adjustment that included sPESI scores, lower extremity ultrasound results, and cardiac biomarker testing, these odds became insignificant.11 In other words, identifying echocardiographic RVD did not improve prognostication in hemodynamically stable PE patients when other commonly available variables were used.
Finally, in hemodynamically stable patients, echocardiographic RVD might create patient anxiety and cause harm. In a recent retrospective cohort study of 64,037 stable patients with PE, exposure to echocardiography was associated with a five-fold increase in likelihood of having received thrombolysis without any significant differences in risk-adjusted mortality.16 These data suggest that when faced with an abnormal echocardiogram, clinicians and patients may opt for more aggressive, time-sensitive therapies. Basing thrombolysis decisions on echocardiographic RVD potentially subjects patients to harm without decreasing mortality.5,14,15 For example, the PEITHO study, which was the largest randomized trial evaluating thrombolysis in intermediate-risk acute PE, enrolled 1,006 patients and demonstrated that treating 29 intermediate-risk patients with thrombolysis prevented one case of hemodynamic decompensation.5 These benefits were counterbalanced by a number needed to harm of 14 to cause stroke or major bleeding. Ominous echocardiographic findings may also bias clinicians toward more intensive monitoring. Rates of echocardiogram utilization in hemodynamically stable PE are linked to higher rates of ICU admission and longer hospital stays without significant impact on patient outcomes.16
WHEN ECHOCARDIOGRAPHY MIGHT BE HELPFUL IN HEMODYNAMICALLY STABLE PATIENTS WITH PE
Echocardiography should be used to exclude other causes of hypotension in patients with presumed PE-related shock7,9 and to improve clinicians’ confidence prescribing systemic thrombolytics in the face of hemodynamic instability.6,7 Otherwise, echocardiography should be reserved for highly selected intermediate-risk patients with acute PE. Among patients with intermediate-risk PE, those most likely to decompensate or die typically satisfy all of the following conditions: (1) highest-risk PESI or sPESI scores, (2) elevated natriuretic peptides, (3) elevated troponin, and (4) proximal deep vein thrombosis (DVT) on lower extremity ultrasound.11,13 In such patients, the echocardiogram may reveal a critical “tipping point,” such as a right atrial or ventricular thrombus-in-transit, that may warrant more intensive monitoring and multidisciplinary input into the most appropriate treatment plan.
Echocardiography could aid therapeutic decisions when the benefits from thrombolysis may outweigh the risks, such as for patients with minimal physiologic reserve and/or a low risk of major bleeding complications. Prognostic models like sPESI utilize binary variables, such as the presence/absence of chronic cardiopulmonary disease or oxygen saturation above/below 90%. Clearly, these variables exist on a spectrum; intuitively, patients with severe comorbidities and more alarming vital signs have a higher risk of death or decompensation than predicted by sPESI. Analogously, echocardiographic findings of RVD also encompass a spectrum. Because prognostic models and clinical trials cannot guide decisions for each individual patient, clinicians could justify using echocardiography to “fine tune” prognostication and to provide a personalized approach for carefully selected patients.
WHAT SHOULD YOU DO INSTEAD?
Clinicians should use a risk prediction model for all hemodynamically stable patients with confirmed PE.6,7 Validated risk calculators include the sPESI,6,7,14 which relies exclusively on the patient’s history and vital signs, and the eStiMaTe© tool (www.peprognosis.org), which enhances prognostication from sPESI by incorporating troponin, natriuretic peptide, and lower- extremity Doppler results. 11 For patients with symptoms or physical signs of RVD, chest CT and cardiac biomarkers (ie, troponin and/or natriuretic peptides) are sufficient for prognostication.11,14 In intermediate-risk patients with the highest risk for decompensation based on risk prediction scores, the echocardiogram should represent a part of a comprehensive clinical evaluation, not the sole criterion for intensive monitoring and aggressive treatment.
RECOMMENDATIONS
- Clinicians should use a validated tool, such as the sPESI, for initial risk stratification of hemodynamically stable patients with acute pulmonary embolism.
- Hemodynamically unstable patients with confirmed or suspected acute PE may benefit from early echocardiography to confirm RVD as the cause of shock.6,7,9
- The majority of normotensive adults with acute PE should not undergo echocardiography. To identify the patients at the greatest risk for decompensation, clinicians may consider using the eStiMaTe© tool (www.peprognosis.org), which augments risk stratification afforded by sPESI.
- For hemodynamically stable patients with PE who have already undergone echocardiography, clinicians should avoid being biased by the finding of RVD, particularly if other prognostic markers are reassuring.
CONCLUSION
In evaluating the patient described earlier, echocardiography has no clear prognostic implications. Her admission sPESI score equals zero, predicting a 30-day mortality of 1.1%. Including her lower extremity ultrasound and troponin T results into the eStiMaTe© calculator (www.peprognosis.org) surprisingly predicts an even lower rate of 30-day mortality (0.4%) and low risk of a complicated course (2.4%). Assessing for RVD on echocardiography may increase her risk of unnecessary and potentially injurious interventions.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailingTWDFNR@hospitalmedicine.org.
Disclosures
The authors have no conflicts of interest relevant to this article.
1. Centers for Disease Control and Prevention (CDC). Venous thromboembolism in adult hospitalizations, United States, 2007-2009. Morbidity and mortality weekly report (MMWR). 2012;61(22):401-40. Available: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6122a1.htm. Accessed May 7, 2018.
2. Stein PD, Matta F, Alrifai A, Rahman A. Trends in case fatality rate in pulmonary embolism according to stability and treatment. Thromb Res. 2012;130(6):841-846. PubMed
3. Jiménez D, de Miguel-Díez J, Guijarro R, et al. Trends in the management and outcomes of acute pulmonary embolism: analysis from the RIETE Registry. J Am Coll Cardiol. 2016;67(2):162-170. PubMed
4. Jaff MR, McMurtry MS, Archer SL, et al. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011;123(16):1788-1830. PubMed
5. Meyer G, Vicaut E, Danays T, et al. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N Engl J Med. 2014;370:1402-1411. PubMed
6. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest. 2016;49(2):315-352. PubMed
7. Konstantinides SV, Torbicki A, Agnelli G, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2014;35(43):3033-69, 3069a-3069k. PubMed
8. Becattini C, Agnelli G, Lankeit M, et al. Acute pulmonary embolism: mortality prediction by the 2014 European Society of Cardiology risk stratification model. Eur Respir J. 2016;48(3):780-786. PubMed
9. Levitov A, Frankel HL, Blaivas M, et al. Guidelines for the appropriate use of bedside general and cardiac ultrasonography in the evaluation of critically ill patients-part II: Cardiac Ultrasonography. Crit Care Med. 2016;44(6):1206-1227. PubMed
10. Cho JH, Kutti Sridharan G, Kim SH, et al. Right ventricular dysfunction as an echocardiographic prognostic factor in hemodynamically stable patients with acute pulmonary embolism: a meta-analysis. BMC Cardiovasc Disord. 2014;14:64. PubMed
11. Jiménez D, Kopecna D, Tapson V, et al. Derivation and validation of multimarker prognostication for normotensive patients with acute symptomatic pulmonary embolism. Am J Respir Crit Care Med. 2014;189(6):718-726. PubMed
12. Sanchez O, Trinquart L, Colombet I, et al. Prognostic value of right ventricular dysfunction in patients with haemodynamically stable pulmonary embolism: a systematic review. Eur Heart J. 2008;29(12):1569-1577. PubMed
13. Elias A, Mallett S, Daoud-Elias M, Poggi JN, Clarke M. Prognostic models in acute pulmonary embolism: a systematic review and meta-analysis. BMJ Open. 2016;6(4):e010324. PubMed
14. Konstantinides S, Geibel A, Heusel G, et al. Heparin plus alteplase compared with heparin alone in patients with submassive pulmonary embolism. N Engl J Med. 2002;347(15):1143-1150. PubMed
15. Kline JA, Nordenholz KE, Courtney DM, et al. Treatment of submassive pulmonary embolism with tenecteplase or placebo: cardiopulmonary outcomes at 3 months: multicenter double-blind, placebo-controlled randomized trial. J Thromb Haemost. 2014;12(4):459-468. PubMed
16. Cohen DM, Winter M, Lindenauer PK, Walkey AJ. Echocardiogram in the evaluation of hemodynamically stable acute pulmonary embolism: national practices and clinical outcomes. Ann Am Thorac Soc. 2018;15(5):581-588. PubMed
Inspired by the ABIM Foundation’s Choosing Wisely® campaign, the “Things We Do for No Reason” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A 28 year-old woman presents to the emergency department with acute onset bilateral chest pain and dyspnea. She has a respiratory rate of 28, a heart rate of 106, blood pressure of 110/65 mm Hg, and pulse oximetry of 92% saturation on room air. She has no history of cardiac or pulmonary disease and no personal history of venous thromboembolism. She takes an estrogen-containing oral contraceptive. On examination, she has no jugular venous distention, normal cardiac tones without murmur, and no lower extremity swelling. D-dimer is elevated at 3.4 mg/L (normal < 0.5 mg/L), and she undergoes computed tomography (CT) of the chest, which demonstrates acute segmental pulmonary emboli (PE) in the right upper and middle lobes as well as multiple bilateral subsegmental PEs. The CT suggests right ventricular dysfunction (RVD), and her troponin T is 0.06 ng/mL (normal < 0.01 ng/mL). Bilateral lower extremity venous Doppler ultrasonography demonstrates no acute thrombus.
BACKGROUND
Acute pulmonary embolism (PE) accounts for more than 300,000 inpatient admissions annually in the United States.1 The vast majority of patients with acute PE who receive adequate anticoagulation will have favorable outcomes.2,3 In the past two decades, for example, mortality has decreased significantly among patients admitted with acute PE,2 with 30-day all-cause mortality falling to approximately 5%.3 The risk-adjusted rate of recurrent venous thromboembolism (VTE) within 30 days has concomitantly dropped below 1%.3
Acute PE severity was previously classified as massive or high risk, submassive or intermediate risk, and low risk.4 Massive PE was defined by RVD and persistent hypotension or shock requiring vasopressors. 4 Intermediate-risk or submassive PE typically referred to normotensive patients with RVD and/or myocardial necrosis (eg, elevated troponin).4,5 Low-risk PEs had neither hemodynamic instability nor RVD. This classification scheme, however, has fallen out of favor as PE severity exists on a risk spectrum.6 Instead, recent guidelines from the European Society of Cardiology and the American College of Chest Physicians recommend first parsing PE severity by the presence or absence of hypotension (Figure 1).6,7 Risk assessment can be subsequently enhanced by validated clinical risk prediction scores, imaging-based assessment of RVD, and cardiac biomarker testing.6
In acute PE, hypotension and/or shock are associated with a 12%-35% risk of short-term mortality.2,3,8 Accordingly, patients with high-risk PE, who comprise 3%-12% of hospitalizations for PE,2,3,8 typically receive more intensive monitoring and treatment.2,8,9 In addition to systemic anticoagulation, thrombolysis is generally recommended for hypotensive patients with PE and no contraindications.6,7
Between 7% and 59% of patients with acute PE are hemodynamically stable but have objective evidence of myocardial necrosis and/or RVD.8,10,11 Among these patients, fewer than 10% will have a complicated course as defined by all-cause death, hemodynamic collapse, or recurrent PE in the first month after diagnosis,11 and short-term PE-related mortality rates range from approximately 2%-5%.5,8,11
WHY YOU MIGHT THINK ECHOCARDIOGRAPHY IS HELPFUL IN HEMODYNAMICALLY STABLE ACUTE PE
Echocardiography is a common method for evaluating RVD, and echocardiographic RVD confers an increased risk of adverse outcomes in PE.10-12 In the earliest meta-analysis to evaluate this association, Sanchez et al. combined data from five studies that included 623 patients from emergency room and inpatient settings. They found that echocardiographic RVD conferred an unadjusted relative risk for short-term mortality of 2.53 (95%CI 1.17-5.50).12 A subsequent meta-analysis by Cho et al. pooled data from both prospective and retrospective cohorts to examine short-term mortality in a total of 3,283 hemodynamically stable patients with PE, of whom 1,223 (37.3%) had RVD diagnosed by echocardiogram.10 In this population, RVD was associated with an odds ratio of 2.29 (95%CI 1.61-3.26) for short-term death. Thus, echocardiography could be viewed as a risk stratification tool, even in hemodynamically stable PE.
WHY ECHOCARDIOGRAPHY IN HEMODYNAMICALLY
STABLE ACUTE PE IS NOT AS HELPFUL AS YOU THINK
For most hemodynamically stable patients, echocardiographic findings will not enhance prognostication and/or have a therapeutic impact. The following four reasons explain why echocardiography adds little value to the care of these patients.
First, phenotypic expression of RVD varies from asymptomatic, despite abnormalities on diagnostic testing, to obstructive shock. Unfortunately, available prognostic models classify echocardiographic RVD in a binary fashion (present/absent)4,7,10 whereas RVD exists on a continuum. Consequently, RVD is commonly found in acute PE8,10,11 and has been identified in more than half of patients hospitalized with PE referred for echocardiography.8 Existing data do not allow clinicians to judge the clinical impact of the severity of echocardiographic RVD,8 and only the phenotypic expression of refractory hypotension has clear therapeutic implications.6,7
Second, while echocardiographic RVD is associated with short-term mortality,10-12 absolute rates of adverse outcomes are quite low when RVD is identified. For example, in a study merging multiple prospective cohorts, Becattini et al. demonstrated that RVD diagnosed by echocardiography or CT occurred in 41% of hospitalized patients stratified to low-risk PE by the simplified Pulmonary Embolism Severity Index (sPESI).8 For these patients, the 30-day mortality was 1.2%,8 which approximates the expected mortality from a low-risk sPESI score alone (1.1%).13 Even among intermediate-risk acute PE patients with RVD and/or elevated troponin enrolled in thrombolysis trials, the overall risk of death at 30 days was approximately 2%-3%, irrespective of the treatment arm.5,14,15
Third, RVD identified by echocardiography does not inform or enhance prognostication as compared with cardiac biomarker testing. In a meta-analysis by Sanchez et al., echocardiographic RVD predicted death with a risk ratio of 2.53 (95% CI 1.17-5.50).12 However, both elevated cardiac troponin and brain natriuretic peptide indicated a significantly worse outcome than imaging findings, with risk ratios of 8.3 (95% CI 3.6-19.3) and 9.5 (95% CI 3.2-28.6), respectively.13 More recently, Jiménez derived and validated a multivariable risk prediction model for stable PE.11 In their data, echocardiographic RVD had an unadjusted odds ratio of 2.62 (95% CI 1.54-4.45) for predicting a 30-day complicated course. After multivariable adjustment that included sPESI scores, lower extremity ultrasound results, and cardiac biomarker testing, these odds became insignificant.11 In other words, identifying echocardiographic RVD did not improve prognostication in hemodynamically stable PE patients when other commonly available variables were used.
Finally, in hemodynamically stable patients, echocardiographic RVD might create patient anxiety and cause harm. In a recent retrospective cohort study of 64,037 stable patients with PE, exposure to echocardiography was associated with a five-fold increase in likelihood of having received thrombolysis without any significant differences in risk-adjusted mortality.16 These data suggest that when faced with an abnormal echocardiogram, clinicians and patients may opt for more aggressive, time-sensitive therapies. Basing thrombolysis decisions on echocardiographic RVD potentially subjects patients to harm without decreasing mortality.5,14,15 For example, the PEITHO study, which was the largest randomized trial evaluating thrombolysis in intermediate-risk acute PE, enrolled 1,006 patients and demonstrated that treating 29 intermediate-risk patients with thrombolysis prevented one case of hemodynamic decompensation.5 These benefits were counterbalanced by a number needed to harm of 14 to cause stroke or major bleeding. Ominous echocardiographic findings may also bias clinicians toward more intensive monitoring. Rates of echocardiogram utilization in hemodynamically stable PE are linked to higher rates of ICU admission and longer hospital stays without significant impact on patient outcomes.16
WHEN ECHOCARDIOGRAPHY MIGHT BE HELPFUL IN HEMODYNAMICALLY STABLE PATIENTS WITH PE
Echocardiography should be used to exclude other causes of hypotension in patients with presumed PE-related shock7,9 and to improve clinicians’ confidence prescribing systemic thrombolytics in the face of hemodynamic instability.6,7 Otherwise, echocardiography should be reserved for highly selected intermediate-risk patients with acute PE. Among patients with intermediate-risk PE, those most likely to decompensate or die typically satisfy all of the following conditions: (1) highest-risk PESI or sPESI scores, (2) elevated natriuretic peptides, (3) elevated troponin, and (4) proximal deep vein thrombosis (DVT) on lower extremity ultrasound.11,13 In such patients, the echocardiogram may reveal a critical “tipping point,” such as a right atrial or ventricular thrombus-in-transit, that may warrant more intensive monitoring and multidisciplinary input into the most appropriate treatment plan.
Echocardiography could aid therapeutic decisions when the benefits from thrombolysis may outweigh the risks, such as for patients with minimal physiologic reserve and/or a low risk of major bleeding complications. Prognostic models like sPESI utilize binary variables, such as the presence/absence of chronic cardiopulmonary disease or oxygen saturation above/below 90%. Clearly, these variables exist on a spectrum; intuitively, patients with severe comorbidities and more alarming vital signs have a higher risk of death or decompensation than predicted by sPESI. Analogously, echocardiographic findings of RVD also encompass a spectrum. Because prognostic models and clinical trials cannot guide decisions for each individual patient, clinicians could justify using echocardiography to “fine tune” prognostication and to provide a personalized approach for carefully selected patients.
WHAT SHOULD YOU DO INSTEAD?
Clinicians should use a risk prediction model for all hemodynamically stable patients with confirmed PE.6,7 Validated risk calculators include the sPESI,6,7,14 which relies exclusively on the patient’s history and vital signs, and the eStiMaTe© tool (www.peprognosis.org), which enhances prognostication from sPESI by incorporating troponin, natriuretic peptide, and lower- extremity Doppler results. 11 For patients with symptoms or physical signs of RVD, chest CT and cardiac biomarkers (ie, troponin and/or natriuretic peptides) are sufficient for prognostication.11,14 In intermediate-risk patients with the highest risk for decompensation based on risk prediction scores, the echocardiogram should represent a part of a comprehensive clinical evaluation, not the sole criterion for intensive monitoring and aggressive treatment.
RECOMMENDATIONS
- Clinicians should use a validated tool, such as the sPESI, for initial risk stratification of hemodynamically stable patients with acute pulmonary embolism.
- Hemodynamically unstable patients with confirmed or suspected acute PE may benefit from early echocardiography to confirm RVD as the cause of shock.6,7,9
- The majority of normotensive adults with acute PE should not undergo echocardiography. To identify the patients at the greatest risk for decompensation, clinicians may consider using the eStiMaTe© tool (www.peprognosis.org), which augments risk stratification afforded by sPESI.
- For hemodynamically stable patients with PE who have already undergone echocardiography, clinicians should avoid being biased by the finding of RVD, particularly if other prognostic markers are reassuring.
CONCLUSION
In evaluating the patient described earlier, echocardiography has no clear prognostic implications. Her admission sPESI score equals zero, predicting a 30-day mortality of 1.1%. Including her lower extremity ultrasound and troponin T results into the eStiMaTe© calculator (www.peprognosis.org) surprisingly predicts an even lower rate of 30-day mortality (0.4%) and low risk of a complicated course (2.4%). Assessing for RVD on echocardiography may increase her risk of unnecessary and potentially injurious interventions.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailingTWDFNR@hospitalmedicine.org.
Disclosures
The authors have no conflicts of interest relevant to this article.
Inspired by the ABIM Foundation’s Choosing Wisely® campaign, the “Things We Do for No Reason” (TWDFNR) series reviews practices that have become common parts of hospital care but may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
CLINICAL SCENARIO
A 28 year-old woman presents to the emergency department with acute onset bilateral chest pain and dyspnea. She has a respiratory rate of 28, a heart rate of 106, blood pressure of 110/65 mm Hg, and pulse oximetry of 92% saturation on room air. She has no history of cardiac or pulmonary disease and no personal history of venous thromboembolism. She takes an estrogen-containing oral contraceptive. On examination, she has no jugular venous distention, normal cardiac tones without murmur, and no lower extremity swelling. D-dimer is elevated at 3.4 mg/L (normal < 0.5 mg/L), and she undergoes computed tomography (CT) of the chest, which demonstrates acute segmental pulmonary emboli (PE) in the right upper and middle lobes as well as multiple bilateral subsegmental PEs. The CT suggests right ventricular dysfunction (RVD), and her troponin T is 0.06 ng/mL (normal < 0.01 ng/mL). Bilateral lower extremity venous Doppler ultrasonography demonstrates no acute thrombus.
BACKGROUND
Acute pulmonary embolism (PE) accounts for more than 300,000 inpatient admissions annually in the United States.1 The vast majority of patients with acute PE who receive adequate anticoagulation will have favorable outcomes.2,3 In the past two decades, for example, mortality has decreased significantly among patients admitted with acute PE,2 with 30-day all-cause mortality falling to approximately 5%.3 The risk-adjusted rate of recurrent venous thromboembolism (VTE) within 30 days has concomitantly dropped below 1%.3
Acute PE severity was previously classified as massive or high risk, submassive or intermediate risk, and low risk.4 Massive PE was defined by RVD and persistent hypotension or shock requiring vasopressors. 4 Intermediate-risk or submassive PE typically referred to normotensive patients with RVD and/or myocardial necrosis (eg, elevated troponin).4,5 Low-risk PEs had neither hemodynamic instability nor RVD. This classification scheme, however, has fallen out of favor as PE severity exists on a risk spectrum.6 Instead, recent guidelines from the European Society of Cardiology and the American College of Chest Physicians recommend first parsing PE severity by the presence or absence of hypotension (Figure 1).6,7 Risk assessment can be subsequently enhanced by validated clinical risk prediction scores, imaging-based assessment of RVD, and cardiac biomarker testing.6
In acute PE, hypotension and/or shock are associated with a 12%-35% risk of short-term mortality.2,3,8 Accordingly, patients with high-risk PE, who comprise 3%-12% of hospitalizations for PE,2,3,8 typically receive more intensive monitoring and treatment.2,8,9 In addition to systemic anticoagulation, thrombolysis is generally recommended for hypotensive patients with PE and no contraindications.6,7
Between 7% and 59% of patients with acute PE are hemodynamically stable but have objective evidence of myocardial necrosis and/or RVD.8,10,11 Among these patients, fewer than 10% will have a complicated course as defined by all-cause death, hemodynamic collapse, or recurrent PE in the first month after diagnosis,11 and short-term PE-related mortality rates range from approximately 2%-5%.5,8,11
WHY YOU MIGHT THINK ECHOCARDIOGRAPHY IS HELPFUL IN HEMODYNAMICALLY STABLE ACUTE PE
Echocardiography is a common method for evaluating RVD, and echocardiographic RVD confers an increased risk of adverse outcomes in PE.10-12 In the earliest meta-analysis to evaluate this association, Sanchez et al. combined data from five studies that included 623 patients from emergency room and inpatient settings. They found that echocardiographic RVD conferred an unadjusted relative risk for short-term mortality of 2.53 (95%CI 1.17-5.50).12 A subsequent meta-analysis by Cho et al. pooled data from both prospective and retrospective cohorts to examine short-term mortality in a total of 3,283 hemodynamically stable patients with PE, of whom 1,223 (37.3%) had RVD diagnosed by echocardiogram.10 In this population, RVD was associated with an odds ratio of 2.29 (95%CI 1.61-3.26) for short-term death. Thus, echocardiography could be viewed as a risk stratification tool, even in hemodynamically stable PE.
WHY ECHOCARDIOGRAPHY IN HEMODYNAMICALLY
STABLE ACUTE PE IS NOT AS HELPFUL AS YOU THINK
For most hemodynamically stable patients, echocardiographic findings will not enhance prognostication and/or have a therapeutic impact. The following four reasons explain why echocardiography adds little value to the care of these patients.
First, phenotypic expression of RVD varies from asymptomatic, despite abnormalities on diagnostic testing, to obstructive shock. Unfortunately, available prognostic models classify echocardiographic RVD in a binary fashion (present/absent)4,7,10 whereas RVD exists on a continuum. Consequently, RVD is commonly found in acute PE8,10,11 and has been identified in more than half of patients hospitalized with PE referred for echocardiography.8 Existing data do not allow clinicians to judge the clinical impact of the severity of echocardiographic RVD,8 and only the phenotypic expression of refractory hypotension has clear therapeutic implications.6,7
Second, while echocardiographic RVD is associated with short-term mortality,10-12 absolute rates of adverse outcomes are quite low when RVD is identified. For example, in a study merging multiple prospective cohorts, Becattini et al. demonstrated that RVD diagnosed by echocardiography or CT occurred in 41% of hospitalized patients stratified to low-risk PE by the simplified Pulmonary Embolism Severity Index (sPESI).8 For these patients, the 30-day mortality was 1.2%,8 which approximates the expected mortality from a low-risk sPESI score alone (1.1%).13 Even among intermediate-risk acute PE patients with RVD and/or elevated troponin enrolled in thrombolysis trials, the overall risk of death at 30 days was approximately 2%-3%, irrespective of the treatment arm.5,14,15
Third, RVD identified by echocardiography does not inform or enhance prognostication as compared with cardiac biomarker testing. In a meta-analysis by Sanchez et al., echocardiographic RVD predicted death with a risk ratio of 2.53 (95% CI 1.17-5.50).12 However, both elevated cardiac troponin and brain natriuretic peptide indicated a significantly worse outcome than imaging findings, with risk ratios of 8.3 (95% CI 3.6-19.3) and 9.5 (95% CI 3.2-28.6), respectively.13 More recently, Jiménez derived and validated a multivariable risk prediction model for stable PE.11 In their data, echocardiographic RVD had an unadjusted odds ratio of 2.62 (95% CI 1.54-4.45) for predicting a 30-day complicated course. After multivariable adjustment that included sPESI scores, lower extremity ultrasound results, and cardiac biomarker testing, these odds became insignificant.11 In other words, identifying echocardiographic RVD did not improve prognostication in hemodynamically stable PE patients when other commonly available variables were used.
Finally, in hemodynamically stable patients, echocardiographic RVD might create patient anxiety and cause harm. In a recent retrospective cohort study of 64,037 stable patients with PE, exposure to echocardiography was associated with a five-fold increase in likelihood of having received thrombolysis without any significant differences in risk-adjusted mortality.16 These data suggest that when faced with an abnormal echocardiogram, clinicians and patients may opt for more aggressive, time-sensitive therapies. Basing thrombolysis decisions on echocardiographic RVD potentially subjects patients to harm without decreasing mortality.5,14,15 For example, the PEITHO study, which was the largest randomized trial evaluating thrombolysis in intermediate-risk acute PE, enrolled 1,006 patients and demonstrated that treating 29 intermediate-risk patients with thrombolysis prevented one case of hemodynamic decompensation.5 These benefits were counterbalanced by a number needed to harm of 14 to cause stroke or major bleeding. Ominous echocardiographic findings may also bias clinicians toward more intensive monitoring. Rates of echocardiogram utilization in hemodynamically stable PE are linked to higher rates of ICU admission and longer hospital stays without significant impact on patient outcomes.16
WHEN ECHOCARDIOGRAPHY MIGHT BE HELPFUL IN HEMODYNAMICALLY STABLE PATIENTS WITH PE
Echocardiography should be used to exclude other causes of hypotension in patients with presumed PE-related shock7,9 and to improve clinicians’ confidence prescribing systemic thrombolytics in the face of hemodynamic instability.6,7 Otherwise, echocardiography should be reserved for highly selected intermediate-risk patients with acute PE. Among patients with intermediate-risk PE, those most likely to decompensate or die typically satisfy all of the following conditions: (1) highest-risk PESI or sPESI scores, (2) elevated natriuretic peptides, (3) elevated troponin, and (4) proximal deep vein thrombosis (DVT) on lower extremity ultrasound.11,13 In such patients, the echocardiogram may reveal a critical “tipping point,” such as a right atrial or ventricular thrombus-in-transit, that may warrant more intensive monitoring and multidisciplinary input into the most appropriate treatment plan.
Echocardiography could aid therapeutic decisions when the benefits from thrombolysis may outweigh the risks, such as for patients with minimal physiologic reserve and/or a low risk of major bleeding complications. Prognostic models like sPESI utilize binary variables, such as the presence/absence of chronic cardiopulmonary disease or oxygen saturation above/below 90%. Clearly, these variables exist on a spectrum; intuitively, patients with severe comorbidities and more alarming vital signs have a higher risk of death or decompensation than predicted by sPESI. Analogously, echocardiographic findings of RVD also encompass a spectrum. Because prognostic models and clinical trials cannot guide decisions for each individual patient, clinicians could justify using echocardiography to “fine tune” prognostication and to provide a personalized approach for carefully selected patients.
WHAT SHOULD YOU DO INSTEAD?
Clinicians should use a risk prediction model for all hemodynamically stable patients with confirmed PE.6,7 Validated risk calculators include the sPESI,6,7,14 which relies exclusively on the patient’s history and vital signs, and the eStiMaTe© tool (www.peprognosis.org), which enhances prognostication from sPESI by incorporating troponin, natriuretic peptide, and lower- extremity Doppler results. 11 For patients with symptoms or physical signs of RVD, chest CT and cardiac biomarkers (ie, troponin and/or natriuretic peptides) are sufficient for prognostication.11,14 In intermediate-risk patients with the highest risk for decompensation based on risk prediction scores, the echocardiogram should represent a part of a comprehensive clinical evaluation, not the sole criterion for intensive monitoring and aggressive treatment.
RECOMMENDATIONS
- Clinicians should use a validated tool, such as the sPESI, for initial risk stratification of hemodynamically stable patients with acute pulmonary embolism.
- Hemodynamically unstable patients with confirmed or suspected acute PE may benefit from early echocardiography to confirm RVD as the cause of shock.6,7,9
- The majority of normotensive adults with acute PE should not undergo echocardiography. To identify the patients at the greatest risk for decompensation, clinicians may consider using the eStiMaTe© tool (www.peprognosis.org), which augments risk stratification afforded by sPESI.
- For hemodynamically stable patients with PE who have already undergone echocardiography, clinicians should avoid being biased by the finding of RVD, particularly if other prognostic markers are reassuring.
CONCLUSION
In evaluating the patient described earlier, echocardiography has no clear prognostic implications. Her admission sPESI score equals zero, predicting a 30-day mortality of 1.1%. Including her lower extremity ultrasound and troponin T results into the eStiMaTe© calculator (www.peprognosis.org) surprisingly predicts an even lower rate of 30-day mortality (0.4%) and low risk of a complicated course (2.4%). Assessing for RVD on echocardiography may increase her risk of unnecessary and potentially injurious interventions.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailingTWDFNR@hospitalmedicine.org.
Disclosures
The authors have no conflicts of interest relevant to this article.
1. Centers for Disease Control and Prevention (CDC). Venous thromboembolism in adult hospitalizations, United States, 2007-2009. Morbidity and mortality weekly report (MMWR). 2012;61(22):401-40. Available: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6122a1.htm. Accessed May 7, 2018.
2. Stein PD, Matta F, Alrifai A, Rahman A. Trends in case fatality rate in pulmonary embolism according to stability and treatment. Thromb Res. 2012;130(6):841-846. PubMed
3. Jiménez D, de Miguel-Díez J, Guijarro R, et al. Trends in the management and outcomes of acute pulmonary embolism: analysis from the RIETE Registry. J Am Coll Cardiol. 2016;67(2):162-170. PubMed
4. Jaff MR, McMurtry MS, Archer SL, et al. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011;123(16):1788-1830. PubMed
5. Meyer G, Vicaut E, Danays T, et al. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N Engl J Med. 2014;370:1402-1411. PubMed
6. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest. 2016;49(2):315-352. PubMed
7. Konstantinides SV, Torbicki A, Agnelli G, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2014;35(43):3033-69, 3069a-3069k. PubMed
8. Becattini C, Agnelli G, Lankeit M, et al. Acute pulmonary embolism: mortality prediction by the 2014 European Society of Cardiology risk stratification model. Eur Respir J. 2016;48(3):780-786. PubMed
9. Levitov A, Frankel HL, Blaivas M, et al. Guidelines for the appropriate use of bedside general and cardiac ultrasonography in the evaluation of critically ill patients-part II: Cardiac Ultrasonography. Crit Care Med. 2016;44(6):1206-1227. PubMed
10. Cho JH, Kutti Sridharan G, Kim SH, et al. Right ventricular dysfunction as an echocardiographic prognostic factor in hemodynamically stable patients with acute pulmonary embolism: a meta-analysis. BMC Cardiovasc Disord. 2014;14:64. PubMed
11. Jiménez D, Kopecna D, Tapson V, et al. Derivation and validation of multimarker prognostication for normotensive patients with acute symptomatic pulmonary embolism. Am J Respir Crit Care Med. 2014;189(6):718-726. PubMed
12. Sanchez O, Trinquart L, Colombet I, et al. Prognostic value of right ventricular dysfunction in patients with haemodynamically stable pulmonary embolism: a systematic review. Eur Heart J. 2008;29(12):1569-1577. PubMed
13. Elias A, Mallett S, Daoud-Elias M, Poggi JN, Clarke M. Prognostic models in acute pulmonary embolism: a systematic review and meta-analysis. BMJ Open. 2016;6(4):e010324. PubMed
14. Konstantinides S, Geibel A, Heusel G, et al. Heparin plus alteplase compared with heparin alone in patients with submassive pulmonary embolism. N Engl J Med. 2002;347(15):1143-1150. PubMed
15. Kline JA, Nordenholz KE, Courtney DM, et al. Treatment of submassive pulmonary embolism with tenecteplase or placebo: cardiopulmonary outcomes at 3 months: multicenter double-blind, placebo-controlled randomized trial. J Thromb Haemost. 2014;12(4):459-468. PubMed
16. Cohen DM, Winter M, Lindenauer PK, Walkey AJ. Echocardiogram in the evaluation of hemodynamically stable acute pulmonary embolism: national practices and clinical outcomes. Ann Am Thorac Soc. 2018;15(5):581-588. PubMed
1. Centers for Disease Control and Prevention (CDC). Venous thromboembolism in adult hospitalizations, United States, 2007-2009. Morbidity and mortality weekly report (MMWR). 2012;61(22):401-40. Available: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6122a1.htm. Accessed May 7, 2018.
2. Stein PD, Matta F, Alrifai A, Rahman A. Trends in case fatality rate in pulmonary embolism according to stability and treatment. Thromb Res. 2012;130(6):841-846. PubMed
3. Jiménez D, de Miguel-Díez J, Guijarro R, et al. Trends in the management and outcomes of acute pulmonary embolism: analysis from the RIETE Registry. J Am Coll Cardiol. 2016;67(2):162-170. PubMed
4. Jaff MR, McMurtry MS, Archer SL, et al. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011;123(16):1788-1830. PubMed
5. Meyer G, Vicaut E, Danays T, et al. Fibrinolysis for patients with intermediate-risk pulmonary embolism. N Engl J Med. 2014;370:1402-1411. PubMed
6. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest. 2016;49(2):315-352. PubMed
7. Konstantinides SV, Torbicki A, Agnelli G, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2014;35(43):3033-69, 3069a-3069k. PubMed
8. Becattini C, Agnelli G, Lankeit M, et al. Acute pulmonary embolism: mortality prediction by the 2014 European Society of Cardiology risk stratification model. Eur Respir J. 2016;48(3):780-786. PubMed
9. Levitov A, Frankel HL, Blaivas M, et al. Guidelines for the appropriate use of bedside general and cardiac ultrasonography in the evaluation of critically ill patients-part II: Cardiac Ultrasonography. Crit Care Med. 2016;44(6):1206-1227. PubMed
10. Cho JH, Kutti Sridharan G, Kim SH, et al. Right ventricular dysfunction as an echocardiographic prognostic factor in hemodynamically stable patients with acute pulmonary embolism: a meta-analysis. BMC Cardiovasc Disord. 2014;14:64. PubMed
11. Jiménez D, Kopecna D, Tapson V, et al. Derivation and validation of multimarker prognostication for normotensive patients with acute symptomatic pulmonary embolism. Am J Respir Crit Care Med. 2014;189(6):718-726. PubMed
12. Sanchez O, Trinquart L, Colombet I, et al. Prognostic value of right ventricular dysfunction in patients with haemodynamically stable pulmonary embolism: a systematic review. Eur Heart J. 2008;29(12):1569-1577. PubMed
13. Elias A, Mallett S, Daoud-Elias M, Poggi JN, Clarke M. Prognostic models in acute pulmonary embolism: a systematic review and meta-analysis. BMJ Open. 2016;6(4):e010324. PubMed
14. Konstantinides S, Geibel A, Heusel G, et al. Heparin plus alteplase compared with heparin alone in patients with submassive pulmonary embolism. N Engl J Med. 2002;347(15):1143-1150. PubMed
15. Kline JA, Nordenholz KE, Courtney DM, et al. Treatment of submassive pulmonary embolism with tenecteplase or placebo: cardiopulmonary outcomes at 3 months: multicenter double-blind, placebo-controlled randomized trial. J Thromb Haemost. 2014;12(4):459-468. PubMed
16. Cohen DM, Winter M, Lindenauer PK, Walkey AJ. Echocardiogram in the evaluation of hemodynamically stable acute pulmonary embolism: national practices and clinical outcomes. Ann Am Thorac Soc. 2018;15(5):581-588. PubMed
© 2019 Society of Hospital Medicine
The Right Frame
A 65-year-old man was transferred to a tertiary academic medical center with one week of progressive shortness of breath, dry cough, and fevers. He reported no weight loss or night sweats but had experienced mild right upper quadrant pain and anorexia for the preceding three weeks. Several years had passed since he had consulted a physician, and he did not take any medications. He immigrated to the United States from Mexico four decades prior. He traveled back frequently to visit his family, most recently one month before his presentation. He worked as a farming supervisor in the Central Valley of California. He smoked tobacco and had a 30 pack-year history. He drank alcohol occasionally and denied any drug use.
Causes of subacute cough and dyspnea include bronchitis, pneumonia, heart failure, and asthma. Pneumonia and heart failure might cause right upper quadrant pain from diaphragmatic irritation and hepatic congestion, respectively. Metastatic cancer or infection may lead to synchronous pulmonary and hepatic involvement. The patient is at increased risk of lung cancer, given his extensive smoking history.
The patient’s place of residence in the Southwestern United States places him at risk of respiratory illness from coccidioidomycosis. His exact involvement with animals and their products should be further explored. For example, consumption of unpasteurized milk might result in pneumonia, hepatitis, or both from M. bovis, Brucella species, or C. burnetii. His travel to Mexico prompts consideration of tuberculosis, histoplasmosis, and paracoccidiomycosis as causes of respiratory and possible hepatic illness.
Two weeks prior, the patient had initially presented to another hospital with one week of intermittent right upper quadrant pain unrelated to eating. An abdominal ultrasound and hepatobiliary iminodiacetic acid (HIDA) scan were normal. Computed tomography (CT) of the chest, abdomen, and pelvis with contrast demonstrated a left upper lobe lung mass measuring 5.5 × 4.4 × 3.7 cm3 and scattered right-sided pulmonary nodules (Figure 1). He underwent CT-guided biopsy of the mass and was discharged with a presumed diagnosis of primary pulmonary malignancy with plans for outpatient follow-up.
Over the next four days, the patient developed progressive dyspnea with cough and subjective fevers. The patient was readmitted with a diagnosis of postobstructive pneumonia and acute kidney injury (creatinine increased from 0.7 mg/dL to 2.9 mg/dL between admissions), and this finding was attributed to contrast-induced nephropathy from his recent CT scan. He was treated with vancomycin and piperacillin/tazobactam for two days but wished to transfer to a tertiary care hospital for a second opinion.
Postobstructive pneumonia, pulmonary embolism, and pleural effusion are common causes of dyspnea in patients with lung cancer. The patient’s travel and occupational history, lung nodules, acute renal insufficiency, and rapidly progressive respiratory symptoms prompt consideration for radiographic mimickers of lung cancer. Tuberculosis might present as a lung mass (pulmonary tuberculoma) during primary infection or reactivation. Noninfectious causes of pulmonary masses and nodules include metastatic cancer (eg, colon cancer), sarcoidosis, IgG4-related disease, and granulomatous polyangiitis (GPA).
Contrast-induced nephropathy is unusual in patients with normal renal function. More probable explanations include hypovolemia or acute tubular necrosis (ATN) from underlying inflammation. The patient’s CT-negative right upper quadrant pain may be a distinct process or represent another facet of a disseminated illness such as hepatic infiltration from lymphoma.
Upon arrival, the patient’s temperature was 38°C, heart rate (HR) 107 beats per minute, blood pressure (BP) 159/89 mm Hg, respiratory rate 25 breaths per minute, and oxygen saturation 92% on 2 L of oxygen per minute. He showed no signs of distress. Mild scleral icterus was noted. The cardiac exam was normal. Auscultation revealed scattered wheezes and crackles in the left upper lobe. Mild right upper quadrant tenderness without hepatosplenomegaly was noted on the abdominal exam. The patient’s lower extremities exhibited bilateral trace edema. No rash was observed, and his neurologic exam was normal.
The white blood cell (WBC) count was 28,300 per cubic millimeter (87% neutrophils, 3.6% lymphocytes, and 0.03% eosinophils), hemoglobin 11.1 g per deciliter, and platelet count 789,000 per cubic millimeter. Sodium was 127 mmol per liter, potassium 4.6 mmol per liter, chloride 101 mmol per liter, bicarbonate 13 mmol per liter, blood urea nitrogen 60 mg per deciliter, and creatinine 3.4 mg per deciliter. Aspartate aminotransferase and alanine aminotransferase levels were normal. Alkaline phosphatase was 283 units per liter (normal range, 31-95), and total bilirubin was 4.5 mg per deciliter (normal range, 0.2-1.3) with a direct bilirubin of 2.7 mg per deciliter. Urinalysis demonstrated urine protein of 30 mg/dL, specific gravity of 1.013, negative nitrites, 10-21 white cells per high-powered field (normal, < 5), and 21-50 red cells per high-powered field (normal, < 3). Urine microscopy revealed muddy brown casts but no cellular casts or dysmorphic red cells. A chest radiograph (CXR) showed patchy consolidations in the bilateral upper lobes and left lower lobe along with Kerley B lines, a small left pleural effusion, and thickened right horizontal fissure; the left upper lobe mass was re-demonstrated. Vancomycin, piperacillin-tazobactam, and azithromycin were administered.
At this point, the most likely source of sepsis is multifocal pneumonia. The patient is at risk for S. aureus and P. aeruginosa given his recent hospitalization. A severe form of leptospirosis (Weil’s disease) is associated with pulmonary disease, hyperbilirubinemia, and renal failure. Repeat abdominal imaging is necessary to evaluate for cholangitis given the patient’s right upper quadrant pain, fever, and jaundice. It would also help categorize his cholestatic pattern of liver injury as intrahepatic or extrahepatic (eg, stricture). An infiltrative disease such as sarcoidosis may cause both intrahepatic cholestasis and parenchymal lung disease, although the pleural pathology is uncommon.
His normal cardiac exam does not exclude cardiogenic pulmonary edema, a common cause of interstitial edema and pleural effusion. In this setting of systemic inflammation (neutrophilia, thrombocytosis, and hypoalbuminemia), the thickened right horizontal fissure and interlobular septa might represent an infiltrative process, such as lymphangitic carcinomatosis, lymphoma, or sarcoidosis.
Muddy brown casts are characteristic of ATN. The patient’s risk factors for ATN include sepsis and previously administered iodinated contrast. Fluid retention from oliguric renal failure is likely contributing to his hyponatremia and lower extremity edema. Pathology isolated to the tubules, however, would not cause hematuria and pyuria and suggests glomerular or interstitial disease. The lack of cellular casts on a single urinary specimen does not eliminate the likelihood of either disease. Hematuria and diffuse parenchymal lung disease prompt consideration of pulmonary-renal syndromes, such as anti-glomerular basement membrane disease, GPA, and systemic lupus erythematosus, which can all be triggered by infection.
On the night of transfer, the patient experienced acute respiratory distress. Heart rate was 130 beats per minute, BP 170/95 mm Hg, respiratory rate 40 breaths per minute, and oxygen saturation 88% on six liters of supplemental oxygen by nasal cannula. His arterial blood gas demonstrated a pH of 7.23, PaCO2 of 32 mm Hg, and PaO2 of 65 mm Hg. He was emergently intubated for progressive hypoxemic respiratory failure. A small amount of blood was noted in the endotracheal tube. A noncontrast CT of the chest demonstrated multifocal airspace opacities and bilateral pleural effusions. The previously noted left upper lobe mass was unchanged.
Rapid respiratory decline and diffuse alveolar disease commonly result from aspiration, flash pulmonary edema, and acute respiratory distress syndrome (ARDS). Necrotizing pneumonia (eg, S. aureus) and trauma during intubation are possible causes of blood in his endotracheal tube. However, in the setting of multifocal airspace opacity, renal insufficiency, hematuria, and rapid respiratory decline, the blood might represent diffuse alveolar hemorrhage (DAH). Bronchoscopy with bronchioalveolar lavage to evaluate for pulmonary edema, infection, and hemorrhage would be indicated.
The patient subsequently developed oliguria, requiring continuous renal replacement therapy. An echocardiogram demonstrated impaired left ventricular relaxation and a reduced ejection fraction of 45% without segmental wall motion abnormalities or valvular disease, and a right ventricular systolic pressure of 36 mm Hg. Over the next 12 hours, his respiratory status improved, and he was extubated to 15 L per minute of supplemental oxygen by high-flow nasal cannula (HFNC).
The pathology report of the lung biopsy from the other hospital disclosed chronic inflammation and fibrosis with ill-defined areas of necrosis and myxoid degeneration surrounded by nuclear palisading suggestive of granulomatous inflammation. Staining for acid-fast bacilli (AFB) and fungal organisms was negative.
The rapid pulmonary recovery is inconsistent with multifocal pneumonia or ARDS. Flash pulmonary edema might result in sudden hypoxemic respiratory failure that resolves with positive pressure ventilation and ultrafiltration. However, this condition would not explain the biopsy results. Granulomatous lung pathology often results from mycobacterial or fungal disease. Tuberculosis and fungal pneumonia are not excluded with negative staining alone. However, neither would cause self-limited respiratory failure. Histologic evidence of necrosis lessens the likelihood of sarcoidosis, which rarely causes fulminant pulmonary disease. Lymphoma can result in granulomatous inflammation but would not cause transient pulmonary disease. GPA, a cause of necrotizing granulomatous lung disease, might result in a lung mass and worsened hypoxemia through DAH.
The patient continued to require 15 L of oxygen per minute by HFNC. He had persistent bilateral perihilar alveolar and interstitial opacities on CXR. Repeat WBC count was 29,200 per cubic millimeter, hemoglobin 7.8 g per deciliter, and platelets 656,000 per cubic millimeter. The C-reactive protein was 300 mg per L (normal range, <6.3) and erythrocyte sedimentation rate 100 mm per hour (normal range, <10). Legionella urinary antigen, serum immunodiffusion for Coccidiodes imitus, human immunodeficiency virus antibody, respiratory viral panel, and beta-D glucan were negative. Rare acid-fast bacilli were visualized in one out of three concentrated AFB sputum smears. He was started on empiric antituberculous therapy with rifampin, isoniazid, pyrazinamide, and ethambutol.
The sputum sample is suggestive of pulmonary tuberculosis. The salient features of this case include systemic inflammation, pulmonary nodules and mass, necrotizing granulomatous lung pathology, renal insufficiency, and hematuria. Disseminated tuberculosis might explain all these findings. However, a positive AFB smear may signal the presence of a nontuberculous mycobacteria, which is less likely to cause this clinical syndrome.
M. tuberculosis complex polymerase chain reaction (MTB PCR) assay returned negative for M. tuberculosis. Antiproteinase 3 antibody was 1,930 units (normal range, <20). Antimyeloperoxidase and antiglomerular basement membrane antibodies were negative.
Tuberculosis and GPA share several overlapping features, such as necrotizing lung pathology and less commonly antineutrophil cytoplasmic autoantibody (ANCA)-associated antibodies. However, the lung mass, acute renal and respiratory failure, hematuria, and the degree of anti-proteinase 3 level elevation are highly suggestive of GPA. The negative MTB PCR raises the possibility that a nontuberculous mycobacterium was detected on the sputum smear. Nevertheless, continued treatment until finalization of culture results is appropriate given that tuberculosis is endemic in Mexico.
The patient’s presenting features of right upper quadrant tenderness, jaundice, and cholestatic hepatitis remain poorly explained by either of these diagnoses. Neither tuberculosis nor GPA commonly presents with accompanying hepatic involvement, though both have been occasionally described as causing hepatitis. As the greatest concern in this patient remains his progressive renal failure and accompanying pulmonary hemorrhage, a renal biopsy to assess for glomerulonephritis associated with GPA is warranted before further investigation into the cause of his cholestatic hepatitis.
A core renal biopsy demonstrated pauci-immune focal crescentic and necrotizing glomerulonephritis with mixed tubulointerstitial inflammation (Figure 2). In conjunction with the pulmonary syndrome and positive antiproteinase 3 serology, a diagnosis of granulomatosis with polyangiitis was made. The patient was treated with pulse dose steroids, rituximab, and plasma exchange. Two weeks later, the sputum mycobacterial culture returned positive for Mycobacterium llatzerense and anti-tuberculous treatment was discontinued.
Over the following weeks, the patient improved and was transitioned off dialysis prior to hospital discharge. By six months later, he had resolution of his hemoptysis, shortness of breath, liver biochemical test abnormalities, and significant improvement in his renal function. Repeat sputum mycobacterial cultures were negative.
DISCUSSION
A 65-year-old man from Mexico with a significant smoking history presented with an apical lung mass and cough, prioritizing tuberculosis and pulmonary malignancy. As the case unfolded, renal failure, multifocal lung opacities, conflicting tuberculosis test results, positive anti-proteinase 3 antibody, and ultimately a renal biopsy led to the diagnosis of granulomatosis with polyangiitis (GPA).
The correct interpretation of occasionally conflicting mycobacterial testing is crucial. Mycobacterial cultures remain the gold standard for diagnosing tuberculosis. However, results take weeks to return. Rapid tests include acid-fast bacilli (AFB) smear microscopy and nucleic acid-amplification tests (NAAT) of sputum or bronchoalveolar samples.1 When three sputum smears are performed, the sensitivity of AFB smear microscopy for tuberculosis in immunocompetent hosts is 70%.1 The AFB smear does not distinguish between different mycobacterial organisms. Thus, a positive result must be interpreted with the relative prevalence of tuberculosis and nontuberculous mycobacteria (NTM) in mind. The addition of NAAT-based assays has allowed for enhanced sensitivity and specificity in the diagnosis of tuberculosis, such that a negative NAAT in a patient with a positive AFB smear strongly argues for the presence of a NTM.2-4
NTM are widely prevalent environmental microbes, with over 140 species described, and careful consideration is required to determine if an isolate is pathogenic.5 Given their ubiquitous nature, a high rate of asymptomatic respiratory and cutaneous colonization occurs. Correspondingly, the diagnosis of NTM disease requires multiple positive cultures or pathologic features on tissue biopsy, compatible clinical findings, and diligent exclusion of other causes.5 A retrospective study of all NTM isolates in Oregon from 2005-2006 revealed that only 47% of patients met the guideline criteria for having symptomatic NTM disease.6 In our case, the patient’s sputum grew M. llatzerense, an aerobic, nonfermenting mycobacterium found in water sources that has only infrequently been implicated as a human pathogen.7,8 Subsequent AFB sputum cultures were negative, and serial imaging showed resolution of the pulmonary findings without additional antimycobacterial therapy, suggesting that this organism was not responsible for the disease process.
Along with microscopic polyangiitis (MPA) and eosinophilic granulomatosis with polyangiitis (EGPA), GPA is an antineutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis that predominantly affects small to medium sized vessels. Although it can occur at any age, GPA most commonly afflicts older adults, with men and women being diagnosed at roughly equal rates.9 GPA is a multisystem disease with a wide array of clinical manifestations. The most frequently involved sites of disease are the respiratory tract and kidneys, although virtually any organ can be affected. Sino-nasal disease, such as destructive sinusitis, or ear involvement are nearly universal. Lower respiratory manifestations occur in 60% of patients, but are highly diverse and reflect the inherent difficulty in diagnosing this condition.9-11 Additionally, GPA is a frequent cause of the pulmonary-renal syndromes, with glomerulonephritis occurring in 80% of patients.9
The diagnosis of GPA in this case was delayed, in part, due to features suggestive of malignancy and pulmonary tuberculosis. While sino-nasal disease was not noted during this hospitalization, the patient had many different respiratory manifestations, including a dominant pulmonary mass, diffuse nodules, and hypoxemic respiratory failure due to suspected diffuse alveolar hemorrhage (DAH), all of which have been reported in GPA.12 Dysmorphic red cells and red blood cell casts are not sensitive for renal involvement in GPA; their absence does not exclude the possibility of an ANCA-associated vasculitis.13 Hematuria and rapid progression to oliguric renal failure are characteristic of a vasculitic process and should sway clinicians away from a working diagnosis of ATN.
The diagnosis of GPA involves the synthesis of clinical data, radiographic findings, serologic testing, and histopathology. ANCA testing is an essential step in the diagnosis of GPA but has limitations. Patients with GPA more commonly have ANCAs targeting the enzyme proteinase-3 (PR3-ANCA), with MPA being more closely associated with myeloperoxidase (MPO-ANCA), although cross-reactivity and antibody-negative disease can occur.14 Although 90% of patients with GPA with multiorgan involvement will have a positive ANCA, a negative test is more common in localized upper airway disease, where only 50% have a positive ANCA.15 A number of drugs, medications, infections, and nonvasculitic autoimmune diseases have been associated with positive ANCA serologies in the absence of systemic vasculitis.14,16,17 As such, pathologic demonstration of vasculitis is necessary for establishing the diagnosis. Typical sites for biopsy include the kidneys and lungs.9
This case illustrates how clinicians often find themselves at a diagnostic crossroads—being forced to choose which clinical elements to prioritize. At various points, our patient’s presentation could have been framed as “a man from a Tb-endemic country with hemoptysis and an apical opacity,” “an elderly man with extensive smoking history and lung mass,” or “a patient with elevated inflammatory markers and pulmonary-renal syndrome”. In such situations, it is incumbent on the clinician to evaluate how well a given problem representation encompasses or highlights the salient features of a case. As with painting or photography, an essential aspect of appreciating the whole picture involves carefully selecting the right frame.
KEY TEACHING POINTS
- The diagnosis of tuberculosis relies on smear microscopy, nucleic-acid amplification testing (NAAT), and cultures. A positive AFB smear with negative NAAT suggests the presence of a nontuberculous mycobacteria (NTM).
- NTM are common environmental organisms and often exist as nonpathogenic colonizers.6 The diagnosis of NTM disease requires exclusion of other causes and careful clinical, microbiologic, and radiographic correlation.
- Granulomatosis with polyangiitis is a multisystem disease often involving the respiratory track and kidney. Pulmonary disease can present with airway involvement, parenchymal nodules, opacities, pleural findings, and diffuse alveolar hemorrhage.12
Disclosures
Drs. Minter, Geha, Boslett, Chung, and Ramani have no disclosures. Dr. Manesh is supported by the Jeremiah A. Barondess Fellowship in the Clinical Transaction of the New York Academy of Medicine, in collaboration with the Accreditation Council for Graduate Medical Education (ACGME).
1. Lewinsohn DM, Leonard MK, LoBue PA, et al. Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention clinical practice guidelines: diagnosis of tuberculosis in adults and children. Clin Infect Dis. 2017;64(2):e1-e33. PubMed
2. Steingart KR, Sohn H, Schiller I, et al. Xpert(R) MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. 2013;(1):Cd009593. PubMed
3. Luetkemeyer AF, Firnhaber C, Kendall MA, et al. Evaluation of Xpert MTB/RIF versus afb smear and culture to identify pulmonary tuberculosis in patients with suspected tuberculosis from low and higher prevalence settings. Clin Infect Dis. 2016;62(9):1081-1088. PubMed
4. Boehme CC, Nabeta P, Hillemann D, et al. Rapid molecular detection of tuberculosis and rifampin resistance. N Engl J Med. 2010;363(11):1005-1015. PubMed
5. Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175(4):367-416. PubMed
6. Winthrop KL, McNelley E, Kendall B, et al. Pulmonary nontuberculous mycobacterial disease prevalence and clinical features: an emerging public health disease. Am J Respir Crit Care Med. 2010;182(7):977-982. PubMed
7. Teixeira L, Avery RK, Iseman M, et al. Mycobacterium llatzerense lung infection in a liver transplant recipient: case report and review of the literature. Am J Transplant. 2013;13(8):2198-2200. PubMed
8. Cárdenas AM, Gomila M, Lalucat J, Edelstein PH. Abdominal abscess caused by Mycobacterium llatzerense. J Clin Microbiol. 2014;52(4):1287-1289. PubMed
9. Jennette JC, Falk RJ. Small-vessel vasculitis. N Engl J Med. 1997;337(21):1512-1523. PubMed
10. Mahr A, Katsahian S, Varet H, et al. Revisiting the classification of clinical phenotypes of anti-neutrophil cytoplasmic antibody-associated vasculitis: a cluster analysis. Ann Rheum Dis. 2013;72(6):1003-1010. PubMed
11. Holle JU, Gross WL, Latza U, et al. Improved outcome in 445 patients with Wegener’s granulomatosis in a German vasculitis center over four decades. Arthritis Rheum. 2011;63(1):257-266. PubMed
12. Cordier JF, Valeyre D, Guillevin L, Loire R, Brechot JM. Pulmonary Wegener’s granulomatosis. A clinical and imaging study of 77 cases. Chest. 1990;97(4):906-912. PubMed
13. Hamadah AM, Gharaibeh K, Mara KC, et al. Urinalysis for the diagnosis of glomerulonephritis: role of dysmorphic red blood cells. Nephrol Dial Transplant. 2018;33(8):1397-1403. PubMed
14. Jennette JC, Falk RJ. Pathogenesis of antineutrophil cytoplasmic autoantibody-mediated disease. Nat Rev Rheumatol. 2014;10(8):463-473. PubMed
15. Borner U, Landis BN, Banz Y, et al. Diagnostic value of biopsies in identifying cytoplasmic antineutrophil cytoplasmic antibody-negative localized Wegener’s granulomatosis presenting primarily with sinonasal disease. Am J Rhinol Allergy. 2012;26(6):475-480. PubMed
16. Mahr A, Batteux F, Tubiana S, et al. Brief report: prevalence of antineutrophil cytoplasmic antibodies in infective endocarditis. Arthritis Rheumatol. 2014;66(6):1672-1677. PubMed
17. Sherkat R, Mostafavizadeh K, Zeydabadi L, Shoaei P, Rostami S. Antineutrophil cytoplasmic antibodies in patients with pulmonary tuberculosis. Iran J Immunol. 2011;8(1):52-57. PubMed
A 65-year-old man was transferred to a tertiary academic medical center with one week of progressive shortness of breath, dry cough, and fevers. He reported no weight loss or night sweats but had experienced mild right upper quadrant pain and anorexia for the preceding three weeks. Several years had passed since he had consulted a physician, and he did not take any medications. He immigrated to the United States from Mexico four decades prior. He traveled back frequently to visit his family, most recently one month before his presentation. He worked as a farming supervisor in the Central Valley of California. He smoked tobacco and had a 30 pack-year history. He drank alcohol occasionally and denied any drug use.
Causes of subacute cough and dyspnea include bronchitis, pneumonia, heart failure, and asthma. Pneumonia and heart failure might cause right upper quadrant pain from diaphragmatic irritation and hepatic congestion, respectively. Metastatic cancer or infection may lead to synchronous pulmonary and hepatic involvement. The patient is at increased risk of lung cancer, given his extensive smoking history.
The patient’s place of residence in the Southwestern United States places him at risk of respiratory illness from coccidioidomycosis. His exact involvement with animals and their products should be further explored. For example, consumption of unpasteurized milk might result in pneumonia, hepatitis, or both from M. bovis, Brucella species, or C. burnetii. His travel to Mexico prompts consideration of tuberculosis, histoplasmosis, and paracoccidiomycosis as causes of respiratory and possible hepatic illness.
Two weeks prior, the patient had initially presented to another hospital with one week of intermittent right upper quadrant pain unrelated to eating. An abdominal ultrasound and hepatobiliary iminodiacetic acid (HIDA) scan were normal. Computed tomography (CT) of the chest, abdomen, and pelvis with contrast demonstrated a left upper lobe lung mass measuring 5.5 × 4.4 × 3.7 cm3 and scattered right-sided pulmonary nodules (Figure 1). He underwent CT-guided biopsy of the mass and was discharged with a presumed diagnosis of primary pulmonary malignancy with plans for outpatient follow-up.
Over the next four days, the patient developed progressive dyspnea with cough and subjective fevers. The patient was readmitted with a diagnosis of postobstructive pneumonia and acute kidney injury (creatinine increased from 0.7 mg/dL to 2.9 mg/dL between admissions), and this finding was attributed to contrast-induced nephropathy from his recent CT scan. He was treated with vancomycin and piperacillin/tazobactam for two days but wished to transfer to a tertiary care hospital for a second opinion.
Postobstructive pneumonia, pulmonary embolism, and pleural effusion are common causes of dyspnea in patients with lung cancer. The patient’s travel and occupational history, lung nodules, acute renal insufficiency, and rapidly progressive respiratory symptoms prompt consideration for radiographic mimickers of lung cancer. Tuberculosis might present as a lung mass (pulmonary tuberculoma) during primary infection or reactivation. Noninfectious causes of pulmonary masses and nodules include metastatic cancer (eg, colon cancer), sarcoidosis, IgG4-related disease, and granulomatous polyangiitis (GPA).
Contrast-induced nephropathy is unusual in patients with normal renal function. More probable explanations include hypovolemia or acute tubular necrosis (ATN) from underlying inflammation. The patient’s CT-negative right upper quadrant pain may be a distinct process or represent another facet of a disseminated illness such as hepatic infiltration from lymphoma.
Upon arrival, the patient’s temperature was 38°C, heart rate (HR) 107 beats per minute, blood pressure (BP) 159/89 mm Hg, respiratory rate 25 breaths per minute, and oxygen saturation 92% on 2 L of oxygen per minute. He showed no signs of distress. Mild scleral icterus was noted. The cardiac exam was normal. Auscultation revealed scattered wheezes and crackles in the left upper lobe. Mild right upper quadrant tenderness without hepatosplenomegaly was noted on the abdominal exam. The patient’s lower extremities exhibited bilateral trace edema. No rash was observed, and his neurologic exam was normal.
The white blood cell (WBC) count was 28,300 per cubic millimeter (87% neutrophils, 3.6% lymphocytes, and 0.03% eosinophils), hemoglobin 11.1 g per deciliter, and platelet count 789,000 per cubic millimeter. Sodium was 127 mmol per liter, potassium 4.6 mmol per liter, chloride 101 mmol per liter, bicarbonate 13 mmol per liter, blood urea nitrogen 60 mg per deciliter, and creatinine 3.4 mg per deciliter. Aspartate aminotransferase and alanine aminotransferase levels were normal. Alkaline phosphatase was 283 units per liter (normal range, 31-95), and total bilirubin was 4.5 mg per deciliter (normal range, 0.2-1.3) with a direct bilirubin of 2.7 mg per deciliter. Urinalysis demonstrated urine protein of 30 mg/dL, specific gravity of 1.013, negative nitrites, 10-21 white cells per high-powered field (normal, < 5), and 21-50 red cells per high-powered field (normal, < 3). Urine microscopy revealed muddy brown casts but no cellular casts or dysmorphic red cells. A chest radiograph (CXR) showed patchy consolidations in the bilateral upper lobes and left lower lobe along with Kerley B lines, a small left pleural effusion, and thickened right horizontal fissure; the left upper lobe mass was re-demonstrated. Vancomycin, piperacillin-tazobactam, and azithromycin were administered.
At this point, the most likely source of sepsis is multifocal pneumonia. The patient is at risk for S. aureus and P. aeruginosa given his recent hospitalization. A severe form of leptospirosis (Weil’s disease) is associated with pulmonary disease, hyperbilirubinemia, and renal failure. Repeat abdominal imaging is necessary to evaluate for cholangitis given the patient’s right upper quadrant pain, fever, and jaundice. It would also help categorize his cholestatic pattern of liver injury as intrahepatic or extrahepatic (eg, stricture). An infiltrative disease such as sarcoidosis may cause both intrahepatic cholestasis and parenchymal lung disease, although the pleural pathology is uncommon.
His normal cardiac exam does not exclude cardiogenic pulmonary edema, a common cause of interstitial edema and pleural effusion. In this setting of systemic inflammation (neutrophilia, thrombocytosis, and hypoalbuminemia), the thickened right horizontal fissure and interlobular septa might represent an infiltrative process, such as lymphangitic carcinomatosis, lymphoma, or sarcoidosis.
Muddy brown casts are characteristic of ATN. The patient’s risk factors for ATN include sepsis and previously administered iodinated contrast. Fluid retention from oliguric renal failure is likely contributing to his hyponatremia and lower extremity edema. Pathology isolated to the tubules, however, would not cause hematuria and pyuria and suggests glomerular or interstitial disease. The lack of cellular casts on a single urinary specimen does not eliminate the likelihood of either disease. Hematuria and diffuse parenchymal lung disease prompt consideration of pulmonary-renal syndromes, such as anti-glomerular basement membrane disease, GPA, and systemic lupus erythematosus, which can all be triggered by infection.
On the night of transfer, the patient experienced acute respiratory distress. Heart rate was 130 beats per minute, BP 170/95 mm Hg, respiratory rate 40 breaths per minute, and oxygen saturation 88% on six liters of supplemental oxygen by nasal cannula. His arterial blood gas demonstrated a pH of 7.23, PaCO2 of 32 mm Hg, and PaO2 of 65 mm Hg. He was emergently intubated for progressive hypoxemic respiratory failure. A small amount of blood was noted in the endotracheal tube. A noncontrast CT of the chest demonstrated multifocal airspace opacities and bilateral pleural effusions. The previously noted left upper lobe mass was unchanged.
Rapid respiratory decline and diffuse alveolar disease commonly result from aspiration, flash pulmonary edema, and acute respiratory distress syndrome (ARDS). Necrotizing pneumonia (eg, S. aureus) and trauma during intubation are possible causes of blood in his endotracheal tube. However, in the setting of multifocal airspace opacity, renal insufficiency, hematuria, and rapid respiratory decline, the blood might represent diffuse alveolar hemorrhage (DAH). Bronchoscopy with bronchioalveolar lavage to evaluate for pulmonary edema, infection, and hemorrhage would be indicated.
The patient subsequently developed oliguria, requiring continuous renal replacement therapy. An echocardiogram demonstrated impaired left ventricular relaxation and a reduced ejection fraction of 45% without segmental wall motion abnormalities or valvular disease, and a right ventricular systolic pressure of 36 mm Hg. Over the next 12 hours, his respiratory status improved, and he was extubated to 15 L per minute of supplemental oxygen by high-flow nasal cannula (HFNC).
The pathology report of the lung biopsy from the other hospital disclosed chronic inflammation and fibrosis with ill-defined areas of necrosis and myxoid degeneration surrounded by nuclear palisading suggestive of granulomatous inflammation. Staining for acid-fast bacilli (AFB) and fungal organisms was negative.
The rapid pulmonary recovery is inconsistent with multifocal pneumonia or ARDS. Flash pulmonary edema might result in sudden hypoxemic respiratory failure that resolves with positive pressure ventilation and ultrafiltration. However, this condition would not explain the biopsy results. Granulomatous lung pathology often results from mycobacterial or fungal disease. Tuberculosis and fungal pneumonia are not excluded with negative staining alone. However, neither would cause self-limited respiratory failure. Histologic evidence of necrosis lessens the likelihood of sarcoidosis, which rarely causes fulminant pulmonary disease. Lymphoma can result in granulomatous inflammation but would not cause transient pulmonary disease. GPA, a cause of necrotizing granulomatous lung disease, might result in a lung mass and worsened hypoxemia through DAH.
The patient continued to require 15 L of oxygen per minute by HFNC. He had persistent bilateral perihilar alveolar and interstitial opacities on CXR. Repeat WBC count was 29,200 per cubic millimeter, hemoglobin 7.8 g per deciliter, and platelets 656,000 per cubic millimeter. The C-reactive protein was 300 mg per L (normal range, <6.3) and erythrocyte sedimentation rate 100 mm per hour (normal range, <10). Legionella urinary antigen, serum immunodiffusion for Coccidiodes imitus, human immunodeficiency virus antibody, respiratory viral panel, and beta-D glucan were negative. Rare acid-fast bacilli were visualized in one out of three concentrated AFB sputum smears. He was started on empiric antituberculous therapy with rifampin, isoniazid, pyrazinamide, and ethambutol.
The sputum sample is suggestive of pulmonary tuberculosis. The salient features of this case include systemic inflammation, pulmonary nodules and mass, necrotizing granulomatous lung pathology, renal insufficiency, and hematuria. Disseminated tuberculosis might explain all these findings. However, a positive AFB smear may signal the presence of a nontuberculous mycobacteria, which is less likely to cause this clinical syndrome.
M. tuberculosis complex polymerase chain reaction (MTB PCR) assay returned negative for M. tuberculosis. Antiproteinase 3 antibody was 1,930 units (normal range, <20). Antimyeloperoxidase and antiglomerular basement membrane antibodies were negative.
Tuberculosis and GPA share several overlapping features, such as necrotizing lung pathology and less commonly antineutrophil cytoplasmic autoantibody (ANCA)-associated antibodies. However, the lung mass, acute renal and respiratory failure, hematuria, and the degree of anti-proteinase 3 level elevation are highly suggestive of GPA. The negative MTB PCR raises the possibility that a nontuberculous mycobacterium was detected on the sputum smear. Nevertheless, continued treatment until finalization of culture results is appropriate given that tuberculosis is endemic in Mexico.
The patient’s presenting features of right upper quadrant tenderness, jaundice, and cholestatic hepatitis remain poorly explained by either of these diagnoses. Neither tuberculosis nor GPA commonly presents with accompanying hepatic involvement, though both have been occasionally described as causing hepatitis. As the greatest concern in this patient remains his progressive renal failure and accompanying pulmonary hemorrhage, a renal biopsy to assess for glomerulonephritis associated with GPA is warranted before further investigation into the cause of his cholestatic hepatitis.
A core renal biopsy demonstrated pauci-immune focal crescentic and necrotizing glomerulonephritis with mixed tubulointerstitial inflammation (Figure 2). In conjunction with the pulmonary syndrome and positive antiproteinase 3 serology, a diagnosis of granulomatosis with polyangiitis was made. The patient was treated with pulse dose steroids, rituximab, and plasma exchange. Two weeks later, the sputum mycobacterial culture returned positive for Mycobacterium llatzerense and anti-tuberculous treatment was discontinued.
Over the following weeks, the patient improved and was transitioned off dialysis prior to hospital discharge. By six months later, he had resolution of his hemoptysis, shortness of breath, liver biochemical test abnormalities, and significant improvement in his renal function. Repeat sputum mycobacterial cultures were negative.
DISCUSSION
A 65-year-old man from Mexico with a significant smoking history presented with an apical lung mass and cough, prioritizing tuberculosis and pulmonary malignancy. As the case unfolded, renal failure, multifocal lung opacities, conflicting tuberculosis test results, positive anti-proteinase 3 antibody, and ultimately a renal biopsy led to the diagnosis of granulomatosis with polyangiitis (GPA).
The correct interpretation of occasionally conflicting mycobacterial testing is crucial. Mycobacterial cultures remain the gold standard for diagnosing tuberculosis. However, results take weeks to return. Rapid tests include acid-fast bacilli (AFB) smear microscopy and nucleic acid-amplification tests (NAAT) of sputum or bronchoalveolar samples.1 When three sputum smears are performed, the sensitivity of AFB smear microscopy for tuberculosis in immunocompetent hosts is 70%.1 The AFB smear does not distinguish between different mycobacterial organisms. Thus, a positive result must be interpreted with the relative prevalence of tuberculosis and nontuberculous mycobacteria (NTM) in mind. The addition of NAAT-based assays has allowed for enhanced sensitivity and specificity in the diagnosis of tuberculosis, such that a negative NAAT in a patient with a positive AFB smear strongly argues for the presence of a NTM.2-4
NTM are widely prevalent environmental microbes, with over 140 species described, and careful consideration is required to determine if an isolate is pathogenic.5 Given their ubiquitous nature, a high rate of asymptomatic respiratory and cutaneous colonization occurs. Correspondingly, the diagnosis of NTM disease requires multiple positive cultures or pathologic features on tissue biopsy, compatible clinical findings, and diligent exclusion of other causes.5 A retrospective study of all NTM isolates in Oregon from 2005-2006 revealed that only 47% of patients met the guideline criteria for having symptomatic NTM disease.6 In our case, the patient’s sputum grew M. llatzerense, an aerobic, nonfermenting mycobacterium found in water sources that has only infrequently been implicated as a human pathogen.7,8 Subsequent AFB sputum cultures were negative, and serial imaging showed resolution of the pulmonary findings without additional antimycobacterial therapy, suggesting that this organism was not responsible for the disease process.
Along with microscopic polyangiitis (MPA) and eosinophilic granulomatosis with polyangiitis (EGPA), GPA is an antineutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis that predominantly affects small to medium sized vessels. Although it can occur at any age, GPA most commonly afflicts older adults, with men and women being diagnosed at roughly equal rates.9 GPA is a multisystem disease with a wide array of clinical manifestations. The most frequently involved sites of disease are the respiratory tract and kidneys, although virtually any organ can be affected. Sino-nasal disease, such as destructive sinusitis, or ear involvement are nearly universal. Lower respiratory manifestations occur in 60% of patients, but are highly diverse and reflect the inherent difficulty in diagnosing this condition.9-11 Additionally, GPA is a frequent cause of the pulmonary-renal syndromes, with glomerulonephritis occurring in 80% of patients.9
The diagnosis of GPA in this case was delayed, in part, due to features suggestive of malignancy and pulmonary tuberculosis. While sino-nasal disease was not noted during this hospitalization, the patient had many different respiratory manifestations, including a dominant pulmonary mass, diffuse nodules, and hypoxemic respiratory failure due to suspected diffuse alveolar hemorrhage (DAH), all of which have been reported in GPA.12 Dysmorphic red cells and red blood cell casts are not sensitive for renal involvement in GPA; their absence does not exclude the possibility of an ANCA-associated vasculitis.13 Hematuria and rapid progression to oliguric renal failure are characteristic of a vasculitic process and should sway clinicians away from a working diagnosis of ATN.
The diagnosis of GPA involves the synthesis of clinical data, radiographic findings, serologic testing, and histopathology. ANCA testing is an essential step in the diagnosis of GPA but has limitations. Patients with GPA more commonly have ANCAs targeting the enzyme proteinase-3 (PR3-ANCA), with MPA being more closely associated with myeloperoxidase (MPO-ANCA), although cross-reactivity and antibody-negative disease can occur.14 Although 90% of patients with GPA with multiorgan involvement will have a positive ANCA, a negative test is more common in localized upper airway disease, where only 50% have a positive ANCA.15 A number of drugs, medications, infections, and nonvasculitic autoimmune diseases have been associated with positive ANCA serologies in the absence of systemic vasculitis.14,16,17 As such, pathologic demonstration of vasculitis is necessary for establishing the diagnosis. Typical sites for biopsy include the kidneys and lungs.9
This case illustrates how clinicians often find themselves at a diagnostic crossroads—being forced to choose which clinical elements to prioritize. At various points, our patient’s presentation could have been framed as “a man from a Tb-endemic country with hemoptysis and an apical opacity,” “an elderly man with extensive smoking history and lung mass,” or “a patient with elevated inflammatory markers and pulmonary-renal syndrome”. In such situations, it is incumbent on the clinician to evaluate how well a given problem representation encompasses or highlights the salient features of a case. As with painting or photography, an essential aspect of appreciating the whole picture involves carefully selecting the right frame.
KEY TEACHING POINTS
- The diagnosis of tuberculosis relies on smear microscopy, nucleic-acid amplification testing (NAAT), and cultures. A positive AFB smear with negative NAAT suggests the presence of a nontuberculous mycobacteria (NTM).
- NTM are common environmental organisms and often exist as nonpathogenic colonizers.6 The diagnosis of NTM disease requires exclusion of other causes and careful clinical, microbiologic, and radiographic correlation.
- Granulomatosis with polyangiitis is a multisystem disease often involving the respiratory track and kidney. Pulmonary disease can present with airway involvement, parenchymal nodules, opacities, pleural findings, and diffuse alveolar hemorrhage.12
Disclosures
Drs. Minter, Geha, Boslett, Chung, and Ramani have no disclosures. Dr. Manesh is supported by the Jeremiah A. Barondess Fellowship in the Clinical Transaction of the New York Academy of Medicine, in collaboration with the Accreditation Council for Graduate Medical Education (ACGME).
A 65-year-old man was transferred to a tertiary academic medical center with one week of progressive shortness of breath, dry cough, and fevers. He reported no weight loss or night sweats but had experienced mild right upper quadrant pain and anorexia for the preceding three weeks. Several years had passed since he had consulted a physician, and he did not take any medications. He immigrated to the United States from Mexico four decades prior. He traveled back frequently to visit his family, most recently one month before his presentation. He worked as a farming supervisor in the Central Valley of California. He smoked tobacco and had a 30 pack-year history. He drank alcohol occasionally and denied any drug use.
Causes of subacute cough and dyspnea include bronchitis, pneumonia, heart failure, and asthma. Pneumonia and heart failure might cause right upper quadrant pain from diaphragmatic irritation and hepatic congestion, respectively. Metastatic cancer or infection may lead to synchronous pulmonary and hepatic involvement. The patient is at increased risk of lung cancer, given his extensive smoking history.
The patient’s place of residence in the Southwestern United States places him at risk of respiratory illness from coccidioidomycosis. His exact involvement with animals and their products should be further explored. For example, consumption of unpasteurized milk might result in pneumonia, hepatitis, or both from M. bovis, Brucella species, or C. burnetii. His travel to Mexico prompts consideration of tuberculosis, histoplasmosis, and paracoccidiomycosis as causes of respiratory and possible hepatic illness.
Two weeks prior, the patient had initially presented to another hospital with one week of intermittent right upper quadrant pain unrelated to eating. An abdominal ultrasound and hepatobiliary iminodiacetic acid (HIDA) scan were normal. Computed tomography (CT) of the chest, abdomen, and pelvis with contrast demonstrated a left upper lobe lung mass measuring 5.5 × 4.4 × 3.7 cm3 and scattered right-sided pulmonary nodules (Figure 1). He underwent CT-guided biopsy of the mass and was discharged with a presumed diagnosis of primary pulmonary malignancy with plans for outpatient follow-up.
Over the next four days, the patient developed progressive dyspnea with cough and subjective fevers. The patient was readmitted with a diagnosis of postobstructive pneumonia and acute kidney injury (creatinine increased from 0.7 mg/dL to 2.9 mg/dL between admissions), and this finding was attributed to contrast-induced nephropathy from his recent CT scan. He was treated with vancomycin and piperacillin/tazobactam for two days but wished to transfer to a tertiary care hospital for a second opinion.
Postobstructive pneumonia, pulmonary embolism, and pleural effusion are common causes of dyspnea in patients with lung cancer. The patient’s travel and occupational history, lung nodules, acute renal insufficiency, and rapidly progressive respiratory symptoms prompt consideration for radiographic mimickers of lung cancer. Tuberculosis might present as a lung mass (pulmonary tuberculoma) during primary infection or reactivation. Noninfectious causes of pulmonary masses and nodules include metastatic cancer (eg, colon cancer), sarcoidosis, IgG4-related disease, and granulomatous polyangiitis (GPA).
Contrast-induced nephropathy is unusual in patients with normal renal function. More probable explanations include hypovolemia or acute tubular necrosis (ATN) from underlying inflammation. The patient’s CT-negative right upper quadrant pain may be a distinct process or represent another facet of a disseminated illness such as hepatic infiltration from lymphoma.
Upon arrival, the patient’s temperature was 38°C, heart rate (HR) 107 beats per minute, blood pressure (BP) 159/89 mm Hg, respiratory rate 25 breaths per minute, and oxygen saturation 92% on 2 L of oxygen per minute. He showed no signs of distress. Mild scleral icterus was noted. The cardiac exam was normal. Auscultation revealed scattered wheezes and crackles in the left upper lobe. Mild right upper quadrant tenderness without hepatosplenomegaly was noted on the abdominal exam. The patient’s lower extremities exhibited bilateral trace edema. No rash was observed, and his neurologic exam was normal.
The white blood cell (WBC) count was 28,300 per cubic millimeter (87% neutrophils, 3.6% lymphocytes, and 0.03% eosinophils), hemoglobin 11.1 g per deciliter, and platelet count 789,000 per cubic millimeter. Sodium was 127 mmol per liter, potassium 4.6 mmol per liter, chloride 101 mmol per liter, bicarbonate 13 mmol per liter, blood urea nitrogen 60 mg per deciliter, and creatinine 3.4 mg per deciliter. Aspartate aminotransferase and alanine aminotransferase levels were normal. Alkaline phosphatase was 283 units per liter (normal range, 31-95), and total bilirubin was 4.5 mg per deciliter (normal range, 0.2-1.3) with a direct bilirubin of 2.7 mg per deciliter. Urinalysis demonstrated urine protein of 30 mg/dL, specific gravity of 1.013, negative nitrites, 10-21 white cells per high-powered field (normal, < 5), and 21-50 red cells per high-powered field (normal, < 3). Urine microscopy revealed muddy brown casts but no cellular casts or dysmorphic red cells. A chest radiograph (CXR) showed patchy consolidations in the bilateral upper lobes and left lower lobe along with Kerley B lines, a small left pleural effusion, and thickened right horizontal fissure; the left upper lobe mass was re-demonstrated. Vancomycin, piperacillin-tazobactam, and azithromycin were administered.
At this point, the most likely source of sepsis is multifocal pneumonia. The patient is at risk for S. aureus and P. aeruginosa given his recent hospitalization. A severe form of leptospirosis (Weil’s disease) is associated with pulmonary disease, hyperbilirubinemia, and renal failure. Repeat abdominal imaging is necessary to evaluate for cholangitis given the patient’s right upper quadrant pain, fever, and jaundice. It would also help categorize his cholestatic pattern of liver injury as intrahepatic or extrahepatic (eg, stricture). An infiltrative disease such as sarcoidosis may cause both intrahepatic cholestasis and parenchymal lung disease, although the pleural pathology is uncommon.
His normal cardiac exam does not exclude cardiogenic pulmonary edema, a common cause of interstitial edema and pleural effusion. In this setting of systemic inflammation (neutrophilia, thrombocytosis, and hypoalbuminemia), the thickened right horizontal fissure and interlobular septa might represent an infiltrative process, such as lymphangitic carcinomatosis, lymphoma, or sarcoidosis.
Muddy brown casts are characteristic of ATN. The patient’s risk factors for ATN include sepsis and previously administered iodinated contrast. Fluid retention from oliguric renal failure is likely contributing to his hyponatremia and lower extremity edema. Pathology isolated to the tubules, however, would not cause hematuria and pyuria and suggests glomerular or interstitial disease. The lack of cellular casts on a single urinary specimen does not eliminate the likelihood of either disease. Hematuria and diffuse parenchymal lung disease prompt consideration of pulmonary-renal syndromes, such as anti-glomerular basement membrane disease, GPA, and systemic lupus erythematosus, which can all be triggered by infection.
On the night of transfer, the patient experienced acute respiratory distress. Heart rate was 130 beats per minute, BP 170/95 mm Hg, respiratory rate 40 breaths per minute, and oxygen saturation 88% on six liters of supplemental oxygen by nasal cannula. His arterial blood gas demonstrated a pH of 7.23, PaCO2 of 32 mm Hg, and PaO2 of 65 mm Hg. He was emergently intubated for progressive hypoxemic respiratory failure. A small amount of blood was noted in the endotracheal tube. A noncontrast CT of the chest demonstrated multifocal airspace opacities and bilateral pleural effusions. The previously noted left upper lobe mass was unchanged.
Rapid respiratory decline and diffuse alveolar disease commonly result from aspiration, flash pulmonary edema, and acute respiratory distress syndrome (ARDS). Necrotizing pneumonia (eg, S. aureus) and trauma during intubation are possible causes of blood in his endotracheal tube. However, in the setting of multifocal airspace opacity, renal insufficiency, hematuria, and rapid respiratory decline, the blood might represent diffuse alveolar hemorrhage (DAH). Bronchoscopy with bronchioalveolar lavage to evaluate for pulmonary edema, infection, and hemorrhage would be indicated.
The patient subsequently developed oliguria, requiring continuous renal replacement therapy. An echocardiogram demonstrated impaired left ventricular relaxation and a reduced ejection fraction of 45% without segmental wall motion abnormalities or valvular disease, and a right ventricular systolic pressure of 36 mm Hg. Over the next 12 hours, his respiratory status improved, and he was extubated to 15 L per minute of supplemental oxygen by high-flow nasal cannula (HFNC).
The pathology report of the lung biopsy from the other hospital disclosed chronic inflammation and fibrosis with ill-defined areas of necrosis and myxoid degeneration surrounded by nuclear palisading suggestive of granulomatous inflammation. Staining for acid-fast bacilli (AFB) and fungal organisms was negative.
The rapid pulmonary recovery is inconsistent with multifocal pneumonia or ARDS. Flash pulmonary edema might result in sudden hypoxemic respiratory failure that resolves with positive pressure ventilation and ultrafiltration. However, this condition would not explain the biopsy results. Granulomatous lung pathology often results from mycobacterial or fungal disease. Tuberculosis and fungal pneumonia are not excluded with negative staining alone. However, neither would cause self-limited respiratory failure. Histologic evidence of necrosis lessens the likelihood of sarcoidosis, which rarely causes fulminant pulmonary disease. Lymphoma can result in granulomatous inflammation but would not cause transient pulmonary disease. GPA, a cause of necrotizing granulomatous lung disease, might result in a lung mass and worsened hypoxemia through DAH.
The patient continued to require 15 L of oxygen per minute by HFNC. He had persistent bilateral perihilar alveolar and interstitial opacities on CXR. Repeat WBC count was 29,200 per cubic millimeter, hemoglobin 7.8 g per deciliter, and platelets 656,000 per cubic millimeter. The C-reactive protein was 300 mg per L (normal range, <6.3) and erythrocyte sedimentation rate 100 mm per hour (normal range, <10). Legionella urinary antigen, serum immunodiffusion for Coccidiodes imitus, human immunodeficiency virus antibody, respiratory viral panel, and beta-D glucan were negative. Rare acid-fast bacilli were visualized in one out of three concentrated AFB sputum smears. He was started on empiric antituberculous therapy with rifampin, isoniazid, pyrazinamide, and ethambutol.
The sputum sample is suggestive of pulmonary tuberculosis. The salient features of this case include systemic inflammation, pulmonary nodules and mass, necrotizing granulomatous lung pathology, renal insufficiency, and hematuria. Disseminated tuberculosis might explain all these findings. However, a positive AFB smear may signal the presence of a nontuberculous mycobacteria, which is less likely to cause this clinical syndrome.
M. tuberculosis complex polymerase chain reaction (MTB PCR) assay returned negative for M. tuberculosis. Antiproteinase 3 antibody was 1,930 units (normal range, <20). Antimyeloperoxidase and antiglomerular basement membrane antibodies were negative.
Tuberculosis and GPA share several overlapping features, such as necrotizing lung pathology and less commonly antineutrophil cytoplasmic autoantibody (ANCA)-associated antibodies. However, the lung mass, acute renal and respiratory failure, hematuria, and the degree of anti-proteinase 3 level elevation are highly suggestive of GPA. The negative MTB PCR raises the possibility that a nontuberculous mycobacterium was detected on the sputum smear. Nevertheless, continued treatment until finalization of culture results is appropriate given that tuberculosis is endemic in Mexico.
The patient’s presenting features of right upper quadrant tenderness, jaundice, and cholestatic hepatitis remain poorly explained by either of these diagnoses. Neither tuberculosis nor GPA commonly presents with accompanying hepatic involvement, though both have been occasionally described as causing hepatitis. As the greatest concern in this patient remains his progressive renal failure and accompanying pulmonary hemorrhage, a renal biopsy to assess for glomerulonephritis associated with GPA is warranted before further investigation into the cause of his cholestatic hepatitis.
A core renal biopsy demonstrated pauci-immune focal crescentic and necrotizing glomerulonephritis with mixed tubulointerstitial inflammation (Figure 2). In conjunction with the pulmonary syndrome and positive antiproteinase 3 serology, a diagnosis of granulomatosis with polyangiitis was made. The patient was treated with pulse dose steroids, rituximab, and plasma exchange. Two weeks later, the sputum mycobacterial culture returned positive for Mycobacterium llatzerense and anti-tuberculous treatment was discontinued.
Over the following weeks, the patient improved and was transitioned off dialysis prior to hospital discharge. By six months later, he had resolution of his hemoptysis, shortness of breath, liver biochemical test abnormalities, and significant improvement in his renal function. Repeat sputum mycobacterial cultures were negative.
DISCUSSION
A 65-year-old man from Mexico with a significant smoking history presented with an apical lung mass and cough, prioritizing tuberculosis and pulmonary malignancy. As the case unfolded, renal failure, multifocal lung opacities, conflicting tuberculosis test results, positive anti-proteinase 3 antibody, and ultimately a renal biopsy led to the diagnosis of granulomatosis with polyangiitis (GPA).
The correct interpretation of occasionally conflicting mycobacterial testing is crucial. Mycobacterial cultures remain the gold standard for diagnosing tuberculosis. However, results take weeks to return. Rapid tests include acid-fast bacilli (AFB) smear microscopy and nucleic acid-amplification tests (NAAT) of sputum or bronchoalveolar samples.1 When three sputum smears are performed, the sensitivity of AFB smear microscopy for tuberculosis in immunocompetent hosts is 70%.1 The AFB smear does not distinguish between different mycobacterial organisms. Thus, a positive result must be interpreted with the relative prevalence of tuberculosis and nontuberculous mycobacteria (NTM) in mind. The addition of NAAT-based assays has allowed for enhanced sensitivity and specificity in the diagnosis of tuberculosis, such that a negative NAAT in a patient with a positive AFB smear strongly argues for the presence of a NTM.2-4
NTM are widely prevalent environmental microbes, with over 140 species described, and careful consideration is required to determine if an isolate is pathogenic.5 Given their ubiquitous nature, a high rate of asymptomatic respiratory and cutaneous colonization occurs. Correspondingly, the diagnosis of NTM disease requires multiple positive cultures or pathologic features on tissue biopsy, compatible clinical findings, and diligent exclusion of other causes.5 A retrospective study of all NTM isolates in Oregon from 2005-2006 revealed that only 47% of patients met the guideline criteria for having symptomatic NTM disease.6 In our case, the patient’s sputum grew M. llatzerense, an aerobic, nonfermenting mycobacterium found in water sources that has only infrequently been implicated as a human pathogen.7,8 Subsequent AFB sputum cultures were negative, and serial imaging showed resolution of the pulmonary findings without additional antimycobacterial therapy, suggesting that this organism was not responsible for the disease process.
Along with microscopic polyangiitis (MPA) and eosinophilic granulomatosis with polyangiitis (EGPA), GPA is an antineutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis that predominantly affects small to medium sized vessels. Although it can occur at any age, GPA most commonly afflicts older adults, with men and women being diagnosed at roughly equal rates.9 GPA is a multisystem disease with a wide array of clinical manifestations. The most frequently involved sites of disease are the respiratory tract and kidneys, although virtually any organ can be affected. Sino-nasal disease, such as destructive sinusitis, or ear involvement are nearly universal. Lower respiratory manifestations occur in 60% of patients, but are highly diverse and reflect the inherent difficulty in diagnosing this condition.9-11 Additionally, GPA is a frequent cause of the pulmonary-renal syndromes, with glomerulonephritis occurring in 80% of patients.9
The diagnosis of GPA in this case was delayed, in part, due to features suggestive of malignancy and pulmonary tuberculosis. While sino-nasal disease was not noted during this hospitalization, the patient had many different respiratory manifestations, including a dominant pulmonary mass, diffuse nodules, and hypoxemic respiratory failure due to suspected diffuse alveolar hemorrhage (DAH), all of which have been reported in GPA.12 Dysmorphic red cells and red blood cell casts are not sensitive for renal involvement in GPA; their absence does not exclude the possibility of an ANCA-associated vasculitis.13 Hematuria and rapid progression to oliguric renal failure are characteristic of a vasculitic process and should sway clinicians away from a working diagnosis of ATN.
The diagnosis of GPA involves the synthesis of clinical data, radiographic findings, serologic testing, and histopathology. ANCA testing is an essential step in the diagnosis of GPA but has limitations. Patients with GPA more commonly have ANCAs targeting the enzyme proteinase-3 (PR3-ANCA), with MPA being more closely associated with myeloperoxidase (MPO-ANCA), although cross-reactivity and antibody-negative disease can occur.14 Although 90% of patients with GPA with multiorgan involvement will have a positive ANCA, a negative test is more common in localized upper airway disease, where only 50% have a positive ANCA.15 A number of drugs, medications, infections, and nonvasculitic autoimmune diseases have been associated with positive ANCA serologies in the absence of systemic vasculitis.14,16,17 As such, pathologic demonstration of vasculitis is necessary for establishing the diagnosis. Typical sites for biopsy include the kidneys and lungs.9
This case illustrates how clinicians often find themselves at a diagnostic crossroads—being forced to choose which clinical elements to prioritize. At various points, our patient’s presentation could have been framed as “a man from a Tb-endemic country with hemoptysis and an apical opacity,” “an elderly man with extensive smoking history and lung mass,” or “a patient with elevated inflammatory markers and pulmonary-renal syndrome”. In such situations, it is incumbent on the clinician to evaluate how well a given problem representation encompasses or highlights the salient features of a case. As with painting or photography, an essential aspect of appreciating the whole picture involves carefully selecting the right frame.
KEY TEACHING POINTS
- The diagnosis of tuberculosis relies on smear microscopy, nucleic-acid amplification testing (NAAT), and cultures. A positive AFB smear with negative NAAT suggests the presence of a nontuberculous mycobacteria (NTM).
- NTM are common environmental organisms and often exist as nonpathogenic colonizers.6 The diagnosis of NTM disease requires exclusion of other causes and careful clinical, microbiologic, and radiographic correlation.
- Granulomatosis with polyangiitis is a multisystem disease often involving the respiratory track and kidney. Pulmonary disease can present with airway involvement, parenchymal nodules, opacities, pleural findings, and diffuse alveolar hemorrhage.12
Disclosures
Drs. Minter, Geha, Boslett, Chung, and Ramani have no disclosures. Dr. Manesh is supported by the Jeremiah A. Barondess Fellowship in the Clinical Transaction of the New York Academy of Medicine, in collaboration with the Accreditation Council for Graduate Medical Education (ACGME).
1. Lewinsohn DM, Leonard MK, LoBue PA, et al. Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention clinical practice guidelines: diagnosis of tuberculosis in adults and children. Clin Infect Dis. 2017;64(2):e1-e33. PubMed
2. Steingart KR, Sohn H, Schiller I, et al. Xpert(R) MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. 2013;(1):Cd009593. PubMed
3. Luetkemeyer AF, Firnhaber C, Kendall MA, et al. Evaluation of Xpert MTB/RIF versus afb smear and culture to identify pulmonary tuberculosis in patients with suspected tuberculosis from low and higher prevalence settings. Clin Infect Dis. 2016;62(9):1081-1088. PubMed
4. Boehme CC, Nabeta P, Hillemann D, et al. Rapid molecular detection of tuberculosis and rifampin resistance. N Engl J Med. 2010;363(11):1005-1015. PubMed
5. Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175(4):367-416. PubMed
6. Winthrop KL, McNelley E, Kendall B, et al. Pulmonary nontuberculous mycobacterial disease prevalence and clinical features: an emerging public health disease. Am J Respir Crit Care Med. 2010;182(7):977-982. PubMed
7. Teixeira L, Avery RK, Iseman M, et al. Mycobacterium llatzerense lung infection in a liver transplant recipient: case report and review of the literature. Am J Transplant. 2013;13(8):2198-2200. PubMed
8. Cárdenas AM, Gomila M, Lalucat J, Edelstein PH. Abdominal abscess caused by Mycobacterium llatzerense. J Clin Microbiol. 2014;52(4):1287-1289. PubMed
9. Jennette JC, Falk RJ. Small-vessel vasculitis. N Engl J Med. 1997;337(21):1512-1523. PubMed
10. Mahr A, Katsahian S, Varet H, et al. Revisiting the classification of clinical phenotypes of anti-neutrophil cytoplasmic antibody-associated vasculitis: a cluster analysis. Ann Rheum Dis. 2013;72(6):1003-1010. PubMed
11. Holle JU, Gross WL, Latza U, et al. Improved outcome in 445 patients with Wegener’s granulomatosis in a German vasculitis center over four decades. Arthritis Rheum. 2011;63(1):257-266. PubMed
12. Cordier JF, Valeyre D, Guillevin L, Loire R, Brechot JM. Pulmonary Wegener’s granulomatosis. A clinical and imaging study of 77 cases. Chest. 1990;97(4):906-912. PubMed
13. Hamadah AM, Gharaibeh K, Mara KC, et al. Urinalysis for the diagnosis of glomerulonephritis: role of dysmorphic red blood cells. Nephrol Dial Transplant. 2018;33(8):1397-1403. PubMed
14. Jennette JC, Falk RJ. Pathogenesis of antineutrophil cytoplasmic autoantibody-mediated disease. Nat Rev Rheumatol. 2014;10(8):463-473. PubMed
15. Borner U, Landis BN, Banz Y, et al. Diagnostic value of biopsies in identifying cytoplasmic antineutrophil cytoplasmic antibody-negative localized Wegener’s granulomatosis presenting primarily with sinonasal disease. Am J Rhinol Allergy. 2012;26(6):475-480. PubMed
16. Mahr A, Batteux F, Tubiana S, et al. Brief report: prevalence of antineutrophil cytoplasmic antibodies in infective endocarditis. Arthritis Rheumatol. 2014;66(6):1672-1677. PubMed
17. Sherkat R, Mostafavizadeh K, Zeydabadi L, Shoaei P, Rostami S. Antineutrophil cytoplasmic antibodies in patients with pulmonary tuberculosis. Iran J Immunol. 2011;8(1):52-57. PubMed
1. Lewinsohn DM, Leonard MK, LoBue PA, et al. Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention clinical practice guidelines: diagnosis of tuberculosis in adults and children. Clin Infect Dis. 2017;64(2):e1-e33. PubMed
2. Steingart KR, Sohn H, Schiller I, et al. Xpert(R) MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. 2013;(1):Cd009593. PubMed
3. Luetkemeyer AF, Firnhaber C, Kendall MA, et al. Evaluation of Xpert MTB/RIF versus afb smear and culture to identify pulmonary tuberculosis in patients with suspected tuberculosis from low and higher prevalence settings. Clin Infect Dis. 2016;62(9):1081-1088. PubMed
4. Boehme CC, Nabeta P, Hillemann D, et al. Rapid molecular detection of tuberculosis and rifampin resistance. N Engl J Med. 2010;363(11):1005-1015. PubMed
5. Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175(4):367-416. PubMed
6. Winthrop KL, McNelley E, Kendall B, et al. Pulmonary nontuberculous mycobacterial disease prevalence and clinical features: an emerging public health disease. Am J Respir Crit Care Med. 2010;182(7):977-982. PubMed
7. Teixeira L, Avery RK, Iseman M, et al. Mycobacterium llatzerense lung infection in a liver transplant recipient: case report and review of the literature. Am J Transplant. 2013;13(8):2198-2200. PubMed
8. Cárdenas AM, Gomila M, Lalucat J, Edelstein PH. Abdominal abscess caused by Mycobacterium llatzerense. J Clin Microbiol. 2014;52(4):1287-1289. PubMed
9. Jennette JC, Falk RJ. Small-vessel vasculitis. N Engl J Med. 1997;337(21):1512-1523. PubMed
10. Mahr A, Katsahian S, Varet H, et al. Revisiting the classification of clinical phenotypes of anti-neutrophil cytoplasmic antibody-associated vasculitis: a cluster analysis. Ann Rheum Dis. 2013;72(6):1003-1010. PubMed
11. Holle JU, Gross WL, Latza U, et al. Improved outcome in 445 patients with Wegener’s granulomatosis in a German vasculitis center over four decades. Arthritis Rheum. 2011;63(1):257-266. PubMed
12. Cordier JF, Valeyre D, Guillevin L, Loire R, Brechot JM. Pulmonary Wegener’s granulomatosis. A clinical and imaging study of 77 cases. Chest. 1990;97(4):906-912. PubMed
13. Hamadah AM, Gharaibeh K, Mara KC, et al. Urinalysis for the diagnosis of glomerulonephritis: role of dysmorphic red blood cells. Nephrol Dial Transplant. 2018;33(8):1397-1403. PubMed
14. Jennette JC, Falk RJ. Pathogenesis of antineutrophil cytoplasmic autoantibody-mediated disease. Nat Rev Rheumatol. 2014;10(8):463-473. PubMed
15. Borner U, Landis BN, Banz Y, et al. Diagnostic value of biopsies in identifying cytoplasmic antineutrophil cytoplasmic antibody-negative localized Wegener’s granulomatosis presenting primarily with sinonasal disease. Am J Rhinol Allergy. 2012;26(6):475-480. PubMed
16. Mahr A, Batteux F, Tubiana S, et al. Brief report: prevalence of antineutrophil cytoplasmic antibodies in infective endocarditis. Arthritis Rheumatol. 2014;66(6):1672-1677. PubMed
17. Sherkat R, Mostafavizadeh K, Zeydabadi L, Shoaei P, Rostami S. Antineutrophil cytoplasmic antibodies in patients with pulmonary tuberculosis. Iran J Immunol. 2011;8(1):52-57. PubMed
© 2019 Society of Hospital Medicine
Reimagining Inpatient Care in Canadian Teaching Hospitals: Bold Initiatives or Tinkering at the Margins?
Canada’s 17 medical schools and their affiliated teaching hospitals are instrumental in serving local communities and providing regional and national access to specialized therapies. Akin to many other countries, patients in Canadian teaching hospitals typically receive care from trainees supervised by attending physicians on teams that Canadians refer to as clinical teaching units (CTUs).1 For more than 50 years, the CTU model has served trainees, attendings, and patients well.2 The success of the CTU model has been dependent on several factors including the crucial balance between the number of trainees and volume of patients. However, Canadian teaching hospitals are increasingly challenged by an imbalance in the trainee-to-patient volume equilibrium spurred by increasing patient volumes and declining house staff availability. The challenges we are facing today in Canada are similar to those teaching hospitals in the United States have faced and adapted to over the last 15 years. Can we build a new, sustainable model of inpatient care through attending-directed inpatient services much as has happened in the US?
Canada’s population of 36 million people is growing by approximately 1% per year, largely driven by immigration.3 At the same time, Canada’s population is aging and becoming increasingly medically complex; the percentage of Canadians age 65 years and older is anticipated to rise from approximately 17% today to 25% in 2035.4 Canada’s healthcare system historically functioned with relatively few inpatient beds, encouraging efficiency particularly with respect to which patients require hospital admission and which do not.5 Although data suggest that the number of hospital admissions declined in Canada between 1980 and 1995, recent data documented that General Internal Medicine admissions increased by 32% between 2010 and 2015 and accounted for 24% of total hospital bed days.6,7 The effects of population growth and aging on admission volumes might be mitigated to some extent by innovations in healthcare delivery such as improved access to primary care (largely family physicians in Canada). However, even with these innovations, a growing and aging population is likely to have a disproportionate effect on the types of undifferentiated illnesses that are typically admitted to General Internal Medicine in Canadian teaching hospitals.
Increasing volumes and complexity are occurring at the same time that residency training in Canada is undergoing an extraordinary shift, mirroring trends in other countries.8 CTUs in Canada typically have a census of 20 or more patients and are staffed by an attending, one senior resident, two to three junior residents, and medical students. Recognition that physician fatigue is associated with patient safety events and physician burnout has led to shorter resident shifts, though Canadian hospitals typically operate without concrete work hour limits or “hard” caps on team size.8 To fulfill accreditation standards set by the Royal College of Physicians and Surgeons of Canada, residency programs have required increases in formal teaching sessions during working hours, further reducing resident presence at the bedside. Many specialty training programs (eg, anesthesiology and ophthalmology) that traditionally required trainees to rotate through General Medicine have eliminated this requirement. Moreover, postgraduate training now requires additional time be spent in ambulatory and community hospital settings to better prepare residents for practice.9 There is little enthusiasm for increasing the number of residents, as postgraduate training spots increased by 85% between 2000 and 2013, before stabilizing in recent years.10
These factors are leading to a substantial decline in resident availability on CTUs, shifting increasing amounts of direct patient care to attending physicians in Canadian teaching hospitals across virtually all specialties. Unsurprisingly, increased rates of burnout and decreases in job satisfactio
Canadian teaching hospitals currently find themselves facing a confluence of factors nearly identical to those faced by teaching hospitals in the United States during 2003 when the Accreditation Council for Graduate Medical Education instituted resident duty hour restrictions to address concerns over trainee wellness, shift length, and patient safety.8 Instantly, hundreds of US teaching hospitals faced uncertainty over who would provide patient care when residents were unavailable. Virtually all US teaching hospitals responded with a creativity and speed that we are unaccustomed to in academic medicine. Hospitals reallocated money to finance attending-directed services where patient care was provided directly by attending physicians often working without trainees12 but frequently supported by nurse practitioners or physician assistants.13 Despite the differences between US and Canadian healthcare, 15 years later, we in Canada can and should learn from the US experience.14
Attending-directed services offer several advantages. First, attending-directed services offer patient outcomes including ICU transfer, mortality, readmissions, and satisfaction that are similar, if not modestly improved, when compared with traditional teaching services.15 Results also suggest potential reductions in hospital length of stay and diagnostic testing.16 Attending-directed services can enhance trainee education by insuring attending physician presence and oversight in-hospital 24-hours per day.17 Although not well studied, attending-directed services may reduce variation in CTU patient census so that excess volumes can be absorbed by attending-directed teams even with seasonal surges (eg, influenza). Recognizing that many specialties were experiencing the same challenges as General Medicine in 2003, attending-directed services in the US have been designed to care for a wide spectrum of patients drawn from an array of different specialties with evidence of improved outcomes.12 Building attending-directed services in Canadian teaching hospitals may expand to include patients from multiple specialties and subspecialties (surgery, orthopedics, and cardiology) where patient volumes are increasing and resident coverage is increasingly scarce.
The challenges that accompany the implementation of attending-directed teams must be acknowledged. First, while attending-directed teams solve many problems for teaching hospitals, physician billings may not generate sufficient income to be self-sustaining and require additional financial support.18 Without investment from hospitals or government, attending-directed models cannot flourish in teaching hospitals. US hospitals typically provide substantial financial support ($50,000-$100,000 per physician) to hospitalist programs, but Canadian teaching hospitals have been reluctant to follow suit.
Second, attending-directed services require a sustainable workforce. In Canada, inpatient care is provided predominately by family physician hospitalists in community hospitals, whereas internists typically fulfill these roles in teaching hospitals.19 Family physician hospitalists are commonly represented by the Canadian Society for Hospital Medicine, which is the Canadian branch of the Society of Hospital Medicine. Hospital medicine in Canada is typically organized around physician training (family physician vs internist) rather than clinical focus (outpatient vs inpatient). Collaborative models of care that unite hospitalists from all training streams (family physician, internist, and pediatrics) are only just emerging in Canadian teaching hospitals. How these programs are developed will be critical to the successful growth of attending-directed services. Third, if attending-directed services expand in teaching hospitals, the physicians who staff these services must come from somewhere. Either the “production” of physicians will need to increase or physicians will migrate to attending-directed services from outpatient practice or from community hospitals.20 Canadian teaching hospitals can also explore nurse practitioners and physician assistants, a previously underutilized resource. Though the costs of such programs can be significant,21 the payoff in safety, quality, and efficiency may be worth it—as demonstrated in the US system. Fourth, teaching hospitals and medical schools must create academic homes to support and mentor the physicians working on attending-directed services. Although physicians hired for attending-directed services primarily provide direct patient care, few will join academic medical centers solely for this purpose. Teaching hospitals and medical schools need to carefully consider job descriptions, mentoring, and career advancement opportunities as they build attending-directed services. Finally, the interactions between teaching and attending-directed services are complex. There is an inevitable learning curve as clinical operations and protocols are built and developed. For example, decisions need to be made about how patients are divided between services and whether nocturnists are responsible for teaching overnight residents.17 Successful programs have the potential to benefit hospitals, patients, learners, and faculty alike.
The risks associated with the status quo in Canada must also be addressed. Patient volumes and complexity in Canada are likely to continue to slowly increase, while the number of trainees in Canadian teaching hospitals will remain stable at best. Forcing more patients onto already overtaxed teaching services is likely to worsen hospital efficiency, patient outcomes, and educational experiences.22 Forcing additional patient care onto overstretched faculty will slowly erode the academic work (teaching and research) that has characterized excellence in Canadian medicine.
The changes we propose to overcome the challenges facing Canadian teaching hospitals are neither cheap nor easy (Table). We expect resistance on many fronts. Implementing them will likely require concerted advocacy from a diverse group of champions shining a bright spotlight on the sizable challenges Canadian teaching hospitals are confronting. We believe that each challenge maps to a discrete group of champions with discrete targets within hospital leadership, medical school administration, and government who will need to be engaged. In our opinion, organizing around these challenges offers the best opportunity to overcome the perpetual resistance around costs. Canadian teaching hospitals and their CTUs are under unprecedented pressure. Do we act boldly and embrace attending-directed models of care or continue tinkering at the margins?
Acknowledgments
The authors thank Chaim Bell for his advice and suggestions.
Disclosures
The authors have nothing to disclose.
1. Schrewe B, Pratt DD, McKellin WH. Adapting the forms of yesterday to the functions of today and the needs of tomorrow: a genealogical case study of clinical teaching units in Canada. Adv Health Sci Educ Theory Pract. 2016;21(2):475-499. PubMed
2. Maudsley RF. The clinical teaching unit in transition. CMAJ. 1993;148(9):1564-1566. PubMed
3. Statistics Canada. Recent Changes in Demographic Trends in Canada. Ottawa: Ontario, 2015. https://www150.statcan.gc.ca/n1/pub/75-006-x/2015001/article/14240-eng.htm. Accessed December 9, 2018
4. Statistics Canada. Census, Age and Sex. Ottawa: Ontario, 2016. https://www12.statcan.gc.ca/census-recensement/2016/rt-td/as-eng.cfm. Accessed December 10, 2018.
5. Papanicolas I, Woskie LR, Jha AK. Health care spending in the United States and other high-income countries. JAMA. 2018;319(10):1024-1039. PubMed
6. van Walraven C. Trends in 1-year survival of people admitted to hospital in Ontario, 1994-2009. CMAJ. 2013;185(16):E755-E762. PubMed
7. Verma AA, Guo Y, Kwan JL, et al. Patient characteristics, resource use and outcomes associated with general internal medicine hospital care: the General Medicine Inpatient Initiative (GEMINI) retrospective cohort study. CMAJ Open. 2017;5(4):E842-E849. PubMed
8. Pattani R, Wu PE, Dhalla IA. Resident duty hours in Canada: past, present and future. CMAJ. 2014;186(10):761-765. PubMed
9. Royal College of Physicians and Surgeons. Specialty Training Requirements in Internal medicine 2015. http://www.royalcollege.ca/cs/groups/public/documents/document/mdaw/mdg4/~edisp/088402.pdf. Accessed December 12, 2018.
10. Freeman TR, Petterson S, Finnegan S, Bazemore A. Shifting tides in the emigration patterns of Canadian physicians to the United States: a cross-sectional secondary data analysis. BMC Health Serv Res. 2016;16(1):678. PubMed
11. Wong BM, Imrie K. Why resident duty hours regulations must address attending physicians’ workload. Acad Med. 2013;88(9):1209-1211. PubMed
12. Flanders SA, Centor B, Weber V, McGinn T, DeSalvo K, Auerbach A. Challenges and opportunities in academic hospital medicine: report from the Academic Hospital Medicine Summit. J Hosp Med. 2009;4(4):240-246. PubMed
13. Torok H, Lackner C, Landis R, Wright S. Learning needs of physician assistants working in hospital medicine. J Hosp Med. 2012;7(3):190-194. PubMed
14. Ivers N, Brown AD, Detsky AS. Lessons from the Canadian experience with single-payer health insurance: just comfortable enough with the status quo. JAMA Intern Med. 2018;178(9):1250-1255. PubMed
15. Wray CM, Flores A, Padula WV, Prochaska MT, Meltzer DO, Arora VM. Measuring patient experiences on hospitalist and teaching services: patient responses to a 30-day postdischarge questionnaire. J Hosp Med. 2016;11(2):99-104. PubMed
16. Auerbach AD, Wachter RM, Katz P, Showstack J, Baron RB, Goldman L. Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes. Ann Intern Med. 2002;137(11):859-865. PubMed
17. Farnan JM, Burger A, Boonyasai RT, et al. Survey of overnight academic hospitalist supervision of trainees. J Hosp Med. 2012;7(7):521-523. PubMed
18. Gonzalo JD, Kuperman EF, Chuang CH, Lehman E, Glasser F, Abendroth T. Impact of an overnight internal medicine academic hospitalist program on patient outcomes. J Gen Intern Med. 2015;30(12):1795-1802. PubMed
19. Soong C, Fan E, Howell EE, et al. Characteristics of Hospitalists and Hospitalist Programs in the United States and Canada 2009. J Clin Outcomes Meas. 2009; 16 (2): 69-74.
20. Yousefi V, Maslowski R. Health system drivers of hospital medicine in Canada: systematic review. Can Fam Phys Med Fam Can. 2013;59(7):762-767. PubMed
21. Nuckols TK, Escarce JJ. Cost implications of ACGME’s 2011 changes to resident duty hours and the training environment. J Gen Intern Med. 2012;27(2):241-249. PubMed
22. Elliott DJ, Young RS, Brice J, Aguiar R, Kolm P. Effect of hospitalist workload on the quality and efficiency of care. JAMA Intern Med. 2014;174(5):786-793. PubMed
Canada’s 17 medical schools and their affiliated teaching hospitals are instrumental in serving local communities and providing regional and national access to specialized therapies. Akin to many other countries, patients in Canadian teaching hospitals typically receive care from trainees supervised by attending physicians on teams that Canadians refer to as clinical teaching units (CTUs).1 For more than 50 years, the CTU model has served trainees, attendings, and patients well.2 The success of the CTU model has been dependent on several factors including the crucial balance between the number of trainees and volume of patients. However, Canadian teaching hospitals are increasingly challenged by an imbalance in the trainee-to-patient volume equilibrium spurred by increasing patient volumes and declining house staff availability. The challenges we are facing today in Canada are similar to those teaching hospitals in the United States have faced and adapted to over the last 15 years. Can we build a new, sustainable model of inpatient care through attending-directed inpatient services much as has happened in the US?
Canada’s population of 36 million people is growing by approximately 1% per year, largely driven by immigration.3 At the same time, Canada’s population is aging and becoming increasingly medically complex; the percentage of Canadians age 65 years and older is anticipated to rise from approximately 17% today to 25% in 2035.4 Canada’s healthcare system historically functioned with relatively few inpatient beds, encouraging efficiency particularly with respect to which patients require hospital admission and which do not.5 Although data suggest that the number of hospital admissions declined in Canada between 1980 and 1995, recent data documented that General Internal Medicine admissions increased by 32% between 2010 and 2015 and accounted for 24% of total hospital bed days.6,7 The effects of population growth and aging on admission volumes might be mitigated to some extent by innovations in healthcare delivery such as improved access to primary care (largely family physicians in Canada). However, even with these innovations, a growing and aging population is likely to have a disproportionate effect on the types of undifferentiated illnesses that are typically admitted to General Internal Medicine in Canadian teaching hospitals.
Increasing volumes and complexity are occurring at the same time that residency training in Canada is undergoing an extraordinary shift, mirroring trends in other countries.8 CTUs in Canada typically have a census of 20 or more patients and are staffed by an attending, one senior resident, two to three junior residents, and medical students. Recognition that physician fatigue is associated with patient safety events and physician burnout has led to shorter resident shifts, though Canadian hospitals typically operate without concrete work hour limits or “hard” caps on team size.8 To fulfill accreditation standards set by the Royal College of Physicians and Surgeons of Canada, residency programs have required increases in formal teaching sessions during working hours, further reducing resident presence at the bedside. Many specialty training programs (eg, anesthesiology and ophthalmology) that traditionally required trainees to rotate through General Medicine have eliminated this requirement. Moreover, postgraduate training now requires additional time be spent in ambulatory and community hospital settings to better prepare residents for practice.9 There is little enthusiasm for increasing the number of residents, as postgraduate training spots increased by 85% between 2000 and 2013, before stabilizing in recent years.10
These factors are leading to a substantial decline in resident availability on CTUs, shifting increasing amounts of direct patient care to attending physicians in Canadian teaching hospitals across virtually all specialties. Unsurprisingly, increased rates of burnout and decreases in job satisfactio
Canadian teaching hospitals currently find themselves facing a confluence of factors nearly identical to those faced by teaching hospitals in the United States during 2003 when the Accreditation Council for Graduate Medical Education instituted resident duty hour restrictions to address concerns over trainee wellness, shift length, and patient safety.8 Instantly, hundreds of US teaching hospitals faced uncertainty over who would provide patient care when residents were unavailable. Virtually all US teaching hospitals responded with a creativity and speed that we are unaccustomed to in academic medicine. Hospitals reallocated money to finance attending-directed services where patient care was provided directly by attending physicians often working without trainees12 but frequently supported by nurse practitioners or physician assistants.13 Despite the differences between US and Canadian healthcare, 15 years later, we in Canada can and should learn from the US experience.14
Attending-directed services offer several advantages. First, attending-directed services offer patient outcomes including ICU transfer, mortality, readmissions, and satisfaction that are similar, if not modestly improved, when compared with traditional teaching services.15 Results also suggest potential reductions in hospital length of stay and diagnostic testing.16 Attending-directed services can enhance trainee education by insuring attending physician presence and oversight in-hospital 24-hours per day.17 Although not well studied, attending-directed services may reduce variation in CTU patient census so that excess volumes can be absorbed by attending-directed teams even with seasonal surges (eg, influenza). Recognizing that many specialties were experiencing the same challenges as General Medicine in 2003, attending-directed services in the US have been designed to care for a wide spectrum of patients drawn from an array of different specialties with evidence of improved outcomes.12 Building attending-directed services in Canadian teaching hospitals may expand to include patients from multiple specialties and subspecialties (surgery, orthopedics, and cardiology) where patient volumes are increasing and resident coverage is increasingly scarce.
The challenges that accompany the implementation of attending-directed teams must be acknowledged. First, while attending-directed teams solve many problems for teaching hospitals, physician billings may not generate sufficient income to be self-sustaining and require additional financial support.18 Without investment from hospitals or government, attending-directed models cannot flourish in teaching hospitals. US hospitals typically provide substantial financial support ($50,000-$100,000 per physician) to hospitalist programs, but Canadian teaching hospitals have been reluctant to follow suit.
Second, attending-directed services require a sustainable workforce. In Canada, inpatient care is provided predominately by family physician hospitalists in community hospitals, whereas internists typically fulfill these roles in teaching hospitals.19 Family physician hospitalists are commonly represented by the Canadian Society for Hospital Medicine, which is the Canadian branch of the Society of Hospital Medicine. Hospital medicine in Canada is typically organized around physician training (family physician vs internist) rather than clinical focus (outpatient vs inpatient). Collaborative models of care that unite hospitalists from all training streams (family physician, internist, and pediatrics) are only just emerging in Canadian teaching hospitals. How these programs are developed will be critical to the successful growth of attending-directed services. Third, if attending-directed services expand in teaching hospitals, the physicians who staff these services must come from somewhere. Either the “production” of physicians will need to increase or physicians will migrate to attending-directed services from outpatient practice or from community hospitals.20 Canadian teaching hospitals can also explore nurse practitioners and physician assistants, a previously underutilized resource. Though the costs of such programs can be significant,21 the payoff in safety, quality, and efficiency may be worth it—as demonstrated in the US system. Fourth, teaching hospitals and medical schools must create academic homes to support and mentor the physicians working on attending-directed services. Although physicians hired for attending-directed services primarily provide direct patient care, few will join academic medical centers solely for this purpose. Teaching hospitals and medical schools need to carefully consider job descriptions, mentoring, and career advancement opportunities as they build attending-directed services. Finally, the interactions between teaching and attending-directed services are complex. There is an inevitable learning curve as clinical operations and protocols are built and developed. For example, decisions need to be made about how patients are divided between services and whether nocturnists are responsible for teaching overnight residents.17 Successful programs have the potential to benefit hospitals, patients, learners, and faculty alike.
The risks associated with the status quo in Canada must also be addressed. Patient volumes and complexity in Canada are likely to continue to slowly increase, while the number of trainees in Canadian teaching hospitals will remain stable at best. Forcing more patients onto already overtaxed teaching services is likely to worsen hospital efficiency, patient outcomes, and educational experiences.22 Forcing additional patient care onto overstretched faculty will slowly erode the academic work (teaching and research) that has characterized excellence in Canadian medicine.
The changes we propose to overcome the challenges facing Canadian teaching hospitals are neither cheap nor easy (Table). We expect resistance on many fronts. Implementing them will likely require concerted advocacy from a diverse group of champions shining a bright spotlight on the sizable challenges Canadian teaching hospitals are confronting. We believe that each challenge maps to a discrete group of champions with discrete targets within hospital leadership, medical school administration, and government who will need to be engaged. In our opinion, organizing around these challenges offers the best opportunity to overcome the perpetual resistance around costs. Canadian teaching hospitals and their CTUs are under unprecedented pressure. Do we act boldly and embrace attending-directed models of care or continue tinkering at the margins?
Acknowledgments
The authors thank Chaim Bell for his advice and suggestions.
Disclosures
The authors have nothing to disclose.
Canada’s 17 medical schools and their affiliated teaching hospitals are instrumental in serving local communities and providing regional and national access to specialized therapies. Akin to many other countries, patients in Canadian teaching hospitals typically receive care from trainees supervised by attending physicians on teams that Canadians refer to as clinical teaching units (CTUs).1 For more than 50 years, the CTU model has served trainees, attendings, and patients well.2 The success of the CTU model has been dependent on several factors including the crucial balance between the number of trainees and volume of patients. However, Canadian teaching hospitals are increasingly challenged by an imbalance in the trainee-to-patient volume equilibrium spurred by increasing patient volumes and declining house staff availability. The challenges we are facing today in Canada are similar to those teaching hospitals in the United States have faced and adapted to over the last 15 years. Can we build a new, sustainable model of inpatient care through attending-directed inpatient services much as has happened in the US?
Canada’s population of 36 million people is growing by approximately 1% per year, largely driven by immigration.3 At the same time, Canada’s population is aging and becoming increasingly medically complex; the percentage of Canadians age 65 years and older is anticipated to rise from approximately 17% today to 25% in 2035.4 Canada’s healthcare system historically functioned with relatively few inpatient beds, encouraging efficiency particularly with respect to which patients require hospital admission and which do not.5 Although data suggest that the number of hospital admissions declined in Canada between 1980 and 1995, recent data documented that General Internal Medicine admissions increased by 32% between 2010 and 2015 and accounted for 24% of total hospital bed days.6,7 The effects of population growth and aging on admission volumes might be mitigated to some extent by innovations in healthcare delivery such as improved access to primary care (largely family physicians in Canada). However, even with these innovations, a growing and aging population is likely to have a disproportionate effect on the types of undifferentiated illnesses that are typically admitted to General Internal Medicine in Canadian teaching hospitals.
Increasing volumes and complexity are occurring at the same time that residency training in Canada is undergoing an extraordinary shift, mirroring trends in other countries.8 CTUs in Canada typically have a census of 20 or more patients and are staffed by an attending, one senior resident, two to three junior residents, and medical students. Recognition that physician fatigue is associated with patient safety events and physician burnout has led to shorter resident shifts, though Canadian hospitals typically operate without concrete work hour limits or “hard” caps on team size.8 To fulfill accreditation standards set by the Royal College of Physicians and Surgeons of Canada, residency programs have required increases in formal teaching sessions during working hours, further reducing resident presence at the bedside. Many specialty training programs (eg, anesthesiology and ophthalmology) that traditionally required trainees to rotate through General Medicine have eliminated this requirement. Moreover, postgraduate training now requires additional time be spent in ambulatory and community hospital settings to better prepare residents for practice.9 There is little enthusiasm for increasing the number of residents, as postgraduate training spots increased by 85% between 2000 and 2013, before stabilizing in recent years.10
These factors are leading to a substantial decline in resident availability on CTUs, shifting increasing amounts of direct patient care to attending physicians in Canadian teaching hospitals across virtually all specialties. Unsurprisingly, increased rates of burnout and decreases in job satisfactio
Canadian teaching hospitals currently find themselves facing a confluence of factors nearly identical to those faced by teaching hospitals in the United States during 2003 when the Accreditation Council for Graduate Medical Education instituted resident duty hour restrictions to address concerns over trainee wellness, shift length, and patient safety.8 Instantly, hundreds of US teaching hospitals faced uncertainty over who would provide patient care when residents were unavailable. Virtually all US teaching hospitals responded with a creativity and speed that we are unaccustomed to in academic medicine. Hospitals reallocated money to finance attending-directed services where patient care was provided directly by attending physicians often working without trainees12 but frequently supported by nurse practitioners or physician assistants.13 Despite the differences between US and Canadian healthcare, 15 years later, we in Canada can and should learn from the US experience.14
Attending-directed services offer several advantages. First, attending-directed services offer patient outcomes including ICU transfer, mortality, readmissions, and satisfaction that are similar, if not modestly improved, when compared with traditional teaching services.15 Results also suggest potential reductions in hospital length of stay and diagnostic testing.16 Attending-directed services can enhance trainee education by insuring attending physician presence and oversight in-hospital 24-hours per day.17 Although not well studied, attending-directed services may reduce variation in CTU patient census so that excess volumes can be absorbed by attending-directed teams even with seasonal surges (eg, influenza). Recognizing that many specialties were experiencing the same challenges as General Medicine in 2003, attending-directed services in the US have been designed to care for a wide spectrum of patients drawn from an array of different specialties with evidence of improved outcomes.12 Building attending-directed services in Canadian teaching hospitals may expand to include patients from multiple specialties and subspecialties (surgery, orthopedics, and cardiology) where patient volumes are increasing and resident coverage is increasingly scarce.
The challenges that accompany the implementation of attending-directed teams must be acknowledged. First, while attending-directed teams solve many problems for teaching hospitals, physician billings may not generate sufficient income to be self-sustaining and require additional financial support.18 Without investment from hospitals or government, attending-directed models cannot flourish in teaching hospitals. US hospitals typically provide substantial financial support ($50,000-$100,000 per physician) to hospitalist programs, but Canadian teaching hospitals have been reluctant to follow suit.
Second, attending-directed services require a sustainable workforce. In Canada, inpatient care is provided predominately by family physician hospitalists in community hospitals, whereas internists typically fulfill these roles in teaching hospitals.19 Family physician hospitalists are commonly represented by the Canadian Society for Hospital Medicine, which is the Canadian branch of the Society of Hospital Medicine. Hospital medicine in Canada is typically organized around physician training (family physician vs internist) rather than clinical focus (outpatient vs inpatient). Collaborative models of care that unite hospitalists from all training streams (family physician, internist, and pediatrics) are only just emerging in Canadian teaching hospitals. How these programs are developed will be critical to the successful growth of attending-directed services. Third, if attending-directed services expand in teaching hospitals, the physicians who staff these services must come from somewhere. Either the “production” of physicians will need to increase or physicians will migrate to attending-directed services from outpatient practice or from community hospitals.20 Canadian teaching hospitals can also explore nurse practitioners and physician assistants, a previously underutilized resource. Though the costs of such programs can be significant,21 the payoff in safety, quality, and efficiency may be worth it—as demonstrated in the US system. Fourth, teaching hospitals and medical schools must create academic homes to support and mentor the physicians working on attending-directed services. Although physicians hired for attending-directed services primarily provide direct patient care, few will join academic medical centers solely for this purpose. Teaching hospitals and medical schools need to carefully consider job descriptions, mentoring, and career advancement opportunities as they build attending-directed services. Finally, the interactions between teaching and attending-directed services are complex. There is an inevitable learning curve as clinical operations and protocols are built and developed. For example, decisions need to be made about how patients are divided between services and whether nocturnists are responsible for teaching overnight residents.17 Successful programs have the potential to benefit hospitals, patients, learners, and faculty alike.
The risks associated with the status quo in Canada must also be addressed. Patient volumes and complexity in Canada are likely to continue to slowly increase, while the number of trainees in Canadian teaching hospitals will remain stable at best. Forcing more patients onto already overtaxed teaching services is likely to worsen hospital efficiency, patient outcomes, and educational experiences.22 Forcing additional patient care onto overstretched faculty will slowly erode the academic work (teaching and research) that has characterized excellence in Canadian medicine.
The changes we propose to overcome the challenges facing Canadian teaching hospitals are neither cheap nor easy (Table). We expect resistance on many fronts. Implementing them will likely require concerted advocacy from a diverse group of champions shining a bright spotlight on the sizable challenges Canadian teaching hospitals are confronting. We believe that each challenge maps to a discrete group of champions with discrete targets within hospital leadership, medical school administration, and government who will need to be engaged. In our opinion, organizing around these challenges offers the best opportunity to overcome the perpetual resistance around costs. Canadian teaching hospitals and their CTUs are under unprecedented pressure. Do we act boldly and embrace attending-directed models of care or continue tinkering at the margins?
Acknowledgments
The authors thank Chaim Bell for his advice and suggestions.
Disclosures
The authors have nothing to disclose.
1. Schrewe B, Pratt DD, McKellin WH. Adapting the forms of yesterday to the functions of today and the needs of tomorrow: a genealogical case study of clinical teaching units in Canada. Adv Health Sci Educ Theory Pract. 2016;21(2):475-499. PubMed
2. Maudsley RF. The clinical teaching unit in transition. CMAJ. 1993;148(9):1564-1566. PubMed
3. Statistics Canada. Recent Changes in Demographic Trends in Canada. Ottawa: Ontario, 2015. https://www150.statcan.gc.ca/n1/pub/75-006-x/2015001/article/14240-eng.htm. Accessed December 9, 2018
4. Statistics Canada. Census, Age and Sex. Ottawa: Ontario, 2016. https://www12.statcan.gc.ca/census-recensement/2016/rt-td/as-eng.cfm. Accessed December 10, 2018.
5. Papanicolas I, Woskie LR, Jha AK. Health care spending in the United States and other high-income countries. JAMA. 2018;319(10):1024-1039. PubMed
6. van Walraven C. Trends in 1-year survival of people admitted to hospital in Ontario, 1994-2009. CMAJ. 2013;185(16):E755-E762. PubMed
7. Verma AA, Guo Y, Kwan JL, et al. Patient characteristics, resource use and outcomes associated with general internal medicine hospital care: the General Medicine Inpatient Initiative (GEMINI) retrospective cohort study. CMAJ Open. 2017;5(4):E842-E849. PubMed
8. Pattani R, Wu PE, Dhalla IA. Resident duty hours in Canada: past, present and future. CMAJ. 2014;186(10):761-765. PubMed
9. Royal College of Physicians and Surgeons. Specialty Training Requirements in Internal medicine 2015. http://www.royalcollege.ca/cs/groups/public/documents/document/mdaw/mdg4/~edisp/088402.pdf. Accessed December 12, 2018.
10. Freeman TR, Petterson S, Finnegan S, Bazemore A. Shifting tides in the emigration patterns of Canadian physicians to the United States: a cross-sectional secondary data analysis. BMC Health Serv Res. 2016;16(1):678. PubMed
11. Wong BM, Imrie K. Why resident duty hours regulations must address attending physicians’ workload. Acad Med. 2013;88(9):1209-1211. PubMed
12. Flanders SA, Centor B, Weber V, McGinn T, DeSalvo K, Auerbach A. Challenges and opportunities in academic hospital medicine: report from the Academic Hospital Medicine Summit. J Hosp Med. 2009;4(4):240-246. PubMed
13. Torok H, Lackner C, Landis R, Wright S. Learning needs of physician assistants working in hospital medicine. J Hosp Med. 2012;7(3):190-194. PubMed
14. Ivers N, Brown AD, Detsky AS. Lessons from the Canadian experience with single-payer health insurance: just comfortable enough with the status quo. JAMA Intern Med. 2018;178(9):1250-1255. PubMed
15. Wray CM, Flores A, Padula WV, Prochaska MT, Meltzer DO, Arora VM. Measuring patient experiences on hospitalist and teaching services: patient responses to a 30-day postdischarge questionnaire. J Hosp Med. 2016;11(2):99-104. PubMed
16. Auerbach AD, Wachter RM, Katz P, Showstack J, Baron RB, Goldman L. Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes. Ann Intern Med. 2002;137(11):859-865. PubMed
17. Farnan JM, Burger A, Boonyasai RT, et al. Survey of overnight academic hospitalist supervision of trainees. J Hosp Med. 2012;7(7):521-523. PubMed
18. Gonzalo JD, Kuperman EF, Chuang CH, Lehman E, Glasser F, Abendroth T. Impact of an overnight internal medicine academic hospitalist program on patient outcomes. J Gen Intern Med. 2015;30(12):1795-1802. PubMed
19. Soong C, Fan E, Howell EE, et al. Characteristics of Hospitalists and Hospitalist Programs in the United States and Canada 2009. J Clin Outcomes Meas. 2009; 16 (2): 69-74.
20. Yousefi V, Maslowski R. Health system drivers of hospital medicine in Canada: systematic review. Can Fam Phys Med Fam Can. 2013;59(7):762-767. PubMed
21. Nuckols TK, Escarce JJ. Cost implications of ACGME’s 2011 changes to resident duty hours and the training environment. J Gen Intern Med. 2012;27(2):241-249. PubMed
22. Elliott DJ, Young RS, Brice J, Aguiar R, Kolm P. Effect of hospitalist workload on the quality and efficiency of care. JAMA Intern Med. 2014;174(5):786-793. PubMed
1. Schrewe B, Pratt DD, McKellin WH. Adapting the forms of yesterday to the functions of today and the needs of tomorrow: a genealogical case study of clinical teaching units in Canada. Adv Health Sci Educ Theory Pract. 2016;21(2):475-499. PubMed
2. Maudsley RF. The clinical teaching unit in transition. CMAJ. 1993;148(9):1564-1566. PubMed
3. Statistics Canada. Recent Changes in Demographic Trends in Canada. Ottawa: Ontario, 2015. https://www150.statcan.gc.ca/n1/pub/75-006-x/2015001/article/14240-eng.htm. Accessed December 9, 2018
4. Statistics Canada. Census, Age and Sex. Ottawa: Ontario, 2016. https://www12.statcan.gc.ca/census-recensement/2016/rt-td/as-eng.cfm. Accessed December 10, 2018.
5. Papanicolas I, Woskie LR, Jha AK. Health care spending in the United States and other high-income countries. JAMA. 2018;319(10):1024-1039. PubMed
6. van Walraven C. Trends in 1-year survival of people admitted to hospital in Ontario, 1994-2009. CMAJ. 2013;185(16):E755-E762. PubMed
7. Verma AA, Guo Y, Kwan JL, et al. Patient characteristics, resource use and outcomes associated with general internal medicine hospital care: the General Medicine Inpatient Initiative (GEMINI) retrospective cohort study. CMAJ Open. 2017;5(4):E842-E849. PubMed
8. Pattani R, Wu PE, Dhalla IA. Resident duty hours in Canada: past, present and future. CMAJ. 2014;186(10):761-765. PubMed
9. Royal College of Physicians and Surgeons. Specialty Training Requirements in Internal medicine 2015. http://www.royalcollege.ca/cs/groups/public/documents/document/mdaw/mdg4/~edisp/088402.pdf. Accessed December 12, 2018.
10. Freeman TR, Petterson S, Finnegan S, Bazemore A. Shifting tides in the emigration patterns of Canadian physicians to the United States: a cross-sectional secondary data analysis. BMC Health Serv Res. 2016;16(1):678. PubMed
11. Wong BM, Imrie K. Why resident duty hours regulations must address attending physicians’ workload. Acad Med. 2013;88(9):1209-1211. PubMed
12. Flanders SA, Centor B, Weber V, McGinn T, DeSalvo K, Auerbach A. Challenges and opportunities in academic hospital medicine: report from the Academic Hospital Medicine Summit. J Hosp Med. 2009;4(4):240-246. PubMed
13. Torok H, Lackner C, Landis R, Wright S. Learning needs of physician assistants working in hospital medicine. J Hosp Med. 2012;7(3):190-194. PubMed
14. Ivers N, Brown AD, Detsky AS. Lessons from the Canadian experience with single-payer health insurance: just comfortable enough with the status quo. JAMA Intern Med. 2018;178(9):1250-1255. PubMed
15. Wray CM, Flores A, Padula WV, Prochaska MT, Meltzer DO, Arora VM. Measuring patient experiences on hospitalist and teaching services: patient responses to a 30-day postdischarge questionnaire. J Hosp Med. 2016;11(2):99-104. PubMed
16. Auerbach AD, Wachter RM, Katz P, Showstack J, Baron RB, Goldman L. Implementation of a voluntary hospitalist service at a community teaching hospital: improved clinical efficiency and patient outcomes. Ann Intern Med. 2002;137(11):859-865. PubMed
17. Farnan JM, Burger A, Boonyasai RT, et al. Survey of overnight academic hospitalist supervision of trainees. J Hosp Med. 2012;7(7):521-523. PubMed
18. Gonzalo JD, Kuperman EF, Chuang CH, Lehman E, Glasser F, Abendroth T. Impact of an overnight internal medicine academic hospitalist program on patient outcomes. J Gen Intern Med. 2015;30(12):1795-1802. PubMed
19. Soong C, Fan E, Howell EE, et al. Characteristics of Hospitalists and Hospitalist Programs in the United States and Canada 2009. J Clin Outcomes Meas. 2009; 16 (2): 69-74.
20. Yousefi V, Maslowski R. Health system drivers of hospital medicine in Canada: systematic review. Can Fam Phys Med Fam Can. 2013;59(7):762-767. PubMed
21. Nuckols TK, Escarce JJ. Cost implications of ACGME’s 2011 changes to resident duty hours and the training environment. J Gen Intern Med. 2012;27(2):241-249. PubMed
22. Elliott DJ, Young RS, Brice J, Aguiar R, Kolm P. Effect of hospitalist workload on the quality and efficiency of care. JAMA Intern Med. 2014;174(5):786-793. PubMed
© 2019 Society of Hospital Medicine
Leadership & Professional Development: Be the Change You Want to See
“…a truly strong, powerful man isn’t threatened by a strong, powerful woman. Instead, he is challenged by her, he is inspired by her, he is pleased to relate to her as an equal.”
—Michelle Obama
Mentorship is essential to success in hospital medicine and may be particularly important for women. Cross-gender mentorship is especially salient since roughly equal proportions of women and men enter the medical pipeline, but men occupy over 75% of senior leadership roles in healthcare companies.
Cross-gender mentorship poses challenges but can be done successfully.1 We’ve made cross-gender mentoring work well in our own mentoring relationship. We describe three practices for effective mentoring that are especially important for men who mentor women given how common the female mentee-male mentor dyad is in medicine. We make generalizations that don’t apply universally but illustrate the social context in which such mentorship resides.
BE MINDFUL OF GENDER SCRIPTS
Gender scripts refer to social norms relating to gender identities and behaviors. Archetypal scripts include the father/daughter relationship and the knight/damsel-in-distress. Gender scripts often frame women as powerless—waiting to be rescued. By unconsciously activating a gender script, a mentor may reinforce a stereotype that women need rescuing (eg, “She’s really upset—I’ll email her Division Chief and help fix it for her”) or underestimate a mentee’s readiness for independence (eg, “She’s written four papers on this, but she’s still not ready to be senior author”). Astute mentors use reflection to combat gender scripts, asking themselves, “Am I allowing latent biases to affect my judgement?” They also consider when to intervene and when to let the mentee “rescue” herself (eg, “This is challenging, but I trust your judgement. What do you think you should do next?”).
PROMOTE RECIPROCAL LEARNING
Many women value collaborative behaviors and gravitate towards egalitarian learning environments at odds with a traditional, “top-down” mentorship model. Additionally, women may be penalized for demonstrating competitive behaviors, while identical behaviors are chalked up to confidence in men. A critical task, then, is for mentors to coach women to hone their natural leadership style, whether it be more commanding or more communal. A mentor can provide key feedback to the mentee about how her approach might be perceived and how to tweak it for optimal success. Mentors may wish to share missteps and even ask the mentee for advice. Pointing to her competence promotes “relational mentoring” and reciprocal learning, where mentor and mentee can learn positive behaviors from each other.
BE THE CHANGE YOU WANT TO SEE
Mentors will ideally wield their social capital to advance policies that promote gender equity—including fair recruiting, promotion, salary, paid leave, and breastfeeding policies. Exceptional mentors recognize that women may generally have less social capital than men in many organizations, and they proactively make women’s accomplishments more visible.2 They broadcast women’s strengths and nominate women for talks, national committees, honorific societies, and leadership positions. Effective mentors recognize that 30% of female medical faculty report experiencing sexual harassment at work,3 and thus maintain extremely high standards for professional integrity, for both themselves and others who interact with their mentees. They call out sexist remarks in the workplace as unacceptable, making it clear that such behavior won’t be tolerated. As Mohandas Gandhi said: “Be the change that you wish to see in the world.”
Cross-gender mentorship is critical to get right—nearly half our medical workforce depends on it. Men who mentor women help their organizations and gain satisfaction from playing a pivotal role in women’s advancement. When women succeed, we all do.
Disclosures
Dr. Moniz and Dr. Saint have nothing to disclose.
1. Byerley JS. Mentoring in the Era of #MeToo. JAMA. 2018;319(12):1199-1200. PubMed
2. Chopra V, Arora VM, Saint S. Will You Be My Mentor?-Four Archetypes to Help Mentees Succeed in Academic Medicine. JAMA Intern Med. 2018;178(2):175-176. PubMed
3. Jagsi R, Griffith KA, Jones R, Perumalswami CR, Ubel P, Stewart A. Sexual Harassment and Discrimination Experiences of Academic Medical Faculty. JAMA. 2016;315(19):2120-2121. PubMed
“…a truly strong, powerful man isn’t threatened by a strong, powerful woman. Instead, he is challenged by her, he is inspired by her, he is pleased to relate to her as an equal.”
—Michelle Obama
Mentorship is essential to success in hospital medicine and may be particularly important for women. Cross-gender mentorship is especially salient since roughly equal proportions of women and men enter the medical pipeline, but men occupy over 75% of senior leadership roles in healthcare companies.
Cross-gender mentorship poses challenges but can be done successfully.1 We’ve made cross-gender mentoring work well in our own mentoring relationship. We describe three practices for effective mentoring that are especially important for men who mentor women given how common the female mentee-male mentor dyad is in medicine. We make generalizations that don’t apply universally but illustrate the social context in which such mentorship resides.
BE MINDFUL OF GENDER SCRIPTS
Gender scripts refer to social norms relating to gender identities and behaviors. Archetypal scripts include the father/daughter relationship and the knight/damsel-in-distress. Gender scripts often frame women as powerless—waiting to be rescued. By unconsciously activating a gender script, a mentor may reinforce a stereotype that women need rescuing (eg, “She’s really upset—I’ll email her Division Chief and help fix it for her”) or underestimate a mentee’s readiness for independence (eg, “She’s written four papers on this, but she’s still not ready to be senior author”). Astute mentors use reflection to combat gender scripts, asking themselves, “Am I allowing latent biases to affect my judgement?” They also consider when to intervene and when to let the mentee “rescue” herself (eg, “This is challenging, but I trust your judgement. What do you think you should do next?”).
PROMOTE RECIPROCAL LEARNING
Many women value collaborative behaviors and gravitate towards egalitarian learning environments at odds with a traditional, “top-down” mentorship model. Additionally, women may be penalized for demonstrating competitive behaviors, while identical behaviors are chalked up to confidence in men. A critical task, then, is for mentors to coach women to hone their natural leadership style, whether it be more commanding or more communal. A mentor can provide key feedback to the mentee about how her approach might be perceived and how to tweak it for optimal success. Mentors may wish to share missteps and even ask the mentee for advice. Pointing to her competence promotes “relational mentoring” and reciprocal learning, where mentor and mentee can learn positive behaviors from each other.
BE THE CHANGE YOU WANT TO SEE
Mentors will ideally wield their social capital to advance policies that promote gender equity—including fair recruiting, promotion, salary, paid leave, and breastfeeding policies. Exceptional mentors recognize that women may generally have less social capital than men in many organizations, and they proactively make women’s accomplishments more visible.2 They broadcast women’s strengths and nominate women for talks, national committees, honorific societies, and leadership positions. Effective mentors recognize that 30% of female medical faculty report experiencing sexual harassment at work,3 and thus maintain extremely high standards for professional integrity, for both themselves and others who interact with their mentees. They call out sexist remarks in the workplace as unacceptable, making it clear that such behavior won’t be tolerated. As Mohandas Gandhi said: “Be the change that you wish to see in the world.”
Cross-gender mentorship is critical to get right—nearly half our medical workforce depends on it. Men who mentor women help their organizations and gain satisfaction from playing a pivotal role in women’s advancement. When women succeed, we all do.
Disclosures
Dr. Moniz and Dr. Saint have nothing to disclose.
“…a truly strong, powerful man isn’t threatened by a strong, powerful woman. Instead, he is challenged by her, he is inspired by her, he is pleased to relate to her as an equal.”
—Michelle Obama
Mentorship is essential to success in hospital medicine and may be particularly important for women. Cross-gender mentorship is especially salient since roughly equal proportions of women and men enter the medical pipeline, but men occupy over 75% of senior leadership roles in healthcare companies.
Cross-gender mentorship poses challenges but can be done successfully.1 We’ve made cross-gender mentoring work well in our own mentoring relationship. We describe three practices for effective mentoring that are especially important for men who mentor women given how common the female mentee-male mentor dyad is in medicine. We make generalizations that don’t apply universally but illustrate the social context in which such mentorship resides.
BE MINDFUL OF GENDER SCRIPTS
Gender scripts refer to social norms relating to gender identities and behaviors. Archetypal scripts include the father/daughter relationship and the knight/damsel-in-distress. Gender scripts often frame women as powerless—waiting to be rescued. By unconsciously activating a gender script, a mentor may reinforce a stereotype that women need rescuing (eg, “She’s really upset—I’ll email her Division Chief and help fix it for her”) or underestimate a mentee’s readiness for independence (eg, “She’s written four papers on this, but she’s still not ready to be senior author”). Astute mentors use reflection to combat gender scripts, asking themselves, “Am I allowing latent biases to affect my judgement?” They also consider when to intervene and when to let the mentee “rescue” herself (eg, “This is challenging, but I trust your judgement. What do you think you should do next?”).
PROMOTE RECIPROCAL LEARNING
Many women value collaborative behaviors and gravitate towards egalitarian learning environments at odds with a traditional, “top-down” mentorship model. Additionally, women may be penalized for demonstrating competitive behaviors, while identical behaviors are chalked up to confidence in men. A critical task, then, is for mentors to coach women to hone their natural leadership style, whether it be more commanding or more communal. A mentor can provide key feedback to the mentee about how her approach might be perceived and how to tweak it for optimal success. Mentors may wish to share missteps and even ask the mentee for advice. Pointing to her competence promotes “relational mentoring” and reciprocal learning, where mentor and mentee can learn positive behaviors from each other.
BE THE CHANGE YOU WANT TO SEE
Mentors will ideally wield their social capital to advance policies that promote gender equity—including fair recruiting, promotion, salary, paid leave, and breastfeeding policies. Exceptional mentors recognize that women may generally have less social capital than men in many organizations, and they proactively make women’s accomplishments more visible.2 They broadcast women’s strengths and nominate women for talks, national committees, honorific societies, and leadership positions. Effective mentors recognize that 30% of female medical faculty report experiencing sexual harassment at work,3 and thus maintain extremely high standards for professional integrity, for both themselves and others who interact with their mentees. They call out sexist remarks in the workplace as unacceptable, making it clear that such behavior won’t be tolerated. As Mohandas Gandhi said: “Be the change that you wish to see in the world.”
Cross-gender mentorship is critical to get right—nearly half our medical workforce depends on it. Men who mentor women help their organizations and gain satisfaction from playing a pivotal role in women’s advancement. When women succeed, we all do.
Disclosures
Dr. Moniz and Dr. Saint have nothing to disclose.
1. Byerley JS. Mentoring in the Era of #MeToo. JAMA. 2018;319(12):1199-1200. PubMed
2. Chopra V, Arora VM, Saint S. Will You Be My Mentor?-Four Archetypes to Help Mentees Succeed in Academic Medicine. JAMA Intern Med. 2018;178(2):175-176. PubMed
3. Jagsi R, Griffith KA, Jones R, Perumalswami CR, Ubel P, Stewart A. Sexual Harassment and Discrimination Experiences of Academic Medical Faculty. JAMA. 2016;315(19):2120-2121. PubMed
1. Byerley JS. Mentoring in the Era of #MeToo. JAMA. 2018;319(12):1199-1200. PubMed
2. Chopra V, Arora VM, Saint S. Will You Be My Mentor?-Four Archetypes to Help Mentees Succeed in Academic Medicine. JAMA Intern Med. 2018;178(2):175-176. PubMed
3. Jagsi R, Griffith KA, Jones R, Perumalswami CR, Ubel P, Stewart A. Sexual Harassment and Discrimination Experiences of Academic Medical Faculty. JAMA. 2016;315(19):2120-2121. PubMed
© 2019 Society of Hospital Medicine
In Reply to: “Practical Application of Pediatric Hospital Medicine Workforce Data: In Reference to ‘Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey’”
We appreciate the query by Drs. Douglas and Wilson. We hereby supply additional information that is critical for creating and administering sustainable staffing models.
For programs with a census cap, the majority cited 16 or fewer patients as the trigger for that cap. Nearly all programs with back-up used a census of 16 or fewer. Over 80% of programs cited a “safe 7
Regarding clinical weighting of nights, nighttime shifts were often more heavily weighted than day shifts, but approaches to weighting varied and have not been validated. Alternate staffing models for overnight pager calls varied greatly by individual program.
This is a time of significant growth for pediatric hospital medicine, and national workforce data are essential to hospitalists, administrators, and most importantly, patients. Our study1 provides pediatric hospital medicine leaders with data for discussions regarding appropriate FTE and staffing model considerations. The insights generated by our study are particularly relevant in expanding programs and solving problems related to recruitment and retention.
Disclosures
The authors have nothing to disclose.
1. Fromme HB, Chen C, Fine B, Gosdin C, Shaughnessy E. Pediatric hospitalist workload and sustainability in university-based programs: Results from a national interview-based survey. J Hosp Med. 2018;13(10):702-705. PubMed
We appreciate the query by Drs. Douglas and Wilson. We hereby supply additional information that is critical for creating and administering sustainable staffing models.
For programs with a census cap, the majority cited 16 or fewer patients as the trigger for that cap. Nearly all programs with back-up used a census of 16 or fewer. Over 80% of programs cited a “safe 7
Regarding clinical weighting of nights, nighttime shifts were often more heavily weighted than day shifts, but approaches to weighting varied and have not been validated. Alternate staffing models for overnight pager calls varied greatly by individual program.
This is a time of significant growth for pediatric hospital medicine, and national workforce data are essential to hospitalists, administrators, and most importantly, patients. Our study1 provides pediatric hospital medicine leaders with data for discussions regarding appropriate FTE and staffing model considerations. The insights generated by our study are particularly relevant in expanding programs and solving problems related to recruitment and retention.
Disclosures
The authors have nothing to disclose.
We appreciate the query by Drs. Douglas and Wilson. We hereby supply additional information that is critical for creating and administering sustainable staffing models.
For programs with a census cap, the majority cited 16 or fewer patients as the trigger for that cap. Nearly all programs with back-up used a census of 16 or fewer. Over 80% of programs cited a “safe 7
Regarding clinical weighting of nights, nighttime shifts were often more heavily weighted than day shifts, but approaches to weighting varied and have not been validated. Alternate staffing models for overnight pager calls varied greatly by individual program.
This is a time of significant growth for pediatric hospital medicine, and national workforce data are essential to hospitalists, administrators, and most importantly, patients. Our study1 provides pediatric hospital medicine leaders with data for discussions regarding appropriate FTE and staffing model considerations. The insights generated by our study are particularly relevant in expanding programs and solving problems related to recruitment and retention.
Disclosures
The authors have nothing to disclose.
1. Fromme HB, Chen C, Fine B, Gosdin C, Shaughnessy E. Pediatric hospitalist workload and sustainability in university-based programs: Results from a national interview-based survey. J Hosp Med. 2018;13(10):702-705. PubMed
1. Fromme HB, Chen C, Fine B, Gosdin C, Shaughnessy E. Pediatric hospitalist workload and sustainability in university-based programs: Results from a national interview-based survey. J Hosp Med. 2018;13(10):702-705. PubMed
© 2019 Society of Hospital Medicine
Practical Application of Pediatric Hospital Medicine Workforce Data. In Reference to: “Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey”
As leaders of a new Pediatric Hospital Medicine program in New York City, we were pleased to read the Brief Report from Dr. Fromme and colleagues, “Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey.”
Although the study has greatly assisted us in developing our program, the manuscript lacked some data necessary for workforce planning. The authors report census caps for a majority of programs, but neither the actual number of patients in each cap nor whether programs with caps reported an association with patient safety or program sustainability. In addition, although overnight pager calls were calculated in median hours, there were no data on whether nights were weighted or alternate staffing models were used for overnight pager calls.
While the article will help guide our field’s continued understanding of our workforce, without additional detailed data, we found that we were unable to apply staffing models practically in the real world to our new program. Pediatric Hospital Medicine is one of the fastest growing fields in medicine; however, support of new programs and sustainability of existing ones, require benchmark details to create proposals that are acceptable to
Disclosures
Drs. Douglas and Wilson have nothing to disclose.
1. Fromme HB, Chen C, Fine B, Gosdin C, Shaughnessy E. Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey. J Hosp Med. 2018. 13:702-705. PubMed
As leaders of a new Pediatric Hospital Medicine program in New York City, we were pleased to read the Brief Report from Dr. Fromme and colleagues, “Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey.”
Although the study has greatly assisted us in developing our program, the manuscript lacked some data necessary for workforce planning. The authors report census caps for a majority of programs, but neither the actual number of patients in each cap nor whether programs with caps reported an association with patient safety or program sustainability. In addition, although overnight pager calls were calculated in median hours, there were no data on whether nights were weighted or alternate staffing models were used for overnight pager calls.
While the article will help guide our field’s continued understanding of our workforce, without additional detailed data, we found that we were unable to apply staffing models practically in the real world to our new program. Pediatric Hospital Medicine is one of the fastest growing fields in medicine; however, support of new programs and sustainability of existing ones, require benchmark details to create proposals that are acceptable to
Disclosures
Drs. Douglas and Wilson have nothing to disclose.
As leaders of a new Pediatric Hospital Medicine program in New York City, we were pleased to read the Brief Report from Dr. Fromme and colleagues, “Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey.”
Although the study has greatly assisted us in developing our program, the manuscript lacked some data necessary for workforce planning. The authors report census caps for a majority of programs, but neither the actual number of patients in each cap nor whether programs with caps reported an association with patient safety or program sustainability. In addition, although overnight pager calls were calculated in median hours, there were no data on whether nights were weighted or alternate staffing models were used for overnight pager calls.
While the article will help guide our field’s continued understanding of our workforce, without additional detailed data, we found that we were unable to apply staffing models practically in the real world to our new program. Pediatric Hospital Medicine is one of the fastest growing fields in medicine; however, support of new programs and sustainability of existing ones, require benchmark details to create proposals that are acceptable to
Disclosures
Drs. Douglas and Wilson have nothing to disclose.
1. Fromme HB, Chen C, Fine B, Gosdin C, Shaughnessy E. Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey. J Hosp Med. 2018. 13:702-705. PubMed
1. Fromme HB, Chen C, Fine B, Gosdin C, Shaughnessy E. Pediatric Hospitalist Workload and Sustainability in University-Based Programs: Results from a National Interview-Based Survey. J Hosp Med. 2018. 13:702-705. PubMed
© 2019 Society of Hospital Medicine DOI 10.12788/jhm.3149
Lactobionic acid
Lactobionic acid (4-O-beta-galactopyranosyl-D-gluconic acid), a disaccharide formed from gluconic acid and galactose, has been established as a potent antioxidant well suited for use in solutions intended to preserve organs stored for transplantation.1,2 This polyhydroxy bionic acid is used as an excipient agent in some pharmaceutical products and has been the object of increasing interest and use in cosmetics and cosmeceuticals.3 It is included in skin care formulations for its strong humectant and antiaging effects.3,4 Lactobionic acid has been shown to suppress the synthesis of hydroxyl radicals by dint of iron-chelating activity and hinders the production of matrix metalloproteinases (MMPs), which promote photoaging.2,3,5 It may also present an advantage over the class of alpha-hydroxy acids used to treat photoaging by engendering less or no irritation, because of its larger molecular size and corresponding slower penetration rate.6 This column will focus on some recent research on the application of this strong antioxidant in dermatologic practice.
Lactobionic acid as an ingredient and vehicle
In 2010, Tasic-Kostov et al. compared the efficacy and irritation potential of lactobionic and glycolic acids (in gel and emulsion vehicles). In 77 healthy volunteers, the investigators found that , insofar as the former caused no irritation or skin barrier damage. In a second part to the study, they determined that efficacy of the acids was improved through the use of vehicles based on the natural emulsifier, alkyl polyglucoside (APG). They concluded that lactobionic acid in a 6% concentration in an APG vehicle warranted consideration as a low-molecular option in cosmeceutical products.6
In a subsequent study, the same team found supportive evidence that APG-based emulsions are safe cosmetic/dermopharmaceutical vehicles and carriers for extremely acidic and hygroscopic AHAs, particularly lactobionic acid. They did note, however, that lactobionic acid markedly affected the colloidal structure of the emulsion and fostered the development of lamellar structures, which could influence water distribution within the cream. They concluded, therefore, that such an emulsion, which was stabilized by lamellar liquid crystalline structures, would not be a viable carrier for the hygroscopic actives to achieve optimal moisturizing potential.7More recently, Tasic-Kostov et al. investigated the antioxidant and moisturizing traits of lactobionic acid in solution as well as in a natural APG emulsifier–based system using 1,1-diphenyl-2-picrylhydrazyl free radical scavenging and lipid peroxidation inhibition assays. The researchers found that lactobionic acid exhibited suitable physical stability (though it exerted notable impact on the colloidal structure of the vehicle) as well as antioxidant activity in both formats, suggesting its application as a versatile cosmeceutical agent for treating photoaged skin.2
In 2017, Chaouat et al. found that lactobionic acid was a key component in a green microparticle carrier system for cosmetics also containing chitosan and linoleic acid (as the skin penetration–enhancing constituent). Chitosan and lactobionic acid made up the shell surrounding the linoleic acid core. The carrier system, in an aqueous solution, was found to be stable and able to encapsulate the hydrophobic skin lightener phenylethyl resorcinol.8
Potential in atopic dermatitis treatment
Using an oxazolone-induced, atopic dermatitis–like murine dermatitis model, Sakai et al. demonstrated in 2016 that the coapplication of a PAR2 inhibitor and lactobionic acid, which maintained stratum corneum acidity, could target skin barrier abnormality and allergic inflammation, the key mechanisms in atopic dermatitis etiology.9
Lactobionic acid in chemical peels
Early this year, Algiert-Zielinska et al. reported on the results of a split-face study with 20 white women in which the effects of a 20% lactobionic acid peel were compared with those of the 20% peel combined with aluminum oxide crystal microdermabrasion. Treatments were administered weekly over 6 weeks, with the peel alone performed on the left side and the combination therapy on the right. The combination was found to achieve a significantly higher hydration level as well as skin elasticity measurements. There were no statistically significant differences between the tested therapies in transepidermal water loss, which decreased for both approaches. Both the lactobionic acid peel and combination procedure delivered notable moisturizing effects.10
Previously, this team performed a comparative evaluation of the skin-moisturizing activities of lactobionic acid in 10% and 30% concentrations in 10 white subjects between 26 and 73 years old. In this split-face study, 10% lactobionic acid was applied on the left side and 30% on the right on a weekly basis through eight treatments. A 5% lactobionic acid cream was supplied for overnight use. Skin hydration levels were measured before each weekly treatment. Although any differences between cutaneous hydration between the lactobionic acid preparations could not be ascertained, the investigators identified a statistically significant enhancement of hydration levels for both concentrations after the full series of treatments. They concluded that lactobionic is a potent moisturizing compound.11The same authors also conducted a literature review on the moisturizing properties of lactobionic and lactic acids, noting that both acids are capable of binding copious amounts of water and display robust chelating characteristics, as well as antioxidant activity, by suppressing MMPs. The authors added that both act as strong moisturizing substances, helping to maintain epidermal barrier integrity, and are suitable for sensitive skin.3
Conclusion
Greater capacity to moisturize and deliver antiaging benefits while causing less or no irritation are desirable qualities in a dermatologic agent. Evidence is limited, but the data available seem to suggest that lactobionic acid exhibits such qualities in comparison to alpha-hydroxy acids. Much more research is needed, though, to determine the most appropriate ways to use this promising compound.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014), and a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems LLC. Write to her at dermnews@mdedge.com.
References
1. Annu Rev Med. 1995;46:235-47.
2. Int J Cosmet Sci. 2012 Oct;34(5):424-34.
3. Int J Dermatol. 2019 Mar;58(3):374-79.
4. Clin Dermatol. 2009 Sep-Oct;27(5):495-501.
5.The next generation hydroxy acids, in “Cosmeceuticals” (New York: Elsevier Saunders, 2005, pp. 205-11).
6. J Cosmet Dermatol. 2010 Mar;9(1):3-10.
7. Pharmazie. 2011 Nov;66(11):862-70.
8. J Microencapsul. 2017 Mar;34(2):162-70.
9. J Invest Dermatol. 2016 Feb;136(2):538-41.
10. J Cosmet Dermatol. 2019 Jan 20. doi: 10.1111/jocd.12859. [Epub ahead of print].
11. J Cosmet Dermatol. 2018 Dec;17(6):1096-1100.
Lactobionic acid (4-O-beta-galactopyranosyl-D-gluconic acid), a disaccharide formed from gluconic acid and galactose, has been established as a potent antioxidant well suited for use in solutions intended to preserve organs stored for transplantation.1,2 This polyhydroxy bionic acid is used as an excipient agent in some pharmaceutical products and has been the object of increasing interest and use in cosmetics and cosmeceuticals.3 It is included in skin care formulations for its strong humectant and antiaging effects.3,4 Lactobionic acid has been shown to suppress the synthesis of hydroxyl radicals by dint of iron-chelating activity and hinders the production of matrix metalloproteinases (MMPs), which promote photoaging.2,3,5 It may also present an advantage over the class of alpha-hydroxy acids used to treat photoaging by engendering less or no irritation, because of its larger molecular size and corresponding slower penetration rate.6 This column will focus on some recent research on the application of this strong antioxidant in dermatologic practice.
Lactobionic acid as an ingredient and vehicle
In 2010, Tasic-Kostov et al. compared the efficacy and irritation potential of lactobionic and glycolic acids (in gel and emulsion vehicles). In 77 healthy volunteers, the investigators found that , insofar as the former caused no irritation or skin barrier damage. In a second part to the study, they determined that efficacy of the acids was improved through the use of vehicles based on the natural emulsifier, alkyl polyglucoside (APG). They concluded that lactobionic acid in a 6% concentration in an APG vehicle warranted consideration as a low-molecular option in cosmeceutical products.6
In a subsequent study, the same team found supportive evidence that APG-based emulsions are safe cosmetic/dermopharmaceutical vehicles and carriers for extremely acidic and hygroscopic AHAs, particularly lactobionic acid. They did note, however, that lactobionic acid markedly affected the colloidal structure of the emulsion and fostered the development of lamellar structures, which could influence water distribution within the cream. They concluded, therefore, that such an emulsion, which was stabilized by lamellar liquid crystalline structures, would not be a viable carrier for the hygroscopic actives to achieve optimal moisturizing potential.7More recently, Tasic-Kostov et al. investigated the antioxidant and moisturizing traits of lactobionic acid in solution as well as in a natural APG emulsifier–based system using 1,1-diphenyl-2-picrylhydrazyl free radical scavenging and lipid peroxidation inhibition assays. The researchers found that lactobionic acid exhibited suitable physical stability (though it exerted notable impact on the colloidal structure of the vehicle) as well as antioxidant activity in both formats, suggesting its application as a versatile cosmeceutical agent for treating photoaged skin.2
In 2017, Chaouat et al. found that lactobionic acid was a key component in a green microparticle carrier system for cosmetics also containing chitosan and linoleic acid (as the skin penetration–enhancing constituent). Chitosan and lactobionic acid made up the shell surrounding the linoleic acid core. The carrier system, in an aqueous solution, was found to be stable and able to encapsulate the hydrophobic skin lightener phenylethyl resorcinol.8
Potential in atopic dermatitis treatment
Using an oxazolone-induced, atopic dermatitis–like murine dermatitis model, Sakai et al. demonstrated in 2016 that the coapplication of a PAR2 inhibitor and lactobionic acid, which maintained stratum corneum acidity, could target skin barrier abnormality and allergic inflammation, the key mechanisms in atopic dermatitis etiology.9
Lactobionic acid in chemical peels
Early this year, Algiert-Zielinska et al. reported on the results of a split-face study with 20 white women in which the effects of a 20% lactobionic acid peel were compared with those of the 20% peel combined with aluminum oxide crystal microdermabrasion. Treatments were administered weekly over 6 weeks, with the peel alone performed on the left side and the combination therapy on the right. The combination was found to achieve a significantly higher hydration level as well as skin elasticity measurements. There were no statistically significant differences between the tested therapies in transepidermal water loss, which decreased for both approaches. Both the lactobionic acid peel and combination procedure delivered notable moisturizing effects.10
Previously, this team performed a comparative evaluation of the skin-moisturizing activities of lactobionic acid in 10% and 30% concentrations in 10 white subjects between 26 and 73 years old. In this split-face study, 10% lactobionic acid was applied on the left side and 30% on the right on a weekly basis through eight treatments. A 5% lactobionic acid cream was supplied for overnight use. Skin hydration levels were measured before each weekly treatment. Although any differences between cutaneous hydration between the lactobionic acid preparations could not be ascertained, the investigators identified a statistically significant enhancement of hydration levels for both concentrations after the full series of treatments. They concluded that lactobionic is a potent moisturizing compound.11The same authors also conducted a literature review on the moisturizing properties of lactobionic and lactic acids, noting that both acids are capable of binding copious amounts of water and display robust chelating characteristics, as well as antioxidant activity, by suppressing MMPs. The authors added that both act as strong moisturizing substances, helping to maintain epidermal barrier integrity, and are suitable for sensitive skin.3
Conclusion
Greater capacity to moisturize and deliver antiaging benefits while causing less or no irritation are desirable qualities in a dermatologic agent. Evidence is limited, but the data available seem to suggest that lactobionic acid exhibits such qualities in comparison to alpha-hydroxy acids. Much more research is needed, though, to determine the most appropriate ways to use this promising compound.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014), and a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems LLC. Write to her at dermnews@mdedge.com.
References
1. Annu Rev Med. 1995;46:235-47.
2. Int J Cosmet Sci. 2012 Oct;34(5):424-34.
3. Int J Dermatol. 2019 Mar;58(3):374-79.
4. Clin Dermatol. 2009 Sep-Oct;27(5):495-501.
5.The next generation hydroxy acids, in “Cosmeceuticals” (New York: Elsevier Saunders, 2005, pp. 205-11).
6. J Cosmet Dermatol. 2010 Mar;9(1):3-10.
7. Pharmazie. 2011 Nov;66(11):862-70.
8. J Microencapsul. 2017 Mar;34(2):162-70.
9. J Invest Dermatol. 2016 Feb;136(2):538-41.
10. J Cosmet Dermatol. 2019 Jan 20. doi: 10.1111/jocd.12859. [Epub ahead of print].
11. J Cosmet Dermatol. 2018 Dec;17(6):1096-1100.
Lactobionic acid (4-O-beta-galactopyranosyl-D-gluconic acid), a disaccharide formed from gluconic acid and galactose, has been established as a potent antioxidant well suited for use in solutions intended to preserve organs stored for transplantation.1,2 This polyhydroxy bionic acid is used as an excipient agent in some pharmaceutical products and has been the object of increasing interest and use in cosmetics and cosmeceuticals.3 It is included in skin care formulations for its strong humectant and antiaging effects.3,4 Lactobionic acid has been shown to suppress the synthesis of hydroxyl radicals by dint of iron-chelating activity and hinders the production of matrix metalloproteinases (MMPs), which promote photoaging.2,3,5 It may also present an advantage over the class of alpha-hydroxy acids used to treat photoaging by engendering less or no irritation, because of its larger molecular size and corresponding slower penetration rate.6 This column will focus on some recent research on the application of this strong antioxidant in dermatologic practice.
Lactobionic acid as an ingredient and vehicle
In 2010, Tasic-Kostov et al. compared the efficacy and irritation potential of lactobionic and glycolic acids (in gel and emulsion vehicles). In 77 healthy volunteers, the investigators found that , insofar as the former caused no irritation or skin barrier damage. In a second part to the study, they determined that efficacy of the acids was improved through the use of vehicles based on the natural emulsifier, alkyl polyglucoside (APG). They concluded that lactobionic acid in a 6% concentration in an APG vehicle warranted consideration as a low-molecular option in cosmeceutical products.6
In a subsequent study, the same team found supportive evidence that APG-based emulsions are safe cosmetic/dermopharmaceutical vehicles and carriers for extremely acidic and hygroscopic AHAs, particularly lactobionic acid. They did note, however, that lactobionic acid markedly affected the colloidal structure of the emulsion and fostered the development of lamellar structures, which could influence water distribution within the cream. They concluded, therefore, that such an emulsion, which was stabilized by lamellar liquid crystalline structures, would not be a viable carrier for the hygroscopic actives to achieve optimal moisturizing potential.7More recently, Tasic-Kostov et al. investigated the antioxidant and moisturizing traits of lactobionic acid in solution as well as in a natural APG emulsifier–based system using 1,1-diphenyl-2-picrylhydrazyl free radical scavenging and lipid peroxidation inhibition assays. The researchers found that lactobionic acid exhibited suitable physical stability (though it exerted notable impact on the colloidal structure of the vehicle) as well as antioxidant activity in both formats, suggesting its application as a versatile cosmeceutical agent for treating photoaged skin.2
In 2017, Chaouat et al. found that lactobionic acid was a key component in a green microparticle carrier system for cosmetics also containing chitosan and linoleic acid (as the skin penetration–enhancing constituent). Chitosan and lactobionic acid made up the shell surrounding the linoleic acid core. The carrier system, in an aqueous solution, was found to be stable and able to encapsulate the hydrophobic skin lightener phenylethyl resorcinol.8
Potential in atopic dermatitis treatment
Using an oxazolone-induced, atopic dermatitis–like murine dermatitis model, Sakai et al. demonstrated in 2016 that the coapplication of a PAR2 inhibitor and lactobionic acid, which maintained stratum corneum acidity, could target skin barrier abnormality and allergic inflammation, the key mechanisms in atopic dermatitis etiology.9
Lactobionic acid in chemical peels
Early this year, Algiert-Zielinska et al. reported on the results of a split-face study with 20 white women in which the effects of a 20% lactobionic acid peel were compared with those of the 20% peel combined with aluminum oxide crystal microdermabrasion. Treatments were administered weekly over 6 weeks, with the peel alone performed on the left side and the combination therapy on the right. The combination was found to achieve a significantly higher hydration level as well as skin elasticity measurements. There were no statistically significant differences between the tested therapies in transepidermal water loss, which decreased for both approaches. Both the lactobionic acid peel and combination procedure delivered notable moisturizing effects.10
Previously, this team performed a comparative evaluation of the skin-moisturizing activities of lactobionic acid in 10% and 30% concentrations in 10 white subjects between 26 and 73 years old. In this split-face study, 10% lactobionic acid was applied on the left side and 30% on the right on a weekly basis through eight treatments. A 5% lactobionic acid cream was supplied for overnight use. Skin hydration levels were measured before each weekly treatment. Although any differences between cutaneous hydration between the lactobionic acid preparations could not be ascertained, the investigators identified a statistically significant enhancement of hydration levels for both concentrations after the full series of treatments. They concluded that lactobionic is a potent moisturizing compound.11The same authors also conducted a literature review on the moisturizing properties of lactobionic and lactic acids, noting that both acids are capable of binding copious amounts of water and display robust chelating characteristics, as well as antioxidant activity, by suppressing MMPs. The authors added that both act as strong moisturizing substances, helping to maintain epidermal barrier integrity, and are suitable for sensitive skin.3
Conclusion
Greater capacity to moisturize and deliver antiaging benefits while causing less or no irritation are desirable qualities in a dermatologic agent. Evidence is limited, but the data available seem to suggest that lactobionic acid exhibits such qualities in comparison to alpha-hydroxy acids. Much more research is needed, though, to determine the most appropriate ways to use this promising compound.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014), and a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems LLC. Write to her at dermnews@mdedge.com.
References
1. Annu Rev Med. 1995;46:235-47.
2. Int J Cosmet Sci. 2012 Oct;34(5):424-34.
3. Int J Dermatol. 2019 Mar;58(3):374-79.
4. Clin Dermatol. 2009 Sep-Oct;27(5):495-501.
5.The next generation hydroxy acids, in “Cosmeceuticals” (New York: Elsevier Saunders, 2005, pp. 205-11).
6. J Cosmet Dermatol. 2010 Mar;9(1):3-10.
7. Pharmazie. 2011 Nov;66(11):862-70.
8. J Microencapsul. 2017 Mar;34(2):162-70.
9. J Invest Dermatol. 2016 Feb;136(2):538-41.
10. J Cosmet Dermatol. 2019 Jan 20. doi: 10.1111/jocd.12859. [Epub ahead of print].
11. J Cosmet Dermatol. 2018 Dec;17(6):1096-1100.