User login
Infected Bronchogenic Cyst With Left Atrial, Pulmonary Artery, and Esophageal Compression
Bronchogenic cyst is a rare foregut malformation that typically presents during the second decade of life that arises due to aberrant development from the tracheobronchial tree.1 Mediastinal bronchogenic cyst is the most common primary cystic lesion of the mediastinum, and bronchogenic cysts of the mediastinum represent 18% of all primary mediastinal malformations.2 Patients with mediastinal bronchogenic cysts may present with symptoms of cough, dyspnea, or wheezing if there is encroachment on surrounding structures.
Rarely, bronchogenic cysts can become infected. Definitive treatment of bronchogenic cysts is surgical excision; however, endobronchial ultrasound (EBUS)-guided drainage also can be employed. EBUS-guided drainage may be used when the cyst cannot be distinguished from solid mass on computed tomography (CT) images, to relieve symptomatic compression of surrounding structures, or to provide a histologic or microbial diagnosis in cases where surgical excision is not immediately available. We present the first-ever described case of bronchogenic cyst infected with Actinomyces, diagnosed by EBUS-guided drainage as well as a review of the literature regarding infected bronchogenic cysts and management of cysts affecting mediastinal structures.
Case Presentation
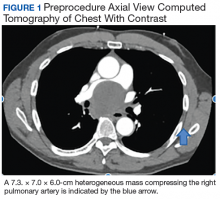
A 57-year-old African American male presented with a 4-day history of continuous, sharp, substernal chest pain accompanied by dyspnea. Additionally, the patient reported progressive dysphagia to solids. The posteroanterior view of a chest X-ray showed a widened mediastinum with splaying of the carina. A contrast-enhanced CT of the chest showed a large, middle mediastinal mass of heterogenous density measuring 7.3. × 7.0 × 6.0 cm with compression of the right pulmonary artery, left atria, superior vena cava and esophagus (Figure 1).
The mass demonstrated neither clear fluid-fluid level nor rounded structure with a distinct wall and uniform attenuation consistent with pure cystic structure and, in fact, was concerning for malignant process, such as lymphoma. Due to the malignancy concern and the findings of significant compression of surrounding mediastinal structures, the decision was made to proceed with bronchoscopy and EBUS-guided transbronchial needle aspiration (EBUS-TBNA) to assist in diagnosis and potentially provide symptomatic relief.
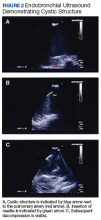
Under general anesthesia a P160 Olympus bronchoscope was advanced into the tracheobronchial tree; bronchoscopy with airway inspection revealed splayed carina with obtuse angle but was otherwise unremarkable. Next, an EBUS P160 fiber optic Olympus bronchoscope was advanced; ultrasound demonstrated a cystic structure. The EBUS-TBNA of cystic structure yielded 20 mL of brown, purulent fluid with decompression bringing pulmonary artery in ultrasound field (Figure 2). Rapid on-site cytology was performed with no preliminary findings of malignancy. The fluid was then sent for cytology and microbiologic evaluation.
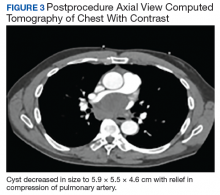
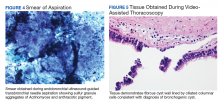
Following EBUS-guided aspiration, the patient reported significant improvement in chest pain, dyspnea, and dysphagia. A repeat chest CT demonstrated decrease in mass size to 5.9 × 5.5 × 4.6 cm with relief of the compression of the right pulmonary artery and decreased mass effect on the carina (Figure 3). Pathology ultimately demonstrated no evidence of malignancy but did demonstrate filamentous material with sulfur granules and anthracotic pigment suggestive of Actinomyces infection (Figure 4).
The patient was placed on amoxicillin/clavulanate 875 mg to 125 mg twice daily for 4 weeks based on antibiotic susceptibility testing to prevent progression to mediastinitis related to Actinomyces infection. The duration of therapy was extrapolated from treatment regimens described in case series of cervicofacial and abdominal Actinomyces infections.3 Thoracic surgery evaluation for definitive excision of cyst was recommended after the patient completed his course of antibiotics.
The patient underwent dental evaluation to identify the source of Actinomyces infection but there appeared to be no odontogenic source. The patient also had extensive skin survey with no findings of overt source of Actinomyces and CT abdomen/pelvis also identified no abscess that could be a potential source. He subsequently underwent thoracoscopic resection with pathology demonstrating a fibrous cyst wall lined with ciliated columnar epithelium consistent with diagnosis of bronchogenic cyst (Figure 5).
Discussion
Bronchogenic cysts can present at birth or later in life; patients may be asymptomatic for decades prior to discovery.4 Cysts located in the mediastinum can cause compression of the trachea and esophagus and cause cough, dyspnea, chest pain, and dysphagia.5 More life-threatening complications include infection, tracheal compression, malignant transformation, superior vena cava syndrome, or spontaneous rupture into the airway.6,7
Infection can occasionally occur, and various bacterial etiologies have been described. Hernandez-Solis and colleagues describe 12 cases of superinfected bronchogenic cysts with Staphylococcus aureus and Pseudomonas aeroginosa, the most commonly described organisms.8 Casal and colleagues describe a case of α-hemolytic Streptococci treated with amoxicillin.9 Liman and colleagues describe 2 cases of bronchogenic cyst infected with Mycobacterium and cite an additional case report by Lin and colleagues similarly infected by Mycobacterium.10,11 Only 1 case was identified to have direct bronchial communication as a potential source of introduction of infection into bronchogenic cyst. In other cases, potential sources of infection were not identified, though it was postulated that direct ventilation could be a potential source of inoculation.
Surgical resection of mediastinal bronchogenic cysts has traditionally been considered the definitive treatment of choice.12,13 However, bronchogenic cysts may sometimes be difficult to differentiate from soft tissue tumors by chest CT, especially in cases of cysts with nonserous fluid. In particular, cysts that are infected are likely to have increased density and high attenuation on imaging; therefore, surgical excision may be delayed until diagnosis is made.14 Due to low complication rates, EBUS is increasingly used in the diagnosis and therapeutic management of bronchogenic cysts as an alternative to surgery, particularly for those who are symptomatic.15,16 Ultrasound guidance can allow for complete aspiration of the cyst, causing complete collapse of the cystic space and can facilitate adhesion between the mucosal surfaces lining the cavity and reduce recurrence.17 Nonetheless, bronchogenic cysts that are found to be infected, recur, or have a malignant component should be resected for definitive treatment.18
The mass discovered on our patient’s imaging appeared to have heterogenous attenuation consistent with malignancy rather than homogenous attenuation surrounded by a clearly demarcated wall consistent with a cystic structure; therefore, EBUS-TBNA was initially pursued and yielded an expedited diagnosis of the first-ever described bronchogenic cyst with Actinomyces superinfection as well as dramatic symptomatic relief of compression of surrounding mediastinal structures, particularly of the right pulmonary artery. As this is a congenital malformation, the patient was likely asymptomatic until the cyst became infected, after which he likely experience cyst growth with subsequent encroachment of surrounding mediastinal structures. Additionally, identification of pathogen by TBNA allowed for treatment before surgical excision, possibly avoiding accidental spread of pathogen intraoperatively.
Conclusions
Our case adds to the literature on the use of EBUS-TBNA as a diagnostic and therapeutic modality for bronchogenic cyst. While cases of mediastinitis and pleural effusion following EBUS-guided aspiration of bronchogenic cysts have been reported, complications are extremely rare.19 EBUS is increasingly favored as a means of immediate diagnosis and treatment in cases where CT imaging may not overtly suggest cystic structure and in patients experiencing compression of critical mediastinal structures.
1. Weber T, Roth TC, Beshay M, Herrmann P, Stein R, Schmid RA. Video-assisted thoracoscopic surgery of mediastinal bronchogenic cysts in adults: a single-center experience. Ann Thorac Surg. 2004;78(3):987-991.
2. Martinod E, Pons F, Azorin J, et al. Thoracoscopic excision of mediastinal bronchogenic cysts: results in 20 cases. Ann Thorac Surg. 2000;69(5):1525-1528.
3. Könönen E, Wade WG. Actinomyces and related organisms in human infections. Clin Microbiol Rev. 2015;28(2):419-442.
4. Ribet ME, Copin MC, Gosselin BH. Bronchogenic cysts of the lung. Ann Thorac Surg. 1996;61(6):1636-1640.
5. Guillem P, Porte H, Marquette CH, Wurtz A. Progressive dysphonia and acute respiratory failure: revealing a bronchogenic cyst. Eur J Cardiothorac Surg. 1997;12(6):925-927.
6. McAdams HP, Kirejczyk WM, Rosado-de-Christenson ML, Matsumoto S. Bronchogenic cyst: imaging features with clinical and histopathologic correlation. Radiology. 2000;217(2):441-446.
7. Rammohan G, Berger HW, Lajam F, Buhain WJ. Superior vena cava syndrome caused by bronchogenic cyst. Chest. 1975;68(4):599-601.
8. Hernández-Solís A, Cruz-Ortiz H, Gutiérrez-Díaz Ceballos ME, Cicero-Sabido R. Quistes broncogénicos. Importancia de la infección en adultos. Estudio de 12 casos [Bronchogenic cysts. Importance of infection in adults. Study of 12 cases]. Cir Cir. 2015;83(2):112-116.
9. Casal RF, Jimenez CA, Mehran RJ, et al. Infected mediastinal bronchogenic cyst successfully treated by endobronchial ultrasound-guided fine-needle aspiration. Ann Thorac Surg. 2010;90(4):e52-e53.
10. Liman ST, Dogan Y, Topcu S, Karabulut N, Demirkan N, Keser Z. Mycobacterial infection of intraparenchymal bronchogenic cysts. Respir Med. 2006;100(11):2060-2062.
11. Lin SH, Lee LN, Chang YL, Lee YC, Ding LW, Hsueh PR. Infected bronchogenic cyst due to Mycobacterium avium in an immunocompetent patient. J Infect. 2005;51(3):e131-e133.
12. Gharagozloo F, Dausmann MJ, McReynolds SD, Sanderson DR, Helmers RA. Recurrent bronchogenic pseudocyst 24 years after incomplete excision. Report of a case. Chest. 1995;108(3):880-883.
13. Bolton JW, Shahian DM. Asymptomatic bronchogenic cysts: what is the best management? Ann Thorac Surg. 1992;53(6):1134-1137.
14. Sarper A, Ayten A, Golbasi I, Demircan A, Isin E. Bronchogenic cyst. Tex Heart Inst J. 2003;30(2):105-108.
15. Varela-Lema L, Fernández-Villar A, Ruano-Ravina A. Effectiveness and safety of endobronchial ultrasound-transbronchial needle aspiration: a systematic review. Eur Respir J. 2009;33(5):1156-1164.
16. Maturu VN, Dhooria S, Agarwal R. Efficacy and safety of transbronchial needle aspiration in diagnosis and treatment of mediastinal bronchogenic cysts: systematic review of case reports. J Bronchology Interv Pulmonol. 2015;22(3):195-203.
17. Galluccio G, Lucantoni G. Mediastinal bronchogenic cyst’s recurrence treated with EBUS-FNA with a long-term follow-up. Eur J Cardiothorac Surg. 2006;29(4):627-629.
18. Lee DH, Park CK, Kum DY, Kim JB, Hwang I. Clinical characteristics and management of intrathoracic bronchogenic cysts: a single center experience. Korean J Thorac Cardiovasc Surg. 2011;44(4):279-284.
19. Onuki T, Kuramochi M, Inagaki M. Mediastinitis of bronchogenic cyst caused by endobronchial ultrasound-guided transbronchial needle aspiration. Respirol Case Rep. 2014;2(2):73-75.
Bronchogenic cyst is a rare foregut malformation that typically presents during the second decade of life that arises due to aberrant development from the tracheobronchial tree.1 Mediastinal bronchogenic cyst is the most common primary cystic lesion of the mediastinum, and bronchogenic cysts of the mediastinum represent 18% of all primary mediastinal malformations.2 Patients with mediastinal bronchogenic cysts may present with symptoms of cough, dyspnea, or wheezing if there is encroachment on surrounding structures.
Rarely, bronchogenic cysts can become infected. Definitive treatment of bronchogenic cysts is surgical excision; however, endobronchial ultrasound (EBUS)-guided drainage also can be employed. EBUS-guided drainage may be used when the cyst cannot be distinguished from solid mass on computed tomography (CT) images, to relieve symptomatic compression of surrounding structures, or to provide a histologic or microbial diagnosis in cases where surgical excision is not immediately available. We present the first-ever described case of bronchogenic cyst infected with Actinomyces, diagnosed by EBUS-guided drainage as well as a review of the literature regarding infected bronchogenic cysts and management of cysts affecting mediastinal structures.
Case Presentation
A 57-year-old African American male presented with a 4-day history of continuous, sharp, substernal chest pain accompanied by dyspnea. Additionally, the patient reported progressive dysphagia to solids. The posteroanterior view of a chest X-ray showed a widened mediastinum with splaying of the carina. A contrast-enhanced CT of the chest showed a large, middle mediastinal mass of heterogenous density measuring 7.3. × 7.0 × 6.0 cm with compression of the right pulmonary artery, left atria, superior vena cava and esophagus (Figure 1).
The mass demonstrated neither clear fluid-fluid level nor rounded structure with a distinct wall and uniform attenuation consistent with pure cystic structure and, in fact, was concerning for malignant process, such as lymphoma. Due to the malignancy concern and the findings of significant compression of surrounding mediastinal structures, the decision was made to proceed with bronchoscopy and EBUS-guided transbronchial needle aspiration (EBUS-TBNA) to assist in diagnosis and potentially provide symptomatic relief.
Under general anesthesia a P160 Olympus bronchoscope was advanced into the tracheobronchial tree; bronchoscopy with airway inspection revealed splayed carina with obtuse angle but was otherwise unremarkable. Next, an EBUS P160 fiber optic Olympus bronchoscope was advanced; ultrasound demonstrated a cystic structure. The EBUS-TBNA of cystic structure yielded 20 mL of brown, purulent fluid with decompression bringing pulmonary artery in ultrasound field (Figure 2). Rapid on-site cytology was performed with no preliminary findings of malignancy. The fluid was then sent for cytology and microbiologic evaluation.
Following EBUS-guided aspiration, the patient reported significant improvement in chest pain, dyspnea, and dysphagia. A repeat chest CT demonstrated decrease in mass size to 5.9 × 5.5 × 4.6 cm with relief of the compression of the right pulmonary artery and decreased mass effect on the carina (Figure 3). Pathology ultimately demonstrated no evidence of malignancy but did demonstrate filamentous material with sulfur granules and anthracotic pigment suggestive of Actinomyces infection (Figure 4).
The patient was placed on amoxicillin/clavulanate 875 mg to 125 mg twice daily for 4 weeks based on antibiotic susceptibility testing to prevent progression to mediastinitis related to Actinomyces infection. The duration of therapy was extrapolated from treatment regimens described in case series of cervicofacial and abdominal Actinomyces infections.3 Thoracic surgery evaluation for definitive excision of cyst was recommended after the patient completed his course of antibiotics.
The patient underwent dental evaluation to identify the source of Actinomyces infection but there appeared to be no odontogenic source. The patient also had extensive skin survey with no findings of overt source of Actinomyces and CT abdomen/pelvis also identified no abscess that could be a potential source. He subsequently underwent thoracoscopic resection with pathology demonstrating a fibrous cyst wall lined with ciliated columnar epithelium consistent with diagnosis of bronchogenic cyst (Figure 5).
Discussion
Bronchogenic cysts can present at birth or later in life; patients may be asymptomatic for decades prior to discovery.4 Cysts located in the mediastinum can cause compression of the trachea and esophagus and cause cough, dyspnea, chest pain, and dysphagia.5 More life-threatening complications include infection, tracheal compression, malignant transformation, superior vena cava syndrome, or spontaneous rupture into the airway.6,7
Infection can occasionally occur, and various bacterial etiologies have been described. Hernandez-Solis and colleagues describe 12 cases of superinfected bronchogenic cysts with Staphylococcus aureus and Pseudomonas aeroginosa, the most commonly described organisms.8 Casal and colleagues describe a case of α-hemolytic Streptococci treated with amoxicillin.9 Liman and colleagues describe 2 cases of bronchogenic cyst infected with Mycobacterium and cite an additional case report by Lin and colleagues similarly infected by Mycobacterium.10,11 Only 1 case was identified to have direct bronchial communication as a potential source of introduction of infection into bronchogenic cyst. In other cases, potential sources of infection were not identified, though it was postulated that direct ventilation could be a potential source of inoculation.
Surgical resection of mediastinal bronchogenic cysts has traditionally been considered the definitive treatment of choice.12,13 However, bronchogenic cysts may sometimes be difficult to differentiate from soft tissue tumors by chest CT, especially in cases of cysts with nonserous fluid. In particular, cysts that are infected are likely to have increased density and high attenuation on imaging; therefore, surgical excision may be delayed until diagnosis is made.14 Due to low complication rates, EBUS is increasingly used in the diagnosis and therapeutic management of bronchogenic cysts as an alternative to surgery, particularly for those who are symptomatic.15,16 Ultrasound guidance can allow for complete aspiration of the cyst, causing complete collapse of the cystic space and can facilitate adhesion between the mucosal surfaces lining the cavity and reduce recurrence.17 Nonetheless, bronchogenic cysts that are found to be infected, recur, or have a malignant component should be resected for definitive treatment.18
The mass discovered on our patient’s imaging appeared to have heterogenous attenuation consistent with malignancy rather than homogenous attenuation surrounded by a clearly demarcated wall consistent with a cystic structure; therefore, EBUS-TBNA was initially pursued and yielded an expedited diagnosis of the first-ever described bronchogenic cyst with Actinomyces superinfection as well as dramatic symptomatic relief of compression of surrounding mediastinal structures, particularly of the right pulmonary artery. As this is a congenital malformation, the patient was likely asymptomatic until the cyst became infected, after which he likely experience cyst growth with subsequent encroachment of surrounding mediastinal structures. Additionally, identification of pathogen by TBNA allowed for treatment before surgical excision, possibly avoiding accidental spread of pathogen intraoperatively.
Conclusions
Our case adds to the literature on the use of EBUS-TBNA as a diagnostic and therapeutic modality for bronchogenic cyst. While cases of mediastinitis and pleural effusion following EBUS-guided aspiration of bronchogenic cysts have been reported, complications are extremely rare.19 EBUS is increasingly favored as a means of immediate diagnosis and treatment in cases where CT imaging may not overtly suggest cystic structure and in patients experiencing compression of critical mediastinal structures.
Bronchogenic cyst is a rare foregut malformation that typically presents during the second decade of life that arises due to aberrant development from the tracheobronchial tree.1 Mediastinal bronchogenic cyst is the most common primary cystic lesion of the mediastinum, and bronchogenic cysts of the mediastinum represent 18% of all primary mediastinal malformations.2 Patients with mediastinal bronchogenic cysts may present with symptoms of cough, dyspnea, or wheezing if there is encroachment on surrounding structures.
Rarely, bronchogenic cysts can become infected. Definitive treatment of bronchogenic cysts is surgical excision; however, endobronchial ultrasound (EBUS)-guided drainage also can be employed. EBUS-guided drainage may be used when the cyst cannot be distinguished from solid mass on computed tomography (CT) images, to relieve symptomatic compression of surrounding structures, or to provide a histologic or microbial diagnosis in cases where surgical excision is not immediately available. We present the first-ever described case of bronchogenic cyst infected with Actinomyces, diagnosed by EBUS-guided drainage as well as a review of the literature regarding infected bronchogenic cysts and management of cysts affecting mediastinal structures.
Case Presentation
A 57-year-old African American male presented with a 4-day history of continuous, sharp, substernal chest pain accompanied by dyspnea. Additionally, the patient reported progressive dysphagia to solids. The posteroanterior view of a chest X-ray showed a widened mediastinum with splaying of the carina. A contrast-enhanced CT of the chest showed a large, middle mediastinal mass of heterogenous density measuring 7.3. × 7.0 × 6.0 cm with compression of the right pulmonary artery, left atria, superior vena cava and esophagus (Figure 1).
The mass demonstrated neither clear fluid-fluid level nor rounded structure with a distinct wall and uniform attenuation consistent with pure cystic structure and, in fact, was concerning for malignant process, such as lymphoma. Due to the malignancy concern and the findings of significant compression of surrounding mediastinal structures, the decision was made to proceed with bronchoscopy and EBUS-guided transbronchial needle aspiration (EBUS-TBNA) to assist in diagnosis and potentially provide symptomatic relief.
Under general anesthesia a P160 Olympus bronchoscope was advanced into the tracheobronchial tree; bronchoscopy with airway inspection revealed splayed carina with obtuse angle but was otherwise unremarkable. Next, an EBUS P160 fiber optic Olympus bronchoscope was advanced; ultrasound demonstrated a cystic structure. The EBUS-TBNA of cystic structure yielded 20 mL of brown, purulent fluid with decompression bringing pulmonary artery in ultrasound field (Figure 2). Rapid on-site cytology was performed with no preliminary findings of malignancy. The fluid was then sent for cytology and microbiologic evaluation.
Following EBUS-guided aspiration, the patient reported significant improvement in chest pain, dyspnea, and dysphagia. A repeat chest CT demonstrated decrease in mass size to 5.9 × 5.5 × 4.6 cm with relief of the compression of the right pulmonary artery and decreased mass effect on the carina (Figure 3). Pathology ultimately demonstrated no evidence of malignancy but did demonstrate filamentous material with sulfur granules and anthracotic pigment suggestive of Actinomyces infection (Figure 4).
The patient was placed on amoxicillin/clavulanate 875 mg to 125 mg twice daily for 4 weeks based on antibiotic susceptibility testing to prevent progression to mediastinitis related to Actinomyces infection. The duration of therapy was extrapolated from treatment regimens described in case series of cervicofacial and abdominal Actinomyces infections.3 Thoracic surgery evaluation for definitive excision of cyst was recommended after the patient completed his course of antibiotics.
The patient underwent dental evaluation to identify the source of Actinomyces infection but there appeared to be no odontogenic source. The patient also had extensive skin survey with no findings of overt source of Actinomyces and CT abdomen/pelvis also identified no abscess that could be a potential source. He subsequently underwent thoracoscopic resection with pathology demonstrating a fibrous cyst wall lined with ciliated columnar epithelium consistent with diagnosis of bronchogenic cyst (Figure 5).
Discussion
Bronchogenic cysts can present at birth or later in life; patients may be asymptomatic for decades prior to discovery.4 Cysts located in the mediastinum can cause compression of the trachea and esophagus and cause cough, dyspnea, chest pain, and dysphagia.5 More life-threatening complications include infection, tracheal compression, malignant transformation, superior vena cava syndrome, or spontaneous rupture into the airway.6,7
Infection can occasionally occur, and various bacterial etiologies have been described. Hernandez-Solis and colleagues describe 12 cases of superinfected bronchogenic cysts with Staphylococcus aureus and Pseudomonas aeroginosa, the most commonly described organisms.8 Casal and colleagues describe a case of α-hemolytic Streptococci treated with amoxicillin.9 Liman and colleagues describe 2 cases of bronchogenic cyst infected with Mycobacterium and cite an additional case report by Lin and colleagues similarly infected by Mycobacterium.10,11 Only 1 case was identified to have direct bronchial communication as a potential source of introduction of infection into bronchogenic cyst. In other cases, potential sources of infection were not identified, though it was postulated that direct ventilation could be a potential source of inoculation.
Surgical resection of mediastinal bronchogenic cysts has traditionally been considered the definitive treatment of choice.12,13 However, bronchogenic cysts may sometimes be difficult to differentiate from soft tissue tumors by chest CT, especially in cases of cysts with nonserous fluid. In particular, cysts that are infected are likely to have increased density and high attenuation on imaging; therefore, surgical excision may be delayed until diagnosis is made.14 Due to low complication rates, EBUS is increasingly used in the diagnosis and therapeutic management of bronchogenic cysts as an alternative to surgery, particularly for those who are symptomatic.15,16 Ultrasound guidance can allow for complete aspiration of the cyst, causing complete collapse of the cystic space and can facilitate adhesion between the mucosal surfaces lining the cavity and reduce recurrence.17 Nonetheless, bronchogenic cysts that are found to be infected, recur, or have a malignant component should be resected for definitive treatment.18
The mass discovered on our patient’s imaging appeared to have heterogenous attenuation consistent with malignancy rather than homogenous attenuation surrounded by a clearly demarcated wall consistent with a cystic structure; therefore, EBUS-TBNA was initially pursued and yielded an expedited diagnosis of the first-ever described bronchogenic cyst with Actinomyces superinfection as well as dramatic symptomatic relief of compression of surrounding mediastinal structures, particularly of the right pulmonary artery. As this is a congenital malformation, the patient was likely asymptomatic until the cyst became infected, after which he likely experience cyst growth with subsequent encroachment of surrounding mediastinal structures. Additionally, identification of pathogen by TBNA allowed for treatment before surgical excision, possibly avoiding accidental spread of pathogen intraoperatively.
Conclusions
Our case adds to the literature on the use of EBUS-TBNA as a diagnostic and therapeutic modality for bronchogenic cyst. While cases of mediastinitis and pleural effusion following EBUS-guided aspiration of bronchogenic cysts have been reported, complications are extremely rare.19 EBUS is increasingly favored as a means of immediate diagnosis and treatment in cases where CT imaging may not overtly suggest cystic structure and in patients experiencing compression of critical mediastinal structures.
1. Weber T, Roth TC, Beshay M, Herrmann P, Stein R, Schmid RA. Video-assisted thoracoscopic surgery of mediastinal bronchogenic cysts in adults: a single-center experience. Ann Thorac Surg. 2004;78(3):987-991.
2. Martinod E, Pons F, Azorin J, et al. Thoracoscopic excision of mediastinal bronchogenic cysts: results in 20 cases. Ann Thorac Surg. 2000;69(5):1525-1528.
3. Könönen E, Wade WG. Actinomyces and related organisms in human infections. Clin Microbiol Rev. 2015;28(2):419-442.
4. Ribet ME, Copin MC, Gosselin BH. Bronchogenic cysts of the lung. Ann Thorac Surg. 1996;61(6):1636-1640.
5. Guillem P, Porte H, Marquette CH, Wurtz A. Progressive dysphonia and acute respiratory failure: revealing a bronchogenic cyst. Eur J Cardiothorac Surg. 1997;12(6):925-927.
6. McAdams HP, Kirejczyk WM, Rosado-de-Christenson ML, Matsumoto S. Bronchogenic cyst: imaging features with clinical and histopathologic correlation. Radiology. 2000;217(2):441-446.
7. Rammohan G, Berger HW, Lajam F, Buhain WJ. Superior vena cava syndrome caused by bronchogenic cyst. Chest. 1975;68(4):599-601.
8. Hernández-Solís A, Cruz-Ortiz H, Gutiérrez-Díaz Ceballos ME, Cicero-Sabido R. Quistes broncogénicos. Importancia de la infección en adultos. Estudio de 12 casos [Bronchogenic cysts. Importance of infection in adults. Study of 12 cases]. Cir Cir. 2015;83(2):112-116.
9. Casal RF, Jimenez CA, Mehran RJ, et al. Infected mediastinal bronchogenic cyst successfully treated by endobronchial ultrasound-guided fine-needle aspiration. Ann Thorac Surg. 2010;90(4):e52-e53.
10. Liman ST, Dogan Y, Topcu S, Karabulut N, Demirkan N, Keser Z. Mycobacterial infection of intraparenchymal bronchogenic cysts. Respir Med. 2006;100(11):2060-2062.
11. Lin SH, Lee LN, Chang YL, Lee YC, Ding LW, Hsueh PR. Infected bronchogenic cyst due to Mycobacterium avium in an immunocompetent patient. J Infect. 2005;51(3):e131-e133.
12. Gharagozloo F, Dausmann MJ, McReynolds SD, Sanderson DR, Helmers RA. Recurrent bronchogenic pseudocyst 24 years after incomplete excision. Report of a case. Chest. 1995;108(3):880-883.
13. Bolton JW, Shahian DM. Asymptomatic bronchogenic cysts: what is the best management? Ann Thorac Surg. 1992;53(6):1134-1137.
14. Sarper A, Ayten A, Golbasi I, Demircan A, Isin E. Bronchogenic cyst. Tex Heart Inst J. 2003;30(2):105-108.
15. Varela-Lema L, Fernández-Villar A, Ruano-Ravina A. Effectiveness and safety of endobronchial ultrasound-transbronchial needle aspiration: a systematic review. Eur Respir J. 2009;33(5):1156-1164.
16. Maturu VN, Dhooria S, Agarwal R. Efficacy and safety of transbronchial needle aspiration in diagnosis and treatment of mediastinal bronchogenic cysts: systematic review of case reports. J Bronchology Interv Pulmonol. 2015;22(3):195-203.
17. Galluccio G, Lucantoni G. Mediastinal bronchogenic cyst’s recurrence treated with EBUS-FNA with a long-term follow-up. Eur J Cardiothorac Surg. 2006;29(4):627-629.
18. Lee DH, Park CK, Kum DY, Kim JB, Hwang I. Clinical characteristics and management of intrathoracic bronchogenic cysts: a single center experience. Korean J Thorac Cardiovasc Surg. 2011;44(4):279-284.
19. Onuki T, Kuramochi M, Inagaki M. Mediastinitis of bronchogenic cyst caused by endobronchial ultrasound-guided transbronchial needle aspiration. Respirol Case Rep. 2014;2(2):73-75.
1. Weber T, Roth TC, Beshay M, Herrmann P, Stein R, Schmid RA. Video-assisted thoracoscopic surgery of mediastinal bronchogenic cysts in adults: a single-center experience. Ann Thorac Surg. 2004;78(3):987-991.
2. Martinod E, Pons F, Azorin J, et al. Thoracoscopic excision of mediastinal bronchogenic cysts: results in 20 cases. Ann Thorac Surg. 2000;69(5):1525-1528.
3. Könönen E, Wade WG. Actinomyces and related organisms in human infections. Clin Microbiol Rev. 2015;28(2):419-442.
4. Ribet ME, Copin MC, Gosselin BH. Bronchogenic cysts of the lung. Ann Thorac Surg. 1996;61(6):1636-1640.
5. Guillem P, Porte H, Marquette CH, Wurtz A. Progressive dysphonia and acute respiratory failure: revealing a bronchogenic cyst. Eur J Cardiothorac Surg. 1997;12(6):925-927.
6. McAdams HP, Kirejczyk WM, Rosado-de-Christenson ML, Matsumoto S. Bronchogenic cyst: imaging features with clinical and histopathologic correlation. Radiology. 2000;217(2):441-446.
7. Rammohan G, Berger HW, Lajam F, Buhain WJ. Superior vena cava syndrome caused by bronchogenic cyst. Chest. 1975;68(4):599-601.
8. Hernández-Solís A, Cruz-Ortiz H, Gutiérrez-Díaz Ceballos ME, Cicero-Sabido R. Quistes broncogénicos. Importancia de la infección en adultos. Estudio de 12 casos [Bronchogenic cysts. Importance of infection in adults. Study of 12 cases]. Cir Cir. 2015;83(2):112-116.
9. Casal RF, Jimenez CA, Mehran RJ, et al. Infected mediastinal bronchogenic cyst successfully treated by endobronchial ultrasound-guided fine-needle aspiration. Ann Thorac Surg. 2010;90(4):e52-e53.
10. Liman ST, Dogan Y, Topcu S, Karabulut N, Demirkan N, Keser Z. Mycobacterial infection of intraparenchymal bronchogenic cysts. Respir Med. 2006;100(11):2060-2062.
11. Lin SH, Lee LN, Chang YL, Lee YC, Ding LW, Hsueh PR. Infected bronchogenic cyst due to Mycobacterium avium in an immunocompetent patient. J Infect. 2005;51(3):e131-e133.
12. Gharagozloo F, Dausmann MJ, McReynolds SD, Sanderson DR, Helmers RA. Recurrent bronchogenic pseudocyst 24 years after incomplete excision. Report of a case. Chest. 1995;108(3):880-883.
13. Bolton JW, Shahian DM. Asymptomatic bronchogenic cysts: what is the best management? Ann Thorac Surg. 1992;53(6):1134-1137.
14. Sarper A, Ayten A, Golbasi I, Demircan A, Isin E. Bronchogenic cyst. Tex Heart Inst J. 2003;30(2):105-108.
15. Varela-Lema L, Fernández-Villar A, Ruano-Ravina A. Effectiveness and safety of endobronchial ultrasound-transbronchial needle aspiration: a systematic review. Eur Respir J. 2009;33(5):1156-1164.
16. Maturu VN, Dhooria S, Agarwal R. Efficacy and safety of transbronchial needle aspiration in diagnosis and treatment of mediastinal bronchogenic cysts: systematic review of case reports. J Bronchology Interv Pulmonol. 2015;22(3):195-203.
17. Galluccio G, Lucantoni G. Mediastinal bronchogenic cyst’s recurrence treated with EBUS-FNA with a long-term follow-up. Eur J Cardiothorac Surg. 2006;29(4):627-629.
18. Lee DH, Park CK, Kum DY, Kim JB, Hwang I. Clinical characteristics and management of intrathoracic bronchogenic cysts: a single center experience. Korean J Thorac Cardiovasc Surg. 2011;44(4):279-284.
19. Onuki T, Kuramochi M, Inagaki M. Mediastinitis of bronchogenic cyst caused by endobronchial ultrasound-guided transbronchial needle aspiration. Respirol Case Rep. 2014;2(2):73-75.
Observations From Embedded Health Engagement Team Members
“Whenever possible, we will develop innovative, low-cost, and small-footprint approaches to achieve our security objectives.” 1
Team member and participant observations can deliver valuable insight into the effectiveness of an activity or project. Certainly, documentation of such qualitative assessment through survey questions or narratives can reveal important information for future action. This qualitative aspect was a significant consideration in the formation of an embedded health engagement team (EHET) intended to improve foreign assistance and health outcomes for global humanitarian and security cooperation activities.
Since health activities are centered on human interaction and relationships, some observation or qualitative assessment must be included to truly determine short-term local buy-in and long-term outcomes. The following observations include the direct narrative perspectives of team members from a multidisciplinary primary care EHET that add experiential depth to prior assessment of the pilot test of such teams during Continuing Promise 2011, a 9-country series of health engagement activities employed from the USNS Comfort.2 The embedded team consisted of US Air Force (USAF), US Navy (USN), and nongovernmental organization (NGO) personnel working directly in a primary care clinic of the Costa Rican public health system.
This is small sample of a few team members who responded to a simple, open-ended prompt to record their impression of the EHET concept and experiences. Documenting this information should highlight the importance of seeking similar qualitative mission data for future health engagements. Standardized questionnaires have been used to evaluate health activities and have provided valuable analysis and recommendations that have advanced US Department of Defense (DoD) global health engagement.3 Captured narrative observation from the EHET pilot study is a complementary qualitative method that supports the concept of small, well prepared, culturally competent, EHETs tailored to work within a partner system rather than outside of it will achieve greater mutual benefit, including the application of better, more equitable health and health system principles.4 In this embedded manner, health care professionals may readily contribute to host nation health sector plans and goals while achieving military objectives, political goals, and mutual strategic interests through both military-military and military-civilian applications.
Observations and Reflections
Family Physician (Maj, Second Physician, USAF)
“Overall, the experience I had with the embedded team was truly rewarding. I hope this becomes a tool used to augment humanitarian missions. There is no way to truly understand a systems strengths and weakness except by being embedded in the clinic or hospital. For 3 days I worked alongside a bilingual physician at a local family practice clinic. The clinic did full spectrum family practice, including prenatal care. The doctor saw between 25 and 35 patients each day plus covered urgent care during lunch. Paper charting was used although the clinic is looking into electronic records. The clinic was very efficient. All team members were very aware of their roles and did their jobs with a smile and worked well together.
“Most patient encounters took between 10 and 15 minutes although the patient might stay around for IV therapy, intramuscular pain medications, or other treatments that were carried out by the nursing staff. There was a small procedure room and procedures would be performed on the same day they were identified. The nursing staff would set up everything, and in between patients the provider would complete the procedure. On the first day I mostly shadowed, but in the afternoon, I was asked to consult on some of the more complicated patients with diabetes mellitus or hypertension. On the second day I shadowed a health care provider who did not speak English and through an interpreter he asked for my input. In the afternoon the nursing staff asked me to discuss the treatment of abscesses. I discussed techniques of incision and drainage and importance of packing and proper wound care, worked with one of their wound care nurses on packing of several wounds, and consulted on a patient with a venous stasis ulcer.
“We identified an educational opportunity for the nursing staff. On the third day I brought a US certified wound care specialist and I gave a Microsoft PowerPoint presentation on venous stasis ulcers and proper wound care. The nursing staff and clinic were very receptive and asked if we would develop a patient-based educational presentation. The wound care specialist spent the afternoon giving hands-on demonstrations in the wound care clinic, and I taught technique for excisional biopsy of skin tags and moles to physicians. One of the host physicians arranged for more consultations on more of the clinic’s complicated patients, which included a staff member and a relative.”
Medical Technician (MSgt, E-7, Independent Duty Medical Technician, USAF)
“The first day I was assigned to work with the ‘auxiliaries,’ nurses working in the urgent care area at the clinic. Their urgent care area had limited equipment and supplies and included equipment such as mercury thermometers, a few stethoscopes and 1 blood pressure cuff. Their duties consisted of screening patients, starting IVs, giving injections and breathing treatments. They also had a minor surgery room where the nurses helped.
“During the observation of the placement of an IV catheter, I noticed that they were using a port and attaching a needle to the IV tubing and leaving the needle attached to the patient. I asked them about their procedure and incidents with needlesticks since they had to be pretty accurate in getting the needle through the port. The nurse stated there were a significant number of cases of needlesticks. The following day, we brought 18-g, 20-g, and 23-g IV catheters, saline locks, syringes, and our team’s junior physician and I instructed the nurses how to set up an IV without using the needle port.
“The third day at the clinic, I assisted in checking in patients (blood pressure, weight, interviews). I also helped run the immunizations clinic, assisting in giving both pediatric and adult immunizations. Since there was only 1 nurse on shift that day, we multitasked and also gave injections prescribed by the providers, such as medroxyprogesterone and dexamethasone. By far, this was the most rewarding part of the mission. I really felt as though we were part of the team and believe we truly made a difference.”
Administrator (LTC, Medical Service Corps, USN)
“I learned many items from our visit to Clinica Dr. Francisco Quintanas Area de Salud 4 Chacarita. I reviewed the business plan contained in two 1.5-inch hardbound books. Their business plan outlined the population served, projections for upcoming year, and contracts. Area 4 served 21,344 people (11,197 men and 10,147 women). The business plan reviewed historical encounter information (ie, average patient is seen 2.6 times annually, 203,285 laboratory tests were performed in 2010, no radiology capabilities) and contained metrics for key programs for upcoming year (eg, vaccinations, women wellness) that seemed similar to US Healthcare Effectiveness Data and Information Set (HEDIS) measures.
“Our partners discussed financing of the health care they provide, including money flows to and from the government, the work center, and the employees. The business plan contains contract information and costs for maintenance, utilities, personnel, and other issues that would be typical for US-based operations as well. Housekeeping, some of the secretaries, and security staff are not employees—they are contracted personnel. Money is shifted to meet unexpected needs (ie, in 2009/2010–H1N1 influenza was unanticipated). Money was taken from other programs to meet the need.
“Within the Area 4 clinics there are 94 personnel, including 15 physicians. They have a document that is similar to our Activity Manning Document, which outlines personnel billet code, name, and specialty. The Asistentes Técnicos de Atención Primaria are the personnel who conduct home visits and are a unique capability—we do not have an exact equivalent in most US health care systems. Pregnant workers are released from work 1 month prior to the due date and are expected to return to work 3 months postdelivery.”
Medical Logistics (Capt, Medical Service Corps, USAF)
“Costa Rica is still growing in aspects of national health care but has a reliable system in place it seems. Similar to many of the countries visited, it has great capacity for building, but is challenged to increase its infrastructure. In 2011, part of this was due to a recent economic decline in the nation and its health care sector. They have interaction both with other regional clinics managed under the same national health system construct (Caja Costarricense del Seguro Social) as well as with private practices and specialty services. The clinics are open only daytime business hours. Only the regional hospital is open 24/7 for emergent care.
“Supplies are distributed to the regional clinics primarily from San José (the capital and largest city), but also there are some smaller warehousing of clinical materials located around the region. One of these warehouses was in Puntarenas where our clinic was located. To get better information for future supply chain management support we would need to speak with the central distribution/suppliers of all nationalized clinic-run entities. What our partners did teach is that at a higher, national level the clinics are standardized with what they will carry and need to keep on-hand depending upon the clinic classification (ie, level 1, 2, or 3).
“Equipment is purchased similar to the DoD method: Requests are submitted toward the end of the year, the administration prioritizes the lists, and then buys what they feel is most beneficial to the clinic with the resources available. Our hosts stated that before the end of the year, it is very difficult to prioritize needs other than some of the items that they ‘always need’ because they are unlikely to receive items very low on their list. The hosts stated that they would be very interested in having a chance to receive any excess US military equipment from their priority lists if there was a mechanism to do so. In future EHET missions, advance coordination would need to occur to see if (locally compatible) equipment needs could be met through the Defense Reutilization and Marketing Office (DRMO). Alternatively, an embedded team focused on Biomedical Equipment repair could work alongside partners such as at this clinic to develop a sustainable preventive maintenance and equipment testing program. Advance coordination on equipment status would foster improvement for resourceful partner clinics such as Chacarita, with targeted involvement from US military biomedical equipment technicians.”
Discussion
These 4 firsthand accounts from a multidisciplinary, primary-care focused, EHET offers multiple preliminary evidence of the value of this small-scale embedded approach. The accounts are responses to an open-ended prompt for personal impressions and key thoughts as part of an EHET. Three of the advantages gleaned from these accounts are greater personal satisfaction, detailed insight into local operations and health systems, and deeper empathy and respect for common challenges despite health system differences compared with the US military health system.
These advantages are critical to afford the US military personnel the ability to more effectively execute engagement goals, such as meeting health needs in humanitarian assistance, advancing interoperable capacity for security cooperation, or achieving targeted training to enhance US medical operational skills. The greater personal satisfaction was evident in the team member responses that, despite mission stops in 7 prior countries, “This by far was the most rewarding part of the Continuing Promise 2011 mission” and “I hope this becomes a tool used to augment humanitarian missions.”
The descriptions by both the administrator and the logistician on the intimate details that the hosts shared with them is a testament to the rapid trust engendered by the embedded approach. There was a trust to share information as a result of acknowledged local strengths and legitimate interest in local challenges. Peer appreciation was evident; although they did not speak the same literal language, they spoke the same professional language, which was apparent even through the use of an interpreter.
A third advantage, evident from these written exchanges is a regular acknowledgement that health system issues, pursued processes, and desired outcomes are similar between different systems. There may be significant differences in actual resources and infrastructure, but some of the bureaucracy is similar. This last insight is essential to grasp in order to seek capacity building and interoperable solutions toward common goals; empathy is needed to encourage local ownership and sustainability while respecting local challenges and different problem-solving approaches and processes.
Conclusions
The EHET concept afforded deep insight by team members into ways to partner with their hosts to target better health outcomes and meaningful partnership for potential long-term geopolitical impact. Long duration embedded teams, or recurrent insertion, in a single location will achieve greater long-term benefits because of greater health system and cultural understanding. EHETs, once accepted and refined from prototype to standard employment tool, should prove to be a more effective tool in building partnerships, building capacity, and increased security cooperation by using US military resources to support legitimate health needs either in a military-military or military-civilian setting.5 These firsthand accounts provide preliminary evidence that embedded teams may be a critical and needed tool to “ensure that military health engagement is appropriate, constructive, effective, and coordinated with other actors.”6
Acknowledgments
Additional original EHET team members included LCDR Jeanne Jimenez, RN; CDR Francine Worthington, Health Administrator; Maj Tony McClung, RN; Mrs. Romero, RN of LDS Charities, and the staff of the Chacarita clinics in Costa Rica.
1. US Department of Defense. Sustaining U.S. global leadership: priorities for 21st century defense. https://archive.defense.gov/news/Defense_Strategic_Guidance.pdf. Published January 2012. Accessed March 18, 2020.
2. Burkett EK. An embedded health engagement team pilot test, Mil Med. 2019;184(11-12):606-610.
3. Center for Disaster and Humanitarian Assistance Medicine. U.S. participants perspectives on military humanitarian assistance. https://www.hsdl.org/?view&did=446168. Accessed March 18, 2020.
4. Burkett EK. Embedded health engagement teams for improved health outcomes and foreign assistance, Poster presented at: AMSUS Annual Meeting November 30, 2015; San Antonio, TX. http://cdm16005.contentdm.oclc.org/cdm/singleitem/collection/p16005coll8/id/14. Accessed March 18, 2020.
5. Burkett EK, Ubiera J, Vess, J, Griffay T, Neese B, Lawrence C. Developing the prototype embedded health engagement team, Poster presented at: Military Health System Research Symposium, August 21, 2018; Orlando, FL. https://cdm16005.contentdm.oclc.org/digital/collection/p16005coll8/id/61/rec/1. Accessed March 18, 2020.
6. Michaud J, Moss K, Licina D, et al. Security and public health: the interface. Lancet. 2019;393(10168):P276-P286. http://glham.org/wp-content/uploads/Militaries-and-Global-Health-Lancet-Series.pdf. Accessed March 18, 2020.
“Whenever possible, we will develop innovative, low-cost, and small-footprint approaches to achieve our security objectives.” 1
Team member and participant observations can deliver valuable insight into the effectiveness of an activity or project. Certainly, documentation of such qualitative assessment through survey questions or narratives can reveal important information for future action. This qualitative aspect was a significant consideration in the formation of an embedded health engagement team (EHET) intended to improve foreign assistance and health outcomes for global humanitarian and security cooperation activities.
Since health activities are centered on human interaction and relationships, some observation or qualitative assessment must be included to truly determine short-term local buy-in and long-term outcomes. The following observations include the direct narrative perspectives of team members from a multidisciplinary primary care EHET that add experiential depth to prior assessment of the pilot test of such teams during Continuing Promise 2011, a 9-country series of health engagement activities employed from the USNS Comfort.2 The embedded team consisted of US Air Force (USAF), US Navy (USN), and nongovernmental organization (NGO) personnel working directly in a primary care clinic of the Costa Rican public health system.
This is small sample of a few team members who responded to a simple, open-ended prompt to record their impression of the EHET concept and experiences. Documenting this information should highlight the importance of seeking similar qualitative mission data for future health engagements. Standardized questionnaires have been used to evaluate health activities and have provided valuable analysis and recommendations that have advanced US Department of Defense (DoD) global health engagement.3 Captured narrative observation from the EHET pilot study is a complementary qualitative method that supports the concept of small, well prepared, culturally competent, EHETs tailored to work within a partner system rather than outside of it will achieve greater mutual benefit, including the application of better, more equitable health and health system principles.4 In this embedded manner, health care professionals may readily contribute to host nation health sector plans and goals while achieving military objectives, political goals, and mutual strategic interests through both military-military and military-civilian applications.
Observations and Reflections
Family Physician (Maj, Second Physician, USAF)
“Overall, the experience I had with the embedded team was truly rewarding. I hope this becomes a tool used to augment humanitarian missions. There is no way to truly understand a systems strengths and weakness except by being embedded in the clinic or hospital. For 3 days I worked alongside a bilingual physician at a local family practice clinic. The clinic did full spectrum family practice, including prenatal care. The doctor saw between 25 and 35 patients each day plus covered urgent care during lunch. Paper charting was used although the clinic is looking into electronic records. The clinic was very efficient. All team members were very aware of their roles and did their jobs with a smile and worked well together.
“Most patient encounters took between 10 and 15 minutes although the patient might stay around for IV therapy, intramuscular pain medications, or other treatments that were carried out by the nursing staff. There was a small procedure room and procedures would be performed on the same day they were identified. The nursing staff would set up everything, and in between patients the provider would complete the procedure. On the first day I mostly shadowed, but in the afternoon, I was asked to consult on some of the more complicated patients with diabetes mellitus or hypertension. On the second day I shadowed a health care provider who did not speak English and through an interpreter he asked for my input. In the afternoon the nursing staff asked me to discuss the treatment of abscesses. I discussed techniques of incision and drainage and importance of packing and proper wound care, worked with one of their wound care nurses on packing of several wounds, and consulted on a patient with a venous stasis ulcer.
“We identified an educational opportunity for the nursing staff. On the third day I brought a US certified wound care specialist and I gave a Microsoft PowerPoint presentation on venous stasis ulcers and proper wound care. The nursing staff and clinic were very receptive and asked if we would develop a patient-based educational presentation. The wound care specialist spent the afternoon giving hands-on demonstrations in the wound care clinic, and I taught technique for excisional biopsy of skin tags and moles to physicians. One of the host physicians arranged for more consultations on more of the clinic’s complicated patients, which included a staff member and a relative.”
Medical Technician (MSgt, E-7, Independent Duty Medical Technician, USAF)
“The first day I was assigned to work with the ‘auxiliaries,’ nurses working in the urgent care area at the clinic. Their urgent care area had limited equipment and supplies and included equipment such as mercury thermometers, a few stethoscopes and 1 blood pressure cuff. Their duties consisted of screening patients, starting IVs, giving injections and breathing treatments. They also had a minor surgery room where the nurses helped.
“During the observation of the placement of an IV catheter, I noticed that they were using a port and attaching a needle to the IV tubing and leaving the needle attached to the patient. I asked them about their procedure and incidents with needlesticks since they had to be pretty accurate in getting the needle through the port. The nurse stated there were a significant number of cases of needlesticks. The following day, we brought 18-g, 20-g, and 23-g IV catheters, saline locks, syringes, and our team’s junior physician and I instructed the nurses how to set up an IV without using the needle port.
“The third day at the clinic, I assisted in checking in patients (blood pressure, weight, interviews). I also helped run the immunizations clinic, assisting in giving both pediatric and adult immunizations. Since there was only 1 nurse on shift that day, we multitasked and also gave injections prescribed by the providers, such as medroxyprogesterone and dexamethasone. By far, this was the most rewarding part of the mission. I really felt as though we were part of the team and believe we truly made a difference.”
Administrator (LTC, Medical Service Corps, USN)
“I learned many items from our visit to Clinica Dr. Francisco Quintanas Area de Salud 4 Chacarita. I reviewed the business plan contained in two 1.5-inch hardbound books. Their business plan outlined the population served, projections for upcoming year, and contracts. Area 4 served 21,344 people (11,197 men and 10,147 women). The business plan reviewed historical encounter information (ie, average patient is seen 2.6 times annually, 203,285 laboratory tests were performed in 2010, no radiology capabilities) and contained metrics for key programs for upcoming year (eg, vaccinations, women wellness) that seemed similar to US Healthcare Effectiveness Data and Information Set (HEDIS) measures.
“Our partners discussed financing of the health care they provide, including money flows to and from the government, the work center, and the employees. The business plan contains contract information and costs for maintenance, utilities, personnel, and other issues that would be typical for US-based operations as well. Housekeeping, some of the secretaries, and security staff are not employees—they are contracted personnel. Money is shifted to meet unexpected needs (ie, in 2009/2010–H1N1 influenza was unanticipated). Money was taken from other programs to meet the need.
“Within the Area 4 clinics there are 94 personnel, including 15 physicians. They have a document that is similar to our Activity Manning Document, which outlines personnel billet code, name, and specialty. The Asistentes Técnicos de Atención Primaria are the personnel who conduct home visits and are a unique capability—we do not have an exact equivalent in most US health care systems. Pregnant workers are released from work 1 month prior to the due date and are expected to return to work 3 months postdelivery.”
Medical Logistics (Capt, Medical Service Corps, USAF)
“Costa Rica is still growing in aspects of national health care but has a reliable system in place it seems. Similar to many of the countries visited, it has great capacity for building, but is challenged to increase its infrastructure. In 2011, part of this was due to a recent economic decline in the nation and its health care sector. They have interaction both with other regional clinics managed under the same national health system construct (Caja Costarricense del Seguro Social) as well as with private practices and specialty services. The clinics are open only daytime business hours. Only the regional hospital is open 24/7 for emergent care.
“Supplies are distributed to the regional clinics primarily from San José (the capital and largest city), but also there are some smaller warehousing of clinical materials located around the region. One of these warehouses was in Puntarenas where our clinic was located. To get better information for future supply chain management support we would need to speak with the central distribution/suppliers of all nationalized clinic-run entities. What our partners did teach is that at a higher, national level the clinics are standardized with what they will carry and need to keep on-hand depending upon the clinic classification (ie, level 1, 2, or 3).
“Equipment is purchased similar to the DoD method: Requests are submitted toward the end of the year, the administration prioritizes the lists, and then buys what they feel is most beneficial to the clinic with the resources available. Our hosts stated that before the end of the year, it is very difficult to prioritize needs other than some of the items that they ‘always need’ because they are unlikely to receive items very low on their list. The hosts stated that they would be very interested in having a chance to receive any excess US military equipment from their priority lists if there was a mechanism to do so. In future EHET missions, advance coordination would need to occur to see if (locally compatible) equipment needs could be met through the Defense Reutilization and Marketing Office (DRMO). Alternatively, an embedded team focused on Biomedical Equipment repair could work alongside partners such as at this clinic to develop a sustainable preventive maintenance and equipment testing program. Advance coordination on equipment status would foster improvement for resourceful partner clinics such as Chacarita, with targeted involvement from US military biomedical equipment technicians.”
Discussion
These 4 firsthand accounts from a multidisciplinary, primary-care focused, EHET offers multiple preliminary evidence of the value of this small-scale embedded approach. The accounts are responses to an open-ended prompt for personal impressions and key thoughts as part of an EHET. Three of the advantages gleaned from these accounts are greater personal satisfaction, detailed insight into local operations and health systems, and deeper empathy and respect for common challenges despite health system differences compared with the US military health system.
These advantages are critical to afford the US military personnel the ability to more effectively execute engagement goals, such as meeting health needs in humanitarian assistance, advancing interoperable capacity for security cooperation, or achieving targeted training to enhance US medical operational skills. The greater personal satisfaction was evident in the team member responses that, despite mission stops in 7 prior countries, “This by far was the most rewarding part of the Continuing Promise 2011 mission” and “I hope this becomes a tool used to augment humanitarian missions.”
The descriptions by both the administrator and the logistician on the intimate details that the hosts shared with them is a testament to the rapid trust engendered by the embedded approach. There was a trust to share information as a result of acknowledged local strengths and legitimate interest in local challenges. Peer appreciation was evident; although they did not speak the same literal language, they spoke the same professional language, which was apparent even through the use of an interpreter.
A third advantage, evident from these written exchanges is a regular acknowledgement that health system issues, pursued processes, and desired outcomes are similar between different systems. There may be significant differences in actual resources and infrastructure, but some of the bureaucracy is similar. This last insight is essential to grasp in order to seek capacity building and interoperable solutions toward common goals; empathy is needed to encourage local ownership and sustainability while respecting local challenges and different problem-solving approaches and processes.
Conclusions
The EHET concept afforded deep insight by team members into ways to partner with their hosts to target better health outcomes and meaningful partnership for potential long-term geopolitical impact. Long duration embedded teams, or recurrent insertion, in a single location will achieve greater long-term benefits because of greater health system and cultural understanding. EHETs, once accepted and refined from prototype to standard employment tool, should prove to be a more effective tool in building partnerships, building capacity, and increased security cooperation by using US military resources to support legitimate health needs either in a military-military or military-civilian setting.5 These firsthand accounts provide preliminary evidence that embedded teams may be a critical and needed tool to “ensure that military health engagement is appropriate, constructive, effective, and coordinated with other actors.”6
Acknowledgments
Additional original EHET team members included LCDR Jeanne Jimenez, RN; CDR Francine Worthington, Health Administrator; Maj Tony McClung, RN; Mrs. Romero, RN of LDS Charities, and the staff of the Chacarita clinics in Costa Rica.
“Whenever possible, we will develop innovative, low-cost, and small-footprint approaches to achieve our security objectives.” 1
Team member and participant observations can deliver valuable insight into the effectiveness of an activity or project. Certainly, documentation of such qualitative assessment through survey questions or narratives can reveal important information for future action. This qualitative aspect was a significant consideration in the formation of an embedded health engagement team (EHET) intended to improve foreign assistance and health outcomes for global humanitarian and security cooperation activities.
Since health activities are centered on human interaction and relationships, some observation or qualitative assessment must be included to truly determine short-term local buy-in and long-term outcomes. The following observations include the direct narrative perspectives of team members from a multidisciplinary primary care EHET that add experiential depth to prior assessment of the pilot test of such teams during Continuing Promise 2011, a 9-country series of health engagement activities employed from the USNS Comfort.2 The embedded team consisted of US Air Force (USAF), US Navy (USN), and nongovernmental organization (NGO) personnel working directly in a primary care clinic of the Costa Rican public health system.
This is small sample of a few team members who responded to a simple, open-ended prompt to record their impression of the EHET concept and experiences. Documenting this information should highlight the importance of seeking similar qualitative mission data for future health engagements. Standardized questionnaires have been used to evaluate health activities and have provided valuable analysis and recommendations that have advanced US Department of Defense (DoD) global health engagement.3 Captured narrative observation from the EHET pilot study is a complementary qualitative method that supports the concept of small, well prepared, culturally competent, EHETs tailored to work within a partner system rather than outside of it will achieve greater mutual benefit, including the application of better, more equitable health and health system principles.4 In this embedded manner, health care professionals may readily contribute to host nation health sector plans and goals while achieving military objectives, political goals, and mutual strategic interests through both military-military and military-civilian applications.
Observations and Reflections
Family Physician (Maj, Second Physician, USAF)
“Overall, the experience I had with the embedded team was truly rewarding. I hope this becomes a tool used to augment humanitarian missions. There is no way to truly understand a systems strengths and weakness except by being embedded in the clinic or hospital. For 3 days I worked alongside a bilingual physician at a local family practice clinic. The clinic did full spectrum family practice, including prenatal care. The doctor saw between 25 and 35 patients each day plus covered urgent care during lunch. Paper charting was used although the clinic is looking into electronic records. The clinic was very efficient. All team members were very aware of their roles and did their jobs with a smile and worked well together.
“Most patient encounters took between 10 and 15 minutes although the patient might stay around for IV therapy, intramuscular pain medications, or other treatments that were carried out by the nursing staff. There was a small procedure room and procedures would be performed on the same day they were identified. The nursing staff would set up everything, and in between patients the provider would complete the procedure. On the first day I mostly shadowed, but in the afternoon, I was asked to consult on some of the more complicated patients with diabetes mellitus or hypertension. On the second day I shadowed a health care provider who did not speak English and through an interpreter he asked for my input. In the afternoon the nursing staff asked me to discuss the treatment of abscesses. I discussed techniques of incision and drainage and importance of packing and proper wound care, worked with one of their wound care nurses on packing of several wounds, and consulted on a patient with a venous stasis ulcer.
“We identified an educational opportunity for the nursing staff. On the third day I brought a US certified wound care specialist and I gave a Microsoft PowerPoint presentation on venous stasis ulcers and proper wound care. The nursing staff and clinic were very receptive and asked if we would develop a patient-based educational presentation. The wound care specialist spent the afternoon giving hands-on demonstrations in the wound care clinic, and I taught technique for excisional biopsy of skin tags and moles to physicians. One of the host physicians arranged for more consultations on more of the clinic’s complicated patients, which included a staff member and a relative.”
Medical Technician (MSgt, E-7, Independent Duty Medical Technician, USAF)
“The first day I was assigned to work with the ‘auxiliaries,’ nurses working in the urgent care area at the clinic. Their urgent care area had limited equipment and supplies and included equipment such as mercury thermometers, a few stethoscopes and 1 blood pressure cuff. Their duties consisted of screening patients, starting IVs, giving injections and breathing treatments. They also had a minor surgery room where the nurses helped.
“During the observation of the placement of an IV catheter, I noticed that they were using a port and attaching a needle to the IV tubing and leaving the needle attached to the patient. I asked them about their procedure and incidents with needlesticks since they had to be pretty accurate in getting the needle through the port. The nurse stated there were a significant number of cases of needlesticks. The following day, we brought 18-g, 20-g, and 23-g IV catheters, saline locks, syringes, and our team’s junior physician and I instructed the nurses how to set up an IV without using the needle port.
“The third day at the clinic, I assisted in checking in patients (blood pressure, weight, interviews). I also helped run the immunizations clinic, assisting in giving both pediatric and adult immunizations. Since there was only 1 nurse on shift that day, we multitasked and also gave injections prescribed by the providers, such as medroxyprogesterone and dexamethasone. By far, this was the most rewarding part of the mission. I really felt as though we were part of the team and believe we truly made a difference.”
Administrator (LTC, Medical Service Corps, USN)
“I learned many items from our visit to Clinica Dr. Francisco Quintanas Area de Salud 4 Chacarita. I reviewed the business plan contained in two 1.5-inch hardbound books. Their business plan outlined the population served, projections for upcoming year, and contracts. Area 4 served 21,344 people (11,197 men and 10,147 women). The business plan reviewed historical encounter information (ie, average patient is seen 2.6 times annually, 203,285 laboratory tests were performed in 2010, no radiology capabilities) and contained metrics for key programs for upcoming year (eg, vaccinations, women wellness) that seemed similar to US Healthcare Effectiveness Data and Information Set (HEDIS) measures.
“Our partners discussed financing of the health care they provide, including money flows to and from the government, the work center, and the employees. The business plan contains contract information and costs for maintenance, utilities, personnel, and other issues that would be typical for US-based operations as well. Housekeeping, some of the secretaries, and security staff are not employees—they are contracted personnel. Money is shifted to meet unexpected needs (ie, in 2009/2010–H1N1 influenza was unanticipated). Money was taken from other programs to meet the need.
“Within the Area 4 clinics there are 94 personnel, including 15 physicians. They have a document that is similar to our Activity Manning Document, which outlines personnel billet code, name, and specialty. The Asistentes Técnicos de Atención Primaria are the personnel who conduct home visits and are a unique capability—we do not have an exact equivalent in most US health care systems. Pregnant workers are released from work 1 month prior to the due date and are expected to return to work 3 months postdelivery.”
Medical Logistics (Capt, Medical Service Corps, USAF)
“Costa Rica is still growing in aspects of national health care but has a reliable system in place it seems. Similar to many of the countries visited, it has great capacity for building, but is challenged to increase its infrastructure. In 2011, part of this was due to a recent economic decline in the nation and its health care sector. They have interaction both with other regional clinics managed under the same national health system construct (Caja Costarricense del Seguro Social) as well as with private practices and specialty services. The clinics are open only daytime business hours. Only the regional hospital is open 24/7 for emergent care.
“Supplies are distributed to the regional clinics primarily from San José (the capital and largest city), but also there are some smaller warehousing of clinical materials located around the region. One of these warehouses was in Puntarenas where our clinic was located. To get better information for future supply chain management support we would need to speak with the central distribution/suppliers of all nationalized clinic-run entities. What our partners did teach is that at a higher, national level the clinics are standardized with what they will carry and need to keep on-hand depending upon the clinic classification (ie, level 1, 2, or 3).
“Equipment is purchased similar to the DoD method: Requests are submitted toward the end of the year, the administration prioritizes the lists, and then buys what they feel is most beneficial to the clinic with the resources available. Our hosts stated that before the end of the year, it is very difficult to prioritize needs other than some of the items that they ‘always need’ because they are unlikely to receive items very low on their list. The hosts stated that they would be very interested in having a chance to receive any excess US military equipment from their priority lists if there was a mechanism to do so. In future EHET missions, advance coordination would need to occur to see if (locally compatible) equipment needs could be met through the Defense Reutilization and Marketing Office (DRMO). Alternatively, an embedded team focused on Biomedical Equipment repair could work alongside partners such as at this clinic to develop a sustainable preventive maintenance and equipment testing program. Advance coordination on equipment status would foster improvement for resourceful partner clinics such as Chacarita, with targeted involvement from US military biomedical equipment technicians.”
Discussion
These 4 firsthand accounts from a multidisciplinary, primary-care focused, EHET offers multiple preliminary evidence of the value of this small-scale embedded approach. The accounts are responses to an open-ended prompt for personal impressions and key thoughts as part of an EHET. Three of the advantages gleaned from these accounts are greater personal satisfaction, detailed insight into local operations and health systems, and deeper empathy and respect for common challenges despite health system differences compared with the US military health system.
These advantages are critical to afford the US military personnel the ability to more effectively execute engagement goals, such as meeting health needs in humanitarian assistance, advancing interoperable capacity for security cooperation, or achieving targeted training to enhance US medical operational skills. The greater personal satisfaction was evident in the team member responses that, despite mission stops in 7 prior countries, “This by far was the most rewarding part of the Continuing Promise 2011 mission” and “I hope this becomes a tool used to augment humanitarian missions.”
The descriptions by both the administrator and the logistician on the intimate details that the hosts shared with them is a testament to the rapid trust engendered by the embedded approach. There was a trust to share information as a result of acknowledged local strengths and legitimate interest in local challenges. Peer appreciation was evident; although they did not speak the same literal language, they spoke the same professional language, which was apparent even through the use of an interpreter.
A third advantage, evident from these written exchanges is a regular acknowledgement that health system issues, pursued processes, and desired outcomes are similar between different systems. There may be significant differences in actual resources and infrastructure, but some of the bureaucracy is similar. This last insight is essential to grasp in order to seek capacity building and interoperable solutions toward common goals; empathy is needed to encourage local ownership and sustainability while respecting local challenges and different problem-solving approaches and processes.
Conclusions
The EHET concept afforded deep insight by team members into ways to partner with their hosts to target better health outcomes and meaningful partnership for potential long-term geopolitical impact. Long duration embedded teams, or recurrent insertion, in a single location will achieve greater long-term benefits because of greater health system and cultural understanding. EHETs, once accepted and refined from prototype to standard employment tool, should prove to be a more effective tool in building partnerships, building capacity, and increased security cooperation by using US military resources to support legitimate health needs either in a military-military or military-civilian setting.5 These firsthand accounts provide preliminary evidence that embedded teams may be a critical and needed tool to “ensure that military health engagement is appropriate, constructive, effective, and coordinated with other actors.”6
Acknowledgments
Additional original EHET team members included LCDR Jeanne Jimenez, RN; CDR Francine Worthington, Health Administrator; Maj Tony McClung, RN; Mrs. Romero, RN of LDS Charities, and the staff of the Chacarita clinics in Costa Rica.
1. US Department of Defense. Sustaining U.S. global leadership: priorities for 21st century defense. https://archive.defense.gov/news/Defense_Strategic_Guidance.pdf. Published January 2012. Accessed March 18, 2020.
2. Burkett EK. An embedded health engagement team pilot test, Mil Med. 2019;184(11-12):606-610.
3. Center for Disaster and Humanitarian Assistance Medicine. U.S. participants perspectives on military humanitarian assistance. https://www.hsdl.org/?view&did=446168. Accessed March 18, 2020.
4. Burkett EK. Embedded health engagement teams for improved health outcomes and foreign assistance, Poster presented at: AMSUS Annual Meeting November 30, 2015; San Antonio, TX. http://cdm16005.contentdm.oclc.org/cdm/singleitem/collection/p16005coll8/id/14. Accessed March 18, 2020.
5. Burkett EK, Ubiera J, Vess, J, Griffay T, Neese B, Lawrence C. Developing the prototype embedded health engagement team, Poster presented at: Military Health System Research Symposium, August 21, 2018; Orlando, FL. https://cdm16005.contentdm.oclc.org/digital/collection/p16005coll8/id/61/rec/1. Accessed March 18, 2020.
6. Michaud J, Moss K, Licina D, et al. Security and public health: the interface. Lancet. 2019;393(10168):P276-P286. http://glham.org/wp-content/uploads/Militaries-and-Global-Health-Lancet-Series.pdf. Accessed March 18, 2020.
1. US Department of Defense. Sustaining U.S. global leadership: priorities for 21st century defense. https://archive.defense.gov/news/Defense_Strategic_Guidance.pdf. Published January 2012. Accessed March 18, 2020.
2. Burkett EK. An embedded health engagement team pilot test, Mil Med. 2019;184(11-12):606-610.
3. Center for Disaster and Humanitarian Assistance Medicine. U.S. participants perspectives on military humanitarian assistance. https://www.hsdl.org/?view&did=446168. Accessed March 18, 2020.
4. Burkett EK. Embedded health engagement teams for improved health outcomes and foreign assistance, Poster presented at: AMSUS Annual Meeting November 30, 2015; San Antonio, TX. http://cdm16005.contentdm.oclc.org/cdm/singleitem/collection/p16005coll8/id/14. Accessed March 18, 2020.
5. Burkett EK, Ubiera J, Vess, J, Griffay T, Neese B, Lawrence C. Developing the prototype embedded health engagement team, Poster presented at: Military Health System Research Symposium, August 21, 2018; Orlando, FL. https://cdm16005.contentdm.oclc.org/digital/collection/p16005coll8/id/61/rec/1. Accessed March 18, 2020.
6. Michaud J, Moss K, Licina D, et al. Security and public health: the interface. Lancet. 2019;393(10168):P276-P286. http://glham.org/wp-content/uploads/Militaries-and-Global-Health-Lancet-Series.pdf. Accessed March 18, 2020.
Belimumab may improve skin in scleroderma
MAUI, HAWAII – Belimumab shows promise as a novel biologic treatment for skin involvement in early diffuse cutaneous systemic sclerosis, Janet E. Pope, MD, said at the 2020 Rheumatology Winter Clinical Symposium.
She highlighted a single-center, double-blind, placebo-controlled, New York pilot study including 20 patients with early diffuse cutaneous systemic sclerosis and moderate skin involvement. Participants had recently started on background mycophenolate mofetil (MMF) at 1,000 mg twice daily and were then randomized to add-on belimumab (Benlysta) at the dosing approved for systemic lupus erythematosus or to infusions of normal saline.
At 52 weeks, the modified Rodnan skin thickness score (mRSS) decreased by a median of 10 points from a baseline of 27 in the belimumab group, compared with just a 3-point reduction in controls on MMF plus placebo.
This small study raises several key points. It definitely warrants confirmation in a large phase 3 trial, according to Dr. Pope, professor of medicine at the University of Western Ontario and head of the division of rheumatology at St. Joseph’s Health Care, both in London.
For one thing, the pilot study makes a good case for multidrug therapy in scleroderma. “In rheumatoid arthritis, if in general one drug is not as good as two, why would we ever think, in our most difficult-to-treat disease, one drug would be okay?” the rheumatologist observed.
The belimumab study also highlights the role of abnormalities in B-cell function in the pathogenesis of skin involvement in early diffuse cutaneous systemic sclerosis. Belimumab is a fully human monoclonal antibody which binds to soluble B-lymphocyte stimulator and inhibits autoantibody production.
Belimumab’s mechanism of benefit was as expected: The improvement in skin scores in the belimumab group was accompanied by decreased expression of profibrotic genes and B-cell signaling, changes that didn’t occur in the controls on MMF alone.
The belimumab study makes another important point: MMF, despite its growing popularity for treatment of skin manifestations of scleroderma, is actually a wimpy drug for that purpose, achieving a mere 3-point reduction in mRSS.
“To be quite honest, mycophenolate mofetil is not all that great on skin,” Dr. Pope said.
Nonetheless, when she and her coworkers recently polled 170 scleroderma experts as to their favored treatments directed at various target organs impaired by the disease, as she had previously done in 2012, a clear trend was evident. “There’s a shift in that mycophenolate mofetil is moving to first-line treatment across the board for skin,” Dr. Pope observed.
Indeed, in the more recent survey, 71% of the experts agreed upon a scleroderma skin involvement treatment algorithm in which the first-line treatment for severe skin disease as defined by an mRSS of 32 was MMF, with methotrexate as second line, intravenous cyclophosphamide third, and autologous stem cell transplantation as fourth line for the small number of patients who qualify for it.
For moderate skin involvement, with an mRSS of 24, methotrexate was endorsed as first line, although by the narrowest of margins, over MMF, with intravenous cyclophosphamide as third line. For mild disease, with an mRSS of 10, methotrexate again narrowly beat out MMF by expert consensus as the preferred first-line therapy.
When asked about concomitant use of corticosteroids for treatment of skin involvement, 35% of experts said they never prescribe them for that indication, 33% do so occasionally, 19% sometimes, and 13% routinely. There was an even split on dosing among those who prescribe steroids: 49% suggested using prednisone at less than 7.5 mg/day, and 51% recommended 7.5-20 mg/day.
The purpose in polling the experts, who were drawn from the Scleroderma Clinical Trials Consortium and the Canadian Scleroderma Research Group, was to provide treatment guidance to general rheumatologists and dermatologists who may not see many patients with scleroderma. In contrast, the great majority of the polled experts see more than 50 scleroderma patients per year. And they had a high level of total agreement for treatment algorithms addressing not only skin disease, but also pulmonary arterial hypertension, interstitial lung disease, Raynaud’s phenomenon, renal crisis, digital ulcers, inflammatory arthritis, cardiac involvement, and gastrointestinal disease, Dr. Pope noted.
She attributed the experts’ rising enthusiasm for MMF for scleroderma skin involvement to the results of the Scleroderma Lung Study II, the first randomized, controlled trial to compare MMF and cyclophosphamide for the treatment of symptomatic scleroderma interstitial lung disease. Two years of MMF improved forced vital capacity as much as 1 year of oral cyclophosphamide. At 2 years of follow-up, the mRSS dropped modestly from baseline by an average of 6.1 points in the cyclophosphamide group and 2.9 points with MMF, a nonsignificant difference. But the incidence of serious adverse events was roughly three times higher and deaths were twice as frequent in the cyclophosphamide group.
“I think mycophenolate mofetil is surging for treatment of skin because of the lung protection and it was safer, but it’s hard for me to know if the deaths were more common in the cyclophosphamide group because of the cyclophosphamide or because of no treatment in year 2,” Dr. Pope commented.
She reported receiving research grants from Bristol-Myers Squibb, Merck, Roche, Seattle Genetics, and UCB, and serving as a consultant to more than a dozen pharmaceutical companies.
MAUI, HAWAII – Belimumab shows promise as a novel biologic treatment for skin involvement in early diffuse cutaneous systemic sclerosis, Janet E. Pope, MD, said at the 2020 Rheumatology Winter Clinical Symposium.
She highlighted a single-center, double-blind, placebo-controlled, New York pilot study including 20 patients with early diffuse cutaneous systemic sclerosis and moderate skin involvement. Participants had recently started on background mycophenolate mofetil (MMF) at 1,000 mg twice daily and were then randomized to add-on belimumab (Benlysta) at the dosing approved for systemic lupus erythematosus or to infusions of normal saline.
At 52 weeks, the modified Rodnan skin thickness score (mRSS) decreased by a median of 10 points from a baseline of 27 in the belimumab group, compared with just a 3-point reduction in controls on MMF plus placebo.
This small study raises several key points. It definitely warrants confirmation in a large phase 3 trial, according to Dr. Pope, professor of medicine at the University of Western Ontario and head of the division of rheumatology at St. Joseph’s Health Care, both in London.
For one thing, the pilot study makes a good case for multidrug therapy in scleroderma. “In rheumatoid arthritis, if in general one drug is not as good as two, why would we ever think, in our most difficult-to-treat disease, one drug would be okay?” the rheumatologist observed.
The belimumab study also highlights the role of abnormalities in B-cell function in the pathogenesis of skin involvement in early diffuse cutaneous systemic sclerosis. Belimumab is a fully human monoclonal antibody which binds to soluble B-lymphocyte stimulator and inhibits autoantibody production.
Belimumab’s mechanism of benefit was as expected: The improvement in skin scores in the belimumab group was accompanied by decreased expression of profibrotic genes and B-cell signaling, changes that didn’t occur in the controls on MMF alone.
The belimumab study makes another important point: MMF, despite its growing popularity for treatment of skin manifestations of scleroderma, is actually a wimpy drug for that purpose, achieving a mere 3-point reduction in mRSS.
“To be quite honest, mycophenolate mofetil is not all that great on skin,” Dr. Pope said.
Nonetheless, when she and her coworkers recently polled 170 scleroderma experts as to their favored treatments directed at various target organs impaired by the disease, as she had previously done in 2012, a clear trend was evident. “There’s a shift in that mycophenolate mofetil is moving to first-line treatment across the board for skin,” Dr. Pope observed.
Indeed, in the more recent survey, 71% of the experts agreed upon a scleroderma skin involvement treatment algorithm in which the first-line treatment for severe skin disease as defined by an mRSS of 32 was MMF, with methotrexate as second line, intravenous cyclophosphamide third, and autologous stem cell transplantation as fourth line for the small number of patients who qualify for it.
For moderate skin involvement, with an mRSS of 24, methotrexate was endorsed as first line, although by the narrowest of margins, over MMF, with intravenous cyclophosphamide as third line. For mild disease, with an mRSS of 10, methotrexate again narrowly beat out MMF by expert consensus as the preferred first-line therapy.
When asked about concomitant use of corticosteroids for treatment of skin involvement, 35% of experts said they never prescribe them for that indication, 33% do so occasionally, 19% sometimes, and 13% routinely. There was an even split on dosing among those who prescribe steroids: 49% suggested using prednisone at less than 7.5 mg/day, and 51% recommended 7.5-20 mg/day.
The purpose in polling the experts, who were drawn from the Scleroderma Clinical Trials Consortium and the Canadian Scleroderma Research Group, was to provide treatment guidance to general rheumatologists and dermatologists who may not see many patients with scleroderma. In contrast, the great majority of the polled experts see more than 50 scleroderma patients per year. And they had a high level of total agreement for treatment algorithms addressing not only skin disease, but also pulmonary arterial hypertension, interstitial lung disease, Raynaud’s phenomenon, renal crisis, digital ulcers, inflammatory arthritis, cardiac involvement, and gastrointestinal disease, Dr. Pope noted.
She attributed the experts’ rising enthusiasm for MMF for scleroderma skin involvement to the results of the Scleroderma Lung Study II, the first randomized, controlled trial to compare MMF and cyclophosphamide for the treatment of symptomatic scleroderma interstitial lung disease. Two years of MMF improved forced vital capacity as much as 1 year of oral cyclophosphamide. At 2 years of follow-up, the mRSS dropped modestly from baseline by an average of 6.1 points in the cyclophosphamide group and 2.9 points with MMF, a nonsignificant difference. But the incidence of serious adverse events was roughly three times higher and deaths were twice as frequent in the cyclophosphamide group.
“I think mycophenolate mofetil is surging for treatment of skin because of the lung protection and it was safer, but it’s hard for me to know if the deaths were more common in the cyclophosphamide group because of the cyclophosphamide or because of no treatment in year 2,” Dr. Pope commented.
She reported receiving research grants from Bristol-Myers Squibb, Merck, Roche, Seattle Genetics, and UCB, and serving as a consultant to more than a dozen pharmaceutical companies.
MAUI, HAWAII – Belimumab shows promise as a novel biologic treatment for skin involvement in early diffuse cutaneous systemic sclerosis, Janet E. Pope, MD, said at the 2020 Rheumatology Winter Clinical Symposium.
She highlighted a single-center, double-blind, placebo-controlled, New York pilot study including 20 patients with early diffuse cutaneous systemic sclerosis and moderate skin involvement. Participants had recently started on background mycophenolate mofetil (MMF) at 1,000 mg twice daily and were then randomized to add-on belimumab (Benlysta) at the dosing approved for systemic lupus erythematosus or to infusions of normal saline.
At 52 weeks, the modified Rodnan skin thickness score (mRSS) decreased by a median of 10 points from a baseline of 27 in the belimumab group, compared with just a 3-point reduction in controls on MMF plus placebo.
This small study raises several key points. It definitely warrants confirmation in a large phase 3 trial, according to Dr. Pope, professor of medicine at the University of Western Ontario and head of the division of rheumatology at St. Joseph’s Health Care, both in London.
For one thing, the pilot study makes a good case for multidrug therapy in scleroderma. “In rheumatoid arthritis, if in general one drug is not as good as two, why would we ever think, in our most difficult-to-treat disease, one drug would be okay?” the rheumatologist observed.
The belimumab study also highlights the role of abnormalities in B-cell function in the pathogenesis of skin involvement in early diffuse cutaneous systemic sclerosis. Belimumab is a fully human monoclonal antibody which binds to soluble B-lymphocyte stimulator and inhibits autoantibody production.
Belimumab’s mechanism of benefit was as expected: The improvement in skin scores in the belimumab group was accompanied by decreased expression of profibrotic genes and B-cell signaling, changes that didn’t occur in the controls on MMF alone.
The belimumab study makes another important point: MMF, despite its growing popularity for treatment of skin manifestations of scleroderma, is actually a wimpy drug for that purpose, achieving a mere 3-point reduction in mRSS.
“To be quite honest, mycophenolate mofetil is not all that great on skin,” Dr. Pope said.
Nonetheless, when she and her coworkers recently polled 170 scleroderma experts as to their favored treatments directed at various target organs impaired by the disease, as she had previously done in 2012, a clear trend was evident. “There’s a shift in that mycophenolate mofetil is moving to first-line treatment across the board for skin,” Dr. Pope observed.
Indeed, in the more recent survey, 71% of the experts agreed upon a scleroderma skin involvement treatment algorithm in which the first-line treatment for severe skin disease as defined by an mRSS of 32 was MMF, with methotrexate as second line, intravenous cyclophosphamide third, and autologous stem cell transplantation as fourth line for the small number of patients who qualify for it.
For moderate skin involvement, with an mRSS of 24, methotrexate was endorsed as first line, although by the narrowest of margins, over MMF, with intravenous cyclophosphamide as third line. For mild disease, with an mRSS of 10, methotrexate again narrowly beat out MMF by expert consensus as the preferred first-line therapy.
When asked about concomitant use of corticosteroids for treatment of skin involvement, 35% of experts said they never prescribe them for that indication, 33% do so occasionally, 19% sometimes, and 13% routinely. There was an even split on dosing among those who prescribe steroids: 49% suggested using prednisone at less than 7.5 mg/day, and 51% recommended 7.5-20 mg/day.
The purpose in polling the experts, who were drawn from the Scleroderma Clinical Trials Consortium and the Canadian Scleroderma Research Group, was to provide treatment guidance to general rheumatologists and dermatologists who may not see many patients with scleroderma. In contrast, the great majority of the polled experts see more than 50 scleroderma patients per year. And they had a high level of total agreement for treatment algorithms addressing not only skin disease, but also pulmonary arterial hypertension, interstitial lung disease, Raynaud’s phenomenon, renal crisis, digital ulcers, inflammatory arthritis, cardiac involvement, and gastrointestinal disease, Dr. Pope noted.
She attributed the experts’ rising enthusiasm for MMF for scleroderma skin involvement to the results of the Scleroderma Lung Study II, the first randomized, controlled trial to compare MMF and cyclophosphamide for the treatment of symptomatic scleroderma interstitial lung disease. Two years of MMF improved forced vital capacity as much as 1 year of oral cyclophosphamide. At 2 years of follow-up, the mRSS dropped modestly from baseline by an average of 6.1 points in the cyclophosphamide group and 2.9 points with MMF, a nonsignificant difference. But the incidence of serious adverse events was roughly three times higher and deaths were twice as frequent in the cyclophosphamide group.
“I think mycophenolate mofetil is surging for treatment of skin because of the lung protection and it was safer, but it’s hard for me to know if the deaths were more common in the cyclophosphamide group because of the cyclophosphamide or because of no treatment in year 2,” Dr. Pope commented.
She reported receiving research grants from Bristol-Myers Squibb, Merck, Roche, Seattle Genetics, and UCB, and serving as a consultant to more than a dozen pharmaceutical companies.
REPORTING FROM RWCS 2020
An Interdisciplinary Clinic for Former Prisoners of War
Since the beginning of the American Republic, servicemen have been captured and held as prisoners of war (POWs), including > 130,000 in World War II , > 7,100 in the Korean War, > 700 in the Vietnam War, and 37 in Operation Desert Storm and recent conflicts.1,2 Also, > 80 servicewomen have been held during these conflicts.1-3 Of those living former POWs (FPOWs), almost all are geriatric (aged > 65 years) with a significant portion aged ≥ 85 years.
The physical hardships and psychological stress endured by FPOWs have lifelong deleterious sequelae on health and social functioning.3-5 The experiences of FPOWs are associated with higher prevalence of chronic diseases and diminished functional performance in later life as demonstrated by a survey of FPOWs from World War II.4 The survey assessed health and functional status in a random sample of 101 FPOWs and a group of 107 non-POW combatants from the same military operations. FPOWs reported a higher mean number of somatic symptoms than did non-POWs (7.2 vs 5.4, respectively; P = .002), a higher mean number of diagnosed health conditions (9.4 vs 7.7, respectively; P < .001), and used a greater mean number of medications (4.5 vs 3.4, respectively; P = .001). Among 15 broad categories of diagnoses, differences were found in gastrointestinal disorders (FPOWs 63% vs non-POWs 49%, P = .032), musculoskeletal disorders (FPOWs 76% vs non-POWs 60%, P = .001), and cognitive disorders (FPOWs 31% vs non-POWs 15%, P = .006). FPOWs had a significantly higher proportion of 7 extrapyramidal signs and 6 signs relating to ataxia. On the Instrumental Activities of Daily Living scale, FPOWs were more likely to be impaired than were non-POWs (33% vs 17%, respectively; P = .01). In addition, FPOWs have an increased risk of developing dementia, and this risk is doubled in FPOWs with posttraumatic stress disorder (PTSD) compared with non-FPOWs without PTSD.5
These data indicate that FPOW status is associated with increased risk of disability and loss of independence. Federal statutes established the presumption of a relationship between FPOW status and many comorbidities for VA disability determinations in recognition of such data and to overcome lack of medical records during POW confinement and to accord benefit of the doubt where medical science cannot conclusively link disease etiology to FPOW status, to FPOWs.
Service-Connected Conditions
The historical development of conditions with a presumption of service connection for adjudication of VA compensation/disability claims began in 1921 with the Act to Establish a Veterans’ Bureau and to Improve the Facilities.1 The act simplified and streamlined the claims adjudication process by eliminating the need to obtain evidence on the part of the veteran. The presumption of service connection also facilitated increased accuracy and consistency in adjudications by requiring similar treatment for similar claims. This “presumptive” process relieved claimants and VA of the necessity of producing direct evidence when it was impractical to do so.
In 1970, the first presumptives specific to FPOWs were legislatively established and covered 17 diseases for a FPOW who had been confined for ≥ 30 days (Pub. L. 91-376). The 30-day confinement requirement was later relaxed, and additional presumptives were established that related to diseases that were more common among FPOWs than they were among non-FPOWs. These disorders included traumatic arthritis, stroke, heart disease, osteoporosis, peripheral neuropathy, cold injuries, as well as a variety of digestive and neuropsychiatric disorders. If a FPOW is diagnosed as having ≥ 1 of these conditions and it is judged to be ≥ 10% disabling, the condition is presumed to be a sequelae of the POW experience, and it is classified as a service-connected disability (Table).
FPOW Care And Benefits Teams
Several Veterans Health Administration (VHA) directives have been issued, including the recent VHA directive 1650, which requires that each VHA medical facility have a special Care and Benefits Team (CBT) that is charged with the evaluation and treatment of FPOWs to ensure that “FPOWs receive the highest quality care and benefit services.”6 CBTs must be composed of a clinician trained in internal medicine or family practice; a clinician who is certified through the VA Office of Disability and Medical Assessment to conduct General Medical Compensation and Pension evaluations; a FPOW advocate who typically is a VHA clinical social worker; and a Veterans Benefits Administration (VBA) FPOW coordinator appointed by the local VBA regional office. CBTs can be expanded to include other members as needed. The CBTs are tasked with facilitating interactions between FPOWs, the VHA, and the VBA.
CBTs face several challenges in meeting their responsibilities. For example, the POW experience often results in psychological trauma that foments denial and distrust; hence, thoughtful sensitivity to the sequelae of captivity when approaching FPOWs about personal issues, such as health care, is required. Establishing trusting relationships with FPOWs is necessary if their needs are to be effectively addressed.
While the VHA is mandated to provide priority treatment for FPOWs, including hospital, nursing home, dental, and outpatient treatment, a significant number of FPOWs do not avail themselves of benefits to which they are entitled. Often these FPOWs have not used VA programs and facilities because they are uninformed or confused about VA benefits for FPOWs. As a result, referrals of eligible FPOWs to appropriate programs can be overlooked. Maximizing the service-connected disability rating of FPOWs not only impacts the disability pensions received by these veterans, but also impacts their eligibility for VHA programs, including long-term care and Dependency and Indemnity Compensation, a monthly benefit paid to spouses, children, and/or surviving parents.
In 2013, the FPOW Committee of the South Texas Veterans Health Care System (STVHCS) noted that 40% of FPOWs in our region had no VA primary care or clinic assignment. In consideration of the commitment of the VA to care for FPOWs, the unique POW-related medical and psychological issues, the geriatric age of many FPOWs, and the surprising number of FPOWs currently not receiving VA care, we expanded the concept of the CBT team to create a specialized interdisciplinary FPOW Clinic to address the unique needs of this predominantly elderly population and to involve more FPOWs in the VA system.
The main purpose of this clinic was to advise FPOWs of all VA benefits and services to which they may be entitled by identifying overlooked FPOW presumptives. As the number of FPOWs continues to decrease, outreach to FPOWs and family members has become critical, especially as increased benefits and special services might be available to this increasingly dependent older population. An informal survey of FPOW advocates across the nation found that 21% of FPOWs had disability ratings from the VA of ≤ 60%, including some who had no VA disability rating at all. Thus, an additional goal of the project was to develop a clinic model that could be disseminated throughout the VHA.
Design
The design of the FPOW Clinic team is based on an interdisciplinary model that has proven successful in geriatric medicine.7 The team comprises a physician, a social worker, and a registered nurse.8 All members have expertise in geriatric medicine and specific training in FPOW-related issues by completing a VA employee education training session on FPOW case management. Completion of this training ensured that team members were:
- Familiar with the experiences of FPOWs as well as about the medical, psychosocial, and mental health conditions that affect FPOWs;
- Knowledgeable about FPOW presumptive conditions;
- Familiar with the VBA process for rating FPOW disability claims; and
- Capable of FPOW case coordination, workflow, and communications between the FPOW Clinic team and the VBA to avail FPOWs and their families of all eligible benefits.
In-person FPOW clinic visits and chart reviews helped identify overlooked FPOW benefits. To facilitate case management, a representative of the VBA attended the initial evaluation of each FPOW in the clinic to confirm any overlooked presumptive benefits and to familiarize FPOWs with the claims process. FPOWs were also given the choice to officially enroll in the FPOW clinic for primary care or to remain with their current health care provider. Special efforts were made to enroll those FPOWs who had no STVHCS assigned primary care clinic.
The clinic was scheduled for 4 hours every week. Initial patient visits were 2 hours each and consisted of separate evaluations by each of the 3 FPOW Clinic team members who then met as a team with the addition of the VBA representative. The purpose of this meeting was to discuss overlooked benefits, address any other specific issues noted, and to devise an appropriate interdisciplinary plan. Findings of overlooked benefits and other relevant outcomes then were conveyed to the FPOW. For FPOWs who opted to continue in the clinic for their primary care, subsequent appointments were 1 hour.
Implementation
STVHCS FPOW advocates identified and sent letters to FPOWs announcing the opening of the clinic and its goals. Phone calls were made to each FPOW to address questions and to ascertain their interest. The FPOW advocates then worked directly with schedulers to make clinic appointments. Forty-one FPOWs responded to this initial invitation and attended the new clinic. Subsequently, this number increased through FPOW consults placed by STVHCS primary care providers.
The service-connected disability rating of clinic patients ranged from none (6% of attendees) to 100% (28% of attendees). For 34% of patients, clinic attendance resulted in identification application for overlooked presumptives. VBA evaluation resulted in increased service-connected disability ratings for nearly one-third of clinic patients. All clinic patients without a service-connected disability prior to FPOW clinic evaluation received an increased service-connected disability rating. Overall, 60% of the FPOWs who attended the clinic opted to receive their primary care at the FPOW clinic.
The FPOW Clinic successfully identified overlooked presumptives and facilitated the determination of appropriate service-connected disabilities. Interestingly, the FPOW Clinic encountered an unanticipated challenge to identifying overlooked FPOW benefits—veterans’ medical conditions that are listed by the VHA as being service-connected in the Computerized Patient Record System did not always reflect those listed officially in VBA records. This led to occasional identification of apparently overlooked FPOW presumptives that were already recognized by the VBA but not reflected in VHA records. This issue was addressed by ensuring that VBA representatives attended postclinic meetings with clinic staff and avoided the need to pursue supposedly unrecognized benefits that were recognized.
Telehealth
At present, FPOWs from World War II outnumber those of all other conflicts; however, this group is rapidly dwindling in numbers. World War II FPOWs are aged > 85 years, and therefore among the most frail and dependent of veterans. Often they are homebound and unable to physically travel to clinics for assessment. To serve these veterans, we are modifying the FPOW Clinic to utilize telehealth. The Telehealth FPOW Clinic will obtain relevant data from review of the electronic health record and telehealth-based clinic visits. Telehealth also may be used for assessments of Vietnam War veterans (eg, Agent Orange exposure), atomic veterans, and Gulf War veterans. Once fully designed and implemented, we believe that telehealth will prove to be a cost-effective way to provide clinic benefits to rural and older veterans.
Conclusions
The VHA provides priority medical treatment to FPOWs as well as timely and appropriate assessment of their eligibility for veterans’ benefits. The complexities benefit programs established for FPOWs is often beyond the ken of VHA physicians, social workers, and nurses. Because of this unfamiliarity, referrals of eligible FPOWs to appropriate programs can be overlooked. We established a clinic-based interdisciplinary team (FPOW Clinic) that was fully trained in FPOW benefit programs to identify overlooked benefits for FPOWs and were able to increase the disability rating on approximately one-third of the FPOWs seen in the FPOW Clinic. A telehealth-based version of the FPOW clinic is now being developed.
1. Henning CA; Congressional Research Service. POWs and MIAs: status and accounting issues. https://fas.org/sgp/crs/natsec/RL33452.pdf. Published June 1, 2006. Accessed March 16, 2020.
2. Klein RE, Wells MR, Somers JM. American Prisoners of War (POWs) and Missing in Action (MIAs). Washington, DC: US Department of Veterans Affairs, Office of Policy, Planning, and Preparedness; 2006.
3. Skelton WP 3rd. American ex-prisoners of war. https://m.vfwilserviceoffice.com/upload/VA%20Report%20on%20Former%20POWs.pdf. Updated April 2002. Accessed March 16, 2020.
4. Creasey H, Sulway MR, Dent O, Broe GA, Jorm A, Tennant C. Is experience as a prisoner of war a risk factor for accelerated age-related illness and disability? J Am Geriatr Soc. 1999;47(1):60-64.
5. Meziab O, Kirby KA, Williams B, Yaffe K, Byers AL, Barnes DE. Prisoner of war status, posttraumatic stress disorder, and dementia in older veterans. Alzheimers Dement. 2014;10(3)(suppl):S236-S241.
6. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1650. Special Care and Benefits Teams Evaluating or Treating Former Prisoners of War. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=7481. Published July 31, 2018. Accessed March 16, 2020.
7. Boult C, Boult LB, Morishita L, Dowd B, Kane RL, Urdangarin CF. A randomized clinical trial of outpatient geriatric evaluation and management. J Am Geriatr Soc. 2001;49(4):351-359.
8. Kellogg, DL Jr. Geriatric Research, Education and Clinical Center (GRECC): former prisoners of war (FPOW) clinic, methods, procedures & training manual. https://www.southtexas.va.gov/grecc/docs/FPOW_toolkit.pdf. Updated January 28, 2015. Accessed March 16, 2020.
Since the beginning of the American Republic, servicemen have been captured and held as prisoners of war (POWs), including > 130,000 in World War II , > 7,100 in the Korean War, > 700 in the Vietnam War, and 37 in Operation Desert Storm and recent conflicts.1,2 Also, > 80 servicewomen have been held during these conflicts.1-3 Of those living former POWs (FPOWs), almost all are geriatric (aged > 65 years) with a significant portion aged ≥ 85 years.
The physical hardships and psychological stress endured by FPOWs have lifelong deleterious sequelae on health and social functioning.3-5 The experiences of FPOWs are associated with higher prevalence of chronic diseases and diminished functional performance in later life as demonstrated by a survey of FPOWs from World War II.4 The survey assessed health and functional status in a random sample of 101 FPOWs and a group of 107 non-POW combatants from the same military operations. FPOWs reported a higher mean number of somatic symptoms than did non-POWs (7.2 vs 5.4, respectively; P = .002), a higher mean number of diagnosed health conditions (9.4 vs 7.7, respectively; P < .001), and used a greater mean number of medications (4.5 vs 3.4, respectively; P = .001). Among 15 broad categories of diagnoses, differences were found in gastrointestinal disorders (FPOWs 63% vs non-POWs 49%, P = .032), musculoskeletal disorders (FPOWs 76% vs non-POWs 60%, P = .001), and cognitive disorders (FPOWs 31% vs non-POWs 15%, P = .006). FPOWs had a significantly higher proportion of 7 extrapyramidal signs and 6 signs relating to ataxia. On the Instrumental Activities of Daily Living scale, FPOWs were more likely to be impaired than were non-POWs (33% vs 17%, respectively; P = .01). In addition, FPOWs have an increased risk of developing dementia, and this risk is doubled in FPOWs with posttraumatic stress disorder (PTSD) compared with non-FPOWs without PTSD.5
These data indicate that FPOW status is associated with increased risk of disability and loss of independence. Federal statutes established the presumption of a relationship between FPOW status and many comorbidities for VA disability determinations in recognition of such data and to overcome lack of medical records during POW confinement and to accord benefit of the doubt where medical science cannot conclusively link disease etiology to FPOW status, to FPOWs.
Service-Connected Conditions
The historical development of conditions with a presumption of service connection for adjudication of VA compensation/disability claims began in 1921 with the Act to Establish a Veterans’ Bureau and to Improve the Facilities.1 The act simplified and streamlined the claims adjudication process by eliminating the need to obtain evidence on the part of the veteran. The presumption of service connection also facilitated increased accuracy and consistency in adjudications by requiring similar treatment for similar claims. This “presumptive” process relieved claimants and VA of the necessity of producing direct evidence when it was impractical to do so.
In 1970, the first presumptives specific to FPOWs were legislatively established and covered 17 diseases for a FPOW who had been confined for ≥ 30 days (Pub. L. 91-376). The 30-day confinement requirement was later relaxed, and additional presumptives were established that related to diseases that were more common among FPOWs than they were among non-FPOWs. These disorders included traumatic arthritis, stroke, heart disease, osteoporosis, peripheral neuropathy, cold injuries, as well as a variety of digestive and neuropsychiatric disorders. If a FPOW is diagnosed as having ≥ 1 of these conditions and it is judged to be ≥ 10% disabling, the condition is presumed to be a sequelae of the POW experience, and it is classified as a service-connected disability (Table).
FPOW Care And Benefits Teams
Several Veterans Health Administration (VHA) directives have been issued, including the recent VHA directive 1650, which requires that each VHA medical facility have a special Care and Benefits Team (CBT) that is charged with the evaluation and treatment of FPOWs to ensure that “FPOWs receive the highest quality care and benefit services.”6 CBTs must be composed of a clinician trained in internal medicine or family practice; a clinician who is certified through the VA Office of Disability and Medical Assessment to conduct General Medical Compensation and Pension evaluations; a FPOW advocate who typically is a VHA clinical social worker; and a Veterans Benefits Administration (VBA) FPOW coordinator appointed by the local VBA regional office. CBTs can be expanded to include other members as needed. The CBTs are tasked with facilitating interactions between FPOWs, the VHA, and the VBA.
CBTs face several challenges in meeting their responsibilities. For example, the POW experience often results in psychological trauma that foments denial and distrust; hence, thoughtful sensitivity to the sequelae of captivity when approaching FPOWs about personal issues, such as health care, is required. Establishing trusting relationships with FPOWs is necessary if their needs are to be effectively addressed.
While the VHA is mandated to provide priority treatment for FPOWs, including hospital, nursing home, dental, and outpatient treatment, a significant number of FPOWs do not avail themselves of benefits to which they are entitled. Often these FPOWs have not used VA programs and facilities because they are uninformed or confused about VA benefits for FPOWs. As a result, referrals of eligible FPOWs to appropriate programs can be overlooked. Maximizing the service-connected disability rating of FPOWs not only impacts the disability pensions received by these veterans, but also impacts their eligibility for VHA programs, including long-term care and Dependency and Indemnity Compensation, a monthly benefit paid to spouses, children, and/or surviving parents.
In 2013, the FPOW Committee of the South Texas Veterans Health Care System (STVHCS) noted that 40% of FPOWs in our region had no VA primary care or clinic assignment. In consideration of the commitment of the VA to care for FPOWs, the unique POW-related medical and psychological issues, the geriatric age of many FPOWs, and the surprising number of FPOWs currently not receiving VA care, we expanded the concept of the CBT team to create a specialized interdisciplinary FPOW Clinic to address the unique needs of this predominantly elderly population and to involve more FPOWs in the VA system.
The main purpose of this clinic was to advise FPOWs of all VA benefits and services to which they may be entitled by identifying overlooked FPOW presumptives. As the number of FPOWs continues to decrease, outreach to FPOWs and family members has become critical, especially as increased benefits and special services might be available to this increasingly dependent older population. An informal survey of FPOW advocates across the nation found that 21% of FPOWs had disability ratings from the VA of ≤ 60%, including some who had no VA disability rating at all. Thus, an additional goal of the project was to develop a clinic model that could be disseminated throughout the VHA.
Design
The design of the FPOW Clinic team is based on an interdisciplinary model that has proven successful in geriatric medicine.7 The team comprises a physician, a social worker, and a registered nurse.8 All members have expertise in geriatric medicine and specific training in FPOW-related issues by completing a VA employee education training session on FPOW case management. Completion of this training ensured that team members were:
- Familiar with the experiences of FPOWs as well as about the medical, psychosocial, and mental health conditions that affect FPOWs;
- Knowledgeable about FPOW presumptive conditions;
- Familiar with the VBA process for rating FPOW disability claims; and
- Capable of FPOW case coordination, workflow, and communications between the FPOW Clinic team and the VBA to avail FPOWs and their families of all eligible benefits.
In-person FPOW clinic visits and chart reviews helped identify overlooked FPOW benefits. To facilitate case management, a representative of the VBA attended the initial evaluation of each FPOW in the clinic to confirm any overlooked presumptive benefits and to familiarize FPOWs with the claims process. FPOWs were also given the choice to officially enroll in the FPOW clinic for primary care or to remain with their current health care provider. Special efforts were made to enroll those FPOWs who had no STVHCS assigned primary care clinic.
The clinic was scheduled for 4 hours every week. Initial patient visits were 2 hours each and consisted of separate evaluations by each of the 3 FPOW Clinic team members who then met as a team with the addition of the VBA representative. The purpose of this meeting was to discuss overlooked benefits, address any other specific issues noted, and to devise an appropriate interdisciplinary plan. Findings of overlooked benefits and other relevant outcomes then were conveyed to the FPOW. For FPOWs who opted to continue in the clinic for their primary care, subsequent appointments were 1 hour.
Implementation
STVHCS FPOW advocates identified and sent letters to FPOWs announcing the opening of the clinic and its goals. Phone calls were made to each FPOW to address questions and to ascertain their interest. The FPOW advocates then worked directly with schedulers to make clinic appointments. Forty-one FPOWs responded to this initial invitation and attended the new clinic. Subsequently, this number increased through FPOW consults placed by STVHCS primary care providers.
The service-connected disability rating of clinic patients ranged from none (6% of attendees) to 100% (28% of attendees). For 34% of patients, clinic attendance resulted in identification application for overlooked presumptives. VBA evaluation resulted in increased service-connected disability ratings for nearly one-third of clinic patients. All clinic patients without a service-connected disability prior to FPOW clinic evaluation received an increased service-connected disability rating. Overall, 60% of the FPOWs who attended the clinic opted to receive their primary care at the FPOW clinic.
The FPOW Clinic successfully identified overlooked presumptives and facilitated the determination of appropriate service-connected disabilities. Interestingly, the FPOW Clinic encountered an unanticipated challenge to identifying overlooked FPOW benefits—veterans’ medical conditions that are listed by the VHA as being service-connected in the Computerized Patient Record System did not always reflect those listed officially in VBA records. This led to occasional identification of apparently overlooked FPOW presumptives that were already recognized by the VBA but not reflected in VHA records. This issue was addressed by ensuring that VBA representatives attended postclinic meetings with clinic staff and avoided the need to pursue supposedly unrecognized benefits that were recognized.
Telehealth
At present, FPOWs from World War II outnumber those of all other conflicts; however, this group is rapidly dwindling in numbers. World War II FPOWs are aged > 85 years, and therefore among the most frail and dependent of veterans. Often they are homebound and unable to physically travel to clinics for assessment. To serve these veterans, we are modifying the FPOW Clinic to utilize telehealth. The Telehealth FPOW Clinic will obtain relevant data from review of the electronic health record and telehealth-based clinic visits. Telehealth also may be used for assessments of Vietnam War veterans (eg, Agent Orange exposure), atomic veterans, and Gulf War veterans. Once fully designed and implemented, we believe that telehealth will prove to be a cost-effective way to provide clinic benefits to rural and older veterans.
Conclusions
The VHA provides priority medical treatment to FPOWs as well as timely and appropriate assessment of their eligibility for veterans’ benefits. The complexities benefit programs established for FPOWs is often beyond the ken of VHA physicians, social workers, and nurses. Because of this unfamiliarity, referrals of eligible FPOWs to appropriate programs can be overlooked. We established a clinic-based interdisciplinary team (FPOW Clinic) that was fully trained in FPOW benefit programs to identify overlooked benefits for FPOWs and were able to increase the disability rating on approximately one-third of the FPOWs seen in the FPOW Clinic. A telehealth-based version of the FPOW clinic is now being developed.
Since the beginning of the American Republic, servicemen have been captured and held as prisoners of war (POWs), including > 130,000 in World War II , > 7,100 in the Korean War, > 700 in the Vietnam War, and 37 in Operation Desert Storm and recent conflicts.1,2 Also, > 80 servicewomen have been held during these conflicts.1-3 Of those living former POWs (FPOWs), almost all are geriatric (aged > 65 years) with a significant portion aged ≥ 85 years.
The physical hardships and psychological stress endured by FPOWs have lifelong deleterious sequelae on health and social functioning.3-5 The experiences of FPOWs are associated with higher prevalence of chronic diseases and diminished functional performance in later life as demonstrated by a survey of FPOWs from World War II.4 The survey assessed health and functional status in a random sample of 101 FPOWs and a group of 107 non-POW combatants from the same military operations. FPOWs reported a higher mean number of somatic symptoms than did non-POWs (7.2 vs 5.4, respectively; P = .002), a higher mean number of diagnosed health conditions (9.4 vs 7.7, respectively; P < .001), and used a greater mean number of medications (4.5 vs 3.4, respectively; P = .001). Among 15 broad categories of diagnoses, differences were found in gastrointestinal disorders (FPOWs 63% vs non-POWs 49%, P = .032), musculoskeletal disorders (FPOWs 76% vs non-POWs 60%, P = .001), and cognitive disorders (FPOWs 31% vs non-POWs 15%, P = .006). FPOWs had a significantly higher proportion of 7 extrapyramidal signs and 6 signs relating to ataxia. On the Instrumental Activities of Daily Living scale, FPOWs were more likely to be impaired than were non-POWs (33% vs 17%, respectively; P = .01). In addition, FPOWs have an increased risk of developing dementia, and this risk is doubled in FPOWs with posttraumatic stress disorder (PTSD) compared with non-FPOWs without PTSD.5
These data indicate that FPOW status is associated with increased risk of disability and loss of independence. Federal statutes established the presumption of a relationship between FPOW status and many comorbidities for VA disability determinations in recognition of such data and to overcome lack of medical records during POW confinement and to accord benefit of the doubt where medical science cannot conclusively link disease etiology to FPOW status, to FPOWs.
Service-Connected Conditions
The historical development of conditions with a presumption of service connection for adjudication of VA compensation/disability claims began in 1921 with the Act to Establish a Veterans’ Bureau and to Improve the Facilities.1 The act simplified and streamlined the claims adjudication process by eliminating the need to obtain evidence on the part of the veteran. The presumption of service connection also facilitated increased accuracy and consistency in adjudications by requiring similar treatment for similar claims. This “presumptive” process relieved claimants and VA of the necessity of producing direct evidence when it was impractical to do so.
In 1970, the first presumptives specific to FPOWs were legislatively established and covered 17 diseases for a FPOW who had been confined for ≥ 30 days (Pub. L. 91-376). The 30-day confinement requirement was later relaxed, and additional presumptives were established that related to diseases that were more common among FPOWs than they were among non-FPOWs. These disorders included traumatic arthritis, stroke, heart disease, osteoporosis, peripheral neuropathy, cold injuries, as well as a variety of digestive and neuropsychiatric disorders. If a FPOW is diagnosed as having ≥ 1 of these conditions and it is judged to be ≥ 10% disabling, the condition is presumed to be a sequelae of the POW experience, and it is classified as a service-connected disability (Table).
FPOW Care And Benefits Teams
Several Veterans Health Administration (VHA) directives have been issued, including the recent VHA directive 1650, which requires that each VHA medical facility have a special Care and Benefits Team (CBT) that is charged with the evaluation and treatment of FPOWs to ensure that “FPOWs receive the highest quality care and benefit services.”6 CBTs must be composed of a clinician trained in internal medicine or family practice; a clinician who is certified through the VA Office of Disability and Medical Assessment to conduct General Medical Compensation and Pension evaluations; a FPOW advocate who typically is a VHA clinical social worker; and a Veterans Benefits Administration (VBA) FPOW coordinator appointed by the local VBA regional office. CBTs can be expanded to include other members as needed. The CBTs are tasked with facilitating interactions between FPOWs, the VHA, and the VBA.
CBTs face several challenges in meeting their responsibilities. For example, the POW experience often results in psychological trauma that foments denial and distrust; hence, thoughtful sensitivity to the sequelae of captivity when approaching FPOWs about personal issues, such as health care, is required. Establishing trusting relationships with FPOWs is necessary if their needs are to be effectively addressed.
While the VHA is mandated to provide priority treatment for FPOWs, including hospital, nursing home, dental, and outpatient treatment, a significant number of FPOWs do not avail themselves of benefits to which they are entitled. Often these FPOWs have not used VA programs and facilities because they are uninformed or confused about VA benefits for FPOWs. As a result, referrals of eligible FPOWs to appropriate programs can be overlooked. Maximizing the service-connected disability rating of FPOWs not only impacts the disability pensions received by these veterans, but also impacts their eligibility for VHA programs, including long-term care and Dependency and Indemnity Compensation, a monthly benefit paid to spouses, children, and/or surviving parents.
In 2013, the FPOW Committee of the South Texas Veterans Health Care System (STVHCS) noted that 40% of FPOWs in our region had no VA primary care or clinic assignment. In consideration of the commitment of the VA to care for FPOWs, the unique POW-related medical and psychological issues, the geriatric age of many FPOWs, and the surprising number of FPOWs currently not receiving VA care, we expanded the concept of the CBT team to create a specialized interdisciplinary FPOW Clinic to address the unique needs of this predominantly elderly population and to involve more FPOWs in the VA system.
The main purpose of this clinic was to advise FPOWs of all VA benefits and services to which they may be entitled by identifying overlooked FPOW presumptives. As the number of FPOWs continues to decrease, outreach to FPOWs and family members has become critical, especially as increased benefits and special services might be available to this increasingly dependent older population. An informal survey of FPOW advocates across the nation found that 21% of FPOWs had disability ratings from the VA of ≤ 60%, including some who had no VA disability rating at all. Thus, an additional goal of the project was to develop a clinic model that could be disseminated throughout the VHA.
Design
The design of the FPOW Clinic team is based on an interdisciplinary model that has proven successful in geriatric medicine.7 The team comprises a physician, a social worker, and a registered nurse.8 All members have expertise in geriatric medicine and specific training in FPOW-related issues by completing a VA employee education training session on FPOW case management. Completion of this training ensured that team members were:
- Familiar with the experiences of FPOWs as well as about the medical, psychosocial, and mental health conditions that affect FPOWs;
- Knowledgeable about FPOW presumptive conditions;
- Familiar with the VBA process for rating FPOW disability claims; and
- Capable of FPOW case coordination, workflow, and communications between the FPOW Clinic team and the VBA to avail FPOWs and their families of all eligible benefits.
In-person FPOW clinic visits and chart reviews helped identify overlooked FPOW benefits. To facilitate case management, a representative of the VBA attended the initial evaluation of each FPOW in the clinic to confirm any overlooked presumptive benefits and to familiarize FPOWs with the claims process. FPOWs were also given the choice to officially enroll in the FPOW clinic for primary care or to remain with their current health care provider. Special efforts were made to enroll those FPOWs who had no STVHCS assigned primary care clinic.
The clinic was scheduled for 4 hours every week. Initial patient visits were 2 hours each and consisted of separate evaluations by each of the 3 FPOW Clinic team members who then met as a team with the addition of the VBA representative. The purpose of this meeting was to discuss overlooked benefits, address any other specific issues noted, and to devise an appropriate interdisciplinary plan. Findings of overlooked benefits and other relevant outcomes then were conveyed to the FPOW. For FPOWs who opted to continue in the clinic for their primary care, subsequent appointments were 1 hour.
Implementation
STVHCS FPOW advocates identified and sent letters to FPOWs announcing the opening of the clinic and its goals. Phone calls were made to each FPOW to address questions and to ascertain their interest. The FPOW advocates then worked directly with schedulers to make clinic appointments. Forty-one FPOWs responded to this initial invitation and attended the new clinic. Subsequently, this number increased through FPOW consults placed by STVHCS primary care providers.
The service-connected disability rating of clinic patients ranged from none (6% of attendees) to 100% (28% of attendees). For 34% of patients, clinic attendance resulted in identification application for overlooked presumptives. VBA evaluation resulted in increased service-connected disability ratings for nearly one-third of clinic patients. All clinic patients without a service-connected disability prior to FPOW clinic evaluation received an increased service-connected disability rating. Overall, 60% of the FPOWs who attended the clinic opted to receive their primary care at the FPOW clinic.
The FPOW Clinic successfully identified overlooked presumptives and facilitated the determination of appropriate service-connected disabilities. Interestingly, the FPOW Clinic encountered an unanticipated challenge to identifying overlooked FPOW benefits—veterans’ medical conditions that are listed by the VHA as being service-connected in the Computerized Patient Record System did not always reflect those listed officially in VBA records. This led to occasional identification of apparently overlooked FPOW presumptives that were already recognized by the VBA but not reflected in VHA records. This issue was addressed by ensuring that VBA representatives attended postclinic meetings with clinic staff and avoided the need to pursue supposedly unrecognized benefits that were recognized.
Telehealth
At present, FPOWs from World War II outnumber those of all other conflicts; however, this group is rapidly dwindling in numbers. World War II FPOWs are aged > 85 years, and therefore among the most frail and dependent of veterans. Often they are homebound and unable to physically travel to clinics for assessment. To serve these veterans, we are modifying the FPOW Clinic to utilize telehealth. The Telehealth FPOW Clinic will obtain relevant data from review of the electronic health record and telehealth-based clinic visits. Telehealth also may be used for assessments of Vietnam War veterans (eg, Agent Orange exposure), atomic veterans, and Gulf War veterans. Once fully designed and implemented, we believe that telehealth will prove to be a cost-effective way to provide clinic benefits to rural and older veterans.
Conclusions
The VHA provides priority medical treatment to FPOWs as well as timely and appropriate assessment of their eligibility for veterans’ benefits. The complexities benefit programs established for FPOWs is often beyond the ken of VHA physicians, social workers, and nurses. Because of this unfamiliarity, referrals of eligible FPOWs to appropriate programs can be overlooked. We established a clinic-based interdisciplinary team (FPOW Clinic) that was fully trained in FPOW benefit programs to identify overlooked benefits for FPOWs and were able to increase the disability rating on approximately one-third of the FPOWs seen in the FPOW Clinic. A telehealth-based version of the FPOW clinic is now being developed.
1. Henning CA; Congressional Research Service. POWs and MIAs: status and accounting issues. https://fas.org/sgp/crs/natsec/RL33452.pdf. Published June 1, 2006. Accessed March 16, 2020.
2. Klein RE, Wells MR, Somers JM. American Prisoners of War (POWs) and Missing in Action (MIAs). Washington, DC: US Department of Veterans Affairs, Office of Policy, Planning, and Preparedness; 2006.
3. Skelton WP 3rd. American ex-prisoners of war. https://m.vfwilserviceoffice.com/upload/VA%20Report%20on%20Former%20POWs.pdf. Updated April 2002. Accessed March 16, 2020.
4. Creasey H, Sulway MR, Dent O, Broe GA, Jorm A, Tennant C. Is experience as a prisoner of war a risk factor for accelerated age-related illness and disability? J Am Geriatr Soc. 1999;47(1):60-64.
5. Meziab O, Kirby KA, Williams B, Yaffe K, Byers AL, Barnes DE. Prisoner of war status, posttraumatic stress disorder, and dementia in older veterans. Alzheimers Dement. 2014;10(3)(suppl):S236-S241.
6. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1650. Special Care and Benefits Teams Evaluating or Treating Former Prisoners of War. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=7481. Published July 31, 2018. Accessed March 16, 2020.
7. Boult C, Boult LB, Morishita L, Dowd B, Kane RL, Urdangarin CF. A randomized clinical trial of outpatient geriatric evaluation and management. J Am Geriatr Soc. 2001;49(4):351-359.
8. Kellogg, DL Jr. Geriatric Research, Education and Clinical Center (GRECC): former prisoners of war (FPOW) clinic, methods, procedures & training manual. https://www.southtexas.va.gov/grecc/docs/FPOW_toolkit.pdf. Updated January 28, 2015. Accessed March 16, 2020.
1. Henning CA; Congressional Research Service. POWs and MIAs: status and accounting issues. https://fas.org/sgp/crs/natsec/RL33452.pdf. Published June 1, 2006. Accessed March 16, 2020.
2. Klein RE, Wells MR, Somers JM. American Prisoners of War (POWs) and Missing in Action (MIAs). Washington, DC: US Department of Veterans Affairs, Office of Policy, Planning, and Preparedness; 2006.
3. Skelton WP 3rd. American ex-prisoners of war. https://m.vfwilserviceoffice.com/upload/VA%20Report%20on%20Former%20POWs.pdf. Updated April 2002. Accessed March 16, 2020.
4. Creasey H, Sulway MR, Dent O, Broe GA, Jorm A, Tennant C. Is experience as a prisoner of war a risk factor for accelerated age-related illness and disability? J Am Geriatr Soc. 1999;47(1):60-64.
5. Meziab O, Kirby KA, Williams B, Yaffe K, Byers AL, Barnes DE. Prisoner of war status, posttraumatic stress disorder, and dementia in older veterans. Alzheimers Dement. 2014;10(3)(suppl):S236-S241.
6. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1650. Special Care and Benefits Teams Evaluating or Treating Former Prisoners of War. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=7481. Published July 31, 2018. Accessed March 16, 2020.
7. Boult C, Boult LB, Morishita L, Dowd B, Kane RL, Urdangarin CF. A randomized clinical trial of outpatient geriatric evaluation and management. J Am Geriatr Soc. 2001;49(4):351-359.
8. Kellogg, DL Jr. Geriatric Research, Education and Clinical Center (GRECC): former prisoners of war (FPOW) clinic, methods, procedures & training manual. https://www.southtexas.va.gov/grecc/docs/FPOW_toolkit.pdf. Updated January 28, 2015. Accessed March 16, 2020.
Treatment for RA, SpA may not affect COVID-19 severity
Patients being treated for RA or spondyloarthritis who develop symptoms of COVID-19 do not appear to be at higher risk of respiratory or life-threatening complications, results from a new study in Italy suggest.
Such patients, the study authors wrote, do not need to be taken off their immunosuppressive medications if they develop COVID-19 symptoms.
In a letter published in Annals of the Rheumatic Diseases, Sara Monti, MD, and colleagues in the rheumatology department of the Fondazione IRCCS Policlinico in San Matteo, Italy, described results from an observational cohort of 320 patients (68% women; mean age, 55 years) with RA or spondyloarthritis from a single outpatient clinic. The vast majority of subjects (92%) were taking biologic disease-modifying antirheumatic drugs (bDMARD), including tumor necrosis factor inhibitors, while the rest were taking targeted synthetic DMARDs (tsDMARD).
Four patients in the cohort developed laboratory-confirmed COVID-19; another four developed symptoms highly suggestive of the disease but did not receive confirmatory testing, and five had contact with a confirmed COVID-19 case but did not develop symptoms of COVID-19.
Among the eight confirmed and suspected COVID-19 patients, only one was hospitalized. All temporarily withdrew bDMARD or tsDMARD treatment at symptom onset.
“To date, there have been no significant relapses of the rheumatic disease,” Dr. Monti and colleagues reported. “None of the patients with a confirmed diagnosis of COVID-19 or with a highly suggestive clinical picture developed severe respiratory complications or died. Only one patient, aged 65, required admission to hospital and low-flow oxygen supplementation for a few days.”
The findings “do not allow any conclusions on the incidence rate of SARS-CoV-2 infection in patients with rheumatic diseases, nor on the overall outcome of immunocompromised patients affected by COVID-19,” the investigators cautioned, adding that such patients should receive careful attention and follow-up. “However, our preliminary experience shows that patients with chronic arthritis treated with bDMARDs or tsDMARDs do not seem to be at increased risk of respiratory or life-threatening complications from SARS-CoV-2, compared with the general population.”
Dr. Monti and colleagues noted that, during previous outbreaks of other coronaviruses, no increased mortality was reported for people taking immunosuppressive drugs for a range of conditions, including autoimmune diseases.
“These data can support rheumatologists [in] avoiding the unjustifiable preventive withdrawal of DMARDs, which could lead to an increased risk of relapses and morbidity from the chronic rheumatological condition,” the researchers concluded.
Dr. Monti and colleagues reported no outside funding or financial conflicts of interest.
SOURCE: Monti S et al. Ann Rheum Dis. 2020 April 2. doi: 10.1136/annrheumdis-2020-217424.
Patients being treated for RA or spondyloarthritis who develop symptoms of COVID-19 do not appear to be at higher risk of respiratory or life-threatening complications, results from a new study in Italy suggest.
Such patients, the study authors wrote, do not need to be taken off their immunosuppressive medications if they develop COVID-19 symptoms.
In a letter published in Annals of the Rheumatic Diseases, Sara Monti, MD, and colleagues in the rheumatology department of the Fondazione IRCCS Policlinico in San Matteo, Italy, described results from an observational cohort of 320 patients (68% women; mean age, 55 years) with RA or spondyloarthritis from a single outpatient clinic. The vast majority of subjects (92%) were taking biologic disease-modifying antirheumatic drugs (bDMARD), including tumor necrosis factor inhibitors, while the rest were taking targeted synthetic DMARDs (tsDMARD).
Four patients in the cohort developed laboratory-confirmed COVID-19; another four developed symptoms highly suggestive of the disease but did not receive confirmatory testing, and five had contact with a confirmed COVID-19 case but did not develop symptoms of COVID-19.
Among the eight confirmed and suspected COVID-19 patients, only one was hospitalized. All temporarily withdrew bDMARD or tsDMARD treatment at symptom onset.
“To date, there have been no significant relapses of the rheumatic disease,” Dr. Monti and colleagues reported. “None of the patients with a confirmed diagnosis of COVID-19 or with a highly suggestive clinical picture developed severe respiratory complications or died. Only one patient, aged 65, required admission to hospital and low-flow oxygen supplementation for a few days.”
The findings “do not allow any conclusions on the incidence rate of SARS-CoV-2 infection in patients with rheumatic diseases, nor on the overall outcome of immunocompromised patients affected by COVID-19,” the investigators cautioned, adding that such patients should receive careful attention and follow-up. “However, our preliminary experience shows that patients with chronic arthritis treated with bDMARDs or tsDMARDs do not seem to be at increased risk of respiratory or life-threatening complications from SARS-CoV-2, compared with the general population.”
Dr. Monti and colleagues noted that, during previous outbreaks of other coronaviruses, no increased mortality was reported for people taking immunosuppressive drugs for a range of conditions, including autoimmune diseases.
“These data can support rheumatologists [in] avoiding the unjustifiable preventive withdrawal of DMARDs, which could lead to an increased risk of relapses and morbidity from the chronic rheumatological condition,” the researchers concluded.
Dr. Monti and colleagues reported no outside funding or financial conflicts of interest.
SOURCE: Monti S et al. Ann Rheum Dis. 2020 April 2. doi: 10.1136/annrheumdis-2020-217424.
Patients being treated for RA or spondyloarthritis who develop symptoms of COVID-19 do not appear to be at higher risk of respiratory or life-threatening complications, results from a new study in Italy suggest.
Such patients, the study authors wrote, do not need to be taken off their immunosuppressive medications if they develop COVID-19 symptoms.
In a letter published in Annals of the Rheumatic Diseases, Sara Monti, MD, and colleagues in the rheumatology department of the Fondazione IRCCS Policlinico in San Matteo, Italy, described results from an observational cohort of 320 patients (68% women; mean age, 55 years) with RA or spondyloarthritis from a single outpatient clinic. The vast majority of subjects (92%) were taking biologic disease-modifying antirheumatic drugs (bDMARD), including tumor necrosis factor inhibitors, while the rest were taking targeted synthetic DMARDs (tsDMARD).
Four patients in the cohort developed laboratory-confirmed COVID-19; another four developed symptoms highly suggestive of the disease but did not receive confirmatory testing, and five had contact with a confirmed COVID-19 case but did not develop symptoms of COVID-19.
Among the eight confirmed and suspected COVID-19 patients, only one was hospitalized. All temporarily withdrew bDMARD or tsDMARD treatment at symptom onset.
“To date, there have been no significant relapses of the rheumatic disease,” Dr. Monti and colleagues reported. “None of the patients with a confirmed diagnosis of COVID-19 or with a highly suggestive clinical picture developed severe respiratory complications or died. Only one patient, aged 65, required admission to hospital and low-flow oxygen supplementation for a few days.”
The findings “do not allow any conclusions on the incidence rate of SARS-CoV-2 infection in patients with rheumatic diseases, nor on the overall outcome of immunocompromised patients affected by COVID-19,” the investigators cautioned, adding that such patients should receive careful attention and follow-up. “However, our preliminary experience shows that patients with chronic arthritis treated with bDMARDs or tsDMARDs do not seem to be at increased risk of respiratory or life-threatening complications from SARS-CoV-2, compared with the general population.”
Dr. Monti and colleagues noted that, during previous outbreaks of other coronaviruses, no increased mortality was reported for people taking immunosuppressive drugs for a range of conditions, including autoimmune diseases.
“These data can support rheumatologists [in] avoiding the unjustifiable preventive withdrawal of DMARDs, which could lead to an increased risk of relapses and morbidity from the chronic rheumatological condition,” the researchers concluded.
Dr. Monti and colleagues reported no outside funding or financial conflicts of interest.
SOURCE: Monti S et al. Ann Rheum Dis. 2020 April 2. doi: 10.1136/annrheumdis-2020-217424.
FROM ANNALS OF THE RHEUMATIC DISEASES
Conducting cancer trials amid the COVID-19 pandemic
More than three-quarters of cancer clinical research programs have experienced operational changes during the COVID-19 pandemic, according to a survey conducted by the Association of Community Cancer Centers (ACCC) during a recent webinar.
The webinar included insights into how some cancer research programs have adapted to the pandemic, a review of guidance for conducting cancer trials during this time, and a discussion of how the cancer research landscape may be affected by COVID-19 going forward.
The webinar was led by Randall A. Oyer, MD, president of the ACCC and medical director of the oncology program at Penn Medicine Lancaster General Health in Pennsylvania.
The impact of COVID-19 on cancer research
Dr. Oyer observed that planning and implementation for COVID-19–related illness at U.S. health care institutions has had a predictable effect of limiting patient access and staff availability for nonessential services.
Coronavirus-related exposure and/or illness has relegated cancer research to a lower-level priority. As a result, ACCC institutions have made adjustments in their cancer research programs, including moving clinical research coordinators off-campus and deploying them in clinical areas.
New clinical trials have not been opened. In some cases, new accruals have been halted, particularly for registry, prevention, and symptom control trials.
Standards that have changed and those that have not
Guidance documents for conducting clinical trials during the pandemic have been developed by the Food and Drug Administration, the National Cancer Institute’s Cancer Therapy Evaluation Program and Central Institutional Review Board, and the National Institutes of Health’s Office of Extramural Research. Industry sponsors and parent institutions of research programs have also disseminated guidance.
Among other topics, guidance documents have addressed:
- How COVID-19-related protocol deviations will be judged at monitoring visits and audits
- Missed office visits and endpoint evaluations
- Providing investigational oral medications to patients via mail and potential issues of medication unavailability
- Processes for patients to have interim visits with providers at external institutions, including providers who may not be personally engaged in or credentialed for the research trial
- Potential delays in submitting protocol amendments for institutional review board (IRB) review
- Recommendations for patients confirmed or suspected of having a coronavirus infection.
Dr. Oyer emphasized that patient safety must remain the highest priority for patient management, on or off study. He advised continuing investigational therapy when potential benefit from treatment is anticipated and identifying alternative methods to face-to-face visits for monitoring and access to treatment.
Dr. Oyer urged programs to:
- Maintain good clinical practice standards
- Consult with sponsors and IRBs when questions arise but implement changes that affect patient safety prior to IRB review if necessary
- Document all deviations and COVID-19 related adaptations in a log or spreadsheet in anticipation of future questions from sponsors, monitors, and other entities.
New questions and considerations
In the short-term, Dr. Oyer predicts fewer available trials and a decreased rate of accrual to existing studies. This may result in delays in trial completion and the possibility of redesign for some trials.
He predicts the emergence of COVID-19-focused research questions, including those assessing the course of coronavirus infection in various malignant settings and the impact of cancer-directed treatments and supportive care interventions (e.g., treatment for graft-versus-host disease) on response to COVID-19.
To facilitate developing a clinically and research-relevant database, Dr. Oyer stressed the importance of documentation in the research record, reporting infections as serious adverse events. Documentation should specify whether the infection was confirmed or suspected coronavirus or related to another organism.
In general, when coronavirus infection is strongly suspected, Dr. Oyer said investigational treatments should be interrupted, but study-specific criteria will be forthcoming on that issue.
Looking to the future
For patients with advanced cancers, clinical trials provide an important option for hope and clinical benefit. Disrupting the conduct of clinical trials could endanger the lives of participants and delay the emergence of promising treatments and diagnostic tests.
When the coronavirus pandemic recedes, advancing knowledge and treatments for cancer will demand renewed commitment across the oncology care community.
Going forward, Dr. Oyer advised that clinical research staff protect their own health and the safety of trial participants. He encouraged programs to work with sponsors and IRBs to solve logistical problems and clarify individual issues.
He was optimistic that resumption of more normal conduct of studies will enable the successful completion of ongoing trials, enhanced by the creative solutions that were devised during the crisis and by additional prospective, clinically annotated, carefully recorded data from academic and community research sites.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
More than three-quarters of cancer clinical research programs have experienced operational changes during the COVID-19 pandemic, according to a survey conducted by the Association of Community Cancer Centers (ACCC) during a recent webinar.
The webinar included insights into how some cancer research programs have adapted to the pandemic, a review of guidance for conducting cancer trials during this time, and a discussion of how the cancer research landscape may be affected by COVID-19 going forward.
The webinar was led by Randall A. Oyer, MD, president of the ACCC and medical director of the oncology program at Penn Medicine Lancaster General Health in Pennsylvania.
The impact of COVID-19 on cancer research
Dr. Oyer observed that planning and implementation for COVID-19–related illness at U.S. health care institutions has had a predictable effect of limiting patient access and staff availability for nonessential services.
Coronavirus-related exposure and/or illness has relegated cancer research to a lower-level priority. As a result, ACCC institutions have made adjustments in their cancer research programs, including moving clinical research coordinators off-campus and deploying them in clinical areas.
New clinical trials have not been opened. In some cases, new accruals have been halted, particularly for registry, prevention, and symptom control trials.
Standards that have changed and those that have not
Guidance documents for conducting clinical trials during the pandemic have been developed by the Food and Drug Administration, the National Cancer Institute’s Cancer Therapy Evaluation Program and Central Institutional Review Board, and the National Institutes of Health’s Office of Extramural Research. Industry sponsors and parent institutions of research programs have also disseminated guidance.
Among other topics, guidance documents have addressed:
- How COVID-19-related protocol deviations will be judged at monitoring visits and audits
- Missed office visits and endpoint evaluations
- Providing investigational oral medications to patients via mail and potential issues of medication unavailability
- Processes for patients to have interim visits with providers at external institutions, including providers who may not be personally engaged in or credentialed for the research trial
- Potential delays in submitting protocol amendments for institutional review board (IRB) review
- Recommendations for patients confirmed or suspected of having a coronavirus infection.
Dr. Oyer emphasized that patient safety must remain the highest priority for patient management, on or off study. He advised continuing investigational therapy when potential benefit from treatment is anticipated and identifying alternative methods to face-to-face visits for monitoring and access to treatment.
Dr. Oyer urged programs to:
- Maintain good clinical practice standards
- Consult with sponsors and IRBs when questions arise but implement changes that affect patient safety prior to IRB review if necessary
- Document all deviations and COVID-19 related adaptations in a log or spreadsheet in anticipation of future questions from sponsors, monitors, and other entities.
New questions and considerations
In the short-term, Dr. Oyer predicts fewer available trials and a decreased rate of accrual to existing studies. This may result in delays in trial completion and the possibility of redesign for some trials.
He predicts the emergence of COVID-19-focused research questions, including those assessing the course of coronavirus infection in various malignant settings and the impact of cancer-directed treatments and supportive care interventions (e.g., treatment for graft-versus-host disease) on response to COVID-19.
To facilitate developing a clinically and research-relevant database, Dr. Oyer stressed the importance of documentation in the research record, reporting infections as serious adverse events. Documentation should specify whether the infection was confirmed or suspected coronavirus or related to another organism.
In general, when coronavirus infection is strongly suspected, Dr. Oyer said investigational treatments should be interrupted, but study-specific criteria will be forthcoming on that issue.
Looking to the future
For patients with advanced cancers, clinical trials provide an important option for hope and clinical benefit. Disrupting the conduct of clinical trials could endanger the lives of participants and delay the emergence of promising treatments and diagnostic tests.
When the coronavirus pandemic recedes, advancing knowledge and treatments for cancer will demand renewed commitment across the oncology care community.
Going forward, Dr. Oyer advised that clinical research staff protect their own health and the safety of trial participants. He encouraged programs to work with sponsors and IRBs to solve logistical problems and clarify individual issues.
He was optimistic that resumption of more normal conduct of studies will enable the successful completion of ongoing trials, enhanced by the creative solutions that were devised during the crisis and by additional prospective, clinically annotated, carefully recorded data from academic and community research sites.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
More than three-quarters of cancer clinical research programs have experienced operational changes during the COVID-19 pandemic, according to a survey conducted by the Association of Community Cancer Centers (ACCC) during a recent webinar.
The webinar included insights into how some cancer research programs have adapted to the pandemic, a review of guidance for conducting cancer trials during this time, and a discussion of how the cancer research landscape may be affected by COVID-19 going forward.
The webinar was led by Randall A. Oyer, MD, president of the ACCC and medical director of the oncology program at Penn Medicine Lancaster General Health in Pennsylvania.
The impact of COVID-19 on cancer research
Dr. Oyer observed that planning and implementation for COVID-19–related illness at U.S. health care institutions has had a predictable effect of limiting patient access and staff availability for nonessential services.
Coronavirus-related exposure and/or illness has relegated cancer research to a lower-level priority. As a result, ACCC institutions have made adjustments in their cancer research programs, including moving clinical research coordinators off-campus and deploying them in clinical areas.
New clinical trials have not been opened. In some cases, new accruals have been halted, particularly for registry, prevention, and symptom control trials.
Standards that have changed and those that have not
Guidance documents for conducting clinical trials during the pandemic have been developed by the Food and Drug Administration, the National Cancer Institute’s Cancer Therapy Evaluation Program and Central Institutional Review Board, and the National Institutes of Health’s Office of Extramural Research. Industry sponsors and parent institutions of research programs have also disseminated guidance.
Among other topics, guidance documents have addressed:
- How COVID-19-related protocol deviations will be judged at monitoring visits and audits
- Missed office visits and endpoint evaluations
- Providing investigational oral medications to patients via mail and potential issues of medication unavailability
- Processes for patients to have interim visits with providers at external institutions, including providers who may not be personally engaged in or credentialed for the research trial
- Potential delays in submitting protocol amendments for institutional review board (IRB) review
- Recommendations for patients confirmed or suspected of having a coronavirus infection.
Dr. Oyer emphasized that patient safety must remain the highest priority for patient management, on or off study. He advised continuing investigational therapy when potential benefit from treatment is anticipated and identifying alternative methods to face-to-face visits for monitoring and access to treatment.
Dr. Oyer urged programs to:
- Maintain good clinical practice standards
- Consult with sponsors and IRBs when questions arise but implement changes that affect patient safety prior to IRB review if necessary
- Document all deviations and COVID-19 related adaptations in a log or spreadsheet in anticipation of future questions from sponsors, monitors, and other entities.
New questions and considerations
In the short-term, Dr. Oyer predicts fewer available trials and a decreased rate of accrual to existing studies. This may result in delays in trial completion and the possibility of redesign for some trials.
He predicts the emergence of COVID-19-focused research questions, including those assessing the course of coronavirus infection in various malignant settings and the impact of cancer-directed treatments and supportive care interventions (e.g., treatment for graft-versus-host disease) on response to COVID-19.
To facilitate developing a clinically and research-relevant database, Dr. Oyer stressed the importance of documentation in the research record, reporting infections as serious adverse events. Documentation should specify whether the infection was confirmed or suspected coronavirus or related to another organism.
In general, when coronavirus infection is strongly suspected, Dr. Oyer said investigational treatments should be interrupted, but study-specific criteria will be forthcoming on that issue.
Looking to the future
For patients with advanced cancers, clinical trials provide an important option for hope and clinical benefit. Disrupting the conduct of clinical trials could endanger the lives of participants and delay the emergence of promising treatments and diagnostic tests.
When the coronavirus pandemic recedes, advancing knowledge and treatments for cancer will demand renewed commitment across the oncology care community.
Going forward, Dr. Oyer advised that clinical research staff protect their own health and the safety of trial participants. He encouraged programs to work with sponsors and IRBs to solve logistical problems and clarify individual issues.
He was optimistic that resumption of more normal conduct of studies will enable the successful completion of ongoing trials, enhanced by the creative solutions that were devised during the crisis and by additional prospective, clinically annotated, carefully recorded data from academic and community research sites.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
‘The kids will be all right,’ won’t they?
Pediatric patients and COVID-19
The coronavirus disease 2019 (COVID-19) pandemic affects us in many ways. Pediatric patients, interestingly, are largely unaffected clinically by this disease. Less than 1% of documented infections occur in children under 10 years old, according to a review of over 72,000 cases from China.1 In that review, most children were asymptomatic or had mild illness, only three required intensive care, and only one death had been reported as of March 10, 2020. This is in stark contrast to the shocking morbidity and mortality statistics we are becoming all too familiar with on the adult side.
From a social standpoint, however, our pediatric patients’ lives have been turned upside down. Their schedules and routines upended, their education and friendships interrupted, and many are likely experiencing real anxiety and fear.2 For countless children, school is a major source of social, emotional, and nutritional support that has been cut off. Some will lose parents, grandparents, or other loved ones to this disease. Parents will lose jobs and will be unable to afford necessities. Pediatric patients will experience delays of procedures or treatments because of the pandemic. Some have projected that rates of child abuse will increase as has been reported during natural disasters.3
Pediatricians around the country are coming together to tackle these issues in creative ways, including the rapid expansion of virtual/telehealth programs. The school systems are developing strategies to deliver online content, and even food, to their students’ homes. Hopefully these tactics will mitigate some of the potential effects on the mental and physical well-being of these patients.
How about my kids? Will they be all right? I am lucky that my husband and I will have jobs throughout this ordeal. Unfortunately, given my role as a hospitalist and my husband’s as a pulmonary/critical care physician, these same jobs that will keep our kids nourished and supported pose the greatest threat to them. As health care workers, we are worried about protecting our families, which may include vulnerable members. The Spanish health ministry announced that medical professionals account for approximately one in eight documented COVID-19 infections in Spain.4 With inadequate supplies of personal protective equipment (PPE) in our own nation, we are concerned that our statistics could be similar.
There are multiple strategies to protect ourselves and our families during this difficult time. First, appropriate PPE is essential and integrity with the process must be maintained always. Hospital leaders can protect us by tirelessly working to acquire PPE. In Grand Rapids, Mich., our health system has partnered with multiple local manufacturing companies, including Steelcase, who are producing PPE for our workforce.5 Leaders can diligently update their system’s PPE recommendations to be in line with the latest CDC recommendations and disseminate the information regularly. Hospitalists should frequently check with their Infection Prevention department to make sure they understand if there have been any changes to the recommendations. Innovative solutions for sterilization of PPE, stethoscopes, badges and other equipment, such as with the use of UV boxes or hydrogen peroxide vapor,6 should be explored to minimize contamination. Hospitalists should bring a set of clothes and shoes to change into upon arrival to work and to change out of prior to leaving the hospital.
We must also keep our heads strong. Currently the anxiety amongst physicians is palpable but there is solidarity. Hospital leaders must ensure that hospitalists have easy access to free mental health resources, such as virtual counseling. Wellness teams must rise to the occasion with innovative tactics to support us. For example, Spectrum Health’s wellness team is sponsoring a blog where physicians can discuss COVID-19–related challenges openly. Hospitalist leaders should ensure that there is a structure for debriefing after critical incidents, which are sure to increase in frequency. Email lists and discussion boards sponsored by professional society also provide a collaborative venue for some of these discussions. We must take advantage of these resources and communicate with each other.
For me, in the end it comes back to the kids. My kids and most pediatric patients are not likely to be hospitalized from COVID-19, but they are also not immune to the toll that fighting this pandemic will take on our families. We took an oath to protect our patients, but what do we owe to our own children? At a minimum we can optimize how we protect ourselves every day, both physically and mentally. As we come together as a strong community to fight this pandemic, in addition to saving lives, we are working to ensure that, in the end, the kids will be all right.
Dr. Hadley is chief of pediatric hospital medicine at Spectrum Health/Helen DeVos Children’s Hospital in Grand Rapids, Mich., and clinical assistant professor at Michigan State University, East Lansing.
References
1. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020 Feb 24. doi: 10.1001/jama.2020.2648.
2. Hagan JF Jr; American Academy of Pediatrics Committee on Psychosocial Aspects of Child and Family Health; Task Force on Terrorism. Psychosocial implications of disaster or terrorism on children: A guide for the pediatrician. Pediatrics. 2005;116(3):787-795.
3. Gearhart S et al. The impact of natural disasters on domestic violence: An analysis of reports of simple assault in Florida (1997-2007). Violence Gend. 2018 Jun. doi: 10.1089/vio.2017.0077.
4. Minder R, Peltier E. Virus knocks thousands of health workers out of action in Europe. The New York Times. March 24, 2020.
5. McVicar B. West Michigan businesses hustle to produce medical supplies amid coronavirus pandemic. MLive. March 25, 2020.
6. Kenney PA et al. Hydrogen Peroxide Vapor sterilization of N95 respirators for reuse. medRxiv preprint. 2020 Mar. doi: 10.1101/2020.03.24.20041087.
Pediatric patients and COVID-19
Pediatric patients and COVID-19
The coronavirus disease 2019 (COVID-19) pandemic affects us in many ways. Pediatric patients, interestingly, are largely unaffected clinically by this disease. Less than 1% of documented infections occur in children under 10 years old, according to a review of over 72,000 cases from China.1 In that review, most children were asymptomatic or had mild illness, only three required intensive care, and only one death had been reported as of March 10, 2020. This is in stark contrast to the shocking morbidity and mortality statistics we are becoming all too familiar with on the adult side.
From a social standpoint, however, our pediatric patients’ lives have been turned upside down. Their schedules and routines upended, their education and friendships interrupted, and many are likely experiencing real anxiety and fear.2 For countless children, school is a major source of social, emotional, and nutritional support that has been cut off. Some will lose parents, grandparents, or other loved ones to this disease. Parents will lose jobs and will be unable to afford necessities. Pediatric patients will experience delays of procedures or treatments because of the pandemic. Some have projected that rates of child abuse will increase as has been reported during natural disasters.3
Pediatricians around the country are coming together to tackle these issues in creative ways, including the rapid expansion of virtual/telehealth programs. The school systems are developing strategies to deliver online content, and even food, to their students’ homes. Hopefully these tactics will mitigate some of the potential effects on the mental and physical well-being of these patients.
How about my kids? Will they be all right? I am lucky that my husband and I will have jobs throughout this ordeal. Unfortunately, given my role as a hospitalist and my husband’s as a pulmonary/critical care physician, these same jobs that will keep our kids nourished and supported pose the greatest threat to them. As health care workers, we are worried about protecting our families, which may include vulnerable members. The Spanish health ministry announced that medical professionals account for approximately one in eight documented COVID-19 infections in Spain.4 With inadequate supplies of personal protective equipment (PPE) in our own nation, we are concerned that our statistics could be similar.
There are multiple strategies to protect ourselves and our families during this difficult time. First, appropriate PPE is essential and integrity with the process must be maintained always. Hospital leaders can protect us by tirelessly working to acquire PPE. In Grand Rapids, Mich., our health system has partnered with multiple local manufacturing companies, including Steelcase, who are producing PPE for our workforce.5 Leaders can diligently update their system’s PPE recommendations to be in line with the latest CDC recommendations and disseminate the information regularly. Hospitalists should frequently check with their Infection Prevention department to make sure they understand if there have been any changes to the recommendations. Innovative solutions for sterilization of PPE, stethoscopes, badges and other equipment, such as with the use of UV boxes or hydrogen peroxide vapor,6 should be explored to minimize contamination. Hospitalists should bring a set of clothes and shoes to change into upon arrival to work and to change out of prior to leaving the hospital.
We must also keep our heads strong. Currently the anxiety amongst physicians is palpable but there is solidarity. Hospital leaders must ensure that hospitalists have easy access to free mental health resources, such as virtual counseling. Wellness teams must rise to the occasion with innovative tactics to support us. For example, Spectrum Health’s wellness team is sponsoring a blog where physicians can discuss COVID-19–related challenges openly. Hospitalist leaders should ensure that there is a structure for debriefing after critical incidents, which are sure to increase in frequency. Email lists and discussion boards sponsored by professional society also provide a collaborative venue for some of these discussions. We must take advantage of these resources and communicate with each other.
For me, in the end it comes back to the kids. My kids and most pediatric patients are not likely to be hospitalized from COVID-19, but they are also not immune to the toll that fighting this pandemic will take on our families. We took an oath to protect our patients, but what do we owe to our own children? At a minimum we can optimize how we protect ourselves every day, both physically and mentally. As we come together as a strong community to fight this pandemic, in addition to saving lives, we are working to ensure that, in the end, the kids will be all right.
Dr. Hadley is chief of pediatric hospital medicine at Spectrum Health/Helen DeVos Children’s Hospital in Grand Rapids, Mich., and clinical assistant professor at Michigan State University, East Lansing.
References
1. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020 Feb 24. doi: 10.1001/jama.2020.2648.
2. Hagan JF Jr; American Academy of Pediatrics Committee on Psychosocial Aspects of Child and Family Health; Task Force on Terrorism. Psychosocial implications of disaster or terrorism on children: A guide for the pediatrician. Pediatrics. 2005;116(3):787-795.
3. Gearhart S et al. The impact of natural disasters on domestic violence: An analysis of reports of simple assault in Florida (1997-2007). Violence Gend. 2018 Jun. doi: 10.1089/vio.2017.0077.
4. Minder R, Peltier E. Virus knocks thousands of health workers out of action in Europe. The New York Times. March 24, 2020.
5. McVicar B. West Michigan businesses hustle to produce medical supplies amid coronavirus pandemic. MLive. March 25, 2020.
6. Kenney PA et al. Hydrogen Peroxide Vapor sterilization of N95 respirators for reuse. medRxiv preprint. 2020 Mar. doi: 10.1101/2020.03.24.20041087.
The coronavirus disease 2019 (COVID-19) pandemic affects us in many ways. Pediatric patients, interestingly, are largely unaffected clinically by this disease. Less than 1% of documented infections occur in children under 10 years old, according to a review of over 72,000 cases from China.1 In that review, most children were asymptomatic or had mild illness, only three required intensive care, and only one death had been reported as of March 10, 2020. This is in stark contrast to the shocking morbidity and mortality statistics we are becoming all too familiar with on the adult side.
From a social standpoint, however, our pediatric patients’ lives have been turned upside down. Their schedules and routines upended, their education and friendships interrupted, and many are likely experiencing real anxiety and fear.2 For countless children, school is a major source of social, emotional, and nutritional support that has been cut off. Some will lose parents, grandparents, or other loved ones to this disease. Parents will lose jobs and will be unable to afford necessities. Pediatric patients will experience delays of procedures or treatments because of the pandemic. Some have projected that rates of child abuse will increase as has been reported during natural disasters.3
Pediatricians around the country are coming together to tackle these issues in creative ways, including the rapid expansion of virtual/telehealth programs. The school systems are developing strategies to deliver online content, and even food, to their students’ homes. Hopefully these tactics will mitigate some of the potential effects on the mental and physical well-being of these patients.
How about my kids? Will they be all right? I am lucky that my husband and I will have jobs throughout this ordeal. Unfortunately, given my role as a hospitalist and my husband’s as a pulmonary/critical care physician, these same jobs that will keep our kids nourished and supported pose the greatest threat to them. As health care workers, we are worried about protecting our families, which may include vulnerable members. The Spanish health ministry announced that medical professionals account for approximately one in eight documented COVID-19 infections in Spain.4 With inadequate supplies of personal protective equipment (PPE) in our own nation, we are concerned that our statistics could be similar.
There are multiple strategies to protect ourselves and our families during this difficult time. First, appropriate PPE is essential and integrity with the process must be maintained always. Hospital leaders can protect us by tirelessly working to acquire PPE. In Grand Rapids, Mich., our health system has partnered with multiple local manufacturing companies, including Steelcase, who are producing PPE for our workforce.5 Leaders can diligently update their system’s PPE recommendations to be in line with the latest CDC recommendations and disseminate the information regularly. Hospitalists should frequently check with their Infection Prevention department to make sure they understand if there have been any changes to the recommendations. Innovative solutions for sterilization of PPE, stethoscopes, badges and other equipment, such as with the use of UV boxes or hydrogen peroxide vapor,6 should be explored to minimize contamination. Hospitalists should bring a set of clothes and shoes to change into upon arrival to work and to change out of prior to leaving the hospital.
We must also keep our heads strong. Currently the anxiety amongst physicians is palpable but there is solidarity. Hospital leaders must ensure that hospitalists have easy access to free mental health resources, such as virtual counseling. Wellness teams must rise to the occasion with innovative tactics to support us. For example, Spectrum Health’s wellness team is sponsoring a blog where physicians can discuss COVID-19–related challenges openly. Hospitalist leaders should ensure that there is a structure for debriefing after critical incidents, which are sure to increase in frequency. Email lists and discussion boards sponsored by professional society also provide a collaborative venue for some of these discussions. We must take advantage of these resources and communicate with each other.
For me, in the end it comes back to the kids. My kids and most pediatric patients are not likely to be hospitalized from COVID-19, but they are also not immune to the toll that fighting this pandemic will take on our families. We took an oath to protect our patients, but what do we owe to our own children? At a minimum we can optimize how we protect ourselves every day, both physically and mentally. As we come together as a strong community to fight this pandemic, in addition to saving lives, we are working to ensure that, in the end, the kids will be all right.
Dr. Hadley is chief of pediatric hospital medicine at Spectrum Health/Helen DeVos Children’s Hospital in Grand Rapids, Mich., and clinical assistant professor at Michigan State University, East Lansing.
References
1. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020 Feb 24. doi: 10.1001/jama.2020.2648.
2. Hagan JF Jr; American Academy of Pediatrics Committee on Psychosocial Aspects of Child and Family Health; Task Force on Terrorism. Psychosocial implications of disaster or terrorism on children: A guide for the pediatrician. Pediatrics. 2005;116(3):787-795.
3. Gearhart S et al. The impact of natural disasters on domestic violence: An analysis of reports of simple assault in Florida (1997-2007). Violence Gend. 2018 Jun. doi: 10.1089/vio.2017.0077.
4. Minder R, Peltier E. Virus knocks thousands of health workers out of action in Europe. The New York Times. March 24, 2020.
5. McVicar B. West Michigan businesses hustle to produce medical supplies amid coronavirus pandemic. MLive. March 25, 2020.
6. Kenney PA et al. Hydrogen Peroxide Vapor sterilization of N95 respirators for reuse. medRxiv preprint. 2020 Mar. doi: 10.1101/2020.03.24.20041087.
Hospital Medicine Management in the Time of COVID-19: Preparing for a Sprint and a Marathon
The pandemic of coronavirus disease 2019 (COVID-19) is confronting the modern world like nothing else before. With over 20 million individuals expected to require hospitalization in the US, this health crisis may become a generation-defining moment for healthcare systems and the field of hospital medicine.1 The specific challenges facing hospital medicine are comparable to running a sprint and a marathon—at the same time. For the sprint underway, hospitalists must learn to respond to a rapidly changing environment in which critical decisions are made within hours and days. At the same time, hospitalists need to plan for the marathon of increased clinical needs over the coming months, the possibility of burnout, and concerns about staff well-being. Although runners typically focus on either the sprint or the marathon, healthcare systems and hospital medicine providers will need to simultaneously prepare for both types of races.
GET READY FOR THE SPRINT
Over the past several weeks, hospital medicine leaders have been rapidly responding to an evolving crisis. Leaders and clinicians are quickly learning how to restructure clinical operations, negotiate the short supply of personal protective equipment (PPE), and manage delays in COVID-19 testing. In these areas, our hospitalist group has experienced a steep learning curve. In addition to the strategies outlined in the Table, we will share here our experiences and insights on managing and preparing for the COVID-19 pandemic.
Communication Is Central
During the sprint, focused, regular communication is imperative to ameliorate anxiety and fear. A study of crisis communication after 9/11 found that, for employees, good communication from leadership was one of the most valued factors.2 Communications experts also note that, in times of crisis, leaders have a special role in communication, specifically around demystifying the situation, providing hope, and maintaining transparency.3
Mental bandwidth may be limited in a stressful environment, so efforts should be taken to maximize the value of each communication. Information on hospital metrics should be provided regularly, including the number of COVID-19 cases, the status of clinical services and staffing, hospital capacity, and resource availability.4 Although the ubiquity and ease of email is convenient, recognize that providers are likely receiving email updates from multiple layers within your healthcare organization. To guard against losing important information, we use the same templated format for daily email updates with changes highlighted, which allows busy clinicians to digest pertinent information easily.5 Finally, consider having a single individual be responsible for collating COVID-19–related emails sent to your group. Although clinicians may want to share the most recent studies or their clinical experiences with a group email, instead have them send this information to a single individual who can organize these materials and share them on a regular basis.
To keep two-way communication channels open in a busy, asynchronous environment, consider having a centralized shared document in which providers can give real-time feedback to capture on-the-ground experiences or share questions they would like answered. Within our group, we found that centralizing our conversation in a shared document eliminated redundancy, focused our meetings, and kept everyone up to date. Additionally, regularly scheduled meetings may need to be adapted to a remote format (eg, Zoom, WebEx) as clinicians are asked to work from home when not on clinical service. Finally, recognize that virtual meetings require a different skill set than that required by in-person meetings, including reestablishment of social norms and technology preparation.6
Optimize Your Staffing
Hospital volumes could increase to as high as 270% of current hospital bed capacities during this pandemic.1 This surge is further complicated by the effort involved in caring for these patients, given their increased medical complexity, the use of new protocols, and the extra time needed to update staff and family. As the workload intensifies, staffing models and operations will also need to adapt.
First, optimize your inpatient resources based on the changes your hospital system is making. For instance, as elective surgeries were cancelled, we dissolved our surgical comanagement and consult services to better accommodate our hospitals’ needs. Further, consider using advanced practice providers (eg, physician assistants and nurse practitioners) released from their clinical duties to help with inpatient care in the event of a surge. If your hospital has trainees (eg, residents or fellows), consider reassigning those whose rotations have been postponed to newly created inpatient teams; trainees often have strong institutional knowledge and understanding of hospital protocols and resources.
Second, use hospitalists for their most relevant skills. Hospitalists are pluripotent clinicians who are comfortable with high-acuity patients and can fit into a myriad of clinical positions. The initial instinct at our institution was to mobilize hospitalists across all areas of increasing needs in the hospital (eg, screening clinics,7 advice phone lines for patients, or in the Emergency Department), but we quickly recognized that the hospitalist group is a finite resource. We focused our hospitalists’ clinical work on the expanding inpatient needs and allowed other outpatient or procedure-based specialties that have less inpatient experience to fill the broader institutional gaps.
Finally, consider long-term implications of staffing decisions. Leaders are making challenging coverage decisions that can affect the morale and autonomy of staff. Does backup staffing happen on a volunteer basis? Who fills the need—those with less clinical time or those with fewer personal obligations? When a staffing model is challenged and your group is making such decisions, engaged communication again becomes paramount.
PREPARE FOR THE MARATHON
Experts believe that we are only at the beginning of this crisis, one for which we don’t know what the end looks like or when it will come. With this in mind, hospital medicine leadership must plan for the long-term implications of the lengthy race ahead. Recognizing that morale, motivation, and burnout will be issues to deal with on the horizon, a focus on sustainability and wellness will become increasingly important as the marathon continues. To date, we’ve found the following principles to be helpful.
Delegate Responsibilities
Hospitals will not be able to survive COVID-19 through the efforts of single individuals. Instead, consider creating “operational champion” roles for frontline clinicians. These individuals can lead in specific areas (eg, PPE, updates on COVID-19 testing, discharge protocols) and act as conduits for information, updates, and resources for your group. At our institution, such operational meetings and activities take hours out of each day. By creating a breadth of leadership roles, our group has spread the operational workload while still allowing clinicians to care for patients, avoid burnout, and build autonomy and opportunities for both personal and professional growth. While for most institutions, these positions are temporary and not compensated with salary or time, the contribution to the group should be recognized both now and in the future.
Focus on Wellness
Providers are battling a laundry list of both clinical and personal stressors. The Centers for Disease Control and Prevention has already recognized that stress and mental health are going to be large hurdles for both patients and providers during this crisis.8 From the beginning, hospitalist leadership should be attuned to physician wellness and be aware that burnout, mental and physical exhaustion, and the possibility of contracting COVID-19 will be issues in the coming weeks and months. Volunteerism is built into the physician’s work ethic, but we must be mindful about its cost for long-term staffing demands. In addition, scarce medical resources add an additional moral strain for clinicians as they face tough allocation decisions, as we’ve seen with our Italian colleagues.9
As regular meetings around COVID-19 have become commonplace, we’ve made sure to set aside defined time for staff to discuss and reflect on their experiences. Doing so has allowed our clinicians to feel heard and to acknowledge the difficulties they are facing in their clinical duties. Leaders should also consider frequent check-ins with individual providers. At our institution, the first positive COVID-19 patient did not radically change any protocol that was in place, but a check-in with the hospitalist on service that day proved helpful for a debrief and processing opportunity. Individual conversations can help those on the front lines feel supported and remind them they are not operating alone in an anonymous vacuum.
Continue by celebrating small victories because this marathon is not going to end with an obvious finish line or a singular moment in which everyone can rejoice. A negative test, a patient with a good outcome, and a donation of PPE are all opportunities to celebrate. It may be what keeps us going when there is no end in sight. We have relied on these celebrations and moments of levity as an integral part of our regular group meetings.
CONCLUSION
At the end of this pandemic, just as we hope that our social distancing feels like an overreaction, we similarly hope that our sprint to build capacity ends up being unnecessary as well. As we wrote this Perspectives piece, uncertainty about the extent, length, and impact of this pandemic still existed. By the time it is published it may be that the sprint is over, and the marathon is beginning. Or, if our wildest hopes come true, there will be no marathon to run at all.
1. Tsai TC, Jacobson BH, Jha AK. American Hospital Capacity and Projected Need for COVID-19. Health Affairs. March 17, 2020. https://www.healthaffairs.org/do/10.1377/hblog20200317.457910/full/. Accessed April 1, 2020.
2. Argenti PA. Crisis communication: lessons from 9/11. Harvard Business Review. December 2002. https://hbr.org/2002/12/crisis-communication-lessons-from-911. Accessed April 2, 2020.
3. Argenti PA. Communicating through the coronavirus crisis. Harvard Business Review. March 2020. https://hbr.org/2020/03/communicating-through-the-coronavirus-crisis. Accessed April 2, 2020.
4. Chopra V, Toner E, Waldhorn R, Washer L. How should US hospitals prepare for COVID-19? Ann Intern Med. 2020. https://doi.org/10.7326/M20-0907.
5. National Institutes of Health. Formatting and Visual Clarity. Published July 1, 2015. Updated March 27, 2017. https://www.nih.gov/institutes-nih/nih-office-director/office-communications-public-liaison/clear-communication/plain-language/formatting-visual-clarity. Accessed April 2, 2020.
6. Frisch B, Greene C. What it takes to run a great virtual meeting. Harvard Business Review. March 2020. https://hbr.org/2020/03/what-it-takes-to-run-a-great-virtual-meeting. Accessed April 2, 2020.
7. Yan W. Coronavirus testing goes mobile in Seattle. New York Times. March 13, 2020. https://www.nytimes.com/2020/03/13/us/coronavirus-testing-drive-through-seattle.html. Accessed April 2, 2020.
8. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Stress and Coping. February 11, 2020. https://www.cdc.gov/coronavirus/2019-ncov/prepare/managing-stress-anxiety.html. Accessed April 2, 2020.
9. Rosenbaum L. Facing Covid-19 in Italy—ethics, logistics, and therapeutics on the epidemic’s front line. N Engl J Med. 2020. https://doi.org/10.1056/NEJMp2005492.
The pandemic of coronavirus disease 2019 (COVID-19) is confronting the modern world like nothing else before. With over 20 million individuals expected to require hospitalization in the US, this health crisis may become a generation-defining moment for healthcare systems and the field of hospital medicine.1 The specific challenges facing hospital medicine are comparable to running a sprint and a marathon—at the same time. For the sprint underway, hospitalists must learn to respond to a rapidly changing environment in which critical decisions are made within hours and days. At the same time, hospitalists need to plan for the marathon of increased clinical needs over the coming months, the possibility of burnout, and concerns about staff well-being. Although runners typically focus on either the sprint or the marathon, healthcare systems and hospital medicine providers will need to simultaneously prepare for both types of races.
GET READY FOR THE SPRINT
Over the past several weeks, hospital medicine leaders have been rapidly responding to an evolving crisis. Leaders and clinicians are quickly learning how to restructure clinical operations, negotiate the short supply of personal protective equipment (PPE), and manage delays in COVID-19 testing. In these areas, our hospitalist group has experienced a steep learning curve. In addition to the strategies outlined in the Table, we will share here our experiences and insights on managing and preparing for the COVID-19 pandemic.
Communication Is Central
During the sprint, focused, regular communication is imperative to ameliorate anxiety and fear. A study of crisis communication after 9/11 found that, for employees, good communication from leadership was one of the most valued factors.2 Communications experts also note that, in times of crisis, leaders have a special role in communication, specifically around demystifying the situation, providing hope, and maintaining transparency.3
Mental bandwidth may be limited in a stressful environment, so efforts should be taken to maximize the value of each communication. Information on hospital metrics should be provided regularly, including the number of COVID-19 cases, the status of clinical services and staffing, hospital capacity, and resource availability.4 Although the ubiquity and ease of email is convenient, recognize that providers are likely receiving email updates from multiple layers within your healthcare organization. To guard against losing important information, we use the same templated format for daily email updates with changes highlighted, which allows busy clinicians to digest pertinent information easily.5 Finally, consider having a single individual be responsible for collating COVID-19–related emails sent to your group. Although clinicians may want to share the most recent studies or their clinical experiences with a group email, instead have them send this information to a single individual who can organize these materials and share them on a regular basis.
To keep two-way communication channels open in a busy, asynchronous environment, consider having a centralized shared document in which providers can give real-time feedback to capture on-the-ground experiences or share questions they would like answered. Within our group, we found that centralizing our conversation in a shared document eliminated redundancy, focused our meetings, and kept everyone up to date. Additionally, regularly scheduled meetings may need to be adapted to a remote format (eg, Zoom, WebEx) as clinicians are asked to work from home when not on clinical service. Finally, recognize that virtual meetings require a different skill set than that required by in-person meetings, including reestablishment of social norms and technology preparation.6
Optimize Your Staffing
Hospital volumes could increase to as high as 270% of current hospital bed capacities during this pandemic.1 This surge is further complicated by the effort involved in caring for these patients, given their increased medical complexity, the use of new protocols, and the extra time needed to update staff and family. As the workload intensifies, staffing models and operations will also need to adapt.
First, optimize your inpatient resources based on the changes your hospital system is making. For instance, as elective surgeries were cancelled, we dissolved our surgical comanagement and consult services to better accommodate our hospitals’ needs. Further, consider using advanced practice providers (eg, physician assistants and nurse practitioners) released from their clinical duties to help with inpatient care in the event of a surge. If your hospital has trainees (eg, residents or fellows), consider reassigning those whose rotations have been postponed to newly created inpatient teams; trainees often have strong institutional knowledge and understanding of hospital protocols and resources.
Second, use hospitalists for their most relevant skills. Hospitalists are pluripotent clinicians who are comfortable with high-acuity patients and can fit into a myriad of clinical positions. The initial instinct at our institution was to mobilize hospitalists across all areas of increasing needs in the hospital (eg, screening clinics,7 advice phone lines for patients, or in the Emergency Department), but we quickly recognized that the hospitalist group is a finite resource. We focused our hospitalists’ clinical work on the expanding inpatient needs and allowed other outpatient or procedure-based specialties that have less inpatient experience to fill the broader institutional gaps.
Finally, consider long-term implications of staffing decisions. Leaders are making challenging coverage decisions that can affect the morale and autonomy of staff. Does backup staffing happen on a volunteer basis? Who fills the need—those with less clinical time or those with fewer personal obligations? When a staffing model is challenged and your group is making such decisions, engaged communication again becomes paramount.
PREPARE FOR THE MARATHON
Experts believe that we are only at the beginning of this crisis, one for which we don’t know what the end looks like or when it will come. With this in mind, hospital medicine leadership must plan for the long-term implications of the lengthy race ahead. Recognizing that morale, motivation, and burnout will be issues to deal with on the horizon, a focus on sustainability and wellness will become increasingly important as the marathon continues. To date, we’ve found the following principles to be helpful.
Delegate Responsibilities
Hospitals will not be able to survive COVID-19 through the efforts of single individuals. Instead, consider creating “operational champion” roles for frontline clinicians. These individuals can lead in specific areas (eg, PPE, updates on COVID-19 testing, discharge protocols) and act as conduits for information, updates, and resources for your group. At our institution, such operational meetings and activities take hours out of each day. By creating a breadth of leadership roles, our group has spread the operational workload while still allowing clinicians to care for patients, avoid burnout, and build autonomy and opportunities for both personal and professional growth. While for most institutions, these positions are temporary and not compensated with salary or time, the contribution to the group should be recognized both now and in the future.
Focus on Wellness
Providers are battling a laundry list of both clinical and personal stressors. The Centers for Disease Control and Prevention has already recognized that stress and mental health are going to be large hurdles for both patients and providers during this crisis.8 From the beginning, hospitalist leadership should be attuned to physician wellness and be aware that burnout, mental and physical exhaustion, and the possibility of contracting COVID-19 will be issues in the coming weeks and months. Volunteerism is built into the physician’s work ethic, but we must be mindful about its cost for long-term staffing demands. In addition, scarce medical resources add an additional moral strain for clinicians as they face tough allocation decisions, as we’ve seen with our Italian colleagues.9
As regular meetings around COVID-19 have become commonplace, we’ve made sure to set aside defined time for staff to discuss and reflect on their experiences. Doing so has allowed our clinicians to feel heard and to acknowledge the difficulties they are facing in their clinical duties. Leaders should also consider frequent check-ins with individual providers. At our institution, the first positive COVID-19 patient did not radically change any protocol that was in place, but a check-in with the hospitalist on service that day proved helpful for a debrief and processing opportunity. Individual conversations can help those on the front lines feel supported and remind them they are not operating alone in an anonymous vacuum.
Continue by celebrating small victories because this marathon is not going to end with an obvious finish line or a singular moment in which everyone can rejoice. A negative test, a patient with a good outcome, and a donation of PPE are all opportunities to celebrate. It may be what keeps us going when there is no end in sight. We have relied on these celebrations and moments of levity as an integral part of our regular group meetings.
CONCLUSION
At the end of this pandemic, just as we hope that our social distancing feels like an overreaction, we similarly hope that our sprint to build capacity ends up being unnecessary as well. As we wrote this Perspectives piece, uncertainty about the extent, length, and impact of this pandemic still existed. By the time it is published it may be that the sprint is over, and the marathon is beginning. Or, if our wildest hopes come true, there will be no marathon to run at all.
The pandemic of coronavirus disease 2019 (COVID-19) is confronting the modern world like nothing else before. With over 20 million individuals expected to require hospitalization in the US, this health crisis may become a generation-defining moment for healthcare systems and the field of hospital medicine.1 The specific challenges facing hospital medicine are comparable to running a sprint and a marathon—at the same time. For the sprint underway, hospitalists must learn to respond to a rapidly changing environment in which critical decisions are made within hours and days. At the same time, hospitalists need to plan for the marathon of increased clinical needs over the coming months, the possibility of burnout, and concerns about staff well-being. Although runners typically focus on either the sprint or the marathon, healthcare systems and hospital medicine providers will need to simultaneously prepare for both types of races.
GET READY FOR THE SPRINT
Over the past several weeks, hospital medicine leaders have been rapidly responding to an evolving crisis. Leaders and clinicians are quickly learning how to restructure clinical operations, negotiate the short supply of personal protective equipment (PPE), and manage delays in COVID-19 testing. In these areas, our hospitalist group has experienced a steep learning curve. In addition to the strategies outlined in the Table, we will share here our experiences and insights on managing and preparing for the COVID-19 pandemic.
Communication Is Central
During the sprint, focused, regular communication is imperative to ameliorate anxiety and fear. A study of crisis communication after 9/11 found that, for employees, good communication from leadership was one of the most valued factors.2 Communications experts also note that, in times of crisis, leaders have a special role in communication, specifically around demystifying the situation, providing hope, and maintaining transparency.3
Mental bandwidth may be limited in a stressful environment, so efforts should be taken to maximize the value of each communication. Information on hospital metrics should be provided regularly, including the number of COVID-19 cases, the status of clinical services and staffing, hospital capacity, and resource availability.4 Although the ubiquity and ease of email is convenient, recognize that providers are likely receiving email updates from multiple layers within your healthcare organization. To guard against losing important information, we use the same templated format for daily email updates with changes highlighted, which allows busy clinicians to digest pertinent information easily.5 Finally, consider having a single individual be responsible for collating COVID-19–related emails sent to your group. Although clinicians may want to share the most recent studies or their clinical experiences with a group email, instead have them send this information to a single individual who can organize these materials and share them on a regular basis.
To keep two-way communication channels open in a busy, asynchronous environment, consider having a centralized shared document in which providers can give real-time feedback to capture on-the-ground experiences or share questions they would like answered. Within our group, we found that centralizing our conversation in a shared document eliminated redundancy, focused our meetings, and kept everyone up to date. Additionally, regularly scheduled meetings may need to be adapted to a remote format (eg, Zoom, WebEx) as clinicians are asked to work from home when not on clinical service. Finally, recognize that virtual meetings require a different skill set than that required by in-person meetings, including reestablishment of social norms and technology preparation.6
Optimize Your Staffing
Hospital volumes could increase to as high as 270% of current hospital bed capacities during this pandemic.1 This surge is further complicated by the effort involved in caring for these patients, given their increased medical complexity, the use of new protocols, and the extra time needed to update staff and family. As the workload intensifies, staffing models and operations will also need to adapt.
First, optimize your inpatient resources based on the changes your hospital system is making. For instance, as elective surgeries were cancelled, we dissolved our surgical comanagement and consult services to better accommodate our hospitals’ needs. Further, consider using advanced practice providers (eg, physician assistants and nurse practitioners) released from their clinical duties to help with inpatient care in the event of a surge. If your hospital has trainees (eg, residents or fellows), consider reassigning those whose rotations have been postponed to newly created inpatient teams; trainees often have strong institutional knowledge and understanding of hospital protocols and resources.
Second, use hospitalists for their most relevant skills. Hospitalists are pluripotent clinicians who are comfortable with high-acuity patients and can fit into a myriad of clinical positions. The initial instinct at our institution was to mobilize hospitalists across all areas of increasing needs in the hospital (eg, screening clinics,7 advice phone lines for patients, or in the Emergency Department), but we quickly recognized that the hospitalist group is a finite resource. We focused our hospitalists’ clinical work on the expanding inpatient needs and allowed other outpatient or procedure-based specialties that have less inpatient experience to fill the broader institutional gaps.
Finally, consider long-term implications of staffing decisions. Leaders are making challenging coverage decisions that can affect the morale and autonomy of staff. Does backup staffing happen on a volunteer basis? Who fills the need—those with less clinical time or those with fewer personal obligations? When a staffing model is challenged and your group is making such decisions, engaged communication again becomes paramount.
PREPARE FOR THE MARATHON
Experts believe that we are only at the beginning of this crisis, one for which we don’t know what the end looks like or when it will come. With this in mind, hospital medicine leadership must plan for the long-term implications of the lengthy race ahead. Recognizing that morale, motivation, and burnout will be issues to deal with on the horizon, a focus on sustainability and wellness will become increasingly important as the marathon continues. To date, we’ve found the following principles to be helpful.
Delegate Responsibilities
Hospitals will not be able to survive COVID-19 through the efforts of single individuals. Instead, consider creating “operational champion” roles for frontline clinicians. These individuals can lead in specific areas (eg, PPE, updates on COVID-19 testing, discharge protocols) and act as conduits for information, updates, and resources for your group. At our institution, such operational meetings and activities take hours out of each day. By creating a breadth of leadership roles, our group has spread the operational workload while still allowing clinicians to care for patients, avoid burnout, and build autonomy and opportunities for both personal and professional growth. While for most institutions, these positions are temporary and not compensated with salary or time, the contribution to the group should be recognized both now and in the future.
Focus on Wellness
Providers are battling a laundry list of both clinical and personal stressors. The Centers for Disease Control and Prevention has already recognized that stress and mental health are going to be large hurdles for both patients and providers during this crisis.8 From the beginning, hospitalist leadership should be attuned to physician wellness and be aware that burnout, mental and physical exhaustion, and the possibility of contracting COVID-19 will be issues in the coming weeks and months. Volunteerism is built into the physician’s work ethic, but we must be mindful about its cost for long-term staffing demands. In addition, scarce medical resources add an additional moral strain for clinicians as they face tough allocation decisions, as we’ve seen with our Italian colleagues.9
As regular meetings around COVID-19 have become commonplace, we’ve made sure to set aside defined time for staff to discuss and reflect on their experiences. Doing so has allowed our clinicians to feel heard and to acknowledge the difficulties they are facing in their clinical duties. Leaders should also consider frequent check-ins with individual providers. At our institution, the first positive COVID-19 patient did not radically change any protocol that was in place, but a check-in with the hospitalist on service that day proved helpful for a debrief and processing opportunity. Individual conversations can help those on the front lines feel supported and remind them they are not operating alone in an anonymous vacuum.
Continue by celebrating small victories because this marathon is not going to end with an obvious finish line or a singular moment in which everyone can rejoice. A negative test, a patient with a good outcome, and a donation of PPE are all opportunities to celebrate. It may be what keeps us going when there is no end in sight. We have relied on these celebrations and moments of levity as an integral part of our regular group meetings.
CONCLUSION
At the end of this pandemic, just as we hope that our social distancing feels like an overreaction, we similarly hope that our sprint to build capacity ends up being unnecessary as well. As we wrote this Perspectives piece, uncertainty about the extent, length, and impact of this pandemic still existed. By the time it is published it may be that the sprint is over, and the marathon is beginning. Or, if our wildest hopes come true, there will be no marathon to run at all.
1. Tsai TC, Jacobson BH, Jha AK. American Hospital Capacity and Projected Need for COVID-19. Health Affairs. March 17, 2020. https://www.healthaffairs.org/do/10.1377/hblog20200317.457910/full/. Accessed April 1, 2020.
2. Argenti PA. Crisis communication: lessons from 9/11. Harvard Business Review. December 2002. https://hbr.org/2002/12/crisis-communication-lessons-from-911. Accessed April 2, 2020.
3. Argenti PA. Communicating through the coronavirus crisis. Harvard Business Review. March 2020. https://hbr.org/2020/03/communicating-through-the-coronavirus-crisis. Accessed April 2, 2020.
4. Chopra V, Toner E, Waldhorn R, Washer L. How should US hospitals prepare for COVID-19? Ann Intern Med. 2020. https://doi.org/10.7326/M20-0907.
5. National Institutes of Health. Formatting and Visual Clarity. Published July 1, 2015. Updated March 27, 2017. https://www.nih.gov/institutes-nih/nih-office-director/office-communications-public-liaison/clear-communication/plain-language/formatting-visual-clarity. Accessed April 2, 2020.
6. Frisch B, Greene C. What it takes to run a great virtual meeting. Harvard Business Review. March 2020. https://hbr.org/2020/03/what-it-takes-to-run-a-great-virtual-meeting. Accessed April 2, 2020.
7. Yan W. Coronavirus testing goes mobile in Seattle. New York Times. March 13, 2020. https://www.nytimes.com/2020/03/13/us/coronavirus-testing-drive-through-seattle.html. Accessed April 2, 2020.
8. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Stress and Coping. February 11, 2020. https://www.cdc.gov/coronavirus/2019-ncov/prepare/managing-stress-anxiety.html. Accessed April 2, 2020.
9. Rosenbaum L. Facing Covid-19 in Italy—ethics, logistics, and therapeutics on the epidemic’s front line. N Engl J Med. 2020. https://doi.org/10.1056/NEJMp2005492.
1. Tsai TC, Jacobson BH, Jha AK. American Hospital Capacity and Projected Need for COVID-19. Health Affairs. March 17, 2020. https://www.healthaffairs.org/do/10.1377/hblog20200317.457910/full/. Accessed April 1, 2020.
2. Argenti PA. Crisis communication: lessons from 9/11. Harvard Business Review. December 2002. https://hbr.org/2002/12/crisis-communication-lessons-from-911. Accessed April 2, 2020.
3. Argenti PA. Communicating through the coronavirus crisis. Harvard Business Review. March 2020. https://hbr.org/2020/03/communicating-through-the-coronavirus-crisis. Accessed April 2, 2020.
4. Chopra V, Toner E, Waldhorn R, Washer L. How should US hospitals prepare for COVID-19? Ann Intern Med. 2020. https://doi.org/10.7326/M20-0907.
5. National Institutes of Health. Formatting and Visual Clarity. Published July 1, 2015. Updated March 27, 2017. https://www.nih.gov/institutes-nih/nih-office-director/office-communications-public-liaison/clear-communication/plain-language/formatting-visual-clarity. Accessed April 2, 2020.
6. Frisch B, Greene C. What it takes to run a great virtual meeting. Harvard Business Review. March 2020. https://hbr.org/2020/03/what-it-takes-to-run-a-great-virtual-meeting. Accessed April 2, 2020.
7. Yan W. Coronavirus testing goes mobile in Seattle. New York Times. March 13, 2020. https://www.nytimes.com/2020/03/13/us/coronavirus-testing-drive-through-seattle.html. Accessed April 2, 2020.
8. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Stress and Coping. February 11, 2020. https://www.cdc.gov/coronavirus/2019-ncov/prepare/managing-stress-anxiety.html. Accessed April 2, 2020.
9. Rosenbaum L. Facing Covid-19 in Italy—ethics, logistics, and therapeutics on the epidemic’s front line. N Engl J Med. 2020. https://doi.org/10.1056/NEJMp2005492.
© 2020 Society of Hospital Medicine
COVID-19 linked to multiple cardiovascular presentations
It’s becoming clear that COVID-19 infection can involve the cardiovascular system in many different ways, and this has “evolving” potential implications for treatment, say a team of cardiologists on the frontlines of the COVID-19 battle in New York City.
In an article published online April 3 in Circulation, Justin Fried, MD, Division of Cardiology, Columbia University, New York City, and colleagues present four case studies of COVID-19 patients with various cardiovascular presentations.
Case 1 is a 64-year-old woman whose predominant symptoms on admission were cardiac in nature, including chest pain and ST elevation, but without fever, cough, or other symptoms suggestive of COVID-19.
“In patients presenting with what appears to be a typical cardiac syndrome, COVID-19 infection should be in the differential during the current pandemic, even in the absence of fever or cough,” the clinicians advise.
Case 2 is a 38-year-old man with cardiogenic shock and acute respiratory distress with profound hypoxia who was rescued with veno-arterial-venous extracorporeal membrane oxygenation (VV ECMO).
The initial presentation of this patient was more characteristic of severe COVID-19 disease, and cardiac involvement only became apparent after the initiation of ECMO, Fried and colleagues report.
Based on this case, they advise a “low threshold” to assess for cardiogenic shock in patients with acute systolic heart failure related to COVID-19. If inotropic support fails in these patients, intra-aortic balloon pump should be considered first for mechanical circulatory support because it requires the least maintenance from medical support staff.
In addition, in their experience, when a patient on VV ECMO develops superimposed cardiogenic shock, adding an arterial conduit at a relatively low blood flow rate may provide the necessary circulatory support without inducing left ventricular distension, they note.
“Our experience confirms that rescue of patients even with profound cardiogenic or mixed shock may be possible with temporary hemodynamic support at centers with availability of such devices,” Fried and colleagues report.
Case 3 is a 64-year-old woman with underlying cardiac disease who developed profound decompensation with COVID-19 infection.
This case demonstrates that the infection can cause decompensation of underlying heart failure and may lead to mixed shock, the clinicians say.
“Invasive hemodynamic monitoring, if feasible, may be helpful to manage the cardiac component of shock in such cases. Medications that prolong the QT interval are being considered for COVID-19 patients and may require closer monitoring in patients with underlying structural heart disease,” they note.
Case 4 is a 51-year-old man who underwent a heart transplant in 2007 and a kidney transplant in 2010. He had COVID-19 symptoms akin to those seen in nonimmunosuppressed patients with COVID-19.
The COVID-19 pandemic presents a “unique challenge” for solid organ transplant recipients, with only “limited” data on how to adjust immunosuppression during COVID-19 infection, Fried and colleagues say.
The pandemic also creates a challenge for the management of heart failure patients on the heart transplant wait list; the risks of delaying a transplant need to be balanced against the risks of donor infection and uncertainty regarding the impact of post-transplant immunosuppression protocols, they note.
As reported by Medscape Medical News, the American Heart Association has developed a COVID-19 patient registry to collect data on cardiovascular conditions and outcomes related to COVID-19 infection.
To participate in the registry, contact qualityresearch@heart.org.
This article first appeared on Medscape.com.
It’s becoming clear that COVID-19 infection can involve the cardiovascular system in many different ways, and this has “evolving” potential implications for treatment, say a team of cardiologists on the frontlines of the COVID-19 battle in New York City.
In an article published online April 3 in Circulation, Justin Fried, MD, Division of Cardiology, Columbia University, New York City, and colleagues present four case studies of COVID-19 patients with various cardiovascular presentations.
Case 1 is a 64-year-old woman whose predominant symptoms on admission were cardiac in nature, including chest pain and ST elevation, but without fever, cough, or other symptoms suggestive of COVID-19.
“In patients presenting with what appears to be a typical cardiac syndrome, COVID-19 infection should be in the differential during the current pandemic, even in the absence of fever or cough,” the clinicians advise.
Case 2 is a 38-year-old man with cardiogenic shock and acute respiratory distress with profound hypoxia who was rescued with veno-arterial-venous extracorporeal membrane oxygenation (VV ECMO).
The initial presentation of this patient was more characteristic of severe COVID-19 disease, and cardiac involvement only became apparent after the initiation of ECMO, Fried and colleagues report.
Based on this case, they advise a “low threshold” to assess for cardiogenic shock in patients with acute systolic heart failure related to COVID-19. If inotropic support fails in these patients, intra-aortic balloon pump should be considered first for mechanical circulatory support because it requires the least maintenance from medical support staff.
In addition, in their experience, when a patient on VV ECMO develops superimposed cardiogenic shock, adding an arterial conduit at a relatively low blood flow rate may provide the necessary circulatory support without inducing left ventricular distension, they note.
“Our experience confirms that rescue of patients even with profound cardiogenic or mixed shock may be possible with temporary hemodynamic support at centers with availability of such devices,” Fried and colleagues report.
Case 3 is a 64-year-old woman with underlying cardiac disease who developed profound decompensation with COVID-19 infection.
This case demonstrates that the infection can cause decompensation of underlying heart failure and may lead to mixed shock, the clinicians say.
“Invasive hemodynamic monitoring, if feasible, may be helpful to manage the cardiac component of shock in such cases. Medications that prolong the QT interval are being considered for COVID-19 patients and may require closer monitoring in patients with underlying structural heart disease,” they note.
Case 4 is a 51-year-old man who underwent a heart transplant in 2007 and a kidney transplant in 2010. He had COVID-19 symptoms akin to those seen in nonimmunosuppressed patients with COVID-19.
The COVID-19 pandemic presents a “unique challenge” for solid organ transplant recipients, with only “limited” data on how to adjust immunosuppression during COVID-19 infection, Fried and colleagues say.
The pandemic also creates a challenge for the management of heart failure patients on the heart transplant wait list; the risks of delaying a transplant need to be balanced against the risks of donor infection and uncertainty regarding the impact of post-transplant immunosuppression protocols, they note.
As reported by Medscape Medical News, the American Heart Association has developed a COVID-19 patient registry to collect data on cardiovascular conditions and outcomes related to COVID-19 infection.
To participate in the registry, contact qualityresearch@heart.org.
This article first appeared on Medscape.com.
It’s becoming clear that COVID-19 infection can involve the cardiovascular system in many different ways, and this has “evolving” potential implications for treatment, say a team of cardiologists on the frontlines of the COVID-19 battle in New York City.
In an article published online April 3 in Circulation, Justin Fried, MD, Division of Cardiology, Columbia University, New York City, and colleagues present four case studies of COVID-19 patients with various cardiovascular presentations.
Case 1 is a 64-year-old woman whose predominant symptoms on admission were cardiac in nature, including chest pain and ST elevation, but without fever, cough, or other symptoms suggestive of COVID-19.
“In patients presenting with what appears to be a typical cardiac syndrome, COVID-19 infection should be in the differential during the current pandemic, even in the absence of fever or cough,” the clinicians advise.
Case 2 is a 38-year-old man with cardiogenic shock and acute respiratory distress with profound hypoxia who was rescued with veno-arterial-venous extracorporeal membrane oxygenation (VV ECMO).
The initial presentation of this patient was more characteristic of severe COVID-19 disease, and cardiac involvement only became apparent after the initiation of ECMO, Fried and colleagues report.
Based on this case, they advise a “low threshold” to assess for cardiogenic shock in patients with acute systolic heart failure related to COVID-19. If inotropic support fails in these patients, intra-aortic balloon pump should be considered first for mechanical circulatory support because it requires the least maintenance from medical support staff.
In addition, in their experience, when a patient on VV ECMO develops superimposed cardiogenic shock, adding an arterial conduit at a relatively low blood flow rate may provide the necessary circulatory support without inducing left ventricular distension, they note.
“Our experience confirms that rescue of patients even with profound cardiogenic or mixed shock may be possible with temporary hemodynamic support at centers with availability of such devices,” Fried and colleagues report.
Case 3 is a 64-year-old woman with underlying cardiac disease who developed profound decompensation with COVID-19 infection.
This case demonstrates that the infection can cause decompensation of underlying heart failure and may lead to mixed shock, the clinicians say.
“Invasive hemodynamic monitoring, if feasible, may be helpful to manage the cardiac component of shock in such cases. Medications that prolong the QT interval are being considered for COVID-19 patients and may require closer monitoring in patients with underlying structural heart disease,” they note.
Case 4 is a 51-year-old man who underwent a heart transplant in 2007 and a kidney transplant in 2010. He had COVID-19 symptoms akin to those seen in nonimmunosuppressed patients with COVID-19.
The COVID-19 pandemic presents a “unique challenge” for solid organ transplant recipients, with only “limited” data on how to adjust immunosuppression during COVID-19 infection, Fried and colleagues say.
The pandemic also creates a challenge for the management of heart failure patients on the heart transplant wait list; the risks of delaying a transplant need to be balanced against the risks of donor infection and uncertainty regarding the impact of post-transplant immunosuppression protocols, they note.
As reported by Medscape Medical News, the American Heart Association has developed a COVID-19 patient registry to collect data on cardiovascular conditions and outcomes related to COVID-19 infection.
To participate in the registry, contact qualityresearch@heart.org.
This article first appeared on Medscape.com.
Prescribing aspirin to improve pregnancy outcomes: Expand the indications? Increase the dose?
Authors of a recent Cochrane review concluded that low-dose aspirin treatment of 1,000 pregnant women at risk of developing preeclampsia resulted in 16 fewer cases of preeclampsia, 16 fewer preterm births, 7 fewer cases of small-for-gestational age newborns, and 5 fewer fetal or neonatal deaths.1
The American College of Obstetricians and Gynecologists (ACOG) and the US Preventive Services Task Force (USPSTF) recommend treatment with 81 mg of aspirin daily, initiated before 16 weeks of pregnancy to prevent preeclampsia in women with one major risk factor (personal history of preeclampsia, multifetal gestation, chronic hypertension, type 1 or 2 diabetes, renal or autoimmune disease) or at least two moderate risk factors (nulliparity; obesity; mother or sister with preeclampsia; a sociodemographic characteristic such as African American race or low socioeconomic status; age ≥35 years; personal history factors such as prior low birth weight infant, previous adverse pregnancy outcome, or >10-year interpregnancy interval).2,3 Healthy pregnant women with a previous uncomplicated full-term delivery do not need treatment with low-dose aspirin.2,3
However, evolving data and expert opinion suggest that expanding the indications for aspirin treatment and increasing the recommended dose of aspirin may be warranted.
Nulliparity
Nulliparity is the single clinical characteristic that is associated with the greatest number of cases of preeclampsia.4 Hence, from a public health perspective, reducing the rate of preeclampsia among nulliparous women is a top priority.
ACOG and USPSTF do not recommend aspirin treatment for all nulliparous women because risk factors help to identify those nulliparous women who benefit from aspirin treatment.
However, a recent cost-effectiveness analysis compared the health care costs and rates of preeclampsia for 4 prevention strategies among all pregnant women in the United States (nulliparous and parous)5:
- no aspirin use
- use of aspirin based on biomarker and ultrasound measurements
- use of aspirin based on USPSTF guidelines for identifying women at risk
- prescription of aspirin to all pregnant women.
Health care costs and rates of preeclampsia were lowest with the universal prescription of aspirin to all pregnant women in the United States. Compared with universal prescription of aspirin, the USPSTF approach, the biomarker-ultrasound approach, and the no aspirin approach were associated with 346, 308, and 762 additional cases of preeclampsia per 100,000 women. In sensitivity analyses, universal aspirin was the optimal strategy under most assumptions.
Another cost effectiveness analysis concluded that among nulliparous pregnant women, universal aspirin treatment was superior to aspirin treatment based on biomarker-ultrasound identification of women at high risk.6
In a recent clinical trial performed in India, Guatemala, Pakistan, Democratic Republic of Congo, Kenya, and Zambia, 14,361 nulliparous women were randomly assigned to placebo or 81 mg of aspirin daily between 6 and 14 weeks of gestation.7 Preterm birth (<37 weeks’ gestation) occurred in 13.1% and 11.6% of women treated with placebo or aspirin (relative risk [RR], 0.89; 95% confidence interval [CI], 0.81 to 0.98, P = .012). Most of the decrease in preterm birth appeared to be due to a decrease in the rate of preeclampsia in the aspirin-treated nulliparous women. The investigators also noted that aspirin treatment of nulliparous women resulted in a statistically significant decrease in perinatal mortality (RR, 0.86) and early preterm delivery, <34 weeks’ gestation (RR, 0.75).
Universal prescription of low-dose aspirin to nulliparous women in order to prevent preeclampsia and preterm birth may become recognized as an optimal public health strategy. As a step toward universal prescription of aspirin to nulliparous women, an opt-out rather than a screen-in strategy might be considered.8

Continue to: Booking systolic blood pressure, 120 to 134 mm Hg...
Booking systolic blood pressure, 120 to 134 mm Hg
All obstetricians recognize that women with chronic hypertension should be treated with low-dose aspirin because they are at high risk for preeclampsia. However, there is evidence that nulliparous women with a booking systolic pressure ≥120 mm Hg might also benefit from low-dose aspirin treatment. In one US trial, 3,135 nulliparous normotensive women (booking blood pressure [BP] <135/85 mm Hg) were randomly assigned to treatment with aspirin (60 mg daily) or placebo initiated between 13 and 26 weeks’ gestation. Preeclampsia occurred in 6.3% and 4.6% of the women treated with placebo or aspirin, respectively (RR, 0.7; 95% CI, 0.6–1.0; P = .05).9 A secondary analysis showed that, among 519 nulliparous women with a booking systolic BP from 120 to 134 mm Hg, compared with placebo, low-dose aspirin treatment reduced the rate of preeclampsia from 11.9% to 5.6%.9 Aspirin did not reduce the rate of preeclampsia among nulliparous women with a booking systolic BP <120 mm Hg.9 A systematic review of risk factors for developing preeclampsia reported that a booking diastolic BP of ≥80 mm Hg was associated with an increased risk of developing preeclampsia (RR, 1.38).10
The American Heart Association (AHA) and the American College of Cardiology (ACC) recently updated the definition of hypertension.11 Normal BP is now defined as a systolic pressure <120 mm Hg and diastolic pressure <80 mm Hg. Elevated BP is a systolic pressure of 120 to 129 mm Hg and diastolic pressure of <80 mm Hg. Stage I hypertension is a systolic BP from 130 to 139 mm Hg or diastolic blood pressure from 80 to 89 mm Hg. Stage II hypertension is a systolic BP of ≥140 mm Hg or diastolic blood pressure ≥90 mm Hg.11
A recent study reported that 90% of women at 12 weeks’ gestation have a BP of ≤130 mm Hg systolic and ≤80 mm Hg diastolic, suggesting that the AHA-ACC criteria for stage I hypertension are reasonable.12 Obstetricians have not yet fully adopted the AHA-ACC criteria for defining stage I hypertension in pregnant women. Future research may demonstrate that a booking systolic BP
≥130 mm Hg or a diastolic BP ≥80 mm Hg are major risk factors for developing preeclampsia and warrant treatment with low-dose aspirin.

Continue to: Pregnancy resulting from fertility therapy...
Pregnancy resulting from fertility therapy
Current ACOG and USPSTF guidelines do not specifically identify pregnancies resulting from assisted reproductive technology as a major or moderate risk factor for preeclampsia.2,3 In a study comparing 83,582 births resulting from in vitro fertilization (IVF) and 1,382,311 births to fertile women, treatment with autologous cryopreserved embryos (adjusted odds ratio [aOR], 1.30), fresh donor embryos (aOR, 1.92), and cryopreserved donor embryos (aOR, 1.70) significantly increased the risk of preeclampsia.13 However, use of fresh autologous embryos did not increase the risk of preeclampsia (aOR, 1.04). These associations persisted after controlling for diabetes, hypertension, body mass index, and cause of infertility.13
Other studies also have reported that use of cryopreserved embryos is associated with a higher rate of preeclampsia than use of fresh autologous embryos. In a study of 825 infertile women undergoing IVF and randomly assigned to single embryo cryopreserved or fresh cycles, the rate of preeclampsia was 3.1% and 1.0% in the pregnancies that resulted from cryopreserved versus fresh cycles.14
What is the optimal dose of aspirin?
ACOG and the USPSTF recommend aspirin 81 mg daily for the prevention of preeclampsia.2,3 The International Federation of Gynecology and Obstetrics (FIGO) recommends aspirin 150 mg daily for the prevention of preeclampsia.15 The FIGO recommendation is based, in part, on the results of a large international clinical trial that randomly assigned 1,776 women at high risk for preeclampsia as determined by clinical factors plus biomarker and ultrasound screening to receive aspirin 150 mg daily or placebo daily initiated at 11 to 14 weeks’ gestation and continued until 36 weeks’ gestation.16 Preeclampsia before 37 weeks’ gestation occurred in 4.3% and 1.6% of women in the placebo and aspirin groups (OR, 0.38; 95% CI, 0.20–0.74; P = .004).16 FIGO recommends that women at risk for preeclampsia with a body mass <40 kg take aspirin 100 mg daily and women with a body mass ≥40 kg take aspirin at a dose of 150 mg daily. For women who live in a country where aspirin is not available in a pill containing 150 mg, FIGO recommends taking two 81 mg tablets.15 FIGO recommends initiating aspirin between 11 and 14 weeks and 6 days of gestation and continuing aspirin therapy until 36 weeks of gestation.15
Aspirin is an inexpensive intervention with many possible benefits
For many nulliparous women and some parous women aspirin treatment initiated early in pregnancy will improve maternal and newborn outcomes, including reducing the risk of preeclampsia, preterm birth, and intrauterine growth restriction.1 Obstetricians may want to begin to expand the indications for offering aspirin to prevent preeclampsia from those recommended by ACOG and the USPSTF to include nulliparous women with a booking systolic pressure of 120 to 134 mm Hg and women whose pregnancy was the result of an assisted reproduction treatment that used cryopreserved embryos. In addition, obstetricians who currently prescribe 81 mg of aspirin daily might want to consider increasing the prescribed dose to 162 mg of aspirin daily (two 81 mg tablets daily or one-half of a 325 mg tablet). Aspirin costs about less than 5 cents per 81 mg tablet (according to GoodRx website). It is an inexpensive intervention that could benefit many mothers and newborns. ●
- Duley L, Meher S, Hunter KE, et al. Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2019;CD004659.
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 743: low-dose aspirin use during pregnancy. Obstet Gynecol. 2018;132:e44-e52.
- LeFevre ML; U.S. Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force Recommendation Statement. Ann Int Med. 2014;161: 819-826.
- Bartsch E, Medcalf KE, Park AL, et al. Clinical risk factors for pre-eclampsia determined in early pregnancy: systematic review and meta-analysis of large cohort studies. BMJ. 2016;353:i1753.
- Mallampati D, Grobman W, Rouse DJ, et al. Strategies for prescribing aspirin to prevent preeclampsia: a cost-effectiveness analysis. Obstet Gynecol. 2019;134:537-544.
- Mone F, O’Mahony JF, Tyrrell E, et al. Preeclampsia prevention using routine versus screening test-indicated aspirin in low-risk women. Hypertension. 2018;72:1391-1396.
- Hoffman MK, Goudar SS, Kodkany BS, et al. Low-dose aspirin for the prevention of preterm delivery in nulliparous women with a singleton pregnancy (ASPIRIN): a randomised, double-blind, placebo-controlled trial. Lancet. 2020;395:285-293.
- Ayala NK, Rouse DJ. A nudge toward universal aspirin for preeclampsia prevention. Obstet Gynecol. 2019;133:725-728.
- Sibai BM, Caritis SN, Thom E, et al. Prevention of preeclampsia with low-dose aspirin in healthy, nulliparous pregnant women. The National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. N Engl J Med. 1993;329:1213-1218.
- Duckitt K, Harrington D. Risk factors for preeclampsia at antenatal booking: systematic review of controlled studies. BMJ. 2005;330:565.
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71:2199-2269.
- Green LJ, Mackillop LH, Salvi D, et al. Gestation-specific vital sign reference ranges in pregnancy. Obstet Gynecol. 2020;135:653-664.
- Luke B, Brown MB, Eisenberg ML, et al. In vitro fertilization and risk for hypertensive disorders of pregnancy: associations with treatment parameters. Am J Obstet Gynecol. October 17, 2019. doi:10.1016/j.ajog.2019.10.003.
- Wei D, Liu JY, Sun Y, et al. Frozen versus fresh single blastocyst transfer in ovulatory women: a multicentre, randomised controlled trial. Lancet. 2019;393:1310-1318.
- Poon LC, Shennan A, Hyett JA, et al. International Federation of Gynecology and Obstetrics (FIGO) initiative on preeclampsia: A pragmatic guide for first-trimester screening and prevention. Int J Gynaecol Obstet. 2019;145(suppl 1):1-33.
- Rolnik DL, Wright D, Poon LC, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377:613-622.
Authors of a recent Cochrane review concluded that low-dose aspirin treatment of 1,000 pregnant women at risk of developing preeclampsia resulted in 16 fewer cases of preeclampsia, 16 fewer preterm births, 7 fewer cases of small-for-gestational age newborns, and 5 fewer fetal or neonatal deaths.1
The American College of Obstetricians and Gynecologists (ACOG) and the US Preventive Services Task Force (USPSTF) recommend treatment with 81 mg of aspirin daily, initiated before 16 weeks of pregnancy to prevent preeclampsia in women with one major risk factor (personal history of preeclampsia, multifetal gestation, chronic hypertension, type 1 or 2 diabetes, renal or autoimmune disease) or at least two moderate risk factors (nulliparity; obesity; mother or sister with preeclampsia; a sociodemographic characteristic such as African American race or low socioeconomic status; age ≥35 years; personal history factors such as prior low birth weight infant, previous adverse pregnancy outcome, or >10-year interpregnancy interval).2,3 Healthy pregnant women with a previous uncomplicated full-term delivery do not need treatment with low-dose aspirin.2,3
However, evolving data and expert opinion suggest that expanding the indications for aspirin treatment and increasing the recommended dose of aspirin may be warranted.
Nulliparity
Nulliparity is the single clinical characteristic that is associated with the greatest number of cases of preeclampsia.4 Hence, from a public health perspective, reducing the rate of preeclampsia among nulliparous women is a top priority.
ACOG and USPSTF do not recommend aspirin treatment for all nulliparous women because risk factors help to identify those nulliparous women who benefit from aspirin treatment.
However, a recent cost-effectiveness analysis compared the health care costs and rates of preeclampsia for 4 prevention strategies among all pregnant women in the United States (nulliparous and parous)5:
- no aspirin use
- use of aspirin based on biomarker and ultrasound measurements
- use of aspirin based on USPSTF guidelines for identifying women at risk
- prescription of aspirin to all pregnant women.
Health care costs and rates of preeclampsia were lowest with the universal prescription of aspirin to all pregnant women in the United States. Compared with universal prescription of aspirin, the USPSTF approach, the biomarker-ultrasound approach, and the no aspirin approach were associated with 346, 308, and 762 additional cases of preeclampsia per 100,000 women. In sensitivity analyses, universal aspirin was the optimal strategy under most assumptions.
Another cost effectiveness analysis concluded that among nulliparous pregnant women, universal aspirin treatment was superior to aspirin treatment based on biomarker-ultrasound identification of women at high risk.6
In a recent clinical trial performed in India, Guatemala, Pakistan, Democratic Republic of Congo, Kenya, and Zambia, 14,361 nulliparous women were randomly assigned to placebo or 81 mg of aspirin daily between 6 and 14 weeks of gestation.7 Preterm birth (<37 weeks’ gestation) occurred in 13.1% and 11.6% of women treated with placebo or aspirin (relative risk [RR], 0.89; 95% confidence interval [CI], 0.81 to 0.98, P = .012). Most of the decrease in preterm birth appeared to be due to a decrease in the rate of preeclampsia in the aspirin-treated nulliparous women. The investigators also noted that aspirin treatment of nulliparous women resulted in a statistically significant decrease in perinatal mortality (RR, 0.86) and early preterm delivery, <34 weeks’ gestation (RR, 0.75).
Universal prescription of low-dose aspirin to nulliparous women in order to prevent preeclampsia and preterm birth may become recognized as an optimal public health strategy. As a step toward universal prescription of aspirin to nulliparous women, an opt-out rather than a screen-in strategy might be considered.8

Continue to: Booking systolic blood pressure, 120 to 134 mm Hg...
Booking systolic blood pressure, 120 to 134 mm Hg
All obstetricians recognize that women with chronic hypertension should be treated with low-dose aspirin because they are at high risk for preeclampsia. However, there is evidence that nulliparous women with a booking systolic pressure ≥120 mm Hg might also benefit from low-dose aspirin treatment. In one US trial, 3,135 nulliparous normotensive women (booking blood pressure [BP] <135/85 mm Hg) were randomly assigned to treatment with aspirin (60 mg daily) or placebo initiated between 13 and 26 weeks’ gestation. Preeclampsia occurred in 6.3% and 4.6% of the women treated with placebo or aspirin, respectively (RR, 0.7; 95% CI, 0.6–1.0; P = .05).9 A secondary analysis showed that, among 519 nulliparous women with a booking systolic BP from 120 to 134 mm Hg, compared with placebo, low-dose aspirin treatment reduced the rate of preeclampsia from 11.9% to 5.6%.9 Aspirin did not reduce the rate of preeclampsia among nulliparous women with a booking systolic BP <120 mm Hg.9 A systematic review of risk factors for developing preeclampsia reported that a booking diastolic BP of ≥80 mm Hg was associated with an increased risk of developing preeclampsia (RR, 1.38).10
The American Heart Association (AHA) and the American College of Cardiology (ACC) recently updated the definition of hypertension.11 Normal BP is now defined as a systolic pressure <120 mm Hg and diastolic pressure <80 mm Hg. Elevated BP is a systolic pressure of 120 to 129 mm Hg and diastolic pressure of <80 mm Hg. Stage I hypertension is a systolic BP from 130 to 139 mm Hg or diastolic blood pressure from 80 to 89 mm Hg. Stage II hypertension is a systolic BP of ≥140 mm Hg or diastolic blood pressure ≥90 mm Hg.11
A recent study reported that 90% of women at 12 weeks’ gestation have a BP of ≤130 mm Hg systolic and ≤80 mm Hg diastolic, suggesting that the AHA-ACC criteria for stage I hypertension are reasonable.12 Obstetricians have not yet fully adopted the AHA-ACC criteria for defining stage I hypertension in pregnant women. Future research may demonstrate that a booking systolic BP
≥130 mm Hg or a diastolic BP ≥80 mm Hg are major risk factors for developing preeclampsia and warrant treatment with low-dose aspirin.

Continue to: Pregnancy resulting from fertility therapy...
Pregnancy resulting from fertility therapy
Current ACOG and USPSTF guidelines do not specifically identify pregnancies resulting from assisted reproductive technology as a major or moderate risk factor for preeclampsia.2,3 In a study comparing 83,582 births resulting from in vitro fertilization (IVF) and 1,382,311 births to fertile women, treatment with autologous cryopreserved embryos (adjusted odds ratio [aOR], 1.30), fresh donor embryos (aOR, 1.92), and cryopreserved donor embryos (aOR, 1.70) significantly increased the risk of preeclampsia.13 However, use of fresh autologous embryos did not increase the risk of preeclampsia (aOR, 1.04). These associations persisted after controlling for diabetes, hypertension, body mass index, and cause of infertility.13
Other studies also have reported that use of cryopreserved embryos is associated with a higher rate of preeclampsia than use of fresh autologous embryos. In a study of 825 infertile women undergoing IVF and randomly assigned to single embryo cryopreserved or fresh cycles, the rate of preeclampsia was 3.1% and 1.0% in the pregnancies that resulted from cryopreserved versus fresh cycles.14
What is the optimal dose of aspirin?
ACOG and the USPSTF recommend aspirin 81 mg daily for the prevention of preeclampsia.2,3 The International Federation of Gynecology and Obstetrics (FIGO) recommends aspirin 150 mg daily for the prevention of preeclampsia.15 The FIGO recommendation is based, in part, on the results of a large international clinical trial that randomly assigned 1,776 women at high risk for preeclampsia as determined by clinical factors plus biomarker and ultrasound screening to receive aspirin 150 mg daily or placebo daily initiated at 11 to 14 weeks’ gestation and continued until 36 weeks’ gestation.16 Preeclampsia before 37 weeks’ gestation occurred in 4.3% and 1.6% of women in the placebo and aspirin groups (OR, 0.38; 95% CI, 0.20–0.74; P = .004).16 FIGO recommends that women at risk for preeclampsia with a body mass <40 kg take aspirin 100 mg daily and women with a body mass ≥40 kg take aspirin at a dose of 150 mg daily. For women who live in a country where aspirin is not available in a pill containing 150 mg, FIGO recommends taking two 81 mg tablets.15 FIGO recommends initiating aspirin between 11 and 14 weeks and 6 days of gestation and continuing aspirin therapy until 36 weeks of gestation.15
Aspirin is an inexpensive intervention with many possible benefits
For many nulliparous women and some parous women aspirin treatment initiated early in pregnancy will improve maternal and newborn outcomes, including reducing the risk of preeclampsia, preterm birth, and intrauterine growth restriction.1 Obstetricians may want to begin to expand the indications for offering aspirin to prevent preeclampsia from those recommended by ACOG and the USPSTF to include nulliparous women with a booking systolic pressure of 120 to 134 mm Hg and women whose pregnancy was the result of an assisted reproduction treatment that used cryopreserved embryos. In addition, obstetricians who currently prescribe 81 mg of aspirin daily might want to consider increasing the prescribed dose to 162 mg of aspirin daily (two 81 mg tablets daily or one-half of a 325 mg tablet). Aspirin costs about less than 5 cents per 81 mg tablet (according to GoodRx website). It is an inexpensive intervention that could benefit many mothers and newborns. ●
Authors of a recent Cochrane review concluded that low-dose aspirin treatment of 1,000 pregnant women at risk of developing preeclampsia resulted in 16 fewer cases of preeclampsia, 16 fewer preterm births, 7 fewer cases of small-for-gestational age newborns, and 5 fewer fetal or neonatal deaths.1
The American College of Obstetricians and Gynecologists (ACOG) and the US Preventive Services Task Force (USPSTF) recommend treatment with 81 mg of aspirin daily, initiated before 16 weeks of pregnancy to prevent preeclampsia in women with one major risk factor (personal history of preeclampsia, multifetal gestation, chronic hypertension, type 1 or 2 diabetes, renal or autoimmune disease) or at least two moderate risk factors (nulliparity; obesity; mother or sister with preeclampsia; a sociodemographic characteristic such as African American race or low socioeconomic status; age ≥35 years; personal history factors such as prior low birth weight infant, previous adverse pregnancy outcome, or >10-year interpregnancy interval).2,3 Healthy pregnant women with a previous uncomplicated full-term delivery do not need treatment with low-dose aspirin.2,3
However, evolving data and expert opinion suggest that expanding the indications for aspirin treatment and increasing the recommended dose of aspirin may be warranted.
Nulliparity
Nulliparity is the single clinical characteristic that is associated with the greatest number of cases of preeclampsia.4 Hence, from a public health perspective, reducing the rate of preeclampsia among nulliparous women is a top priority.
ACOG and USPSTF do not recommend aspirin treatment for all nulliparous women because risk factors help to identify those nulliparous women who benefit from aspirin treatment.
However, a recent cost-effectiveness analysis compared the health care costs and rates of preeclampsia for 4 prevention strategies among all pregnant women in the United States (nulliparous and parous)5:
- no aspirin use
- use of aspirin based on biomarker and ultrasound measurements
- use of aspirin based on USPSTF guidelines for identifying women at risk
- prescription of aspirin to all pregnant women.
Health care costs and rates of preeclampsia were lowest with the universal prescription of aspirin to all pregnant women in the United States. Compared with universal prescription of aspirin, the USPSTF approach, the biomarker-ultrasound approach, and the no aspirin approach were associated with 346, 308, and 762 additional cases of preeclampsia per 100,000 women. In sensitivity analyses, universal aspirin was the optimal strategy under most assumptions.
Another cost effectiveness analysis concluded that among nulliparous pregnant women, universal aspirin treatment was superior to aspirin treatment based on biomarker-ultrasound identification of women at high risk.6
In a recent clinical trial performed in India, Guatemala, Pakistan, Democratic Republic of Congo, Kenya, and Zambia, 14,361 nulliparous women were randomly assigned to placebo or 81 mg of aspirin daily between 6 and 14 weeks of gestation.7 Preterm birth (<37 weeks’ gestation) occurred in 13.1% and 11.6% of women treated with placebo or aspirin (relative risk [RR], 0.89; 95% confidence interval [CI], 0.81 to 0.98, P = .012). Most of the decrease in preterm birth appeared to be due to a decrease in the rate of preeclampsia in the aspirin-treated nulliparous women. The investigators also noted that aspirin treatment of nulliparous women resulted in a statistically significant decrease in perinatal mortality (RR, 0.86) and early preterm delivery, <34 weeks’ gestation (RR, 0.75).
Universal prescription of low-dose aspirin to nulliparous women in order to prevent preeclampsia and preterm birth may become recognized as an optimal public health strategy. As a step toward universal prescription of aspirin to nulliparous women, an opt-out rather than a screen-in strategy might be considered.8

Continue to: Booking systolic blood pressure, 120 to 134 mm Hg...
Booking systolic blood pressure, 120 to 134 mm Hg
All obstetricians recognize that women with chronic hypertension should be treated with low-dose aspirin because they are at high risk for preeclampsia. However, there is evidence that nulliparous women with a booking systolic pressure ≥120 mm Hg might also benefit from low-dose aspirin treatment. In one US trial, 3,135 nulliparous normotensive women (booking blood pressure [BP] <135/85 mm Hg) were randomly assigned to treatment with aspirin (60 mg daily) or placebo initiated between 13 and 26 weeks’ gestation. Preeclampsia occurred in 6.3% and 4.6% of the women treated with placebo or aspirin, respectively (RR, 0.7; 95% CI, 0.6–1.0; P = .05).9 A secondary analysis showed that, among 519 nulliparous women with a booking systolic BP from 120 to 134 mm Hg, compared with placebo, low-dose aspirin treatment reduced the rate of preeclampsia from 11.9% to 5.6%.9 Aspirin did not reduce the rate of preeclampsia among nulliparous women with a booking systolic BP <120 mm Hg.9 A systematic review of risk factors for developing preeclampsia reported that a booking diastolic BP of ≥80 mm Hg was associated with an increased risk of developing preeclampsia (RR, 1.38).10
The American Heart Association (AHA) and the American College of Cardiology (ACC) recently updated the definition of hypertension.11 Normal BP is now defined as a systolic pressure <120 mm Hg and diastolic pressure <80 mm Hg. Elevated BP is a systolic pressure of 120 to 129 mm Hg and diastolic pressure of <80 mm Hg. Stage I hypertension is a systolic BP from 130 to 139 mm Hg or diastolic blood pressure from 80 to 89 mm Hg. Stage II hypertension is a systolic BP of ≥140 mm Hg or diastolic blood pressure ≥90 mm Hg.11
A recent study reported that 90% of women at 12 weeks’ gestation have a BP of ≤130 mm Hg systolic and ≤80 mm Hg diastolic, suggesting that the AHA-ACC criteria for stage I hypertension are reasonable.12 Obstetricians have not yet fully adopted the AHA-ACC criteria for defining stage I hypertension in pregnant women. Future research may demonstrate that a booking systolic BP
≥130 mm Hg or a diastolic BP ≥80 mm Hg are major risk factors for developing preeclampsia and warrant treatment with low-dose aspirin.

Continue to: Pregnancy resulting from fertility therapy...
Pregnancy resulting from fertility therapy
Current ACOG and USPSTF guidelines do not specifically identify pregnancies resulting from assisted reproductive technology as a major or moderate risk factor for preeclampsia.2,3 In a study comparing 83,582 births resulting from in vitro fertilization (IVF) and 1,382,311 births to fertile women, treatment with autologous cryopreserved embryos (adjusted odds ratio [aOR], 1.30), fresh donor embryos (aOR, 1.92), and cryopreserved donor embryos (aOR, 1.70) significantly increased the risk of preeclampsia.13 However, use of fresh autologous embryos did not increase the risk of preeclampsia (aOR, 1.04). These associations persisted after controlling for diabetes, hypertension, body mass index, and cause of infertility.13
Other studies also have reported that use of cryopreserved embryos is associated with a higher rate of preeclampsia than use of fresh autologous embryos. In a study of 825 infertile women undergoing IVF and randomly assigned to single embryo cryopreserved or fresh cycles, the rate of preeclampsia was 3.1% and 1.0% in the pregnancies that resulted from cryopreserved versus fresh cycles.14
What is the optimal dose of aspirin?
ACOG and the USPSTF recommend aspirin 81 mg daily for the prevention of preeclampsia.2,3 The International Federation of Gynecology and Obstetrics (FIGO) recommends aspirin 150 mg daily for the prevention of preeclampsia.15 The FIGO recommendation is based, in part, on the results of a large international clinical trial that randomly assigned 1,776 women at high risk for preeclampsia as determined by clinical factors plus biomarker and ultrasound screening to receive aspirin 150 mg daily or placebo daily initiated at 11 to 14 weeks’ gestation and continued until 36 weeks’ gestation.16 Preeclampsia before 37 weeks’ gestation occurred in 4.3% and 1.6% of women in the placebo and aspirin groups (OR, 0.38; 95% CI, 0.20–0.74; P = .004).16 FIGO recommends that women at risk for preeclampsia with a body mass <40 kg take aspirin 100 mg daily and women with a body mass ≥40 kg take aspirin at a dose of 150 mg daily. For women who live in a country where aspirin is not available in a pill containing 150 mg, FIGO recommends taking two 81 mg tablets.15 FIGO recommends initiating aspirin between 11 and 14 weeks and 6 days of gestation and continuing aspirin therapy until 36 weeks of gestation.15
Aspirin is an inexpensive intervention with many possible benefits
For many nulliparous women and some parous women aspirin treatment initiated early in pregnancy will improve maternal and newborn outcomes, including reducing the risk of preeclampsia, preterm birth, and intrauterine growth restriction.1 Obstetricians may want to begin to expand the indications for offering aspirin to prevent preeclampsia from those recommended by ACOG and the USPSTF to include nulliparous women with a booking systolic pressure of 120 to 134 mm Hg and women whose pregnancy was the result of an assisted reproduction treatment that used cryopreserved embryos. In addition, obstetricians who currently prescribe 81 mg of aspirin daily might want to consider increasing the prescribed dose to 162 mg of aspirin daily (two 81 mg tablets daily or one-half of a 325 mg tablet). Aspirin costs about less than 5 cents per 81 mg tablet (according to GoodRx website). It is an inexpensive intervention that could benefit many mothers and newborns. ●
- Duley L, Meher S, Hunter KE, et al. Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2019;CD004659.
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 743: low-dose aspirin use during pregnancy. Obstet Gynecol. 2018;132:e44-e52.
- LeFevre ML; U.S. Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force Recommendation Statement. Ann Int Med. 2014;161: 819-826.
- Bartsch E, Medcalf KE, Park AL, et al. Clinical risk factors for pre-eclampsia determined in early pregnancy: systematic review and meta-analysis of large cohort studies. BMJ. 2016;353:i1753.
- Mallampati D, Grobman W, Rouse DJ, et al. Strategies for prescribing aspirin to prevent preeclampsia: a cost-effectiveness analysis. Obstet Gynecol. 2019;134:537-544.
- Mone F, O’Mahony JF, Tyrrell E, et al. Preeclampsia prevention using routine versus screening test-indicated aspirin in low-risk women. Hypertension. 2018;72:1391-1396.
- Hoffman MK, Goudar SS, Kodkany BS, et al. Low-dose aspirin for the prevention of preterm delivery in nulliparous women with a singleton pregnancy (ASPIRIN): a randomised, double-blind, placebo-controlled trial. Lancet. 2020;395:285-293.
- Ayala NK, Rouse DJ. A nudge toward universal aspirin for preeclampsia prevention. Obstet Gynecol. 2019;133:725-728.
- Sibai BM, Caritis SN, Thom E, et al. Prevention of preeclampsia with low-dose aspirin in healthy, nulliparous pregnant women. The National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. N Engl J Med. 1993;329:1213-1218.
- Duckitt K, Harrington D. Risk factors for preeclampsia at antenatal booking: systematic review of controlled studies. BMJ. 2005;330:565.
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71:2199-2269.
- Green LJ, Mackillop LH, Salvi D, et al. Gestation-specific vital sign reference ranges in pregnancy. Obstet Gynecol. 2020;135:653-664.
- Luke B, Brown MB, Eisenberg ML, et al. In vitro fertilization and risk for hypertensive disorders of pregnancy: associations with treatment parameters. Am J Obstet Gynecol. October 17, 2019. doi:10.1016/j.ajog.2019.10.003.
- Wei D, Liu JY, Sun Y, et al. Frozen versus fresh single blastocyst transfer in ovulatory women: a multicentre, randomised controlled trial. Lancet. 2019;393:1310-1318.
- Poon LC, Shennan A, Hyett JA, et al. International Federation of Gynecology and Obstetrics (FIGO) initiative on preeclampsia: A pragmatic guide for first-trimester screening and prevention. Int J Gynaecol Obstet. 2019;145(suppl 1):1-33.
- Rolnik DL, Wright D, Poon LC, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377:613-622.
- Duley L, Meher S, Hunter KE, et al. Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2019;CD004659.
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 743: low-dose aspirin use during pregnancy. Obstet Gynecol. 2018;132:e44-e52.
- LeFevre ML; U.S. Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force Recommendation Statement. Ann Int Med. 2014;161: 819-826.
- Bartsch E, Medcalf KE, Park AL, et al. Clinical risk factors for pre-eclampsia determined in early pregnancy: systematic review and meta-analysis of large cohort studies. BMJ. 2016;353:i1753.
- Mallampati D, Grobman W, Rouse DJ, et al. Strategies for prescribing aspirin to prevent preeclampsia: a cost-effectiveness analysis. Obstet Gynecol. 2019;134:537-544.
- Mone F, O’Mahony JF, Tyrrell E, et al. Preeclampsia prevention using routine versus screening test-indicated aspirin in low-risk women. Hypertension. 2018;72:1391-1396.
- Hoffman MK, Goudar SS, Kodkany BS, et al. Low-dose aspirin for the prevention of preterm delivery in nulliparous women with a singleton pregnancy (ASPIRIN): a randomised, double-blind, placebo-controlled trial. Lancet. 2020;395:285-293.
- Ayala NK, Rouse DJ. A nudge toward universal aspirin for preeclampsia prevention. Obstet Gynecol. 2019;133:725-728.
- Sibai BM, Caritis SN, Thom E, et al. Prevention of preeclampsia with low-dose aspirin in healthy, nulliparous pregnant women. The National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. N Engl J Med. 1993;329:1213-1218.
- Duckitt K, Harrington D. Risk factors for preeclampsia at antenatal booking: systematic review of controlled studies. BMJ. 2005;330:565.
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71:2199-2269.
- Green LJ, Mackillop LH, Salvi D, et al. Gestation-specific vital sign reference ranges in pregnancy. Obstet Gynecol. 2020;135:653-664.
- Luke B, Brown MB, Eisenberg ML, et al. In vitro fertilization and risk for hypertensive disorders of pregnancy: associations with treatment parameters. Am J Obstet Gynecol. October 17, 2019. doi:10.1016/j.ajog.2019.10.003.
- Wei D, Liu JY, Sun Y, et al. Frozen versus fresh single blastocyst transfer in ovulatory women: a multicentre, randomised controlled trial. Lancet. 2019;393:1310-1318.
- Poon LC, Shennan A, Hyett JA, et al. International Federation of Gynecology and Obstetrics (FIGO) initiative on preeclampsia: A pragmatic guide for first-trimester screening and prevention. Int J Gynaecol Obstet. 2019;145(suppl 1):1-33.
- Rolnik DL, Wright D, Poon LC, et al. Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia. N Engl J Med. 2017;377:613-622.